REVISIONAL BARIATRIC SURGERY
- INTRODUCTION
Morbid obesity is a global chronic disease affecting 13% of people worldwide. Weight loss surgery has been proven to be effective in addressing this chronic disease and its associated comorbidities. In 2016, over 200,000 procedures were performed in the United States, and the volume continues to grow. Cases analyzed between 2015 and 2018 indicate an overall growth rate of 21.9%. The most common surgeries performed in the United States are Roux-en-Y gastric bypass (RYGB), sleeve gastrectomy (SG), and biliopancreatic diversion with duodenal switch (BPD/DS). Estimated mean weight loss is 33% of the initial body weight. Unfortunately, it is estimated that up to 25% of patients will have weight regain after primary surgery. Weight regains or recidivism has emerged as a clinical entity and important public health issue given its association with re-emergence of obesity related comorbidities, worsening quality of life, and increased healthcare costs. With the increased number of primary bariatric surgery performed worldwide, revisional surgery has also increased, and it has been shown to be the fastest-growing category of bariatric procedures, currently representing 7 to 15% of all bariatric operations and long-term rates of revisional surgery have been estimated to be as high as 56%.
2. CAUSES
Weight regain is estimated in up to 25% of patients following primary bariatric surgery. In addition, it is estimated that more than 80% of the weight regain happens within the first 6 years following primary surgery. A major factor contributing to weight regain is lack of adherence to recommended followup visits, observed in approximately 60% of patients 4 years after primary surgery. The etiology of weight regain has been attributed to:
• Noncompliance with dietary recommendations.
• Hormonal/metabolic imbalance.
• Mental health.
• Physical inactivity.
• Anatomic/surgical factors.
• Medications.
3. PREOPERATIVE EVALUATION
A multidisciplinary evaluation is essential prior to recommending revisional surgery to patients presenting with weight gain. As with primary surgery, a nutritional evaluation, behavioral/psychological assessment, and endoscopic and contrast series studies should be obtained. The latter will not only aid in establishing an anatomic etiology for weight regain if present, but it will also aid in choosing the type of revisional surgery.
4. SELECTING THE TYPE OF REVISIONAL SURGERY
There are several revisional procedures following primary bariatric surgery. The choice of revisional surgery is tailored according to initial surgery, cause of failure, and surgeon’s experience. Multiple revisional surgeries have been described for all primary bariatric surgeries, but no standardized guidelines have been established. In June 2019, 70 experts from 27 countries formed a committee and created the first consensus on revisional bariatric surgery. An agreement of 70% or more was considered consensus. Consensus was achieved in several points including but not limited to:
(1) RBS is justified in some patients;
(2) RBS is more technically challenging than the respective primary bariatric surgery;
(3) second or third RBS can be justified in some patients;
(4) candidates should undergo a nutritional assessment, psychological evaluation, endoscopy, and a contrast series;
(5) RYGB, one anastomosis gastric bypass (OAGB), and SADI-S are options after gastric banding; and
(6) OAGB, BPD/DS, and SADI-S are options after sleeve gastrectomy. Regarding revision for primary RYGB, the only consensus obtained was lengthening of the biliopancreatic limb as RBS option for RYGB or OAGB.
Roux-En-Y Gastric Bypass
Roux-en-Y gastric bypass is one of the most common weight loss procedures performed worldwide and is considered by many to be the gold standard. Unfortunately, approximately 10–34% of patients experience inadequate weight loss or weight gain and may ultimately require revision. The most common etiology of weight regain is pouch dilation. Other reported etiologies include enlarged gastric pouch greater than 5 cm in diameter, wide gastro-jejunal anastomosis (GJA), anastomosis greater than 1 cm, GJA > 1.5 cm in diameter, dilated GJA greater than or equal to 2 cm, pouch >30 mL, pouch dilation >120 mL, weight recidivism with or without gastric fistula, gastric fistula, short-limb bypass, and hyperphagic behavior. Multiple revisional surgeries have been described. In a recent systemic review and meta-analysis, distal Roux-en-Y gastric bypass (DRGB) alone showed the highest decrease in BMI at 1-year follow-up versus biliopancreatic diversion with duodenal switch (BPD/DS) or single anastomosis duodeno-ileal bypass and sleeve gastrectomy (SADI-S) at 3-year follow-up. Overall, they found maximal BMI decrease in DRGB alone, followed by BPD/DS or SADI-S, laparoscopic pouch and/or GJA resizing, and endoscopic pouch and/or GJA resizing.
Sleeve Gastrectomy
Sleeve gastrectomy (SG) is currently the most commonly performed bariatric procedure worldwide. Its relatively simple technique and low complication rate contribute to it being preferred over some other procedures. Revision is estimated in up to 30% of cases for multiple etiologies, including weight regain. Loss of restriction is one of the main anatomic factors contributing to weight regain. Although revision to RYGB or DS has been recommended as the standard of care, some studies have described revision with re-sleeve for dilation of the residual stomach as the cause. The overall %EWL following re-sleeve can be up to 57% at 12 months and up to 60% at 20 months. A retrospective study analyzed conversion from SG to either RYGB or SADI for insufficient weight loss or weight regain. Out of 140 patients, 66 patients underwent SG to SADI, and 74 patients underwent SG to RYGB. SADI was found to achieve 8.7%, 12.4%, and 19.4% more total body weight loss at 6, 12, and 24 months compared to RYGB for weight regain alone. RYGB is preferred when symptoms of reflux accompany weight regain. For patients with super morbid obesity and weight regain after SG, in the absence of reflux symptoms, conversion to biliopancreatic diversion with duodenal switch (BPD/DS) is recommended for maximal weight loss, with %EWL ranging from 70 to 80% at 2 years.
5. WEIGHT LOSS FOLLOWING REVISIONAL SURGERY
Weight loss after revisional bariatric surgery leads to significant weight loss in the long term, rates varying per procedure performed. A single-center retrospective study for patients who underwent revisional surgery for weight regain (52.4%) S. Ardila et al. analyzed weight loss at 3-, 6-, 9-, and 12-month intervals. Patients with a primary restrictive procedure and reflux symptoms underwent conversion to either RYGB or BPD/DS and experienced 50–65.3%EWL at 3 months and 50.1–79.1%EWL at 12 months. Patients with initial RYGB underwent GJ revision for pouch or GJ abnormalities. For those without anatomic abnormalities, they underwent conversion to distal bypass. At 3 months, %EWL was 36.6 for GJ revision and 37.5% for distal revision.
6. Complications of Revisional Surgery
Revisional bariatric surgery is complex, is technically demanding, and is therefore associated with higher morbidity and mortality. Compared to primary surgery, revisional surgery has been associated with higher rates of postoperative complications, longer operative times, longer hospital stay, conversion to open surgery, readmission, and unplanned admission to the critical care unit. In comparing primary versus revisional RYGB, revisional surgery was associated with higher rates of leak, hemorrhage, wound infection, stricture, ulcer, perforation, and hernia. A single-center retrospective study analyzed complications after revisional surgeries performed at their center for weight regain between 2012 and 2015. Of 84 patients, 43 presented for weight regain (52.4%). Complications included incarcerated ventral hernia following AGB conversion to SG, anastomotic leak and recurrent intussusception following gastro-jejunostomy revision, and stricture and marginal ulcer following AGB conversion to RYGB . In summary, reported complications of revisional bariatric surgery are:
• Hernia.
• Anastomotic leak.
• Stricture.
• Marginal ulcer.
• Wound infection.
• Hemorrhage.
• Perforation.
• Obstruction.
Conclusion
Weight regain after primary bariatric surgery is multifactorial. It is imperative to establish guidelines for classifying weight gain in order to guide subsequent intervention and thus to aid bariatric teams internationally in the management of this clinical entity. Revisional surgery has been shown to be a successful treatment option for patients presenting with weight gain, with rates of weight loss nearing those seen after primary surgery. Furthermore, the importance of revisional surgery lies in its ability to readdress many of the obesity-related comorbidities which prompted the primary procedure. When indicated, it is important to tailor the type of revisional surgery to each patient. Although Roux-en-Y gastric bypass remains the most common type of revisional surgery after primary surgery of any type, duodenal switch is emerging as the revision procedure of choice for superobese patients and patients with failed sleeve gastrectomy secondary to weight regain in the absence of reflux symptoms.
Revisional Surgery for Weight Regain
- Introduction
Revisional bariatric surgery is a weight loss surgery for people who have not lost enough weight or have regained weight after their first bariatric surgery. It is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain.
- Causes of Weight Regain
There are many factors that can contribute to weight regain after bariatric surgery, including:
* Lack of adherence to dietary recommendations
* Hormonal/metabolic imbalance
* Mental health
* Physical inactivity
* Anatomic/surgical factors
* Medications
- Preoperative Evaluation
Before revisional surgery, patients will undergo a thorough evaluation to determine if they are eligible for the procedure. This evaluation will include a medical history and physical exam, as well as blood tests, imaging studies, and a psychological evaluation.
- Selecting the Type of Revisional Surgery
The type of revisional surgery that is best for a patient will depend on a number of factors, including the type of primary surgery they had, the cause of their weight regain, and their overall health. Some of the most common types of revisional surgery include:
* Roux-en-Y gastric bypass (RYGB)
* Sleeve gastrectomy (SG)
* Biliopancreatic diversion with duodenal switch (BPD/DS)
* Single anastomosis gastric bypass (OAGB)
* Sleeve gastrectomy with duodenal switch (SADI-S)
- Weight Loss Following Revisional Surgery
Weight loss after revisional surgery can be significant. In one study, patients who underwent revisional surgery for weight regain lost an average of 50–65.3% of their excess weight after 3 months and 50.1–79.1% of their excess weight after 12 months.
- Complications of Revisional Surgery
Revisional surgery is more complex and technically demanding than primary bariatric surgery, and therefore carries a higher risk of complications. Some of the most common complications of revisional surgery include:
* Hernia
* Anastomotic leak
* Stricture
* Marginal ulcer
* Wound infection
* Hemorrhage
* Perforation
* Obstruction
- Conclusion
Revisional bariatric surgery can be a successful treatment option for patients with weight regain. However, it is important to carefully consider the risks and benefits of the procedure before making a decision. Patients should also work closely with their healthcare team to choose the best type of revisional surgery for them and to prepare for the procedure and its aftermath. Revisional bariatric surgery is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain. It is important to work closely with your healthcare team to determine if you are eligible for the procedure and to choose the best type of surgery for you.
Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
GASTROSTOMY: INDICATIONS, TECHNICAL DETAILS AND POSTOPERATIVE CARE.
Ozimo Pereira Gama Filho
Adjunct Professor at the Federal University of Maranhão
No conflict of interest
- INTRODUCTION
- HISTORICAL ASPECTS
- ANATOMICAL FUNDAMENTALS
- INDICATIONS
- TYPES & TECHNIQUES
- ADVERSE EVENTS
- CUSTO RATIO x EFFECTIVENESS
- CONCLUSIONS
SUMMARY
In the last decade, the use of gastrostomies has been widely indicated as the preferred form of access to the gastrointestinal tract for feeding in chronic conditions and during recovery from acute conditions such as trauma. Together with this increase in indications, new techniques have been developed that have made gastrostomies simpler and less risky. From the classical technique of Stamm performed by laparotomy, two new alternatives that do not require laparotomy emerged: percutaneous endoscopic gastrostomy (PEG) and fluoroscopy gastrostomy. Its main benefit is to avoid a laparotomy, with less associated postoperative pain and earlier return of gastrointestinal function. Although peg is currently widely accepted as the insertion technique of choice due to its simplicity and efficacy, there are patients who are not candidates for an endoscopic approach. In this article we seek to clarify the indications, technical aspects and perioperative care of patients undergoing gastrostomy.
Keywords: Surgical Procedures; Ostomies; Gastrostomy.
Area of Knowledge: General Surgery
- INTRODUCTION
The main indication for enteral or parenteral feeding in the perioperative period is the provision of nutritional support to supply the metabolism of patients with inadequate oral intake. Enteral feeding is the preferred method in relation to parenteral feeding in patients with gastrointestinal dysfunction in the perioperative period due to the inherent risks associated with parenteral nutritional support, such as: infectious complications of the access routes, higher operational cost, and the inability to parenteral nutrition to provide adequate enteral stimulation and subsequent involvement of the intestinal defense barrier [1,2]. In addition, enteral feeding may decrease the risk of bacterial translocation and corresponding bacteremia [3]. Gastric nutritional support is the most common type used. Access to insert the gastrostomy probe can be achieved using endoscopy, interventional radiologia, or surgical techniques (open or laparoscopic). However, since its description in the 1980s [4], percutaneous endoscopic gastrostomy (PEG) is currently considered the method of choice for medium and long-term enteral support.
1.1 Objective: This article reviews the current knowledge about GOSTROSTOMIA in the medical literature, emphasizing the technical and perioperative aspects.
- HISTORICAL ASPECTS
In 876, Verneoil [5] successfully made the first gastrostomy in humans. Since then, several technical modifications have been suggested, such as witzel’s technique in 1891, in which a subseroso tunnel is made on the probe [6]. Stamm, in 1894 [7], described one of the most performed techniques today and in the history of surgical gastrostomy, which consists in the making of suture in a pouch to invaginate the probe inserted into the stomach [8]. In 1980, percutaneous endoscopic gastrostomy was described by Gauderer et al. [4] , which transformed the technique of making gastrostomy.
- ANATOMICAL FUNDAMENTALS
The stomach is a J-shaped dilated cylindrical organ that rests in the left epigastric and hypochodrial region of the abdomen at the level of the first lumbar vertebra. It is previously limited by the left hemidiaphragm, the left lobe of the liver and a triangular portion of the anterior abdominal wall. Subsequently, the pancreas, left kidney and adrenal delimit the stomach. The spleen is posterolaterally and the transverse colon is inferior. It is fixed at two points of continuity: gastroesophageal, superiorly and the duodenal, retroperitoneally. Its ligament attachments also help you in fixation to adjacent organs: gastrophemic (diaphragm), hepatogastric or minor omentum (liver), gastrosplenic or gastrolienal (spleen), and gastrocholic or omentum major (transverse colon). The anatomical regions of the stomach can be distinguished as this: começa superiorly in the continuity of the abdominal part of the esophagus and dthe gastroesophageal junction, the cardiac part of the stomach. Soon below this portion, lies the bottom of the stomach that expands to the left extending above thegastroesophageal junction, forming an acute angle with the distal esophagus known as cardiac notches. The body s andextends as a distensible reservoir and forms a medial edge called the smallest curvature to the right and a side edge called the largest curvature on the left. The gastric den of the stomach is not anatomically distinguishable, but it is estimated to be a region of the angular isis along the distal minor curvature to a point along a lower line to the distal major curvature. It thus ends bymouthing r into the pyloric canal limited by the pyloric sphincter, a palpable thickened ring of muscle that is continuous with the first part of the duodenum.
- INDICATIONS
Gastrostomy is used in the following situations:
- Gastric decompression: can be obtained by means of temporary gastrostomy, occasionally recommended, as a complement to large abdominal operations for which gastric stems, prolonged “adynamic ileus” and digestive fistulas are foreshadowed.
- Nutritional Support: b.1 Temporary; indicated when access to the digestive tract is temporarily impaired for recovery and maintenance of nutritional status (E.g. CEsophageal EC); b.2 Definitive; as palliative therapy in patients with unresectable malignant neoplasia of the head and neck, as wellas n degenerative neurological diseases that lead to irreversible disorders of deglutition.
However, the decision to perform a gastrostomy, as well as its route (surgical, radiological or endoscopic) should be individualizedaccording to the needs, diagnosis, life expectancy of the patient and the available hospital logistics. The objective is not only to optimize perioperative recovery, to improve survival and nutritional status of the patient, but also to promote quality of life, which is not necessarily correlated with nutritional improvement only [9]. Therefore, the appropriate indication, like any other surgical intervention, must be clearly establishedand informed before it is performed. Some of the absolute contraindications of gastrostomy are summarized in Table 1. In addition to absolute contraindication conditions, other situations such as the presence of non-obstructive oromyctological oresophageal malignancy, hepatomegaly, splenomegaly, peritoneal dialysis, portal hypertension with gastric varicose veins, and a history of partial gastrectomy are also considered relative contraindications.
| ABSOLUTE CONTRA – INDICATIONS |
| Coagulopatia Severa (INR > 5, Plaquetas < 50.000 e TPT > 50s) |
| Hemodynamic Instability |
| Septic Shock |
| Refractory Ascites |
| Peritonitis |
| Dermatological infection in the upper abdomen |
| Carcinomatose Peritoneal |
| Interposition of organs that prevent gastric access |
| History of Total Gastrectomy |
| Stenosis or Pyloric Obstruction |
| Severe gastroparesis, in cases of indication for nutritional support |
| Absence of Informed Consent |
- TYPES & TECHNIQUES
Currently there are three techniques for performing gastrostomy: radiological, through percutaneous gastrostomy by fluoroscopy, percutaneous endoscopic gastrostomy (PEG) and surgical gastrostomy. Due to the didactic characteristics of this material, we will focus on endoscopic and surgical gastrostomy , which becomes the main option in the following situations: 1) when the patient will already undergo a laparotomy due to some abdominal condition ; 2) impossibility of performing gastroscopy to perform gastrostomy endoscopic percutaneous (PEG) ; 3) in case of peg technical failure; 4) unavailability of resources for the preparation of PEG or percutaneous gastrostomy by fluoroscopy.
- ENDOSCOPIC PERCUTANEOUS GASTROSTOMY
The informed consent form must be obtained from patients or their legal representatives. Patients should fast for a minimum of 8 hours and receive prophylactic antibiotics one hour before proceeding and intravenous administration of 1-2 g of cefazolin is recommended. The technique introduced by Gauderer et al [4] is the most used technique to insert the PEG gastrostomy probe. In this method, a guide wire is used, inserted in the distal gastric chamber through a needle puncture n to the anterior abdominal wall. This guide wire is then seized endoscopically with a handle and then removed through the esophagus and mouth. Subsequently, the guide wire is fixed to the end of the gastrostomy probe and then pulled from the mouth to the esophagus, stomach and then out to the abdominal wall, where it will be fixed.
- SURGICAL GASTROSTOMY
Surgical gastrostomy can be performed in two ways: 1) via laparotomy – the predominant form; and 2) laparoscopic approach. The preparations are the same as the endoscopic pathway.
5.2.1. GASTROSTOMIA At STAMM
After adequate asepsis and antisepsis, with the patient under anesthesia and in horizontal dorsal decubitus, the technical steps are as follows: 1. Median laparotomy (supraumbilical median incision); 2. Identification of the gastric body; 3. Stomach hold with Babcock tweezers (to evaluate the approach of the stomach to the peritoneum); 4. Suture in pouch (circular area of 2cm) – atraumatic absorbable thread; 5. Section of the stomach wall (0.5cm) – (incision with scalpel or Electrocautery in the center of the suture, of sufficient size, for the placement of a probe with 20 to 26 French); 6. Placement of the Gastrostomy probe in the extension of 5-6 cm, followed by suture closure in a pouch); 7. Tie the suture threads in a pouch around the probe; 8. Apply a second suture in a pouch 1cm above the first (seromuscular stitches); 9. Externalization of the probe by counter opening on the left flank; 10. Fix the stomach wall to the abdominal wall in 4 cardinal points (external ration with the two Kocher tweezers used for grip of the alba line and against traction by means of the index fingers of the wall of the left hipochondrio to approach the parietal peritoneum of the gastric wall); 11. Fixation of the probe to the skin (point with nonabsorbable wire); 12. Closure of the abdominal wall (synthesis of the wall with approximation of the alba thread by continuous suture with monofilament thread 1-0 or 2-0 and of the skin with separate points of nylon 3-0); 13. Dressing.
5.2.1 GASTROSTOMIA EM WITZEL
The initial technical steps from gastrostomy to Witzel are like those of gastrostomy to Stamm, including fixation of the probe to the stomach by a pouch suture. Then, the probe is placed on the gastric wall and a tunnel of 8-10 cm is made by seromuscular suture (continuous or with separate points of absorbable or nonabsorbable thread) covering it and externalization is performed by counteropening.
- ADVERSE EVENTS
According to the literature, the rate of complications for different procedures varies due to the heterogeneity of the samples evaluated. For surgical gastrostomy, the reported complication rates are between 1% and 35%, while for percutaneous radiological gastrostomy it is 3% to 11%, and for percutaneous endoscopic, 17%–32%, the main related adverse event is surgical site infection [10, 11]. Although considered a basic procedure, gastrostomy is associated with an extensive list of related technical complications, care and use of the probe. Serious problems related to the technique include separation of the stomach from the abdominal wall (leading to peritonitis), separation of wounds, hemorrhage, infection, lesion of the posterior gastric wall or other organs, and placement of the tube in an inappropriate place of gastric position. Separation of the stomach from the abdominal wall usually occurs due to inadvertent and premature displacement of the tube, particularly with balloon-like devices, or a rupture during a catheter change. It requires immediate attention, being treated with laparotomy, although in selected cases laparoscopic correction is possible. Most complications can be avoided with the careful choice of the type of procedure, from the appropriate ostomy device, considering it an important intervention and using meticulous technique with the proper approximation of the stomach to the abdominal wall and outflow of the probe through a counter-incision (in conventional procedures), thus avoiding probes in the midline or awfully close to the costal edge.
- COST VS. EFFECTIVENESS
A recent study [12] compared the cost associated with the different gastrostomy techniques, and the results of the evaluation showed variable the benefits of each of the individual percutaneous procedures, indicating that surgical gastrostomy was the onerous mais of the three modalities due to higher costs, complications, and recovery time, as well as the endoscopic technique presenting the cost effectiveness ratio.
- CONCLUSIONS
Despite the technique employed, the decision to performa gastrostomy is not based only on the patient’s survival expectancy, because the adequate indication provides a better quality of life even when the survival of the patient after the procedure is severely limited. Therefore, understanding of techniques, indications, complication rates is essential to guide the surgical team in the scope of multidisciplinary care, as well as the education of patients and their caregivers is vital to ensure the correct maintenance of the devices, thus ensuring adequate nutritional intake of the patient and minimizes complication rates.
References
1 Alverdy J, Chi HS, Sheldon GF. The effect of parenteral nutrition in gastrointestinal immunity. The importance of de estimulação enteral. Ann Surg, 1985; 202: 681-684 [PMID:3935061]
2 Deitch EA, Ma WJ, Ma L, Berg RD, Specian RD. Protein malnutrition predisposes to inflammation-induced intestinal origin septic states. Ann Surg, 1990; 211: 560-567; discussion 560-567 [PMID: 2111125]
3 Deitch EA, Winterton J, Li M, Berg R. The intestine as a portal of entry to bacteremia. Role of protein malnutrition. Ann Surg 1987; 205: 681-692 [PMID: 3592811]
4 Gauderer MW, Ponsky JL, Izant RJ. Gastrostomia sem laparotomy: percutaneous endoscopic technique. J Pediatrician Surg, 1980; 15: 872-875 [PMID: 6780678]
5 Anselmo CB, Tercioti Júnior V, Lopes LR, Coelho Neto JS, Andreollo NA. Surgical gastrostomy: current indications and complications in patients of a university hospital. Rev Col Bras Cir. [Internet journal] 2013;40(6). Available in URL: http://www.scielo.br/rcbc
6 Witzel O. For gastric fistula technique. Chir Zbl. 1891;18:601-4.
7 Stamm M. Gastrostomy: a new method. Med News. 1894;65:324.
8 JP grant. Comparison of percutaneous endoscopic gastrostomy com gastrostomia strain. Ann Surg. 1988;207(5):598-603
9 Bannerman E, Pendlebury J, Phillips F, Ghosh S. Cross-sectional and longitudinal study of health-related quality of life after percutaneous gastrostomy. Eur J Gastroenterol Hepatol 2000; 12: 1101-1109 [PMID: 11057455]
10 Möller P, Lindberg CG, Zilling T. Gastrostomy by various techniques: evaluation of indications, outcome and complications. Scand J Gastroenterol. 1999;34(10):1050-4.
11 Clarke E, Pitts N, Latchford A, Lewis S. A major prospective audit of morbidity and mortality associated with food gastrostomies in the community. Clin Nutr. 2017 Apr;36(2):485-490. DOI: 10.1016/j.clnu.2016.01.008. EPub 2016 January 21. PMID: 26874913.
12 Wollman B, D’Agostino HB, Walus-Wigle JR, Easter DW, Beale A (1995) Radiological, endoscopic and surgical gastrostomy: an institutional evaluation and meta-analysis of the literature. Radiology 197: 699–704.
A Arte da Anatomia (EBook)
Desde a Antiguidade, os médicos, anatomistas e artistas se dedicaram a descrever e representar o corpo humano, por meio de desenhos, pinturas, esculturas e outras formas de representação artística. Com o passar dos séculos, houve uma evolução significativa na forma como as ilustrações anatômicas eram produzidas, desde as primeiras representações rudimentares até as ilustrações altamente detalhadas e realistas que temos hoje.
Boa Leitura!!!
Intraoperative cholangiography: Selective or Routine?
Intraoperative cholangiography (IOC), described by Mirizzi in 1932, represented a significant advance in the diagnosis of choledocolithiasis during cholecystectomy. The natural history of asymptomatic choledocolithiasis has been investigated in different populations and its therapeutic management continues to be controversial. IOC is traditionally advocated as a procedure to be adopted in all laparoscopic cholecystectomies since it permits to define the anatomy of the biliary tract and to detect common bile duct stones. However, in the laparoscopic era, technological advances in radiologic-endoscopic workup have markedly increased the costs of investigation of patients with suspected choledocolithiasis. The routine use of IOC has raised the question of which cases require the exploration of bile tract anatomy during surgery and whether there are methods to predict preoperatively unsuspected choledocolithiasis. Therefore, the most appropriate management of preoperatively unsuspected choledocolithiasis, i.e., the routine or selective use of IOC, still remains undefined. In this respect, well-defined criteria for the inclusion of patients with possible choledocolithiasis who should be submitted to cholangiography exist in the literature, but there is no safe approach to exclude asymptomatic patients without an indication for contrast examination.
A systematic literature search was performed by KOVACS N, et al (2022) using the following search keys: cholangiogra* and cholecystectomy. The primary outcomes were BDI and retained stone rate. To investigate the differences between the groups (routine IOC vs selective IOC and IOC vs no IOC), they calculated weighted mean differences (WMD) for continuous outcomes and relative risks (RR) for dichotomous outcomes, with 95% confidence intervals (CI). Of the 19,863 articles, 38 were selected and 32 were included in the quantitative synthesis. Routine IOC showed no superiority compared to selective IOC in decreasing BDI (RR = 0.91, 95% CI 0.66; 1.24). Comparing IOC and no IOC, no statistically significant differences were found in the case of BDI, retained stone rate, readmission rate, and length of hospital stay. They found an increased risk of conversion rate to open surgery in the no IOC group (RR = 0.64, CI 0.51; 0.78). The operation time was significantly longer in the IOC group compared to the no IOC group (WMD = 11.25 min, 95% CI 6.57; 15.93). So this data findings suggest that IOC may not be indicated in every case, however, the evidence is very uncertain.
Postpancreatectomy Hemorrhage
Postoperative complications represent one of the most debated topics in pancreatic surgery. Indeed, the rate of complications following pancreatectomy is among the highest in abdominal surgery, with morbidity ranging between 30 and 60%. They are often characterized by elevated clinical burden, with a consequent challenging postoperative management. Mortality rates can exceed 5%. The impact on patient recovery and hospital stay eventually leads to massive utilization of resources and increases costs for the health system. The International Study Group of Pancreatic Surgery (ISGPS) provides standardized definitions and clinically based classifications for the most common complications after pancreatectomy, including postoperative pancreatic fistula (POPF), post pancreatectomy hemorrhage (PPH), delayed gastric emptying (DGE), bile leakage, and chyle leak.
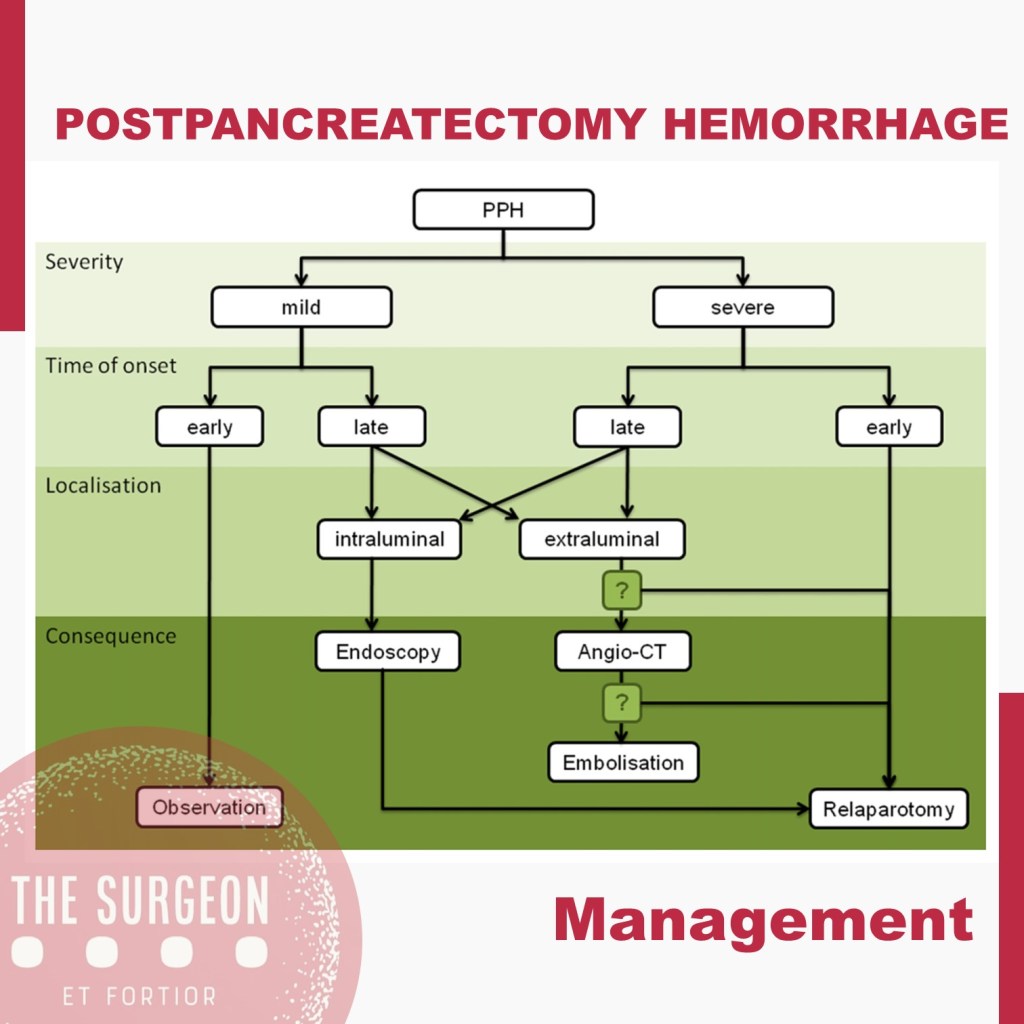
Post Pancreatectomy Hemorrhage
Despite its lower incidence compared with POPF with reported rates of 3–10% after pancreatectomy, PPH remains one of the major of postoperative complications, with mortality rates ranging from 30 to 50%. According to current ISGPS recommendations, PPH is classified in three grades (A, B, and C) based on two main criteria: timing of the hemorrhage and severity of the bleeding. The timing is dichotomized as early, occurring within 24 hours from the index surgery, and late, when it happens afterwards.
Management is tailored according to the clinical picture, the timing of onset, and the presumptive location. Early hemorrhage is generally due to either unsuccessful intraoperative hemostasis or to an underlying coagulopathy. The vast majority of patients are basically asymptomatic, with PPH having no influence on the postoperative course. However, when the bleeding is severe, re-laparotomy is recommended, with the aim of finding and controlling the source of bleeding. This approach usually guarantees an uneventful subsequent course. Late PPH is often challenging and the pathogenesis is diverse. Vascular erosion secondary to POPF or intraabdominal abscess, late failure of intraoperative hemostatic devices, arterial pseudoaneurysm, and intraluminal ulceration are some of the most common causes. In this setting, surgical access to the source of bleeding may be challenging. Angiography (if extraluminal) and endoscopy (if intraluminal) represent the primary approaches to treatment. Surgery is reserved for hemodynamically unstable patients and for those who present with deteriorating condition, multiorgan failure or sepsis. Given these assumptions, except for early mild events, contrast-enhanced abdominal CT is crucial in all cases of PPH, possibly allowing identification of the source and planning the management accordingly. Also, it should be noted that late massive hemorrhages may be preceded by mild self-limiting sentinel bleeds. A prompt abdominal CT scan aimed at excluding vascular lesions is therefore strongly recommended in these cases.
The incidence of complications following pancreatic resection remains high. The ISGPS established standardized definitions and clinical grading systems for POPF, PPH, DGE, and biliary and chyle leak. These classification systems have enabled unbiased comparisons of intraoperative techniques and management decisions. However, the management policies of these complications are most often driven by a patient’s condition and local surgical expertise and is not always based on the available high-level evidence. The development of high-volume specialized units with appropriate resources and multidisciplinary experience in complication management might further improve the evidence and the outcomes.
Subtotal cholecystectomy for difficult acute cholecystitis
Laparoscopic cholecystectomy is considered the gold standard for treatment of benign gallbladder diseases. Cholecystectomy using this method can be completed in 90% of elective cholecystectomies and 70% of emergency cholecystectomies. Acute cholecystitis, especially if difficult, can change the above paradigm, resulting in open conversion or change of technique. The conditions that define a difficult cholecystectomy are as follows: necessity of conversion from laparoscopic to open surgery; duration of procedure greater than 180 min; blood loss greater than 300 ml; and urgent need for involvement of a more experienced surgeon. One of the “rescue” procedures to complete the surgery safely (both for the surgeons and patients) is subtotal cholecystectomy (STC). Open and laparoscopic subtotal cholecystectomy have been reported. For many surgeons, this is considered a bail out technique, and the timing of decision making is crucial to avoid catastrophic complications. Te capability to perform STC in laparoscopy is increasingly requested during difficult laparoscopic cholecystectomy. Difficult LC has a risk of BDI from 3 to 5 times higher in laparoscopy than open surgery. In case of operative difficulties of young surgeons mostly trained in laparoscopy the help of senior surgeons is strongly recommended. The purpose of the present study is to clarify how laparoscopic subtotal cholecystectomy may be used to complete a difficult cholecystectomy for acute cholecystitis without serious complications.

Biliary leakage represents the most frequent complication of incomplete resection of the gallbladder wall in cases of difficult acute cholecystectomy treated with subtotal cholecystectomy. This complication is rarely fatal but requires correct treatment. If bile leakage does not stop spontaneously seven days postoperatively, the possible treatments are endoscopic biliary sphincterotomy, endoscopic plastic stent, and a fully covered self-expanding metal stent. When performing closure of the gallbladder stump, suturing the anterior residual of both anterior and posterior wall represents the best method to have fewer complications. Complications, if not lethal, decrease the patient’s quality of life. Intraoperatively, it is of utmost importance to carefully expose the gallbladder stump to avoid left-in-place stones, wash the entire cavity and drain the abdomen. Bile duct injuries can be a significant complication in this type of surgery. Prevention of the lesions with conversion from laparoscopic to open, or the opinion of older surgeon in case of difficulties is strongly recommended. Mortality is a very rare complication. The limitations of our study are given by the heterogeneity of the techniques used for LSC and the lack of a long-term follow-up analyzing the related complications.
The “BAD” Gallbladder
Once the decision for surgery has been made, an operative plan needs to be discussed and implemented. Should one initially start with laparoscopic surgery for the “bad gallbladder”? If a laparoscopic approach is taken, when should bail-out maneuvers be attempted? Is converting to open operation still the standard next step? A 2016 study published by Ashfaq and colleagues sheds some light on our first question. They studied 2212 patients who underwent laparoscopic cholecystectomy, of which 351 were considered “difficult gallbladders.” A difficult gallbladder was considered one that was necrotic or gangrenous, involved Mirizzi syndrome, had extensive adhesions, was converted to open, lasted more than 120 minutes, had a prior tube cholecystostomy, or had known gallbladder perforation. Seventy of these 351 operations were converted to open. The indications for conversion included severe inflammation and adhesions around the gallbladder rendering dissection of triangle of Calot difficult (n 5 37 [11.1%]), altered anatomy (n 5 14 [4.2%]), and intraoperative bleeding that was difficult to control laparoscopically (n 5 6 [1.8%]). The remaining 13 patients (18.5%) included a combination of cholecystoenteric fistula, concern for malignancy, common bile duct exploration for stones, and inadvertent enterotomy requiring small bowel repair. Comparing the total laparoscopic cholecystectomy group and the conversion groups, operative time and length of hospital stay were significantly different; 147 +- 47 minutes versus 185 +- 71 minutes (P<.005) and 3+-2 days versus 5+-3 days (P 5 .011), respectively. There was no significant difference in postoperative hemorrhage, subhepatic collection, cystic duct leak, wound infection, reoperation, and 30-day mortality.2 From these findings, we can glean that most cholecystectomies should be started laparoscopically, because it is safe to do so. It is the authors’ practice to start laparoscopically in all cases.
BAILOUT PROCEDURES
Despite the best efforts of experienced surgeons, it is sometimes impossible to safely obtain the critical view of safety in a bad gallbladder with dense inflammation and even scarring in the hepatocystic triangle. Continued attempts to dissect in this hazardous region can lead to devastating injury, including transection of 1 or both hepatic ducts, the common bile duct, and/or a major vascular injury (usually the right hepatic artery). Therefore, it is imperative that any surgeon faced with a bad gallbladder have a toolkit of procedures to safely terminate the operation while obtaining maximum symptom and source control, rather than continue to plunge blindly into treacherous terrain. If the critical view of safety cannot be achieved owing to inflammation, and when further dissection in the hepatocystic triangle is dangerous, these authors default to laparoscopic subtotal cholecystectomy as our bail-out procedure of choice. The rationale for this approach is that it resolves symptoms by removing the majority of the gallbladder, leading to low (although not zero) rates of recurrent symptoms. It is safe, and can be easily completed laparoscopically, thus avoiding the longer hospital stay and morbidity of an open operation. There is now significant data supporting this approach. In a series of 168 patients (of whom 153 were laparoscopic) who underwent subtotal cholecystectomy for bad gallbladders, the mean operative time was 150 minutes (range, 70–315 minutes) and the average blood loss was 170 mL (range, 50–1500 mL). The median length of stay for these patients was 4 days (range, 1–68 days), and there were no common bile duct injuries.23 There were 12 postoperative collections (7.1%), 4 wound infections (2.4%), 1 bile leak (0.6%), and 7 retained stones (4.2%), but the 30-day mortality was similar to those who underwent a total laparoscopic cholecystectomy. A systematic review and meta-analysis by Elshaer and colleagues showed that subtotal cholecystectomy achieves comparable morbidity rates compared with total cholecystectomy. These data support the idea that we should move away from the idea that the only acceptable outcome for a cholecystectomy is the complete removal of a gallbladder, especially when it is not safe to do so. This shift toward subtotal cholecystectomy has been appropriately referred to as the safety first, total cholecystectomy second approach.
Liver Procurement
Donor Evaluation and Management
There are very few absolute contraindications for abdominal organ donation, which can be summarized in the short form CHUMP: (1) Creutzfeldt-Jakob disease, (2) active HIV infection, (3) uncontrolled donor sepsis, (4) history of melanoma or other malignancy that poses a risk for transmission regardless of the apparent disease – free period, and (5) past history of non-curable malignancy (curable malignancy such as localized small kidney tumors, localized prostate cancer, localized colon malignancy >5 years previously may be considered after careful risk/benefit assessment). In addition to these general criteria, there are organ-specific criteria for guiding the acceptance of a liver for transplantation. A history of hepatitis or alcoholism is certainly a warning sign, but both livers from HBsAg-positive and/or HCV-positive donors are currently used worldwide, and suitability for transplant must be judged on a case-by-case basis. In general, in the case of a marginal liver donor, the intraoperative assessment by the donor surgeon, in addition to liver biopsy pathological evaluation, is the best single piece of information.
Technical Aspects of Liver Procurement
A midline laparotomy from the xyphoid to the pubis is performed and the round ligament divided. The intra-abdominal organs are explored to check for eventual malignancies, and the quality of the liver is assessed: in the absence of contraindications for a transplant, a sternotomy can be performed. Of note, in the presence of prior heart surgery, the complete warm dissection should be made prior to the sternotomy. It is also prudential to isolate and encircle the aorta prior to sternotomy in order to be ready to cannulate in the event of cardiac arrest/injury at thoracotomy. A blunt dissection behind the sternum just below the jugular notch should be performed until the fingertip can be placed retrosternal around the jugular notch. The sternotomy is then performed in a cranial to caudal direction with the sternum saw to avoid left innominate vein injury. The division of the left triangular ligament allows the mobilization of the left lateral segments of the liver and the exposure of the supraceliac aorta just below the diaphragm to be encircled. The division of the falciform ligament up to the suprahepatic inferior vena cava (IVC) provides more mobility of the liver, necessary when the IVC must be divided from a cardiac graft. Before starting the dissection of the hepatoduodenal ligament, the hepatogastric ligament must be inspected by dividing the lesser omentum. This ligament is usually very thin and transparent so that any replaced or accessory left hepatic artery should be easily visible. In addition, palpation of the ventral border of the foramen of Winslow makes it possible to identify a possible accessory or replaced right hepatic artery. Variations in the hepatic arterial supply can complicate the hilar dissection in up to one third of donors.
THE HILAR STRUCTURES
The hilar structures of the liver are then dissected free; the common bile duct (CBD) is dissected on the level of the edge of the second duodenal portion after opening of the peritoneum and visualization of the duct. In difficult cases, due to a high BMI, following the cystic duct out of the gall bladder can help to identify the CBD. The CBD should be encircled from the lateral border of the hepatoduodenal ligament in order to avoid injury of the portal vein. The CBD and the gallbladder are opened and flushed with normosaline solution. The origins of the gastroduodenal, gastric, and splenic arteries are then identified and encircled and, in the case of liver only procurement, will be taped just before cross-clamping in order to increase flushing through the hepatic artery to the liver.
VASCULAR CANULATION / SOLUTION PRESERVATION
The aorta can be isolated by two approaches. One approach requires mobilization of the right colon on top of Gerota’s fascia and should be extended into a Kocher maneuver to uncover both the inferior vena cava and the abdominal aorta; the other approach is performed by opening the root of the mesentery from the Treitz fascia, along the margin of the duodenum until visualization of the right iliac vessels and ureter is achieved. The inferior mesenteric artery can be tied and divided, and the abdominal aorta, just 2–3 cm above the bifurcation, isolated and encircled. The lumbar arteries could be either tied or clipped and then cut in order to provide mobility of the aorta and facilitate the cannulation. Two umbilical tapes are placed around the dissected segment of the aorta and secured by clamps and will be used to secure aortic cannulae to the vessel. The inferior mesenteric vein (IMV) is most commonly used for access into the portal system by ligating the distal part of it but leaving it uncut to retract the vein with a mosquito clamp. Another tie is then placed around the cranial portion of the vein, using it for occlusion of the vein by retracting it while a partial incision of the vein is performed. The portal cannula can be inserted into the IMV while the tension of the occluding tie is decreased before tying it around the vein and inserted cannula. At this point, 30,000-IU heparin should be given to prevent the blood from clotting after the cross-clamping. Once these preliminary procedures have been completed, the aortic cannulae (20-F armed cannulae) can be inserted into the distal abdominal aorta and secured with the umbilical tapes.
CROSS-CLAMP
The subdiaphragmatic aorta is now clamped (cross-clamp), and cold preservation solution is then rapidly infused through the aortic and portal cannulae; the liver flow is decompressed by dividing the inferior vena cava in the chest. The abdomen is filled with water and ice. The choice of solution for infusion and its amount varies from center to center. The quality of the flush can be assessed by evaluating the outflow of the supradiaphragmatic IVC which should become more transparent with time as the blood in the abdominal organs is replaced by the preservation solution. After the flush is completed, some of the ice is removed from the abdomen to allow the cold dissection of the structures. The gastroduodenal, gastric, and splenic arteries can now be divided. Just below the gastroduodenal artery, the portal vein can be found and can be followed back, if pancreas procurement is not performed, by dividing the head of the pancreas. The cannulae in the IMV can now be removed, the splenic vein ligated and divided, and the venous cannulae replaced in the superior mesenteric vein once it is divided from its distal branches. The superior mesenteric artery (SMA) can now be found in the retro-pancreatic laminae and should be ligated, secured to a clamp and divided in order to find the aortic plane by following back the SMA. This dissection must be made on the left side of the SMA in order to avoid damage to a possible replaced or accessory right hepatic artery. The renal arteries are usually just below the SMA. They should be visualized before the suprarenal aorta is divided. This section must be made in 45°, first looking for ostia of accessory renal arteries before performing complete separation of the aorta. By following back the splenic and gastric arteries, the celiac trunk can be visualized. The aorta must now be divided just below the diaphragm, obtaining a patch containing the celiac trunk and the origin of the mesenteric artery. At this time point, a finger is placed in the supradiaphragmatic IVC helping to identify it while the diaphragm is cut. A portion of the diaphragm should be kept with the liver to ensure that this gross and fast dissection does not damage the organ. The diaphragm is cut to the right, and the incision is then continued between the right kidney and the liver, usually dividing the adrenal gland which is a good sign that none of the adjacent organs are damaged. The location for division of the infrahepatic IVC depends on the renal veins. These are identified on both sides, and the IVC can be safely divided on the virtual line about 1 cm above the renal veins. The only structures now holding the liver in the abdomen are the diaphragmatic pillars. By keeping the liver to the right thoracic cavity and holding the aortic patch, the resected IVC, and the portal vein with its cannulae, the liver removal can be completed by cutting the diaphragmatic muscles. The liver is freed and taken out of the abdomen. A further perfusion with cool preservation solution should be performed on the back table before packing the liver in the transportation box usually with 1 l of preservation solution. The liver can now be packed in the transportation box.
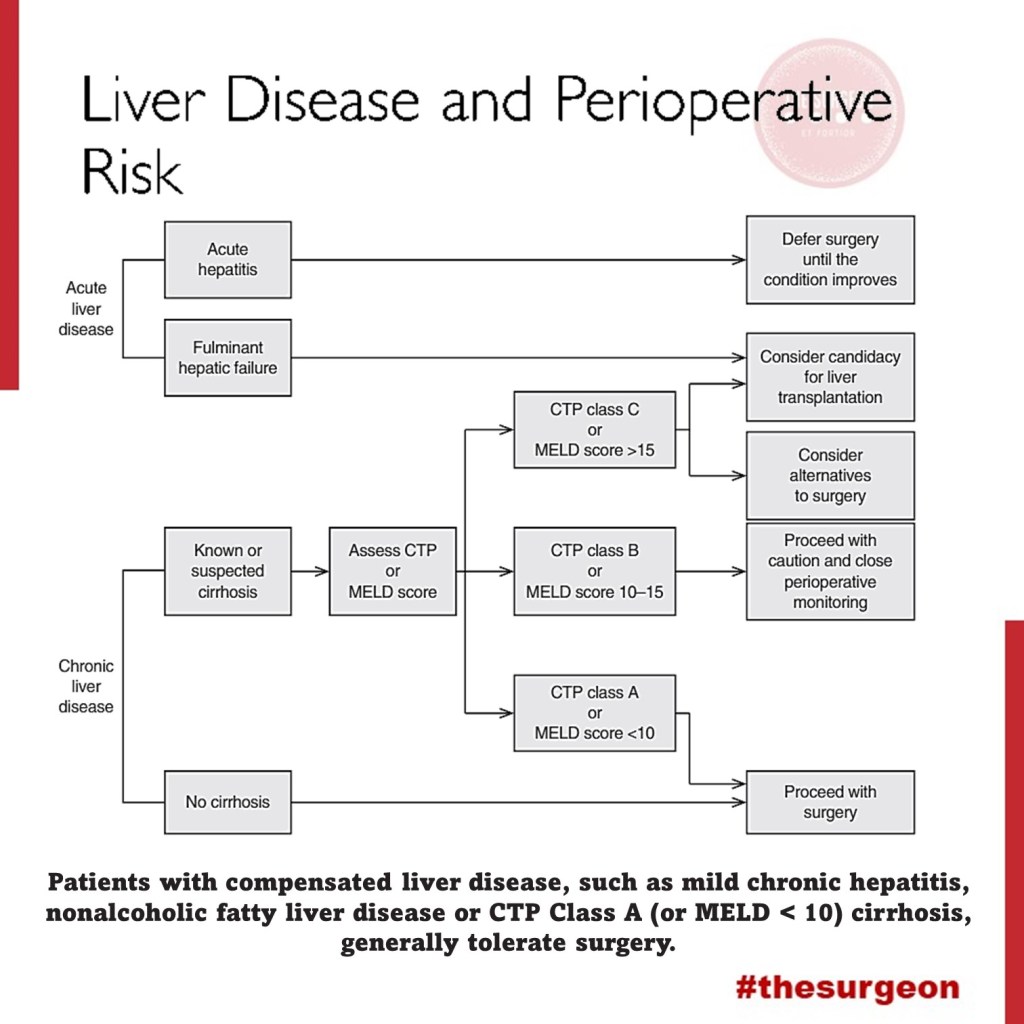
Liver Disease and Perioperative Risk
The liver is the biggest intestinal organ and plays a central role in the homeostasis of different physiological systems including nutrition and drug metabolism, the synthesis of plasma proteins and haemostatic factors, as well as the elimination of different endogenous and exogenous substances. Although the liver contributes with only 3% to total body weight, given its major role in homeostasis and high energy consumption, it receives 25% of total cardiac output (CO). Two vessels contribute to the perfusion of the liver. The majority (70%) of the hepatic perfusion is provided by the portal vein, which contributes 50% of the organ’s oxygen demand. The other 50% is provided by the hepatic artery, which makes up around 30% of total liver perfusion. Hepatic arterial blood flow is mainly dependent on the organ’s metabolic demands and controlled via autoregulatory mechanisms, whereas blood supply through the portal vein depends on the perfusion throughout the whole gastrointestinal tract and the spleen. This unique, dual perfusion system provides constant perfusion rates and oxygen supply, which is crucial for adequate liver function. These high oxygen demands are reflected in a hepatic vein saturation of almost 30%.
The liver is also unique in its ability of regeneration, which allows the performance of major surgery including, amongst others, extended resections of liver tumours, living donations and so on. Many patients have normal liver function parameters when they present for liver surgery, especially when the reason for resection is metastasis or a benign liver tumour. The most common causes of liver resections are the hepatocellular carcinoma (HCC) and the cholangiocellular carcinoma (CCC). Hepatocellular carcinoma (HCC) often develops in patients with underlying liver cirrhosis; many of these patients show signs of chronic liver dysfunction (CLD).
As explained previously, the liver plays a central role in a great deal of physiological systems. Therefore, in case of chronic liver dysfunction (CLD) or liver failure, several effects on other organ systems have to be expected. Consequently liver resections and bile duct surgery as having a high risk for perioperative cardiac events, with an estimated 30-day cardiac event rate (cardiac death and myocardial infarction) of more than 5%. Patients undergoing liver surgery pose a significant challenge to treating physicians in the perioperative period. Due to the improvement of surgical techniques, the “liver patient” is becoming more and more complex, confronting surgeons, anaesthetists and intensive care personnel with difficult intra- and postoperative courses, and considerable multiorgan disorders. The cornerstones of an optimal management are careful selection of the patients, appropriate monitoring and protection of the liver and other vital organs.
Role of The SURGEON
Attributes of a Good Surgeon
Realising the benefits that good leadership and teamwork can deliver requires commitment from all those involved in patient care. From the surgeon’s viewpoint there are numerous desirable attributes which are developed through medical school education, foundation training, core training and into professional practice. These are outlined below:
1. Clinical Care
An obvious consideration of what makes a “good surgeon” is the care provided to patients throughout the patient journey. This includes technical ability in the operating theatre and non-technical skills.
2. Maintenance and Improvement
Remaining up-to-date with innovations in surgical practice and patient are is an important attribute of a good surgeon. In doing so, one is able to inform patients and explain the reasons for and against procedures, allowing them to make an informed decision. Willingness to learn from others and improve from others by reviewing personal practice forms part of Continuing Professional Development; this is a requirement in a portfolio to meet revalidation and recertification criteria.
3. Teaching, Training and Supervision
Educating others forms part of professional development and surgeons frequently oversee projects for medical students or trainees. This requires knowledge of the objectives of the tasks undertaken, knowledge of what technical and non-technical skills should be improved and knowledge of how to encourage the development of these skills. The mentormentee relationship should work both ways, such that the mentee is able to approach their supervisor for assistance and is accepting of any constructive criticism delivered.
4. Relationships with Patients
Relationships with patients are fundamentally based on trust; the patient trusts that the surgeon will do all in their power to help them and their surgical journey. Obtaining informed consent prior to clinical care is based on trust and allows patient autonomy to be upheld. Developing relationships with patients begins from the first consultation and is continued after the day of an operation being undertaken. Acknowledging the needs of the individual and employing effective communication helps in developing an open relationship. In this way patients disclose their medical history and admit underlying fears, allowing better patient care to be delivered.
5. Relationships with Colleagues
Partnership with all members of the multidisciplinary clinical team, management, technicians and support staff fosters healthy working relationships. Consequently, patient care is enhanced through communication, enhanced productivity and an improved team dynamic. Understanding how a colleague works and taking action to facilitate a positive working environment is beneficial to all. Emotional intelligence forms an important component of working relationships, through the ability “to understand and recognize emotional states and to use that understanding to manage one’s self and other individuals or teams”.
6. Health
Maintenance of good personal health and knowing when you must stop working is important in the protection of patient safety. The relevant senior staff must be informed of communicable disease or blood-borne disease transmission. In addition, being vigilant of the health of colleagues forms part of protecting patient safety, for example, failure to report suspicion that the consultant consistently operates after several glasses of wine or that the CT2 has been seen smoking drugs can facilitate the propagation of errors in the workplace. Finally, surgeons are renowned for working at all hours, however acknowledgement that we all need rest is crucial in good patient care.
Adverse events in SURGERY
The surgical domain can be seen as more complex and high risk in its delivery of care than other non-interventional specialities. It is therefore not surprising that in the majority of studies of adverse events in healthcare, at least 50% occurred within the surgical domain and the majority of these in the operating theatre. Furthermore, at least half of these adverse events were also deemed preventable. Just as the multiple studies in the developed world have similar figures for adverse events in hospitalised patients across all specialities, there appears to be a similar rate of harm in surgery. A review of 14 studies, incorporating more than 16000 surgical patients, quoted an adverse event occurring in 14.4% of surgical patients. This was not simply minor harm; a full 3.6% of these adverse events were fatal, 10% severe and 34% moderately harmful. Gawande, a surgeon from Boston, made one of the first attempts to clarify the source of these adverse events.

This paper pioneered the concept that the majority of these adverse events were not due to lack of technical expertise or surgical skill on the part of the surgeon, finding instead that ‘systems factors’ were the main contributing factor in 86% of adverse events. The most common system factors quoted were related to the people involved and how they were functioning in their environment. Communication breakdown was a factor in 43% of incidents, individual cognitive factors (such as decision-making) were cited in 86%, with excessive workload, fatigue and the design or ergonomics of the environment also contributing.
These findings were confirmed in the systematic review of surgical adverse events, where it was found that errors in what were described as ‘nonoperative management’ were implicated in 8.32% of the study population versus only 2.5% contributed to by technical surgical error. In accordance with other high-risk industries, such as commercial aviation, the majority of these adverse events are therefore not caused by failures of technical skill on the part of the individual surgeon, but rather lie within the wider healthcare team, environment and system. Lapses and errors in communication, teamworking, leadership, situational awareness or decision-making all feature highly in post-hoc analysis of surgical adverse events. This knowledge of error causation has been prominent and acknowledged in most other high-risk industries for many years, but it is only recently that healthcare has appreciated this.
Hepatocellular ADENOMA
Benign liver tumours are common and are frequently found coincidentally. Most benign liver lesions are asymptomatic, although larger lesions can cause non-specific complaints such as vague abdominal pain. Although rare, some of the benign lesions, e.g. large hepatic adenomas, can cause complications such as rupture or bleeding. Asymptomatic lesions are often managed conservatively by observation. Surgical resection can be performed for symptomatic lesions or when there is a risk of malignant transformation. The type of resection is variable, from small, simple, peripheral resections or enucleations, to large resections or even liver transplantation for severe polycystic liver disease.
General Considerations
Hepatocellular adenomas (HCA) are rare benign hepatic neoplasms in otherwise normal livers with a prevalence of around 0.04% on abdominal imaging. HCAs are predominantly found in women of child-bearing age (2nd to 4th decade) with a history of oral contraceptive use; they occur less frequently in men. The association between oral contraceptive usage and HCA is strong and the risk for a HCA increases if an oral contraceptive with high hormonal potency is used, and if it is used for over 20 months. Long-term users of oral contraceptives have an estimated annual incidence of HCA of 3–4 per 100000. More recently, an increase in incidence in men has been reported, probably related to the increase in obesity, which is reported as another risk factor for developing HCA. In addition, anabolic steroid usage by body builders and metabolic disorders such as diabetes mellitus or glycogen storage disease type I are associated with HCAs. HCAs in men are generally smaller but have a higher risk of developing into a malignancy. In the majority of patients, only one HCA is found, but in a minority of patients more than 10 lesions have been described (also referred to as liver adenomatosis).
Clinical presentation
Small HCAs are often asymptomatic and found on abdominal imaging being undertaken for other purposes, during abdominal surgery or at autopsy. Some patients present with abdominal discomfort, fullness or (right upper quadrant) pain due to an abdominal mass. It is not uncommon that the initial symptoms of a HCA are acute onset of abdominal pain and hypovolaemic shock due to intraperitoneal rupture. In a series of patients who underwent resection, bleeding was reported in up to 25%. The risk of rupture is related to the size of the adenoma. Exophytic lesions (protruding from the liver) have a higher chance of bleeding compared to intrahepatic or subcapsular lesions (67% vs 11% and 19%, respectively, P<0.001). Lesions in segments II and III are also at higher risk of bleeding compared to lesions in the right liver (35% vs 19%, P = 0.049).
Management
There is no guideline for the treatment of HCAs, although there are general agreements. In men, all lesions should be considered for surgical resection independent of size, given the high risk of malignant transformation, while taking into account comorbidity and location of the lesion. Resection should also be considered in patients with HCAs due to a metabolic disorder. In women, lesions <5 cm can be observed with sequential imaging after cessation of oral contraceptive treatment. In larger tumours, treatment strategies vary. Some clinicians have proposed non-surgical management if hormone therapy is stopped and patients are followed up with serial radiological examinations. The time period of waiting is still under debate, however recent studies indicate that a waiting period of longer than 6 months could be justified.
More recently, the subtypes of the Bordeaux classification of HCA have been studied related to their risk of complications. Some groups report that percutaneous core needle biopsy is of limited value because the therapeutic strategy is based primarily on patient sex and tumour size. Others report a different therapeutic approach based on subtype. Thomeer et al. concluded that there was no evidence to support the use of subtype classification in the stratification and management of individual patients related to risk of bleeding. Size still remains the most important feature to predict those at risk of bleeding during follow-up. However, malignant transformation does seem to be related to differences in subtypes. β-catenin-mutated HCAs trigger a potent mitogenic signalling pathway that is prominent in HCC. Cases of inflammatory HCAs can also show activation of the β-catenin pathway with a risk of developing malignancy. Therefore, β-catenin-mutated and inflammatory HCAs are prone to malignant degeneration, and particularly if >5cm. In these circumstances, invasive treatment should be considered.
POPF after Distal Pancreatectomy
Minimally Invasive Versus Open Techniques
Despite advances in laparoscopic and robotic approaches, the vast majority of distal pancreatectomies continue to be performed via an open approach. Recent retrospective data have demonstrated that minimally invasive distal pancreatectomy is associated with decreased blood loss and shorter hospital stays than open pancreatectomy. A large recent study utilizing the Nationwide Inpatient Sample database suggested, first, that the minimally invasive approach is becoming more widely utilized, increasing from 2.4 to 7.3 % over a study period from 1998 to 2009. Second, that study reported that the minimally invasive approach was associated with decreased length of stay as well as decreased incidence of infectious complications, bleeding complications, and blood transfusions. This population-based study echoes conclusions drawn by a large multi-institutional study performed several years previously. Drawing on a combined patient sample of 667 patients, with 24 % initially attempted laparoscopically, the authors were able to demonstrate lower overall complication rate, decreased blood loss, and shorter hospital stays among patients undergoing laparoscopic approach via a multivariate analysis.
Notably, there was no significant difference in the pancreatic leak rate between the open and laparoscopic approaches, although there was a nonsignificant trend favoring the laparoscopic approach. More recently, the robotic approach has generated significant interest as a technique for performing distal pancreatectomy. Retrospective analysis has suggested that the robotic approach is well suited for pancreatectomy. Fistula rates, however, remain a concern. A retrospective review of patients undergoing robotic pancreatic operations included 83 patients who underwent distal pancreatectomy. About 27 % were identified as having a ISPGF type A pancreatic leak; 12 and 4.8 % were identified as having a grade B or C leak, respectively.
Identifying Risk Factors
For pancreaticoduodenectomy (PTD) , a fistula risk score has been recently developed that has been shown to be highly predictive of POPF. This score assigns points based on gland texture, gland pathology, duct diameter, and intraoperative blood loss. In general, high blood loss, soft gland texture, and smaller duct diameter confer increased risk of POPF, whereas pancreatic adenocarcinoma and pancreatitis as the indication for PTD confer protection for the development of pancreatic fistula versus other diagnoses. Also of note, higher fistula risk scores correlated with greater incidence of clinically relevant (ISGPF grade B or C) fistula. The adaptation of this risk score to patients undergoing distal pancreatectomy is yet to be validated; however, at least one published study indicates that this scoring system may have limitations in the setting of distal pancreatectomy. In that study, risk factors for pancreatic fistula after stapled gland transection in patients undergoing distal pancreatectomy were examined, and in a multivariate analysis, only the presence of diabetes and the use of a 4.1-mm staple cartridge were associated with increased risk of pancreatic fistula formation.
Surgical Management of CHOLANGIOCARCINOMA
Cholangiocarcinoma is an uncommon cancer that occurs within the intrahepatic and extrahepatic portions of the bile duct system. In North America, the incidence of extrahepatic cholangiocarcinoma is 0.5–2 per 100,000 and 0.95 per 100,000 for intrahepatic cholangiocarcinoma. Up to 50% of patients will be lymph node (LN) positive at presentation, 5% are multifocal tumors, and 10–20% will have peritoneal involvement at presentation. Risk factors for cholangiocarcinoma are primary sclerosing cholangitis (PSC) with a lifetime risk 10–40%, parasitic infection, previous sphincteroplasty, congenital anomalies of the biliary tree (choledochal cyst, Caroli’s disease, anomalous pancreaticobiliary duct junction), and chronic biliary inflammatory disease (hepatitis B/C, liver cirrhosis, recurrent pyogenic cholangitis). The most common presentation is painless jaundice and weight loss in the setting of extrahepatic duct involvement. In Western countries, 80% are extrahepatic (20% distal and 60% hilar) and 20% are intrahepatic.
SPECIAL NOTES
Click here and share on TWITTER
• Ca 19–9 can be elevated in up to 85% of patients with cholangiocarcinoma, but is not specific; elevation can also occur in the setting of obstructive jaundice without malignancy. If it remains elevated after biliary decompression, it could indicate the presence of malignancy. Elevated pre- and postoperative Ca 19–9 predict poor survival.
• For perihilar tumors, decisions regarding which side of the liver to resect depend on right- or left-sided dominance, volume of future liver remnant, and the extent of vascular and ductal involvement.
• Some centers report that 30–50% of tumors will be deemed unresectable at the time of surgery, despite accurate preoperative imaging.
• Quality Indicators: Pathologic Analysis—R0 margin, regional lymphadenectomy includes three or more LN.
SURGICAL MANAGEMENT
Intrahepatic cholangiocarcinoma: Surgical resection is the only potential cure with removal of involved liver segments. There is emerging evidence that recommends a routine hilar LN and dissection for its prognostic value, M1 disease includes involvement of celiac,periaortic or caval LN.
Distal bile duct (below the cystic duct): Surgical resection is the only potential cure. Pancreaticoduodenectomy including en bloc resection of extrahepatic bile duct and gallbladder. Regional nodes include: Hilar (CBD, common hepatic, portal, cystic), Posterior and anterior pancreaticoduodenal Nodes along SMV, Nodes along right and lateral wall of SMA.
Hilar (above the cystic duct): En bloc resection of extrahepatic bile duct and gallbladder, including right and left hepatectomy, or extended right/left hepatectomy, Caudate lobe should be removed. Regional nodes include: Hilar (CBD, hepatic,portal, cystic), Pericholedochal nodes in hepatoduodenal ligament.
Liderança Cirúrgica
Tradicionalmente, a marca de um grande cirurgião está relacionada à habilidade técnica, conhecimento e perspicácia diagnóstica, enquanto pouco foco é dado a outras habilidades de Liderança. Hoje, as habilidades não técnicas, como habilidades de comunicação e liderança, acabam se traduzindo em maior segurança do paciente, experiência e resultados. O dia em que o cirurgião inicia seu primeiro trabalho o define como um líder, e isso é particularmente verdadeiro para o cirurgião do Aparelho Digestivo, pois é imediatamente considerado um especialista. Os cirurgiões são líderes preparados para aproveitar as muitas oportunidades de desempenho que as posições acadêmicas fornecem para executar seus interesses fora do atendimento tradicional ao paciente. Algumas pessoas nascem com habilidades de liderança e outras as desenvolvem com o tempo. É claro que esse processo deve começar cedo e que os estudantes de medicina e residentes devem estar preparados para papéis de liderança conforme RODRIGO VIANA explica neste vídeo para ajudar no desenvolvimento das habilidades críticas e não clínicas relacionadas à liderança de uma equipe e ao ensino. Para alguns, os papéis principais podem mudar ou desviar a carreira do atendimento ao paciente. Para outros, um equilíbrio entre as carreiras clínicas e administrativas pode ser cumprido em um papel definido. Independentemente disso, estabelecer metas e trabalhar para alcançá-las dentro da construção de uma equipe é a marca de todos os líderes de sucesso.
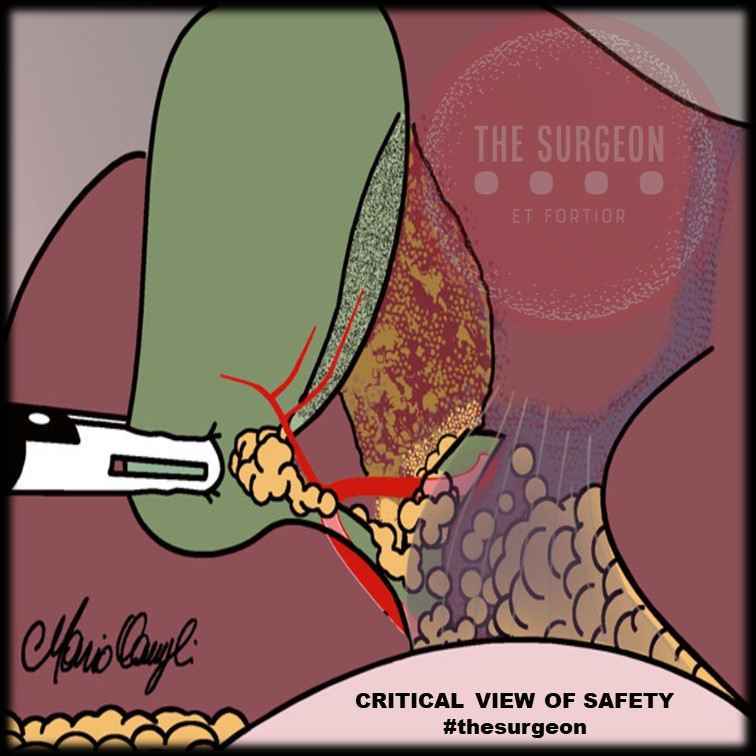
Critical View Of Safety
“The concept of the critical view was described in 1992 but the term CVS was introduced in 1995 in an analytical review of the emerging problem of biliary injury in laparoscopic cholecystectomy. CVS was conceived not as a way to do laparoscopic cholecystectomy but as a way to avoid biliary injury. To achieve this, what was needed was a secure method of identifying the two tubular structures that are divided in a cholecystectomy, i.e., the cystic duct and the cystic artery. CVS is an adoption of a technique of secure identification in open cholecystectomy in which both cystic structures are putatively identified after which the gallbladder is taken off the cystic plate so that it is hanging free and just attached by the two cystic structures. In laparoscopic surgery complete separation of the body of the gallbladder from the cystic plate makes clipping of the cystic structures difficult so for laparoscopy the requirement was that only the lower part of the gallbladder (about one-third) had to be separated from the cystic plate. The other two requirements are that the hepatocystic triangle is cleared of fat and fibrous tissue and that there are two and only two structures attached to the gallbladder and the latter requirements were the same as in the open technique. Not until all three elements of CVS are attained may the cystic structures be clipped and divided. Intraoperatively CVS should be confirmed in a “time-out” in which the 3 elements of CVS are demonstrated. Note again that CVS is not a method of dissection but a method of target identification akin to concepts used in safe hunting procedures. Several years after the CVS was introduced there did not seem to be a lessening of biliary injuries.
Operative notes of biliary injuries were collected and studied in an attempt to determine if CVS was failing to prevent injury. We found that the method of target identification that was failing was not CVS but the infundibular technique in which the cystic duct is identified by exposing the funnel shape where the infundibulum of the gallbladder joins the cystic duct. This seemed to occur most frequently under conditions of severe acute or chronic inflammation. Inflammatory fusion and contraction may cause juxtaposition or adherence of the common hepatic duct to the side of the gallbladder. When the infundibular technique of identification is used under these conditions a compelling visual deception that the common bile duct is the cystic duct may occur. CVS is much less susceptible to this deception because more exposure is needed to achieve CVS, and either the CVS is attained, by which time the anatomic situation is clarified, or operative conditions prevent attainment of CVS and one of several important “bail-out” strategies is used thus avoiding bile duct injury.
CVS must be considered as part of an overall schema of a culture of safety in cholecystectomy. When CVS cannot be attained there are several bailout strategies such a cholecystostomy or in the case of very severe inflammation discontinuation of the procedure and referral to a tertiary center for care. The most satisfactory bailout procedure is subtotal cholecystectomy of which there are two kinds. Subtotal fenestrating cholecystectomy removes the free wall of the gallbladder and ablates the mucosa but does not close the gallbladder remnant. Subtotal reconstituting cholecystectomy closes the gallbladder making a new smaller gallbladder. Such a gallbladder remnant is undesirable since it may become the site of new gallstone formation and recurrent symptoms . Both types may be done laparoscopically.”
Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 1995;180:101-25.
Surgical Management of GERD After Sleeve Gastrectomy
Evaluation of a patient referring GERD after sleeve gastrectomy should start with a detailed history and physical examination; the presence or absence of GERD-related symptoms should be thoroughly documented as well as any prior treatments or therapy used to treat it. Obtaining preoperative and operative records is of paramount importance particularly in those patients who had their index procedure performed elsewhere. Any endoscopic findings and prior imaging available are important to determine what the best course of action would be. If the patient had preoperative and postoperative imaging such as UGI, it is useful to compare those with a recent study to look for anatomical problems that may have been not addressed at the time of the index operation or developed over time. After this information is obtained, we can classify the GERD after sleeve as:
1. De novo GERD
2. Preexisting GERD without improvement
3. Preexisting GERD with worsening/complication
Regardless of how we classify the GERD, an initial evaluation with imaging
studies such as UGI and EGD is recommended. Comparison with any prior films if available is of significant value. Based on the UGI, we can determine if the shape of the sleeve falls into one of the following categories: tubular, dilated bottom, dilated upper, or dumbbell-shaped sleeve; we will also be able to evaluate esophageal peristalsis in real time and if there is associated hiatal hernias. We believe UGI under fluoroscopy provides important physiologic and anatomic information that can help guide our management approach, and therefore we offer it to all patients. We follow the radiologic evaluation with endoscopy, and during endoscopy, we look for objective signs of reflux such as esophagitis, presence of bile in the stomach or esophagus, as well as missed or recurrent hiatal hernias. In patients with evidence of esophagitis or metaplasia, multiple biopsies are taken. During the endoscopy, subtle findings that suggest a kink or a stricture may be present. In the absence of objective signs of gastroesophageal reflux disease on both endoscopy and upper GI series, we pursue physiologic testing followed by highresolution manometry and pH monitoring. In those patients where clear reflux esophagitis is seen, this additional testing may not be necessary or may be performed in selected cases depending on what the surgical or endoscopic therapy would be.

While it is true that most sleeve-related GERD will be effectively treated with a conversion to Roux-en-Y gastric bypass, not every patient with GERD after reflux will require a bypass or would agree to have one. First key step in addressing the patient is to evaluate whether the patient was selected appropriately to have a sleeve and second is to determine the exact sleeve anatomy; are there anatomical factors that will make it more likely for this patient to experience reflux; is there dilated fundus? Is there a kink or stricture in the sleeve or is it an anatomically appropriate operation? We should pay important attention to the weight loss the patient has experienced with the sleeve. Patients who do not have adequate weight loss and have GERD symptoms should not undergo other therapies and should probably undergo a bypass; however it is our unpublished experience that patients with the association of poor weight loss after sleeve and difficult to treat GERD will correct their GERD after conversion, but their weight loss results are still marginal even with a well-constructed bypass.
Covid-19 and Digestive Surgery
The current world Covid-19 pandemic has been the most discussed topic in the media and scientific journals. Fear, uncertainty, and lack of knowledge about the disease may be the significant factors that justify such reality. It has been known that the disease presents with a rapidly spreading, it is significantly more severe among the elderly, and it has a substantial global socioeconomic impact. Besides the challenges associated with the unknown, there are other factors, such as the deluge of information. In this regard, the high number of scientific publications, encompassing in vitro, case studies, observational and randomized clinical studies, and even systematic reviews add up to the uncertainty. Such a situation is even worse when considering that most healthcare professionals lack adequate knowledge to critically appraise the scientific method, something that has been previously addressed by some authors. Therefore, it is of utmost importance that expert societies supported by data provided by the World Health Organization and the National Health Department take the lead in spreading trustworthy and reliable information.
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
#Medicine #Surgery #GeneralSurgery #DigestiveSurgery
#TheSurgeon #OzimoGama
Bariatric Surgery During the COVID-19 Pandemic
Coronavirus disease 2019 (COVID-19) emerged in Wuhan City and rapidly spread throughout China and around the world since December 2019. The World Health Organization (WHO) declared the COVID-19 outbreak a global pandemic on 11 March 2020. Patients with metabolic disorders like cardiovascular diseases, diabetes and obesity may face a greater risk of infection of COVID-19 and it can also greatly affect the development and prognosis of pneumonia.
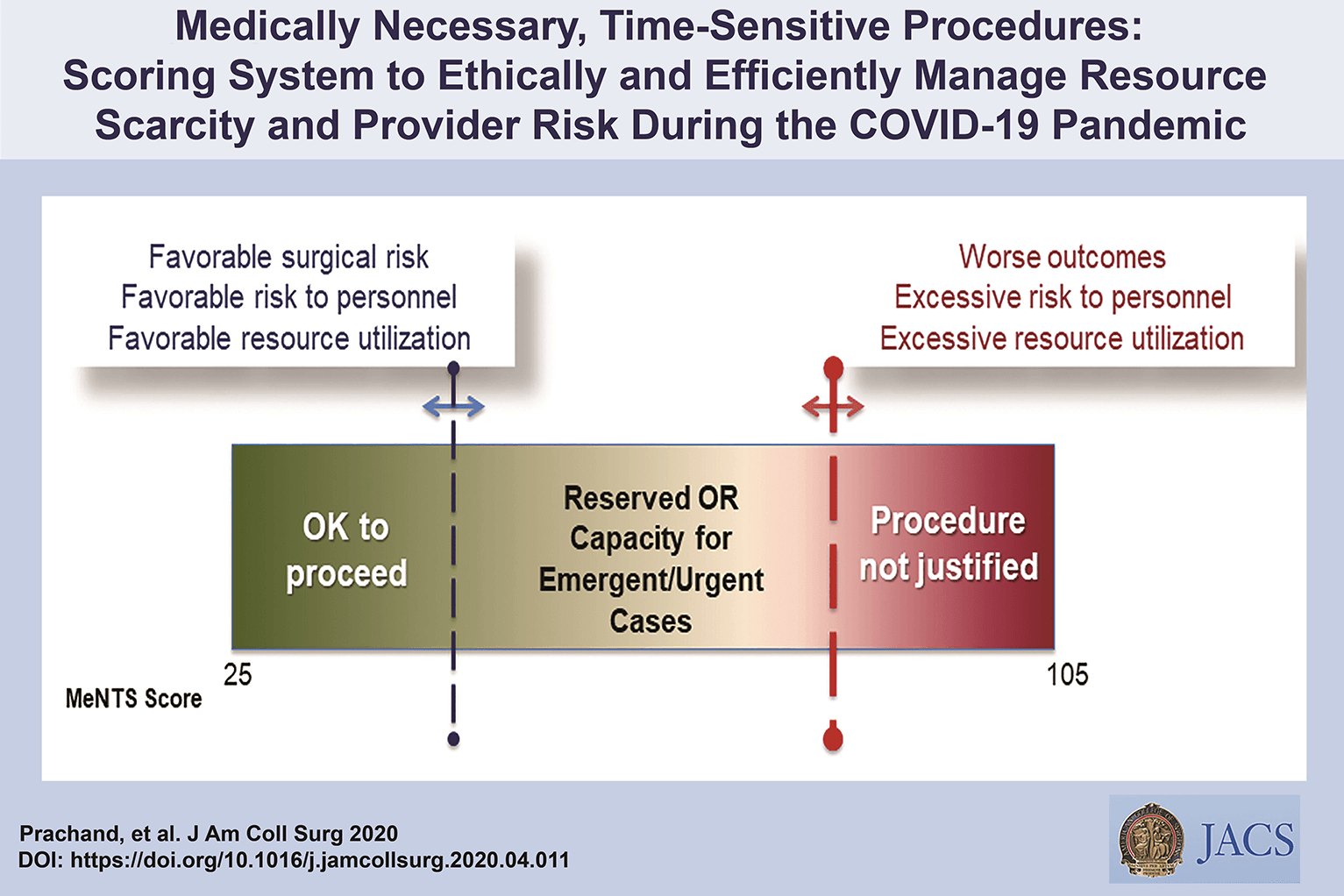
“A higher cumulative MeNTS score, which can range from 21 to 105, is associated with poorer perioperative patient outcome, increased risk of COVID-19 transmission to the health care team, and/or increased hospital resource utilization. Given the need to maintain OR capacity for trauma, emergency, and highly urgent cases, an upper threshold MeNTS score can be designated by surgical and perioperative leadership based on the immediately anticipated conditions and resources at each institution.”
GENERAL RECOMMENDATIONS
All elective surgical and endoscopic cases for metabolic and bariatric surgery should be postponed during the pandemic. This minimises risks to both patient and healthcare team, as well as reducing the utilisation of unnecessary resources, such as beds, ventilators and personal protective equipment (PPE). In addition, postponing these services will minimise potential exposure of the COVID-19 virus to unsuspecting healthcare providers and patients. As the long-term effects or complications of COVID-19 are still unknown, metabolic and bariatric surgeries for patients who were diagnosed and recovered from COVID-19 should be evaluated by a multidisciplinary team. Diet and lifestyle modifications should be advised before surgical treatment.
More information on link below
IFSO RECOMMENDATIONS
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
#Medicine #Surgery #GeneralSurgery #DigestiveSurgery
#TheSurgeon #OzimoGama
Laparoscopic JEJUNOSTOMY
Many oncological patients with upper gastrointestinal (GI) tract tumours, apart from other symptoms, are malnourished or cachectic at the time of presentation. In these patients feeding plays a crucial role, including as part of palliative treatment. Many studies have proved the benefits of enteral feeding over parenteral if feasible. Depending on the tumour’s location and clinical stage there are several options of enteral feeding aids available. Since the introduction of percutaneous endoscopic gastrostomy (PEG) and its relatively easy application in most patients, older techniques such as open gastrostomy or jejunostomy have rather few indications.
The majority of non-PEG techniques are used in patients with upper digestive tract, head and neck tumours or trauma that renders the PEG technique unfeasible or unsafe for the patient. In these patients, especially with advanced disease requiring neoadjuvant chemotherapy or palliative treatment, open gastrostomy and jejunostomy were the only options of enteral access. Since the first report of laparoscopic jejunostomy by O’Regan et al. in 1990 there have been several publications presenting techniques and outcomes of laparoscopic feeding jejunostomy. Laparoscopic jejunostomy can accompany staging or diagnostic laparoscopy for upper GI malignancy when the disease appears advanced, hence avoiding additional anaesthesia and an operation in the near future.
In this video the author describe the technique of laparoscopic feeding jejunostomy applied during the staging laparoscopy in patient with advanced upper gastrointestinal tract cancer with co-morbid cachexy, requiring enteral feeding and neoadjuvant chemotherapy.
Welcome and get to know our Social Networks through the following link
Ozimo Gama’s Social Medias
#AnatomiaHumana #CirurgiaGeral #CirurgiaDigestiva #TheSurgeon
Surgical Management of CHRONIC PANCREATITIS
Chronic pancreatitis (CP) is a progressive, destructive, inflammatory process that ends in total destruction of the pâncreas and results in malabsorption, diabetes mellitus, and severe pain. The incidence and prevalence of CP are increasing in the worldwide and incidence is between 1.6 and 23 per 100 000 with increasing prevalence. The treatment of CP is complex; in the majority of cases na interdisciplinary approach is indicated that includes conservative, endoscopic, and surgical therapy. The surgical treatment of CP is based on two main concepts:preservation of tissue via drainage aims to protect against further loss of pancreatic function, and pancreatic resection is performed for nondilated pancreatic ducts, pancreatic head enlargement,or if a pancreatic carcinoma is suspected in the setting of CP.
RESECTIVE PROCEDURES
The vast majority of patients are seen with a ductal obstruction in the pancreatic head, frequently associated with an inflammatory mass. In these patients, pancreatic head resection is the procedure of choice; The partial pancreatoduodenectomy (PD) or Kausch-Whipple procedure, in its classic or pylorus-preserving variant, has been the procedure of choice for pancreatic head resection in CP for many years (Jimenez et al, 2003). The duodenum-preserving pancreatic head resections and its variants—the Beger (1985), Frey (1987), and Bern procedures (Gloor et al, 2001)—represent less invasive, organsparing techniques with equal long-term results. Only very few patients come to medical attention with smallduct disease (diameter of the pancreatic duct ❤ mm) and no mass in the pancreatic head. Possibly, a large majority of those patients from former series had unknown autoimmune pancreatitis. In these cases, a V-shaped excision of the anterior aspect of the pancreas is a safe approach, with effective pain management (Yekebas et al, 2006). In the rare case of a patient seen with segmental CP in the pancreatic body or tail, such as that seen as a result of posttraumatic ductal stenosis, a middle segment pancreatectomy or a pancreatic left resection may be the best approach.
CONCLUSION
The adequate therapy of CP is adjusted to the symptoms of the patient, the stage of the disease, and the morphology of pathologic changes of the pancreas. The surgical technique must be adjusted to the pathomorphologic changes of the pancreas. For patients with CP and an inflammatory mass in the head of the pancreas, the DPPHR is less invasive than a PD and is associated with comparable long-term results. The Bern modification of the DPPHR represents a technical variation that is equally effective but technically less demanding. Whether total pancreatectomy with islet cell transplantation is a viable therapy of CP remains to be proved by further studies. Surgical therapy provides effective long-term pain relief and improvement of quality of life, but it may not stop the decline of endocrine or exocrine pancreatic function. Strategies to improve or maintain endocrine and exocrine function in CP remain an interesting field of research.
Laparoscopic distal PANCREATECTOMY
Laparoscopic distal pancreatectomy has become a relatively standard operation and has been approached by a similar technique by multiple groups since its original description. Generally, four or five trocars are used to gain entrance to the abdominal cavity, but three-trocar LPD has been described. A “clockwise” technique results in an efficient, reliable, and uniform approach for removing the vast majority of lesions that are located to the left of the neck of the pancreas (Asbun & Stauffer, 2011). The technique begins with the positioning of the patient in a modified right lateral decubitus position. The degree of lateral positioning depends on the patient’s body habitus and the location of the lesion, as well as the tilting capabilities of the operative bed. The use of gravity assisted retraction with the patient in a reverse Trendelenburg position with the left flank elevated is a key component to successful exposure of the tail of the pancreas and the spleen. Four mid- to left-sided abdominal trocars are placed in a semicircle around the body and tail of the pancreas, including two 12 mm and two 5 mm trocars, and a five step clockwise method is used.
Step 1: Mobilization of the splenic flexure of the colon
and exposure of the pancreas
The first step is mobilization of the splenic flexure of the colon. The lateral attachments, splenocolic ligament, and gastrocolic ligament are succes-sively transected to allow access to the lesser sac. If the spleen is to be removed, the dissection proceeds cranially, and the short gastric vessels are transected up to the superior pole of the spleen. Sufficient mobilization of the colon allows for gravity-assisted retraction of the colon, and the stomach is completely freed from the anterior aspect of the body and tail of the pancreas. Infrequently, an additional trocar or tacking stitch is required to elevate the stomach to the anterior abdominal wall off the pancreas and out of the operative field.
Step 2: Dissection along the inferior edge of the pancreas
and choosing the site for pancreatic division
The second step is to identify the inferior border of the pancreas and create a window in the fibroadipose tissue plane between the retroperitoneum and the pancreas. This dissection is carried medially toward the lesion of interest. Intraoperative ultrasound is performed to clearly identify the lesion and the planned site of division of the pancreas.
Step 3: Pancreatic parenchymal division and ligation
of the splenic vein and artery
The third step is pancreatic parenchymal division and ligation of the splenic artery and vein. After dissecting around the pancreas in 360 degrees, a Penrose drain or suture is placed around the proposed site of division of the pancreas and is used to elevate the pancreas from the retroperitoneum. A band passer instrument is helpful for this part of the procedure. For distal pancreatectomy, the splenic vessels will often be dissected, ligated, and divided en bloc with the parenchyma. For subtotal resections with division of the pancreas at the neck, the underlying superior mesenteric vein and splenic vein are dissected away from the posterior aspect of the pancreas, and the celiac trunk is identified individually and dissected free from the neck and proximal body of the pancreas. Parenchymal transection is performed with a linear stapling device by using a slow, gradual, and stepwise compression technique. Thick tissue staples (open staple height of approximately 4 mm) with staple line reinforcement is preferred for almost any pancreas consistency, and the stapler is gradually closed in a stepwise manner over the course of several minutes to allow for parenchymal compression. Parenchymal transection and splenic vessel division are done individually for subtotal pancreatectomy for lesions located between the gastroduodenal artery and the celiac trunk.
Step 4: Dissection along the superior edge of the pancreas
The fourth step is to sweep the pancreas inferiorly and anteriorly off the retroperitoneum toward the splenic hilum. A deeper dissection plane that includes Gerota fascia and the left adrenal gland may be chosen for malig-nancies that appear to have posterior invasion from the pancreas.
Step 5: Mobilization of the spleen and specimen removal
The fifth step is the mobilization of the spleen from its diaphragmatic and retroperitoneal attachments and placement of the specimen within a bag for exteriorization. Major complications were seen in less than 10% of patients, and both the conversion rate and the clinically significant pancreatic fistula (grade B/C) rate by using the gradual stepwise compression stapled technique was seen in fewer than 5%. Operative drains were rarely placed.
Outcomes
The minimally invasive approach to resection of the left-sided pancreas by distal or subtotal pancreatectomy has gained acceptance and been used with an increasing frequency worldwide during the past decade. Multiple systematic reviews have demonstrated the safety of LDP and its superiority versus open distal pancreatectomy (ODP) for selected outcomes, such as blood loss, transfusion rates, and hospital stays; it must be remembered, however, that all these studies are retrospective in nature and therefore severely limited by significant selection bias. All studies showed similar reoperation rates and mortality, but most found a lower overall morbidity for the laparoscopic approach. Some studies identified lower rates of specific complications, such as wound infection and even pancreatic fistula. Although oncologic clearance was similar, most studies have shown that ODP is often the surgery of choice for larger tumors.
Hepatic Hemangioma: Is There an Indication for Surgical Resection?
Hepatic hemangioma (HH) is the most common benign liver tumor. It consists of blood-filled cavities fed by the hepatic arterial circulation, with walls lined by a single layer of endothelial cells, a veritable chaotic entanglement of distorted blood vessels confined to a region as small as a few mm and as large as 10 cm, 20 cm and even 40 cm. The frequency is higher among adults, with a prevalent age at the initial diagnostic in the range of 30-50 years. Literature places the HH incidence at 0.4% to 20% of the total population. At necropsy, the frequency is of 0.4 to 7.3%, all the authors agreeing with an incidence of over 7%. The HH prevalence in the general population varies greatly, most often being discovered incidentally during imaging investigations for various unrelated pathologies. Regarding sex distribution, it seems that women are more susceptible, as confirmed by all pertaining studies, with a reported 4.5:1 to 5:1 ratio of female to male cases. Most often, HH are mono-lesions but multiple-lesions are possible; they account for 2.3% and up to 20-30% of the cases, depending on the source. At the initial diagnosis, the majority of HH measure below 3 cm in size, the so-called capillary hemangiomas; of these, only 10% undergo a size increase with time, for reasons still unknown. The next size class covers lesions between 3 cm and 10 cm in size, referred to as medium hemangiomas. Lastly, giant or cavernous hemangiomas measure up to 10 cm, with occasional literature reports of giant HH reaching 20-40+ cm in size. Location-wise they are most often found in the right liver lobe, often in segment IV, often marginal.
Operative Management
Operative intervention for liver hemangiomas remains a controversial topic. Previous studies from major hepatobiliary centres have proposed varying indications for a hemangioma resection. Findings from the present study demonstrate that operative management of symptomatic hemangiomas remains an effective therapy and can be performed with low morbidity to the patient. However, aside from abdominal symptoms, prophylactic resections in the setting of hemangioma enlargement, size, or patient anxiety is not advised as the risk of developing life-threatening associated complications is rare.
Established Complications. In the minority of cases that present as a surgical emergency due to haemorrhage, rupture, thrombosis and infarction, surgical management may be the only appropriate course of action. There is also a role for the elective surgical management of giant haemangiomata, albeit in a highly selected group of patients. As demonstrated by the data presented above, an operative approach with the objective of preventing future complications of giant haemangiomata is less easy to justify.
Diagnostic Uncertainty. Despite improvements in non-invasive imaging technology, cases of diagnostic uncertainty continue to pose a challenge. In situations where it is not possible to exclude malignancy, surgical intervention by formal liver resection may be indicated. In almost all situations, the use of percutaneous liver biopsy for the differentiation of giant haemangiomata from malignant liver lesions cannot be justified. The risks of haemorrhage as a result of biopsying a giant haemangioma are appreciable and, together with the risks of needle track seeding and intra-abdominal dissemination of a potentially curable malignancy, mean that biopsy in this setting must be avoided.
Incapacitating Symptoms. Having taken all possible steps to ensure that symptoms are attributable to the haemangioma, surgical resection may be justified on grounds of intractable symptoms. Patients with clearly defined abdominal compressive symptoms may be more likely to derive benefit from surgery than patients with non-specific abdominal discomfort, although this is not backed up by a meaningful body of evidence. Management of this group of patients is, by necessity, highly individualised. Despite apparently satisfactory surgical management, symptoms persist in approximately 25% of patients following resection of seemingly symptomatic haemangiomata.
While most people with HH show no sign or symptom, and most HH are non-progressing and do not require treatment, there is a small number of cases with rapid volumetric growth or complications, which prompt for appropriate therapy. The results of clinical and laboratory investigations to date, mostly for imaging techniques, have demonstrated that for small HH, regular follow-up is enough. For cavernous HH, the evolution is unpredictable and often unfavorable, with serious complications requiring particular surgical expertise in difficult cases. Hepatic hemangiomas require a careful diagnosis to differentiate from other focal hepatic lesions, co-occurring diagnoses are also possible.