Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
A Arte da Anatomia (EBook)
Desde a Antiguidade, os médicos, anatomistas e artistas se dedicaram a descrever e representar o corpo humano, por meio de desenhos, pinturas, esculturas e outras formas de representação artística. Com o passar dos séculos, houve uma evolução significativa na forma como as ilustrações anatômicas eram produzidas, desde as primeiras representações rudimentares até as ilustrações altamente detalhadas e realistas que temos hoje.
Boa Leitura!!!
Gallstone Ileus
INTRODUCTION
Gallstone ileus is a misnomer: this condition is not a physiologic ileus at all, but a mechanical obstruction of the intestinal lumen (most commonly the small bowel) by a large gallstone that has passed through a cholecystoenteric fistula. Cholecystoenteric fistulae may occur from the gallbladder to the adjacent luminal viscera-duodenum (most common), stomach, or colon. Gallstone obstruction of the stomach at the pylorus is known as Bouveret’s syndrome. Cholecystocolic fistula is less common. Colonic obstruction in this situation typically occurs at the sigmoid colon. Most common is cholecystoduodenal fistula, with a large (usually >2 cm) gallstone passing through the small bowel and becoming lodged in the terminal ileum. Cholecystoenteric fistula is felt to be caused by a combination of pressure, necrosis, and inflammation with chronic longstanding gallstone disease. Up to 25% of patients who develop gallstone ileus will harbor multiple stones in the alimentary tract; therefore, a close inspection of the entire intestine is important at the time of operation.
CLINICAL PRESENTATION
Over the past few years, however, the incidence of gallstone ileus has been shown to be greater than previously thought. Several recent large population-based series have found that gallstone ileus accounts for approximately 0.1% of all small bowel obstructions. The disease usually affects women (70%) and those in the seventh or eighth decade of life. Most patients present with bloating, crampy abdominal pain, and vomiting, symptoms typical of mechanical small bowel obstruction. A careful history may reveal earlier episodic colicky right upper quadrant abdominal pain consistent with gallstone disease. The classic finding on plain abdominal radiograph is that of Rigler’s triad (pneumobilia, dilated small bowel loops with air-fluid levels, and a large, calcified gallstone in the lumen of the small bowel). Currently, computed tomography (CT) is used ubiquitously. CT has 99% accuracy for diagnosing gallstone ileus. Typical CT findings include pneumobilia, dilated loops of small bowel with air-fluid levels consistent with small bowel obstruction, and transition point with the ectopic stone always visible radiologically.
MANAGEMENT
Operation is required for all patients with gallstone ileus, as spontaneous passage of these large stones is rare once the patient has become symptomatic. It is crucial to optimize the patient physiologically as much as possible in this semi urgent situation, with the understanding that most gallstone ileus patients are elderly and commonly have numerous medical comorbidities. Two contemporary series of registry data have expanded our understanding of gallstone ileus. This condition was once thought to be relatively rare; however, the National Inpatient Sample study identified 3268 gallstone ileus patients, which accounts for approximately 0.1% of all patients admitted to the hospital with mechanical small bowel obstruction during this time period. In this series, overall hospital mortality was substantial at 6.7%. Mortality was significantly higher in patients who underwent cholecystectomy and closure of the biliary fistula compared to those who simply had small bowel obstruction addressed by cholecystolithotomy. Overall, 77% of the 3268 patients had small bowel obstruction pathology treated and the remaining 23% had biliary fistula closed and cholecystectomy at the same operation. An interesting finding was the substantial incidence of postoperative renal insufficiency, or approximately 30% in the entire group of patients. The latter finding highlights the need for preoperative resuscitation and close attention to postoperative fluid management. The surgeon must consider carefully feasibility of same operation intervention to repair biliary pathology: It is safe to defer biliary operation to a later date with a second staged operation. When this strategy of two stage operation is selected, surgeons should consider and counsel their patient regarding the substantial incidence of recurrent biliary symptoms.
Tratamento Cirúrgico da ACALASIA ESOFÁGICA
A acalasia, definida como a falha ou relaxamento incompleto do esfíncter esofágico inferior (EEI), acompanhada de aperistalse do corpo esofágico na ausência de obstrução mecânica, é o tipo mais comum de distúrbio da motilidade esofágica. Tem uma incidência de 1 em 100.000 pessoas, com uma prevalência de 10 em 100.000. Não há diferença na prevalência de gênero entre as idades de 30 e 60 anos. A causa primária da acalasia ainda é indeterminada, mas acredita-se que surja da degeneração das células ganglionares inibitórias no plexo miontérico do EEI e corpo esofágico. Fatores associados a um risco aumentado de acalasia incluem distúrbios virais/neurodegenerativos, síndrome de Down, diabetes mellitus tipo 1, hipotireoidismo e condições autoimunes, como a síndrome de Sjögren, o lúpus eritematoso sistêmico e a uveíte. Os casos familiares são raros.
O diagnóstico de acalasia deve ser suspeitado em pacientes com disfagia para sólidos e líquidos que não melhora com o uso de inibidores da bomba de prótons. Se não for tratada, a acalasia é uma doença progressiva que pode evoluir para megaesôfago e está associada a um aumento do risco de carcinoma de células escamosas do esôfago. Embora não existam critérios padronizados para determinar a gravidade da doença, o diâmetro e a confirmação do esôfago dentro da cavidade torácica são geralmente considerados os dois principais fatores. A escala de Eckardt é uma escala frequentemente usada para avaliar a gravidade da doença e a eficácia da terapia. Qualitativamente, a acalasia grave é definida como um diâmetro esofágico maior que 6 cm; a acalasia em estágio avançado inclui a angulação distal, um esôfago sigmóide/tortuoso com diâmetro maior que 6 cm ou um megaesôfago com diâmetro maior que 10 cm. Aproximadamente 5% a 15% das pessoas com acalasia evoluem para acalasia em estágio avançado, são geralmente resistentes aos tratamentos endoscópicos e cirúrgicos iniciais e, em última instância, requerem uma esofagectomia.
MIOTOMIA Á HELLER
A miotomia laparoscópica de Heller (LHM) com fundoplicatura parcial, desenvolvida como uma alternativa minimamente invasiva à miotomia anterior aberta tradicional e posteriormente à miotomia toracoscópica, tem sido o padrão ouro para o tratamento da acalasia nas últimas três décadas. O objetivo da miotomia é abrir completamente o EEI e aliviar a disfagia. A LHM proporciona alívio sintomático inicial da disfagia em cerca de 90% dos pacientes com tipos I e II de acalasia e em 50% dos pacientes com tipo III de acalasia, enquanto diminui as taxas de refluxo pós-operatório. Em comparação com os procedimentos de miotomia aberta, a LHM está associada a menor dor pós-operatória, menor tempo de internação hospitalar e retorno mais precoce à função. A LHM é indicada como tratamento de primeira linha para todos os candidatos à cirurgia com acalasia que estejam dispostos a se submeter à cirurgia ou para aqueles que falharam na dilatação endoscópica. A operação consiste em dividir os músculos do EEI, seguida por uma fundoplicatura para diminuir o refluxo pós-operatório. A fundoplicatura parcial é favorecida em relação à fundoplicatura total porque reduz a falha do tratamento. As duas principais complicações da cirurgia são perfuração da mucosa e DRGE.
Postpancreatectomy Hemorrhage
Postoperative complications represent one of the most debated topics in pancreatic surgery. Indeed, the rate of complications following pancreatectomy is among the highest in abdominal surgery, with morbidity ranging between 30 and 60%. They are often characterized by elevated clinical burden, with a consequent challenging postoperative management. Mortality rates can exceed 5%. The impact on patient recovery and hospital stay eventually leads to massive utilization of resources and increases costs for the health system. The International Study Group of Pancreatic Surgery (ISGPS) provides standardized definitions and clinically based classifications for the most common complications after pancreatectomy, including postoperative pancreatic fistula (POPF), post pancreatectomy hemorrhage (PPH), delayed gastric emptying (DGE), bile leakage, and chyle leak.
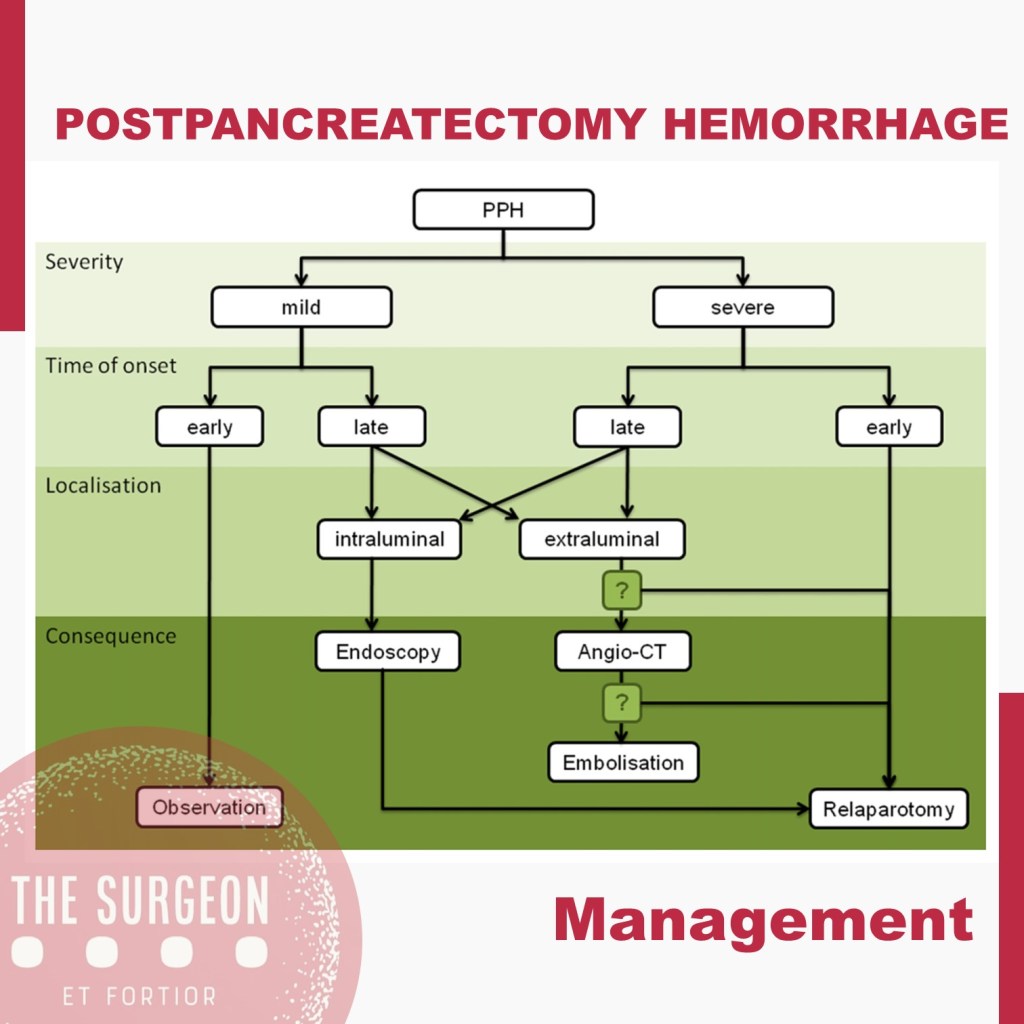
Post Pancreatectomy Hemorrhage
Despite its lower incidence compared with POPF with reported rates of 3–10% after pancreatectomy, PPH remains one of the major of postoperative complications, with mortality rates ranging from 30 to 50%. According to current ISGPS recommendations, PPH is classified in three grades (A, B, and C) based on two main criteria: timing of the hemorrhage and severity of the bleeding. The timing is dichotomized as early, occurring within 24 hours from the index surgery, and late, when it happens afterwards.
Management is tailored according to the clinical picture, the timing of onset, and the presumptive location. Early hemorrhage is generally due to either unsuccessful intraoperative hemostasis or to an underlying coagulopathy. The vast majority of patients are basically asymptomatic, with PPH having no influence on the postoperative course. However, when the bleeding is severe, re-laparotomy is recommended, with the aim of finding and controlling the source of bleeding. This approach usually guarantees an uneventful subsequent course. Late PPH is often challenging and the pathogenesis is diverse. Vascular erosion secondary to POPF or intraabdominal abscess, late failure of intraoperative hemostatic devices, arterial pseudoaneurysm, and intraluminal ulceration are some of the most common causes. In this setting, surgical access to the source of bleeding may be challenging. Angiography (if extraluminal) and endoscopy (if intraluminal) represent the primary approaches to treatment. Surgery is reserved for hemodynamically unstable patients and for those who present with deteriorating condition, multiorgan failure or sepsis. Given these assumptions, except for early mild events, contrast-enhanced abdominal CT is crucial in all cases of PPH, possibly allowing identification of the source and planning the management accordingly. Also, it should be noted that late massive hemorrhages may be preceded by mild self-limiting sentinel bleeds. A prompt abdominal CT scan aimed at excluding vascular lesions is therefore strongly recommended in these cases.
The incidence of complications following pancreatic resection remains high. The ISGPS established standardized definitions and clinical grading systems for POPF, PPH, DGE, and biliary and chyle leak. These classification systems have enabled unbiased comparisons of intraoperative techniques and management decisions. However, the management policies of these complications are most often driven by a patient’s condition and local surgical expertise and is not always based on the available high-level evidence. The development of high-volume specialized units with appropriate resources and multidisciplinary experience in complication management might further improve the evidence and the outcomes.
Subtotal cholecystectomy for difficult acute cholecystitis
Laparoscopic cholecystectomy is considered the gold standard for treatment of benign gallbladder diseases. Cholecystectomy using this method can be completed in 90% of elective cholecystectomies and 70% of emergency cholecystectomies. Acute cholecystitis, especially if difficult, can change the above paradigm, resulting in open conversion or change of technique. The conditions that define a difficult cholecystectomy are as follows: necessity of conversion from laparoscopic to open surgery; duration of procedure greater than 180 min; blood loss greater than 300 ml; and urgent need for involvement of a more experienced surgeon. One of the “rescue” procedures to complete the surgery safely (both for the surgeons and patients) is subtotal cholecystectomy (STC). Open and laparoscopic subtotal cholecystectomy have been reported. For many surgeons, this is considered a bail out technique, and the timing of decision making is crucial to avoid catastrophic complications. Te capability to perform STC in laparoscopy is increasingly requested during difficult laparoscopic cholecystectomy. Difficult LC has a risk of BDI from 3 to 5 times higher in laparoscopy than open surgery. In case of operative difficulties of young surgeons mostly trained in laparoscopy the help of senior surgeons is strongly recommended. The purpose of the present study is to clarify how laparoscopic subtotal cholecystectomy may be used to complete a difficult cholecystectomy for acute cholecystitis without serious complications.

Biliary leakage represents the most frequent complication of incomplete resection of the gallbladder wall in cases of difficult acute cholecystectomy treated with subtotal cholecystectomy. This complication is rarely fatal but requires correct treatment. If bile leakage does not stop spontaneously seven days postoperatively, the possible treatments are endoscopic biliary sphincterotomy, endoscopic plastic stent, and a fully covered self-expanding metal stent. When performing closure of the gallbladder stump, suturing the anterior residual of both anterior and posterior wall represents the best method to have fewer complications. Complications, if not lethal, decrease the patient’s quality of life. Intraoperatively, it is of utmost importance to carefully expose the gallbladder stump to avoid left-in-place stones, wash the entire cavity and drain the abdomen. Bile duct injuries can be a significant complication in this type of surgery. Prevention of the lesions with conversion from laparoscopic to open, or the opinion of older surgeon in case of difficulties is strongly recommended. Mortality is a very rare complication. The limitations of our study are given by the heterogeneity of the techniques used for LSC and the lack of a long-term follow-up analyzing the related complications.
The “BAD” Gallbladder
Once the decision for surgery has been made, an operative plan needs to be discussed and implemented. Should one initially start with laparoscopic surgery for the “bad gallbladder”? If a laparoscopic approach is taken, when should bail-out maneuvers be attempted? Is converting to open operation still the standard next step? A 2016 study published by Ashfaq and colleagues sheds some light on our first question. They studied 2212 patients who underwent laparoscopic cholecystectomy, of which 351 were considered “difficult gallbladders.” A difficult gallbladder was considered one that was necrotic or gangrenous, involved Mirizzi syndrome, had extensive adhesions, was converted to open, lasted more than 120 minutes, had a prior tube cholecystostomy, or had known gallbladder perforation. Seventy of these 351 operations were converted to open. The indications for conversion included severe inflammation and adhesions around the gallbladder rendering dissection of triangle of Calot difficult (n 5 37 [11.1%]), altered anatomy (n 5 14 [4.2%]), and intraoperative bleeding that was difficult to control laparoscopically (n 5 6 [1.8%]). The remaining 13 patients (18.5%) included a combination of cholecystoenteric fistula, concern for malignancy, common bile duct exploration for stones, and inadvertent enterotomy requiring small bowel repair. Comparing the total laparoscopic cholecystectomy group and the conversion groups, operative time and length of hospital stay were significantly different; 147 +- 47 minutes versus 185 +- 71 minutes (P<.005) and 3+-2 days versus 5+-3 days (P 5 .011), respectively. There was no significant difference in postoperative hemorrhage, subhepatic collection, cystic duct leak, wound infection, reoperation, and 30-day mortality.2 From these findings, we can glean that most cholecystectomies should be started laparoscopically, because it is safe to do so. It is the authors’ practice to start laparoscopically in all cases.
BAILOUT PROCEDURES
Despite the best efforts of experienced surgeons, it is sometimes impossible to safely obtain the critical view of safety in a bad gallbladder with dense inflammation and even scarring in the hepatocystic triangle. Continued attempts to dissect in this hazardous region can lead to devastating injury, including transection of 1 or both hepatic ducts, the common bile duct, and/or a major vascular injury (usually the right hepatic artery). Therefore, it is imperative that any surgeon faced with a bad gallbladder have a toolkit of procedures to safely terminate the operation while obtaining maximum symptom and source control, rather than continue to plunge blindly into treacherous terrain. If the critical view of safety cannot be achieved owing to inflammation, and when further dissection in the hepatocystic triangle is dangerous, these authors default to laparoscopic subtotal cholecystectomy as our bail-out procedure of choice. The rationale for this approach is that it resolves symptoms by removing the majority of the gallbladder, leading to low (although not zero) rates of recurrent symptoms. It is safe, and can be easily completed laparoscopically, thus avoiding the longer hospital stay and morbidity of an open operation. There is now significant data supporting this approach. In a series of 168 patients (of whom 153 were laparoscopic) who underwent subtotal cholecystectomy for bad gallbladders, the mean operative time was 150 minutes (range, 70–315 minutes) and the average blood loss was 170 mL (range, 50–1500 mL). The median length of stay for these patients was 4 days (range, 1–68 days), and there were no common bile duct injuries.23 There were 12 postoperative collections (7.1%), 4 wound infections (2.4%), 1 bile leak (0.6%), and 7 retained stones (4.2%), but the 30-day mortality was similar to those who underwent a total laparoscopic cholecystectomy. A systematic review and meta-analysis by Elshaer and colleagues showed that subtotal cholecystectomy achieves comparable morbidity rates compared with total cholecystectomy. These data support the idea that we should move away from the idea that the only acceptable outcome for a cholecystectomy is the complete removal of a gallbladder, especially when it is not safe to do so. This shift toward subtotal cholecystectomy has been appropriately referred to as the safety first, total cholecystectomy second approach.
Managing the “difficult” gallbladder
The gold standard for the surgical treatment of symptomatic cholelithiasis is conventional laparoscopic cholecystectomy (LC). The “difficult gallbladder” is a scenario in which a cholecystectomy turns into an increased surgical risk compared with standard cholecystectomy. The procedure may be difficult due to processes that either obscure normal biliary anatomy (such as acute or chronic inflammation) or operative exposure (obesity or adhesions caused by prior upper abdominal surgery). So, when confronted with a difficult cholecystectomy, the surgeon has a must: to turn the operation into a safe cholecystectomy, which can mean conversion (to an open procedure), cholecystostomy, or partial/ subtotal cholecystectomy. The surgeon should understand that needs to rely on damage control, to prevent more serious complications if choosing to advance and progress to a complete cholecystectomy.
When to Predict a Difficult Laparoscopic Cholecystectomy
A difficult cholecystectomy may be predicted preoperatively based on patient characteristics and ultrasound and laboratory findings. This is probably a very important step in mitigating the high risk associated with a difficult procedure and may serve either to reschedule the procedure or design intraoperative strategies of management to guarantee a safe performance of the surgical procedure.
The following situations are associated with a higher chance of a difficult cholecystectomy:
• Acute cholecystitis (more than 5 days of onset)
• Previous cholecystitis episode
• Male sex
• Obesity
• Cirrhosis
• Sclero-atrophic gallbladder
• Thick walls (>5 mm)
• Previous signs of canalicular dwelling (clinical and laboratory)
Through multivariate analysis, Bourgoin identified these elements of predictive help to identify difficult LC: male sex, previous cholecystitis attack, fibrinogen, neutrophil, and alkaline phosphatase levels. Another important point is the fact of conversion from a laparoscopic procedure to an open and traditional cholecystectomy, usually through a right subcostal incision. Conversion should not be considered as a personal failure, and the surgeon needs to understand the concept of “safety first,” considering that conversion is performed in order to complete the procedure without additional risks and preventing complications and not solving intraoperative complications. It is also useful to define a time threshold to aid in the decision to convert. It is not worth taking an hour and a half and still dissecting adhesions, preventing the correct visualization of the cystic pedicle. This time limit represents a method to prevent inefficiencies in the operating room (OR) schedule as well as additional expenditures.
A smart surgeon should rely to conversion in the following situations:
• Lack of progress in the procedure
• Unclear anatomy/any grade of uncertainty
• CVS not achieved
• Bleeding/vascular injury
• BD injury
• Lack of infrastructure, expertise, and support
Final Remarks
The primary goal of a laparoscopic cholecystectomy in the treatment of symptomatic cholelithiasis is the safe remotion of the gallbladder and the absence of common bile duct injury. Some tips to take into account:
– Never perform a laparoscopic cholecystectomy without a skilled surgeon close by.
– Beware of the easy gallbladder.
– Slow down, take your time.
– Knowledge is power, conversión can be the salvation!
– Do not repair a bile duct injury (unless you have performed at least 25 hepaticojejunostomies).
– Do not ignore postoperative complaints (pain, jaundice, major abdominal discomfort, fever)
Other options when confronted with a difficult laparoscopic cholecystectomy are:
– A percutaneous cholecystostomy, if the risk was identified preoperatively or the patient is a poor surgical candidate;
– An intraoperative cholangiography, which may aid in identifying an injury to the bile duct and solve it, if you are an experienced surgeon;
– A subtotal or partial cholecystectomy;
– Ask for help;
– Conversion to an open procedure;
Liver Procurement
Donor Evaluation and Management
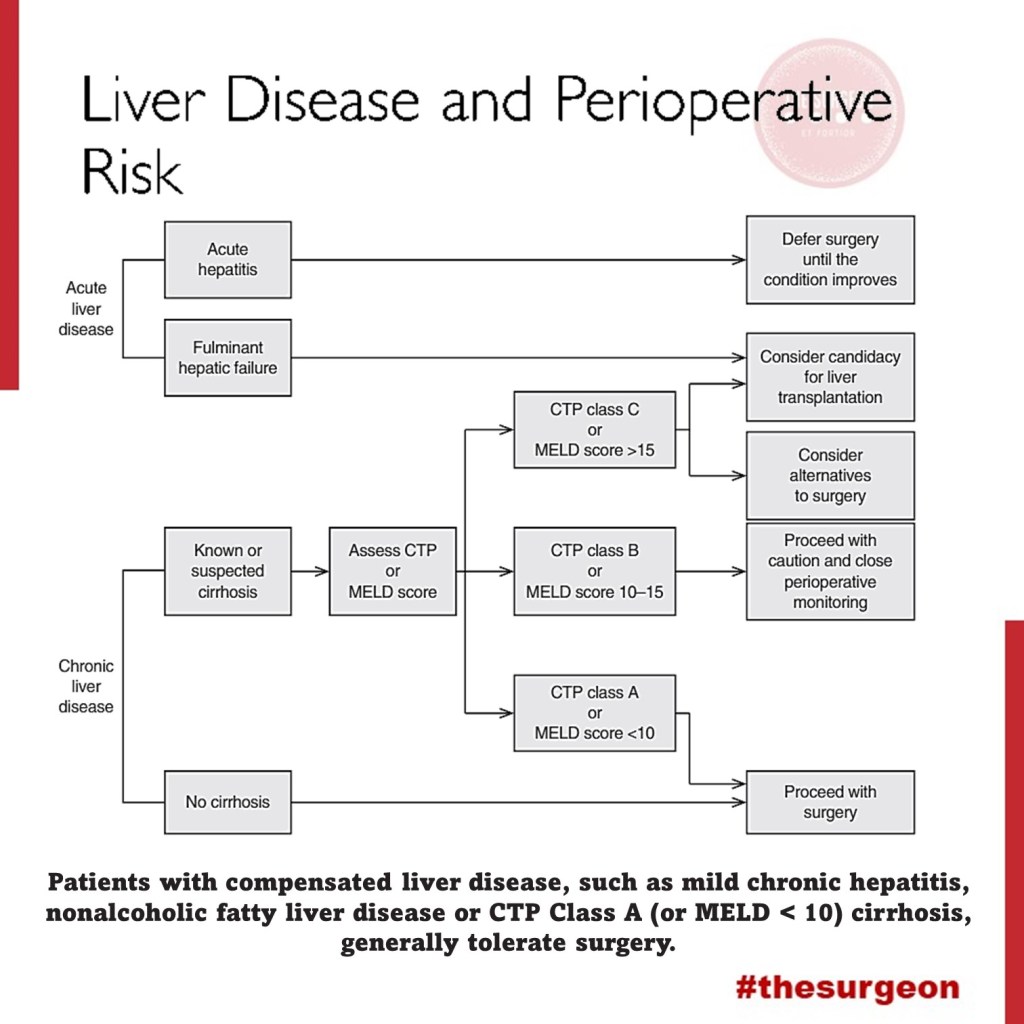
There are very few absolute contraindications for abdominal organ donation, which can be summarized in the short form CHUMP: (1) Creutzfeldt-Jakob disease, (2) active HIV infection, (3) uncontrolled donor sepsis, (4) history of melanoma or other malignancy that poses a risk for transmission regardless of the apparent disease – free period, and (5) past history of non-curable malignancy (curable malignancy such as localized small kidney tumors, localized prostate cancer, localized colon malignancy >5 years previously may be considered after careful risk/benefit assessment). In addition to these general criteria, there are organ-specific criteria for guiding the acceptance of a liver for transplantation. A history of hepatitis or alcoholism is certainly a warning sign, but both livers from HBsAg-positive and/or HCV-positive donors are currently used worldwide, and suitability for transplant must be judged on a case-by-case basis. In general, in the case of a marginal liver donor, the intraoperative assessment by the donor surgeon, in addition to liver biopsy pathological evaluation, is the best single piece of information.
Technical Aspects of Liver Procurement
A midline laparotomy from the xyphoid to the pubis is performed and the round ligament divided. The intra-abdominal organs are explored to check for eventual malignancies, and the quality of the liver is assessed: in the absence of contraindications for a transplant, a sternotomy can be performed. Of note, in the presence of prior heart surgery, the complete warm dissection should be made prior to the sternotomy. It is also prudential to isolate and encircle the aorta prior to sternotomy in order to be ready to cannulate in the event of cardiac arrest/injury at thoracotomy. A blunt dissection behind the sternum just below the jugular notch should be performed until the fingertip can be placed retrosternal around the jugular notch. The sternotomy is then performed in a cranial to caudal direction with the sternum saw to avoid left innominate vein injury. The division of the left triangular ligament allows the mobilization of the left lateral segments of the liver and the exposure of the supraceliac aorta just below the diaphragm to be encircled. The division of the falciform ligament up to the suprahepatic inferior vena cava (IVC) provides more mobility of the liver, necessary when the IVC must be divided from a cardiac graft. Before starting the dissection of the hepatoduodenal ligament, the hepatogastric ligament must be inspected by dividing the lesser omentum. This ligament is usually very thin and transparent so that any replaced or accessory left hepatic artery should be easily visible. In addition, palpation of the ventral border of the foramen of Winslow makes it possible to identify a possible accessory or replaced right hepatic artery. Variations in the hepatic arterial supply can complicate the hilar dissection in up to one third of donors.
THE HILAR STRUCTURES
The hilar structures of the liver are then dissected free; the common bile duct (CBD) is dissected on the level of the edge of the second duodenal portion after opening of the peritoneum and visualization of the duct. In difficult cases, due to a high BMI, following the cystic duct out of the gall bladder can help to identify the CBD. The CBD should be encircled from the lateral border of the hepatoduodenal ligament in order to avoid injury of the portal vein. The CBD and the gallbladder are opened and flushed with normosaline solution. The origins of the gastroduodenal, gastric, and splenic arteries are then identified and encircled and, in the case of liver only procurement, will be taped just before cross-clamping in order to increase flushing through the hepatic artery to the liver.
VASCULAR CANULATION / SOLUTION PRESERVATION
The aorta can be isolated by two approaches. One approach requires mobilization of the right colon on top of Gerota’s fascia and should be extended into a Kocher maneuver to uncover both the inferior vena cava and the abdominal aorta; the other approach is performed by opening the root of the mesentery from the Treitz fascia, along the margin of the duodenum until visualization of the right iliac vessels and ureter is achieved. The inferior mesenteric artery can be tied and divided, and the abdominal aorta, just 2–3 cm above the bifurcation, isolated and encircled. The lumbar arteries could be either tied or clipped and then cut in order to provide mobility of the aorta and facilitate the cannulation. Two umbilical tapes are placed around the dissected segment of the aorta and secured by clamps and will be used to secure aortic cannulae to the vessel. The inferior mesenteric vein (IMV) is most commonly used for access into the portal system by ligating the distal part of it but leaving it uncut to retract the vein with a mosquito clamp. Another tie is then placed around the cranial portion of the vein, using it for occlusion of the vein by retracting it while a partial incision of the vein is performed. The portal cannula can be inserted into the IMV while the tension of the occluding tie is decreased before tying it around the vein and inserted cannula. At this point, 30,000-IU heparin should be given to prevent the blood from clotting after the cross-clamping. Once these preliminary procedures have been completed, the aortic cannulae (20-F armed cannulae) can be inserted into the distal abdominal aorta and secured with the umbilical tapes.
CROSS-CLAMP
The subdiaphragmatic aorta is now clamped (cross-clamp), and cold preservation solution is then rapidly infused through the aortic and portal cannulae; the liver flow is decompressed by dividing the inferior vena cava in the chest. The abdomen is filled with water and ice. The choice of solution for infusion and its amount varies from center to center. The quality of the flush can be assessed by evaluating the outflow of the supradiaphragmatic IVC which should become more transparent with time as the blood in the abdominal organs is replaced by the preservation solution. After the flush is completed, some of the ice is removed from the abdomen to allow the cold dissection of the structures. The gastroduodenal, gastric, and splenic arteries can now be divided. Just below the gastroduodenal artery, the portal vein can be found and can be followed back, if pancreas procurement is not performed, by dividing the head of the pancreas. The cannulae in the IMV can now be removed, the splenic vein ligated and divided, and the venous cannulae replaced in the superior mesenteric vein once it is divided from its distal branches. The superior mesenteric artery (SMA) can now be found in the retro-pancreatic laminae and should be ligated, secured to a clamp and divided in order to find the aortic plane by following back the SMA. This dissection must be made on the left side of the SMA in order to avoid damage to a possible replaced or accessory right hepatic artery. The renal arteries are usually just below the SMA. They should be visualized before the suprarenal aorta is divided. This section must be made in 45°, first looking for ostia of accessory renal arteries before performing complete separation of the aorta. By following back the splenic and gastric arteries, the celiac trunk can be visualized. The aorta must now be divided just below the diaphragm, obtaining a patch containing the celiac trunk and the origin of the mesenteric artery. At this time point, a finger is placed in the supradiaphragmatic IVC helping to identify it while the diaphragm is cut. A portion of the diaphragm should be kept with the liver to ensure that this gross and fast dissection does not damage the organ. The diaphragm is cut to the right, and the incision is then continued between the right kidney and the liver, usually dividing the adrenal gland which is a good sign that none of the adjacent organs are damaged. The location for division of the infrahepatic IVC depends on the renal veins. These are identified on both sides, and the IVC can be safely divided on the virtual line about 1 cm above the renal veins. The only structures now holding the liver in the abdomen are the diaphragmatic pillars. By keeping the liver to the right thoracic cavity and holding the aortic patch, the resected IVC, and the portal vein with its cannulae, the liver removal can be completed by cutting the diaphragmatic muscles. The liver is freed and taken out of the abdomen. A further perfusion with cool preservation solution should be performed on the back table before packing the liver in the transportation box usually with 1 l of preservation solution. The liver can now be packed in the transportation box.
Liver Disease and Perioperative Risk
The liver is the biggest intestinal organ and plays a central role in the homeostasis of different physiological systems including nutrition and drug metabolism, the synthesis of plasma proteins and haemostatic factors, as well as the elimination of different endogenous and exogenous substances. Although the liver contributes with only 3% to total body weight, given its major role in homeostasis and high energy consumption, it receives 25% of total cardiac output (CO). Two vessels contribute to the perfusion of the liver. The majority (70%) of the hepatic perfusion is provided by the portal vein, which contributes 50% of the organ’s oxygen demand. The other 50% is provided by the hepatic artery, which makes up around 30% of total liver perfusion. Hepatic arterial blood flow is mainly dependent on the organ’s metabolic demands and controlled via autoregulatory mechanisms, whereas blood supply through the portal vein depends on the perfusion throughout the whole gastrointestinal tract and the spleen. This unique, dual perfusion system provides constant perfusion rates and oxygen supply, which is crucial for adequate liver function. These high oxygen demands are reflected in a hepatic vein saturation of almost 30%.
The liver is also unique in its ability of regeneration, which allows the performance of major surgery including, amongst others, extended resections of liver tumours, living donations and so on. Many patients have normal liver function parameters when they present for liver surgery, especially when the reason for resection is metastasis or a benign liver tumour. The most common causes of liver resections are the hepatocellular carcinoma (HCC) and the cholangiocellular carcinoma (CCC). Hepatocellular carcinoma (HCC) often develops in patients with underlying liver cirrhosis; many of these patients show signs of chronic liver dysfunction (CLD).
As explained previously, the liver plays a central role in a great deal of physiological systems. Therefore, in case of chronic liver dysfunction (CLD) or liver failure, several effects on other organ systems have to be expected. Consequently liver resections and bile duct surgery as having a high risk for perioperative cardiac events, with an estimated 30-day cardiac event rate (cardiac death and myocardial infarction) of more than 5%. Patients undergoing liver surgery pose a significant challenge to treating physicians in the perioperative period. Due to the improvement of surgical techniques, the “liver patient” is becoming more and more complex, confronting surgeons, anaesthetists and intensive care personnel with difficult intra- and postoperative courses, and considerable multiorgan disorders. The cornerstones of an optimal management are careful selection of the patients, appropriate monitoring and protection of the liver and other vital organs.
Role of The SURGEON
Attributes of a Good Surgeon
Realising the benefits that good leadership and teamwork can deliver requires commitment from all those involved in patient care. From the surgeon’s viewpoint there are numerous desirable attributes which are developed through medical school education, foundation training, core training and into professional practice. These are outlined below:
1. Clinical Care
An obvious consideration of what makes a “good surgeon” is the care provided to patients throughout the patient journey. This includes technical ability in the operating theatre and non-technical skills.
2. Maintenance and Improvement
Remaining up-to-date with innovations in surgical practice and patient are is an important attribute of a good surgeon. In doing so, one is able to inform patients and explain the reasons for and against procedures, allowing them to make an informed decision. Willingness to learn from others and improve from others by reviewing personal practice forms part of Continuing Professional Development; this is a requirement in a portfolio to meet revalidation and recertification criteria.
3. Teaching, Training and Supervision
Educating others forms part of professional development and surgeons frequently oversee projects for medical students or trainees. This requires knowledge of the objectives of the tasks undertaken, knowledge of what technical and non-technical skills should be improved and knowledge of how to encourage the development of these skills. The mentormentee relationship should work both ways, such that the mentee is able to approach their supervisor for assistance and is accepting of any constructive criticism delivered.
4. Relationships with Patients
Relationships with patients are fundamentally based on trust; the patient trusts that the surgeon will do all in their power to help them and their surgical journey. Obtaining informed consent prior to clinical care is based on trust and allows patient autonomy to be upheld. Developing relationships with patients begins from the first consultation and is continued after the day of an operation being undertaken. Acknowledging the needs of the individual and employing effective communication helps in developing an open relationship. In this way patients disclose their medical history and admit underlying fears, allowing better patient care to be delivered.
5. Relationships with Colleagues
Partnership with all members of the multidisciplinary clinical team, management, technicians and support staff fosters healthy working relationships. Consequently, patient care is enhanced through communication, enhanced productivity and an improved team dynamic. Understanding how a colleague works and taking action to facilitate a positive working environment is beneficial to all. Emotional intelligence forms an important component of working relationships, through the ability “to understand and recognize emotional states and to use that understanding to manage one’s self and other individuals or teams”.
6. Health
Maintenance of good personal health and knowing when you must stop working is important in the protection of patient safety. The relevant senior staff must be informed of communicable disease or blood-borne disease transmission. In addition, being vigilant of the health of colleagues forms part of protecting patient safety, for example, failure to report suspicion that the consultant consistently operates after several glasses of wine or that the CT2 has been seen smoking drugs can facilitate the propagation of errors in the workplace. Finally, surgeons are renowned for working at all hours, however acknowledgement that we all need rest is crucial in good patient care.
Surgical Management of PANCREATIC CANCER
INTRODUCTION
Pancreatic ductal adenocarcinoma (PDAC) carries one of the poorest overall prognosis of all human malignancies. The 5-year survival in patients with PDAC, for all stages, remains as low as 6–7%. The low survival rate is attributed to several factors, of which the two most important are aggressive tumor biology and late stage at which most patients are diagnosed. Only 10–20% of patients are eligible for resection at presentation, 30–40% are unresectable/locally advanced, and 50–60% are metastatic. Pancreatic cancer without distant metastasis can be divided into three categories: resectable, borderline resectable, and locally advanced. In absence of metastatic disease, the most important factor for improving survival and possibly offer cure is to achieve a margin-negative resection. Even after potential curative resection, most patients develop recurrences eventually, and 5-year survival of completely resected patients is only up to 25%. The aggressive tumor biology and its inherent resistance to chemotherapy and radiotherapy contributes to early recurrence and metastasis.
SURGICAL ADVANCES
Pancreatic cancer surgery has evolved over the past few decades and remains the cornerstone of treatment of resectable and borderline resectable tumors. Advances in modern imaging give precise information on disease extension and vascular involvement that aids in surgical planning in order to achieve a margin-negative resection.
SURGICAL TECHNIQUES
Surgical techniques for pancreatic cancer include pancreaticoduodenectomy, distal pancreatectomy with splenectomy, and total pancreatectomy. Standard lymphadenectomy for pancreatoduodenectomy should include removal of lymph node stations 5, 6, 8a, 12b1, 12b2, 12c, 13a, 13b, 14a, 14b, 17a, and 17b. Involvement of superior mesenteric vein (SMV)/portal vein(PV) was previously considered as a contraindication for resection. However, curative resection along with SMV/PV with vascular reconstruction has now become a standard practice in specialized high-volume centers. To improve margin-negative resections, specially in borderline resectable tumors with proximity to vascular structures, SMA first approach was proposed as a new modification of standard pancreatico-duodenectomy. In a systematic review, SMA first approach was shown to be associated with better perioperative outcomes, such as blood loss, transfusion requirements, pancreatic fistula, delayed gastric emptying, and reduced local and metastatic recurrence rates. In case of arterial involvement, there is no good evidence at present to justify arterial resections for right-sided pancreatic tumors. However, the modified Appleby procedure, which includes en bloc removal of celiac axis with or without arterial reconstruction, when used in appropriately selected patients, offers margin negative resection with survival benefit for locally advanced pancreatic body and tail tumors and should be performed in high-volume centers. Most evidence does not support advantage of more extended resections such as removal of the para-aortic lymph nodes and nerve plexus and multivisceral resections routinely. Such extended resections are associated with compromised quality of life because of associated higher perioperative morbidity and intractable diarrhea. However, in highly selected patients, with preserved performance status and stable or nonprogressive disease on neoadjuvant treatment, such extended resections can provide survival advantage over palliative treatments. Radical surgery in the presence of oligometastatic disease has also been reported to prolong survival in highly selected patients.
Adverse events in SURGERY
The surgical domain can be seen as more complex and high risk in its delivery of care than other non-interventional specialities. It is therefore not surprising that in the majority of studies of adverse events in healthcare, at least 50% occurred within the surgical domain and the majority of these in the operating theatre. Furthermore, at least half of these adverse events were also deemed preventable. Just as the multiple studies in the developed world have similar figures for adverse events in hospitalised patients across all specialities, there appears to be a similar rate of harm in surgery. A review of 14 studies, incorporating more than 16000 surgical patients, quoted an adverse event occurring in 14.4% of surgical patients. This was not simply minor harm; a full 3.6% of these adverse events were fatal, 10% severe and 34% moderately harmful. Gawande, a surgeon from Boston, made one of the first attempts to clarify the source of these adverse events.

This paper pioneered the concept that the majority of these adverse events were not due to lack of technical expertise or surgical skill on the part of the surgeon, finding instead that ‘systems factors’ were the main contributing factor in 86% of adverse events. The most common system factors quoted were related to the people involved and how they were functioning in their environment. Communication breakdown was a factor in 43% of incidents, individual cognitive factors (such as decision-making) were cited in 86%, with excessive workload, fatigue and the design or ergonomics of the environment also contributing.
These findings were confirmed in the systematic review of surgical adverse events, where it was found that errors in what were described as ‘nonoperative management’ were implicated in 8.32% of the study population versus only 2.5% contributed to by technical surgical error. In accordance with other high-risk industries, such as commercial aviation, the majority of these adverse events are therefore not caused by failures of technical skill on the part of the individual surgeon, but rather lie within the wider healthcare team, environment and system. Lapses and errors in communication, teamworking, leadership, situational awareness or decision-making all feature highly in post-hoc analysis of surgical adverse events. This knowledge of error causation has been prominent and acknowledged in most other high-risk industries for many years, but it is only recently that healthcare has appreciated this.
Hepatocellular ADENOMA
Benign liver tumours are common and are frequently found coincidentally. Most benign liver lesions are asymptomatic, although larger lesions can cause non-specific complaints such as vague abdominal pain. Although rare, some of the benign lesions, e.g. large hepatic adenomas, can cause complications such as rupture or bleeding. Asymptomatic lesions are often managed conservatively by observation. Surgical resection can be performed for symptomatic lesions or when there is a risk of malignant transformation. The type of resection is variable, from small, simple, peripheral resections or enucleations, to large resections or even liver transplantation for severe polycystic liver disease.
General Considerations
Hepatocellular adenomas (HCA) are rare benign hepatic neoplasms in otherwise normal livers with a prevalence of around 0.04% on abdominal imaging. HCAs are predominantly found in women of child-bearing age (2nd to 4th decade) with a history of oral contraceptive use; they occur less frequently in men. The association between oral contraceptive usage and HCA is strong and the risk for a HCA increases if an oral contraceptive with high hormonal potency is used, and if it is used for over 20 months. Long-term users of oral contraceptives have an estimated annual incidence of HCA of 3–4 per 100000. More recently, an increase in incidence in men has been reported, probably related to the increase in obesity, which is reported as another risk factor for developing HCA. In addition, anabolic steroid usage by body builders and metabolic disorders such as diabetes mellitus or glycogen storage disease type I are associated with HCAs. HCAs in men are generally smaller but have a higher risk of developing into a malignancy. In the majority of patients, only one HCA is found, but in a minority of patients more than 10 lesions have been described (also referred to as liver adenomatosis).
Clinical presentation
Small HCAs are often asymptomatic and found on abdominal imaging being undertaken for other purposes, during abdominal surgery or at autopsy. Some patients present with abdominal discomfort, fullness or (right upper quadrant) pain due to an abdominal mass. It is not uncommon that the initial symptoms of a HCA are acute onset of abdominal pain and hypovolaemic shock due to intraperitoneal rupture. In a series of patients who underwent resection, bleeding was reported in up to 25%. The risk of rupture is related to the size of the adenoma. Exophytic lesions (protruding from the liver) have a higher chance of bleeding compared to intrahepatic or subcapsular lesions (67% vs 11% and 19%, respectively, P<0.001). Lesions in segments II and III are also at higher risk of bleeding compared to lesions in the right liver (35% vs 19%, P = 0.049).
Management
There is no guideline for the treatment of HCAs, although there are general agreements. In men, all lesions should be considered for surgical resection independent of size, given the high risk of malignant transformation, while taking into account comorbidity and location of the lesion. Resection should also be considered in patients with HCAs due to a metabolic disorder. In women, lesions <5 cm can be observed with sequential imaging after cessation of oral contraceptive treatment. In larger tumours, treatment strategies vary. Some clinicians have proposed non-surgical management if hormone therapy is stopped and patients are followed up with serial radiological examinations. The time period of waiting is still under debate, however recent studies indicate that a waiting period of longer than 6 months could be justified.
More recently, the subtypes of the Bordeaux classification of HCA have been studied related to their risk of complications. Some groups report that percutaneous core needle biopsy is of limited value because the therapeutic strategy is based primarily on patient sex and tumour size. Others report a different therapeutic approach based on subtype. Thomeer et al. concluded that there was no evidence to support the use of subtype classification in the stratification and management of individual patients related to risk of bleeding. Size still remains the most important feature to predict those at risk of bleeding during follow-up. However, malignant transformation does seem to be related to differences in subtypes. β-catenin-mutated HCAs trigger a potent mitogenic signalling pathway that is prominent in HCC. Cases of inflammatory HCAs can also show activation of the β-catenin pathway with a risk of developing malignancy. Therefore, β-catenin-mutated and inflammatory HCAs are prone to malignant degeneration, and particularly if >5cm. In these circumstances, invasive treatment should be considered.
Liver Uptake on Cadaveric Donors for Transplant
Since the initial descriptions of orthotopic liver transplantation (OLT) in the 1960s, both the number of patients receiving transplants and the indications for the procedure have increased significantly. OLT represents the only treatment modality for many patients with a diverse spectrum of disease, with the predominant common factor end-stage liver failure. Advances in perioperative care of the donor and recipient, organ preservation methods, and surgical techniques have resulted in a 5 year overall survival of 78% for all recipients (Kim et al, 2015).
OPERATIVE TECHNIQUES
The first published description of human liver transplantation was by Starzl and colleagues in 1963 at the University of Colorado. In this seminal paper, the dismal outcomes of three OLT recipients were described, including one intraoperative death from uncorrectable coagulopathy and two survivors of 7 and 22 days. In addition to the pioneering conceptual framework and implementation of LT, the advanced techniques included grafts from non–heart-beating donors, venovenous bypass in the recipients, choledochocholedochostomy, and coagulation monitoring by using thromboelastography (TEG). Many of these concepts remain or have reentered the realm of LT more than 40 years after their initial description. Based largely on the initial body of work by Starzl and colleagues, this section describes the surgical procedures commonly used worlwide.
The typical deceased donor has had a catastrophic head injury or an intracerebral bleed, with brain death but without multisystem organ failure. Electrolyte imbalance and hepatic steatosis in the donor are predictors of graft nonfunction. A “donor risk index” has been derived to assess the likelihood of good graft function. Key adverse factors include older donor age (especially >60 years of age), use of a split or partial graft, and a non–heart-beating donor, from which the organs are harvested after the donor’s cardiac output ceases, in contrast to the more typical deceased donation in which the organs are harvested prior to cardiovascular collapse. Use of non–heart-beating donors is associated with reduced rates of long-term graft survival and increased risk of biliary complications, which correlate with the duration of “warm ischemia” after cardiovascular collapse and before retrieval of the organ. With the critical shortage of deceased organ donors, expansion of the donor pool has included acceptance of donors 70 years of age and older for selected recipients. Prior to hepatectomy, the harvesting team makes a visual and, if necessary, histologic assessment of the donor organ. Particular attention is paid to anatomic variants in the hepatic artery that may complicate the graft arterial anastomosis in the recipient. Once donor circulation is interrupted, the organ is rapidly infused with a cold preservation solution (e.g., University of Wisconsin, histidine-tryptophan-ketoglutarate, or Institut Georges Lopez solution). Donor iliac arteries and veins are also retrieved in case vascular grafting is required. After its arrival at the recipient institution, further vascular dissection, with arterial reconstruction if necessary, is performed before implantation.
Future Perspectives
Major challenges remain in LT, including the shortage of donor organs, threat of recurrent disease, and morbidity associated with lifelong therapeutic immunosuppression. Nevertheless, the availability of LT has transformed the lives of patients with advancing liver disease and their health care providers from an ultimately futile effort to manage the complications of cirrhosis into a life-prolonging and life-enhancing intervention.
Necrotizing Gallstone Pancreatitis
The AGA recently published a Clinical Practice Update reviewing the best available evidence on pancreatic necrosis, regardless of cause. The update provides 15 best practice advice points that include the need for multidisciplinary care coordination and referral to a tertiary-care center as appropriate. The update describes supportive care, avoidance of prophylactic antibiotics, and optimization of nutrition. In addition, there is an included algorithm for the management of pancreatic necrosis requiring debridement. Debridement within the early acute phase of pancreatitis (within the first 2–4 weeks) should be avoided if possible secondary to increased morbidity and mortality. Intervention in the late phase (> 2–4 weeks) is indicated for patients with infected necrosis or persistent organ dysfunction and failure to thrive. Multiple approaches are available for the management of infected necrosis, including but not limited to percutaneous, endoscopic, or laparoscopic transgastric, or open debridement. In addition, a combination approach using percutaneous drainage followed by videoscopic retroperitoneal debridement or step-up approach can also be used. Since the publication of a multicenter RCT (PANTER) in 2010, the step-up approach for necrotizing pancreatitis has been increasingly used.
The step-up approach or video-assisted retroperitoneal debridement (VARD) is a minimally invasive technique that begins with percutaneous drain placement for necrotizing pancreatitis followed by a minimally invasive retroperitoneal necrosectomy. Patients who underwent the step-up approach versus open necrosectomy had less multiple-organ failure, incisional hernias, and newonset diabetes, but no difference in mortality. The 2020 AGA Clinical Practice Update on the management of pancreatic necrosis suggests that best practice is that ‘‘minimally invasive operative approaches to the debridement of acute necrotizing pancreatitis are preferred to open surgical necrosectomy when possible, given lower morbidity’’. However, the update also notes that open necrosectomy still has a role in the modern management of acute necrotizing pancreatitis, particularly for cases whereby less invasive techniques are not feasible.
POPF after Distal Pancreatectomy
Minimally Invasive Versus Open Techniques
Despite advances in laparoscopic and robotic approaches, the vast majority of distal pancreatectomies continue to be performed via an open approach. Recent retrospective data have demonstrated that minimally invasive distal pancreatectomy is associated with decreased blood loss and shorter hospital stays than open pancreatectomy. A large recent study utilizing the Nationwide Inpatient Sample database suggested, first, that the minimally invasive approach is becoming more widely utilized, increasing from 2.4 to 7.3 % over a study period from 1998 to 2009. Second, that study reported that the minimally invasive approach was associated with decreased length of stay as well as decreased incidence of infectious complications, bleeding complications, and blood transfusions. This population-based study echoes conclusions drawn by a large multi-institutional study performed several years previously. Drawing on a combined patient sample of 667 patients, with 24 % initially attempted laparoscopically, the authors were able to demonstrate lower overall complication rate, decreased blood loss, and shorter hospital stays among patients undergoing laparoscopic approach via a multivariate analysis.
Notably, there was no significant difference in the pancreatic leak rate between the open and laparoscopic approaches, although there was a nonsignificant trend favoring the laparoscopic approach. More recently, the robotic approach has generated significant interest as a technique for performing distal pancreatectomy. Retrospective analysis has suggested that the robotic approach is well suited for pancreatectomy. Fistula rates, however, remain a concern. A retrospective review of patients undergoing robotic pancreatic operations included 83 patients who underwent distal pancreatectomy. About 27 % were identified as having a ISPGF type A pancreatic leak; 12 and 4.8 % were identified as having a grade B or C leak, respectively.
Identifying Risk Factors
For pancreaticoduodenectomy (PTD) , a fistula risk score has been recently developed that has been shown to be highly predictive of POPF. This score assigns points based on gland texture, gland pathology, duct diameter, and intraoperative blood loss. In general, high blood loss, soft gland texture, and smaller duct diameter confer increased risk of POPF, whereas pancreatic adenocarcinoma and pancreatitis as the indication for PTD confer protection for the development of pancreatic fistula versus other diagnoses. Also of note, higher fistula risk scores correlated with greater incidence of clinically relevant (ISGPF grade B or C) fistula. The adaptation of this risk score to patients undergoing distal pancreatectomy is yet to be validated; however, at least one published study indicates that this scoring system may have limitations in the setting of distal pancreatectomy. In that study, risk factors for pancreatic fistula after stapled gland transection in patients undergoing distal pancreatectomy were examined, and in a multivariate analysis, only the presence of diabetes and the use of a 4.1-mm staple cartridge were associated with increased risk of pancreatic fistula formation.
Preoperative Biliary Drainage
The most common presenting sign for patients with malignancy of the periampullary region is obstructive jaundice. While a significant proportion of these patients will be asymptomatic, the deleterious systemic consequences of uncontrolled hyperbilirubinemia may still occur. Furthermore, symptoms such as pruritus can be debilitating and have a significant impact on the quality of life. Thus, some have advocated preoperative drainage of the biliary system in patients with resectable periampullary malignancies, given widespread availability of endoscopic retrograde cholangiopancreatography and its perceived safety profile. On the other hand, the purported benefits of routine preoperative drainage in this patient population (namely, resolution of symptoms in symptomatic patients while awaiting surgery, restoration of the enterohepatic cycle, and a potential decrease in postoperative morbidity) have proven to be largely theoretical, and now there are high-quality phase III data that demonstrate the deleterious effects of routine stenting. A seminal study originating from the Netherlands in 2010 evaluated this issue in the only modern randomized controlled trial to date evaluating preoperative endoscopic biliary decompression for these patients. In their multicenter study, they randomized 202 patients with newly diagnosed pancreatic head cancer and bilirubin levels between 2.3 and 14.6 mg/dL to preoperative biliary drainage for 4–6 weeks vs. immediate surgery which was to be performed within a week of enrollment. The primary endpoint was the development of serious complications within 120 days after randomization. Serious complications were defined as complications related to the drainage procedure or the surgical intervention that required additional medical, endoscopic, or surgical management, and that resulted in prolongation of the hospital stay, readmission to the hospital, or death. The reported overall rate of serious complications in this study favored the immediate surgery group (39 vs. 74%; RR 0.54–95% [CI], 0.41–0.71; P < 0.001), complications related to surgery were equivalent (37 vs. 47%; P = 0.14), and there was no difference in mortality rates or length of hospital stay. The observed drainage-related complications included a 15% rate of stent occlusion, 30% need for exchange, and 26% incidence of cholangitis.
“Based on these results, the authors concluded that the morbidity associated with the drainage procedure itself had an additive effect on the postoperative morbidity of patients undergoing pancreatic head resection for cancer and recommended against its routine use in this population.“
A Cochrane systematic review of all available randomized studies (including the abovementioned study by van der Gaag et al.) evaluating preoperative biliary drainage was published in 2012. In this study, Fang et al. assessed the impact of this intervention on survival, serious morbidity (defined as Clavien-Dindo grade 3 or 4), and quality of life. Furthermore, they sought to assess differences in total length of hospital stay and cost. They identified six randomized trials of which four used percutaneous transhepatic biliary drainage and the remaining two used endoscopic sphincterotomy and stenting. The pooled analysis of 520 patients (of which 51% underwent preoperative biliary drainage) showed no difference in mortality, but importantly, it showed a significantly higher incidence of serious morbidity in the preoperative drainage group with a rate ratio (RaR) of 1.66 (95% CI 1.28–2.16;P = 0.002). There was no difference in length of hospital stay and not enough data reported for analysis of cost or quality of life.
“Based on the available level 1 data, the authors concluded that there was no evidence to support or refute routine preoperative biliary drainage in patients with obstructive jaundice.“
However, this review also underscored the fact that preoperative biliary drainage may be associated with an increased rate of adverse events and thus questioned the safety of this practice. This Cochrane review included old studies that evaluated patients undergoing percutaneous drainage, a technique used less frequently today for periampullary malignancies. Furthermore, several of these trials included patients with hilar and other types of biliary obstruction. However, the concept of preoperative decompression, as well as its purported benefits and observed results, may be reasonably extrapolated to patients with periampullary lesions.
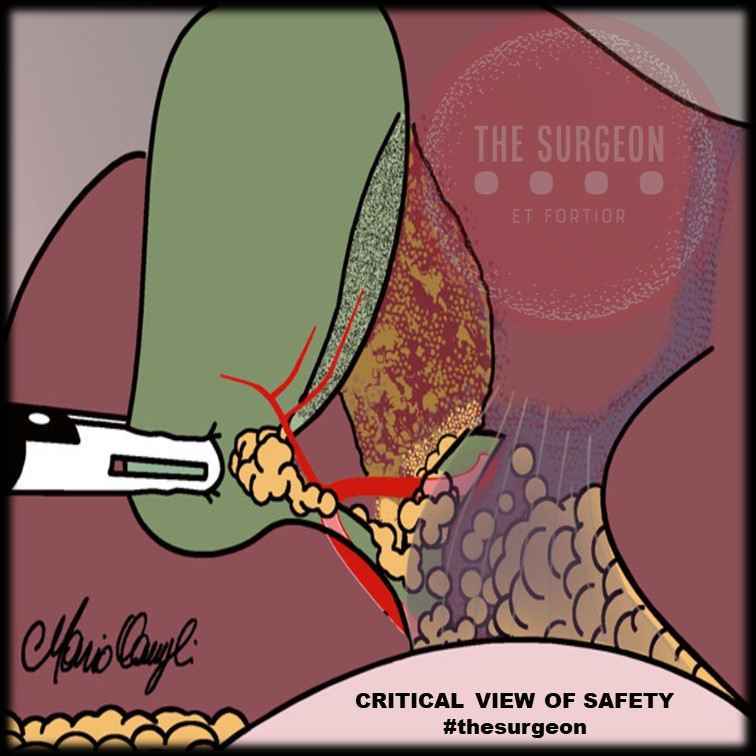
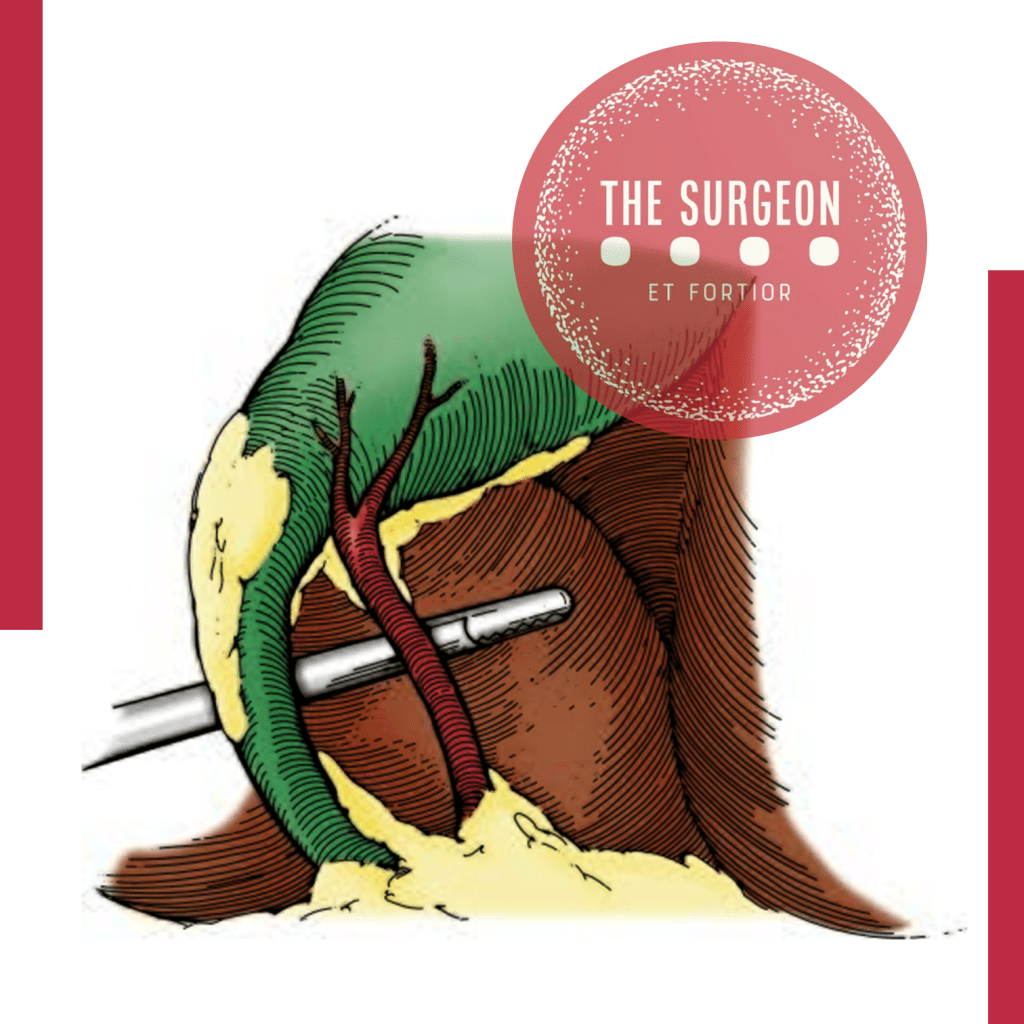
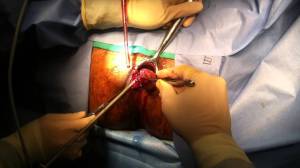
Critical View Of Safety
“The concept of the critical view was described in 1992 but the term CVS was introduced in 1995 in an analytical review of the emerging problem of biliary injury in laparoscopic cholecystectomy. CVS was conceived not as a way to do laparoscopic cholecystectomy but as a way to avoid biliary injury. To achieve this, what was needed was a secure method of identifying the two tubular structures that are divided in a cholecystectomy, i.e., the cystic duct and the cystic artery. CVS is an adoption of a technique of secure identification in open cholecystectomy in which both cystic structures are putatively identified after which the gallbladder is taken off the cystic plate so that it is hanging free and just attached by the two cystic structures. In laparoscopic surgery complete separation of the body of the gallbladder from the cystic plate makes clipping of the cystic structures difficult so for laparoscopy the requirement was that only the lower part of the gallbladder (about one-third) had to be separated from the cystic plate. The other two requirements are that the hepatocystic triangle is cleared of fat and fibrous tissue and that there are two and only two structures attached to the gallbladder and the latter requirements were the same as in the open technique. Not until all three elements of CVS are attained may the cystic structures be clipped and divided. Intraoperatively CVS should be confirmed in a “time-out” in which the 3 elements of CVS are demonstrated. Note again that CVS is not a method of dissection but a method of target identification akin to concepts used in safe hunting procedures. Several years after the CVS was introduced there did not seem to be a lessening of biliary injuries.
Operative notes of biliary injuries were collected and studied in an attempt to determine if CVS was failing to prevent injury. We found that the method of target identification that was failing was not CVS but the infundibular technique in which the cystic duct is identified by exposing the funnel shape where the infundibulum of the gallbladder joins the cystic duct. This seemed to occur most frequently under conditions of severe acute or chronic inflammation. Inflammatory fusion and contraction may cause juxtaposition or adherence of the common hepatic duct to the side of the gallbladder. When the infundibular technique of identification is used under these conditions a compelling visual deception that the common bile duct is the cystic duct may occur. CVS is much less susceptible to this deception because more exposure is needed to achieve CVS, and either the CVS is attained, by which time the anatomic situation is clarified, or operative conditions prevent attainment of CVS and one of several important “bail-out” strategies is used thus avoiding bile duct injury.
CVS must be considered as part of an overall schema of a culture of safety in cholecystectomy. When CVS cannot be attained there are several bailout strategies such a cholecystostomy or in the case of very severe inflammation discontinuation of the procedure and referral to a tertiary center for care. The most satisfactory bailout procedure is subtotal cholecystectomy of which there are two kinds. Subtotal fenestrating cholecystectomy removes the free wall of the gallbladder and ablates the mucosa but does not close the gallbladder remnant. Subtotal reconstituting cholecystectomy closes the gallbladder making a new smaller gallbladder. Such a gallbladder remnant is undesirable since it may become the site of new gallstone formation and recurrent symptoms . Both types may be done laparoscopically.”
Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 1995;180:101-25.
Surgical Management of GERD After Sleeve Gastrectomy
Evaluation of a patient referring GERD after sleeve gastrectomy should start with a detailed history and physical examination; the presence or absence of GERD-related symptoms should be thoroughly documented as well as any prior treatments or therapy used to treat it. Obtaining preoperative and operative records is of paramount importance particularly in those patients who had their index procedure performed elsewhere. Any endoscopic findings and prior imaging available are important to determine what the best course of action would be. If the patient had preoperative and postoperative imaging such as UGI, it is useful to compare those with a recent study to look for anatomical problems that may have been not addressed at the time of the index operation or developed over time. After this information is obtained, we can classify the GERD after sleeve as:
1. De novo GERD
2. Preexisting GERD without improvement
3. Preexisting GERD with worsening/complication
Regardless of how we classify the GERD, an initial evaluation with imaging
studies such as UGI and EGD is recommended. Comparison with any prior films if available is of significant value. Based on the UGI, we can determine if the shape of the sleeve falls into one of the following categories: tubular, dilated bottom, dilated upper, or dumbbell-shaped sleeve; we will also be able to evaluate esophageal peristalsis in real time and if there is associated hiatal hernias. We believe UGI under fluoroscopy provides important physiologic and anatomic information that can help guide our management approach, and therefore we offer it to all patients. We follow the radiologic evaluation with endoscopy, and during endoscopy, we look for objective signs of reflux such as esophagitis, presence of bile in the stomach or esophagus, as well as missed or recurrent hiatal hernias. In patients with evidence of esophagitis or metaplasia, multiple biopsies are taken. During the endoscopy, subtle findings that suggest a kink or a stricture may be present. In the absence of objective signs of gastroesophageal reflux disease on both endoscopy and upper GI series, we pursue physiologic testing followed by highresolution manometry and pH monitoring. In those patients where clear reflux esophagitis is seen, this additional testing may not be necessary or may be performed in selected cases depending on what the surgical or endoscopic therapy would be.

While it is true that most sleeve-related GERD will be effectively treated with a conversion to Roux-en-Y gastric bypass, not every patient with GERD after reflux will require a bypass or would agree to have one. First key step in addressing the patient is to evaluate whether the patient was selected appropriately to have a sleeve and second is to determine the exact sleeve anatomy; are there anatomical factors that will make it more likely for this patient to experience reflux; is there dilated fundus? Is there a kink or stricture in the sleeve or is it an anatomically appropriate operation? We should pay important attention to the weight loss the patient has experienced with the sleeve. Patients who do not have adequate weight loss and have GERD symptoms should not undergo other therapies and should probably undergo a bypass; however it is our unpublished experience that patients with the association of poor weight loss after sleeve and difficult to treat GERD will correct their GERD after conversion, but their weight loss results are still marginal even with a well-constructed bypass.
Complications of HEMORROIDH SURGERY
Symptomatic hemorrhoids require a number of therapeutic interventions each of which has its own complications. Office-based therapy such as rubber band ligation carries the risk of pain and bleeding, which are self-limited, but also carries the risk of rare complications such as sepsis, which may be life threatening. Operative treatment of hemorrhoids includes conventional hemorrhoidectomy, stapled hemorrhoidectomy, and the use of energy devices. Complications of pain and bleeding are common but self-limited. Late complications such as stenosis and fecal incontinence are rare. Recurrent disease is related to the initial grade and therapeutic approach. Treatment of recurrent hemorrhoids should be individualized based on previous treatments and the grade of disease. Anesthetic complications, especially urinary retention, are common and related to the anesthetic technique. Practitioners should council their patients as to the risks of the various approaches to treating symptomatic hemorrhoids.