Cuidados Perioperatórios
O modelo tradicional de cuidado perioperatório, ainda prevalente em parte considerável dos serviços brasileiros, baseia-se em jejum prolongado, preparo intestinal mecânico rotineiro, hidratação venosa abundante, drenos nasogástricos e abdominais por princípio, opioides como pilar analgésico, repouso prolongado e realimentação tardia. O objetivo desta revisão é, portanto, oferecer ao estudantes e residentes em cirurgia do aparelho digestivo uma síntese estruturada, didática e clinicamente útil dos cuidados perioperatórios contemporâneos, articulando história, epidemiologia, evidência protocolada, reconhecimento e manejo de complicações e seguimento ambulatorial, sob o eixo central da segurança e da qualidade assistencial.
Indicações de Cirurgia Bariátrica e Metabólica em Pacientes sob Terapia com Agonistas do Receptor de GLP-1
Critérios de Escalonamento e Prática Clínica
Autor: Prof. Dr. Ozimo Gama
Categoria: Cirurgia Bariátrica e Metabólica / Endocrinologia / Cirurgia do Aparelho Digestivo
Tempo de Leitura: 16 minutos
Introdução
O tratamento da obesidade e da síndrome metabólica atravessa uma mudança de paradigma sem precedentes com a ascensão dos agonistas do receptor de GLP-1 (liraglutida, semaglutida, tirzepatida). Estes fármacos redefiniram as expectativas de perda ponderal e controle glicêmico, desafiando a hegemonia histórica do bisturi como única ferramenta de alta potência. Contudo, na Estratégia Cirúrgica Operacional (ECO), a farmacoterapia não deve ser vista como substituta da cirurgia, mas como um componente de um plano tático integrado. O desafio clínico atual reside em identificar o momento exato da transição do manejo clínico para o cirúrgico. Este artigo propõe uma análise fundamentada nas diretrizes da ASMBS/IFSO 2025 e na literatura de alto impacto, estabelecendo os critérios para o encaminhamento cirúrgico em pacientes que já utilizam estas terapias avançadas.
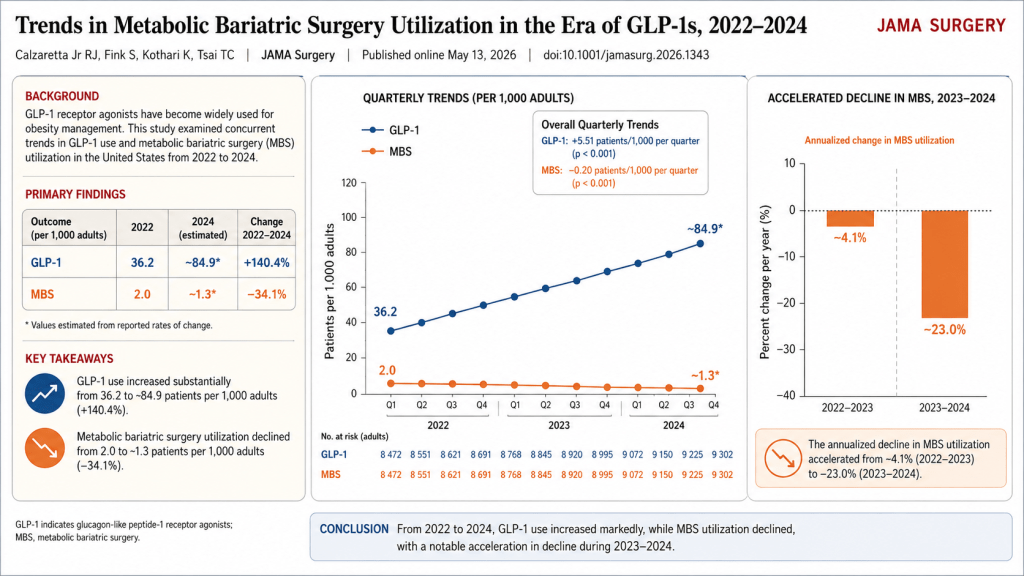
Critérios de Decisão e Resposta Terapêutica
A indicação cirúrgica no paciente sob uso de GLP-1 deve considerar a biologia do “terreno” e o impacto sistêmico da doença.
1. Parâmetros de IMC e Comorbidades
De acordo com o consenso ASMBS/IFSO 2025, a cirurgia bariátrica e metabólica (CBM) é recomendada para indivíduos com IMC ≥ 35 kg/m², independentemente de comorbidades, e deve ser considerada para aqueles com IMC ≥ 30 kg/m² que apresentam doenças metabólicas, como o Diabetes Mellitus Tipo 2 (DM2). No paciente em uso de medicação, o encaminhamento é indicado se:
- IMC inicial persistente: O paciente mantém critérios de elegibilidade apesar da perda de peso induzida pelo fármaco.
- Falha na Remissão: Persistência de DM2 mal controlado ou hipertensão severa mesmo com a otimização da dose de GLP-1.
2. Avaliação da Resposta Terapêutica: O Platô e a Recidiva
A farmacoterapia frequentemente atinge um “platô de eficácia” entre 12 e 18 meses. O cirurgião deve intervir quando a perda ponderal é insuficiente para atingir os objetivos de saúde do paciente ou quando ocorre a recidiva do peso sob vigência da medicação (“atrito” farmacológico).
3. Fatores Funcionais e Anatômicos: Refluxo e Hérnia Hiatal
A escolha da técnica cirúrgica no paciente que usa GLP-1 é ditada pela fisiologia gastroesofágica.
- Doença do Refluxo Gastroesofágico (DRGE): Agonistas de GLP-1 retardam o esvaziamento gástrico, o que pode agravar sintomas de refluxo. Pacientes com DRGE severa e hérnia hiatal volumosa têm contraindicação relativa à Gastrectomia Vertical (Sleeve), sendo o Bypass Gástrico em Y de Roux a manobra tática de escolha por seu efeito antirrefluxo superior.
Aplicação na Cirurgia Digestiva: Diabetes e Esteatose Hepática
A cirurgia metabólica exerce um efeito de “limpeza biológica” em órgãos-alvo que supera a eficácia dos análogos de GLP-1 isolados.
- Diabetes Mellitus Tipo 2 (DM2): Técnicas como o Bypass ou a Bipartição Intestinal promovem uma secreção supra-fisiológica de incretinas endógenas, resultando em taxas de remissão do DM2 superiores a 70% em longo prazo.
- Esteatose Hepática (NASH/MASH): A rápida mobilização de gordura visceral e a redução da resistência insulínica pós-cirúrgica são os métodos mais eficazes para reverter a inflamação e a fibrose hepática, prevenindo a progressão para cirrose.
Pontos-Chave: Sarcopenia e Seguimento
- Risco de Sarcopenia: Perdas rápidas de peso com GLP-1 podem induzir perda de massa muscular. A cirurgia deve ser precedida de avaliação nutricional rigorosa para garantir aporte proteico e preservação funcional.
- Adesão Nutricional: O uso de GLP-1 funciona como um “ensaio tático” para o paciente adaptar-se a volumes menores e maior disciplina alimentar antes da modificação anatômica definitiva.
- Plano de Seguimento: O monitoramento pós-operatório deve ser contínuo, com foco na reposição de micronutrientes e na monitorização do reganho de peso, integrando a equipe multidisciplinar.
Conclusões Sintéticas
A integração entre os agonistas de GLP-1 e a cirurgia bariátrica/metabólica representa o ápice da estratégia multidisciplinar na obesidade. O encaminhamento cirúrgico deve ocorrer quando a farmacoterapia atinge seus limites táticos ou quando a gravidade das comorbidades (DM2 descontrolado, NASH grave, DRGE intratável) exige uma modificação anatômica de alto impacto. Como cirurgiões e professores, devemos ver o GLP-1 como um aliado que “limpa o terreno” para uma cirurgia mais segura e eficaz. A decisão de operar não é uma falha, mas a execução do plano definitivo para a sobrevivência e qualidade de vida do paciente a longo prazo.
“Na guerra contra a obesidade, a medicação é o reconhecimento e a artilharia; a cirurgia é a infantaria que ocupa o terreno e garante a paz metabólica duradoura.” — Adaptado da Estratégia Cirúrgica Operacional do Prof. Dr. Ozimo Gama.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#CirurgiaBariatrica #GLP1 #Obesidade #CirurgiaMetabolica #SaudeMetabolica
Manejo em Unidade de Cuidados Intensivos Pós-Transplante Hepático
Estratégias Operacionais para a Estabilização do Enxerto e Prevenção de Complicações
Autor: Prof. Dr. Ozimo Gama
Categoria: Cuidados Intensivos / Transplante Hepático / Cirurgia Digestiva
Tempo de Leitura: 15 minutos
Introdução
O transplante hepático (TH) consolidou-se como o tratamento padrão para a insuficiência hepática terminal e para determinadas neoplasias hepatobiliares, evoluindo de um procedimento de altíssimo risco para uma intervenção com taxas de sobrevivência que excedem os 85% no primeiro ano. Contudo, a transição do bloco operatório para a Unidade de Cuidados Intensivos (UCI) representa o momento mais crítico da “missão”, onde a “fricção” fisiológica atinge o seu expoente máximo. Nesta fase, o cirurgião e o intensivista atuam num centro de controlo tático, onde a vigilância deve ser ininterrupta. O sucesso não depende apenas da perícia técnica da anastomose, mas do manejo rigoroso do “terreno” biológico (o recetor) para que a “semente” (o enxerto) possa florescer. Com a mudança demográfica nas indicações de transplante — com o aumento da esteato-hepatite não alcoólica (NASH) superando a hepatite C — o perfil do paciente tornou-se mais complexo, exigindo um planeamento operacional ainda mais sofisticado.
A Gestão Multimodal na UCI
O manejo imediato do pós-operatório (PO) de TH deve ser estruturado em eixos de intervenção rápida, visando a estabilização hemodinâmica, a monitorização da função do enxerto e a prevenção de falhas sistémicas.
1. Hemodinâmica e Gestão de Volume
A monitorização da volemia é o pilar da perfusão do enxerto. O estado de “hiperfluxo” ou a congestão venosa podem ser letais para o novo fígado.
- Monitorização: Além da pressão arterial invasiva e do ECG contínuo, a avaliação do volume sistólico e da variação da pressão de pulso oferece uma visão mais precisa do que a pressão venosa central (PVC) isolada.
- Estratégia: O objetivo é a euvolemia. A hipovolémia compromete o influxo pela artéria hepática, enquanto a hipervolémia excessiva aumenta a pressão venosa de saída, promovendo o edema do enxerto e a disfunção sinusoidal.
2. Avaliação da Função e Integridade do Enxerto
A “inteligência de campo” na UCI baseia-se em indicadores laboratoriais e de imagem que confirmam se o enxerto está “online”.
- Marcadores Laboratoriais: A queda progressiva das transaminases (AST/ALT) e a normalização do INR e do lactato são os sinais de que o enxerto assumiu a sua função metabólica. Um aumento súbito das enzimas no PO1 deve levantar suspeita imediata de complicação vascular.
- Doppler da Artéria Hepática e Veia Porta: É o “reconhecimento aéreo” obrigatório. O Doppler deve ser realizado precocemente para confirmar a patência das anastomoses vasculares. A trombose da artéria hepática é uma emergência tática que exige reintervenção imediata para salvar o enxerto.
3. Controlo Metabólico e Eletrolítico
O fígado é o centro logístico do metabolismo. A sua disfunção temporária reflete-se em desequilíbrios graves:
- Sódio: A hiponatremia pré-transplante deve ser corrigida de forma extremamente lenta para evitar a mielinólise pontina central.
- Glicemia: O enxerto funcional deve ser capaz de manter a homeostase da glucose. A hipoglicemia persistente é um sinal ominoso de falência primária do enxerto (Primary Non-Function – PNF).
4. Imunossupressão e Profilaxia
A estratégia moderna foca-se na redução da morbilidade através da retirada precoce de corticosteroides e da minimização dos inibidores da calcineurina em pacientes com disfunção renal. A “superioridade tática” imunológica é alcançada quando se evita a rejeição sem expor o paciente a infeções oportunistas fatais.
Aplicação na Cirurgia Digestiva
Na prática do cirurgião digestivo e transplantador, a UCI é a extensão do bloco operatório. A aplicação da Estratégia de Operações Especiais exige que a equipa multidisciplinar mantenha uma “consciência situacional” absoluta. No Brasil, onde o sistema de transplantes é um dos maiores do mundo e financiado majoritariamente pelo SUS, a eficiência na UCI é o que garante a sustentabilidade do programa. Complicações como a hemorragia pós-operatória ou a disfunção renal aguda devem ser interceptadas antes que desencadeiem a “névoa da guerra” — aquele estado de caos clínico onde as decisões se tornam reativas e não proativas.
Pontos-Chave (Checklist de UCI)
- Patência Vascular: Doppler precoce e seriado da artéria hepática e veia porta.
- Perfusão Tecidual: Débito urinário > 0,5 ml/kg/h e lactato em queda.
- Estabilidade Metabólica: Monitorização rigorosa do sódio, potássio e glicemia.
- Proteção Renal: Evitar nefrotóxicos e manter pressão de perfusão adequada.
- Vigilância de Sangramento: Monitorização dos drenos e do hematócrito. O sangramento é a principal causa de reoperação precoce.
Conclusões Aplicadas à Prática
Os cuidados intensivos após um transplante hepático são um exercício de precisão e paciência estratégica. O cirurgião deve atuar como o estrategista que harmoniza a fisiologia do receptor com a vitalidade do enxerto. A última década de evidências mostrou que a agressividade no suporte inicial, combinada com uma modulação criteriosa da imunossupressão, é o caminho para reduzir a mortalidade. O sucesso da missão de transplante não é declarado na última sutura, mas no momento em que o paciente atinge a estabilidade metabólica e o enxerto demonstra plena autonomia funcional. Na UCI, a vigilância clínica é a nossa arma mais poderosa contra as complicações.
“O transplante hepático é a prova definitiva de que a medicina é uma equipa de equipas. Na UCI, o cirurgião deve ter a humildade de ouvir o intensivista e a coragem de intervir quando o instinto clínico aponta para a falha do enxerto.” — Thomas Starzl, pioneiro do transplante hepático mundial.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#TransplanteHepatico #CuidadosIntensivos #CirurgiaDigestiva #PosOperatorio #SegurancaDoPaciente
Biópsia do Adenocarcinoma de Pâncreas
Com a crescente adoção da quimioterapia neoadjuvante como estratégia pré-operatória, a confirmação histológica tornou-se obrigatória antes do início do tratamento sistêmico. Este artigo compara os principais métodos de biópsia — EUS-FNB, EUS-FNA, biópsia percutânea guiada por TC e biópsia líquida — em termos de acurácia diagnóstica, taxa de complicações, risco de semeadura tumoral e capacidade de fornecer material para análise molecular.
Boa Leitura !
Avanços da Oncologia Clínica nos Tumores do Aparelho Digestivo
Resumo
Os últimos dez anos representaram uma ruptura no tratamento sistêmico dos tumores gastrointestinais. O estabelecimento da quimioterapia tripla perioperatória (FLOT), o papel da imunoterapia adjuvante pós-ressecção trimodal no esôfago (CheckMate-577), a total neoadjuvância no reto (RAPIDO, PRODIGE 23), a introdução do mFOLFIRINOX adjuvante no pâncreas (PRODIGE 24), e o bloqueio de PD-L1 nas vias biliares (TOPAZ-1, KEYNOTE-966) transformaram fundamentalmente o cenário perioperatório. Esta revisão organiza esses marcos por topografia e tipo histológico, com destaque para os estudos que modificaram diretriz.
Boa Leitura !
Manejo Clínico-Cirúrgico das Fístulas Digestivas Pós-Operatórias
Uma Abordagem Estratégica Baseada em Evidências
Autor: Prof. Dr. Ozimo Gama
Categoria: Cirurgia Geral / Emergências Cirúrgicas / Terapia Intensiva / Cirurgia Digestiva
Tempo de Leitura: 15 minutos
Introdução
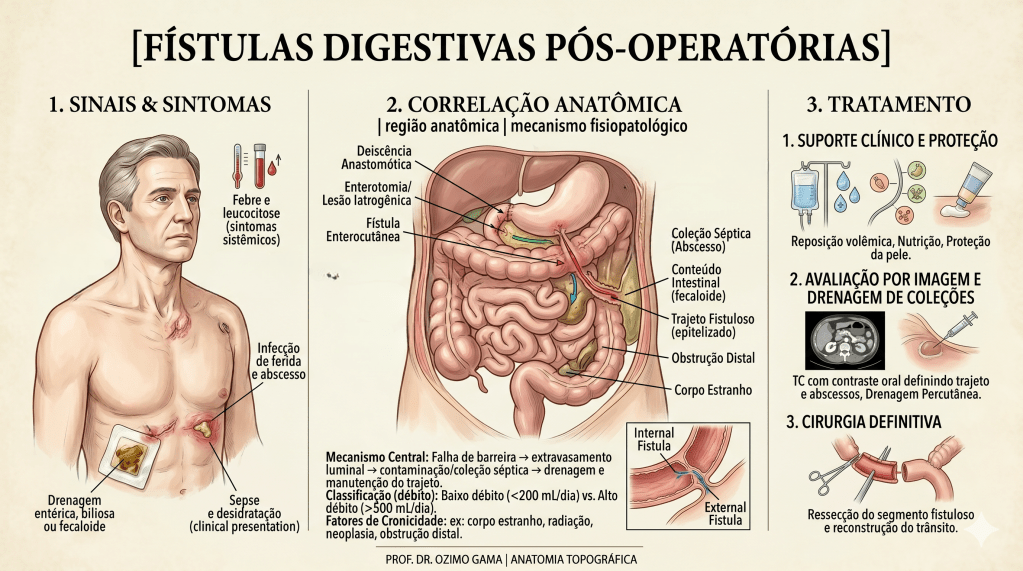
As fístulas digestivas pós-operatórias representam uma das complicações mais temidas e desafiadoras no teatro de operações da cirurgia do aparelho digestivo. Definidas como comunicações anormais entre o lúmen intestinal e o meio externo (fístulas enterocutâneas) ou outros órgãos, elas sinalizam a falência de uma anastomose ou sutura. Historicamente, essa condição era acompanhada por taxas de mortalidade proibitivas. O paradigma do tratamento mudou radicalmente após o trabalho clássico de Chapman et al. (1964), que demonstrou que a desnutrição era o principal determinante de óbito nestes pacientes, e não apenas a falha técnica em si. Antes dessa viragem, a indicação cirúrgica precoce para “fechar o buraco” resultava frequentemente em desastres, devido ao precário estado geral e à inflamação tecidual intensa. Este artigo propõe uma sistematização tática do manejo das fístulas, integrando o suporte metabólico à precisão diagnóstica e à decisão cirúrgica parcimoniosa.
Classificação e Fases do Manejo
A fístula não é um evento isolado, mas um processo dinâmico que exige do cirurgião uma “consciência situacional” aguçada. A classificação baseia-se no débito (volume em 24h): as de Alto Débito (> 500 ml/dia) são as mais críticas, pois provocam distúrbios hidroeletrolíticos e desnutrição acelerada.
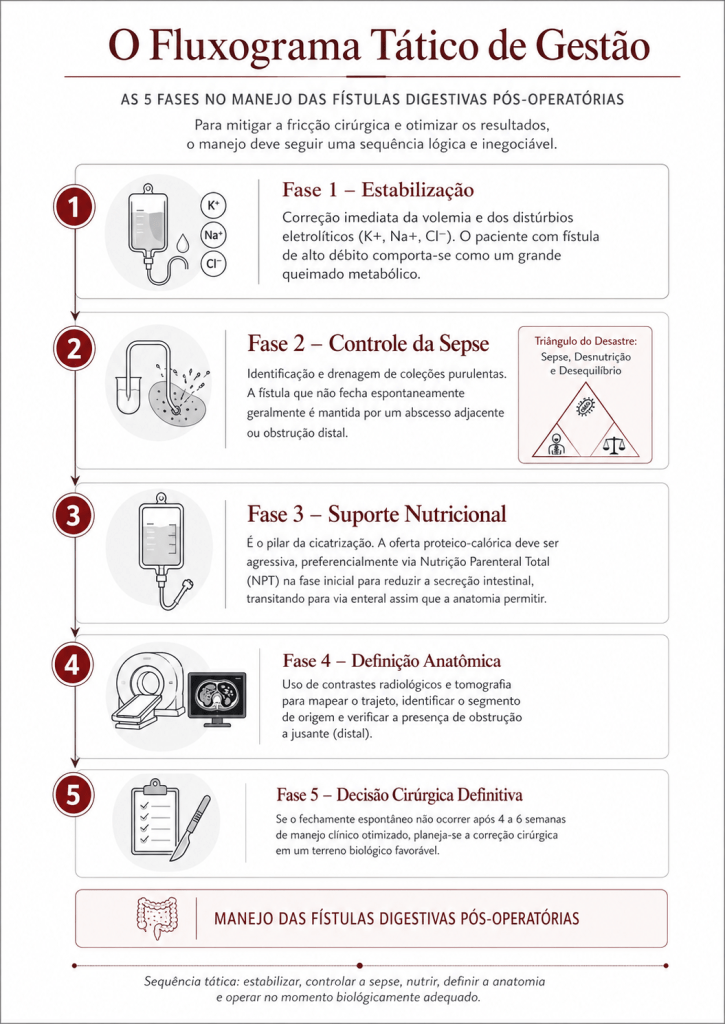
O Fluxograma Tático de Gestão (As 5 Fases)
Para mitigar a fricção cirúrgica e otimizar os resultados, o manejo deve seguir uma sequência lógica e inegociável:
- Fase 1 – Estabilização: Correção imediata da volemia e dos distúrbios eletrolíticos (K+, Na+, Cl-). O paciente com fístula de alto débito comporta-se como um grande queimado metabólico.
- Fase 2 – Controle da Sepse: Identificação e drenagem de coleções purulentas. A fístula que não fecha espontaneamente geralmente é mantida por um abcesso adjacente ou obstrução distal (O Triângulo de Desastre: Sepse, Desnutrição e Desequilíbrio).
- Fase 3 – Suporte Nutricional: É o pilar da cicatrização. A oferta proteico-calórica deve ser agressiva, preferencialmente via Nutrição Parenteral Total (NPT) na fase inicial para reduzir a secreção intestinal, transitando para via enteral assim que a anatomia permitir.
- Fase 4 – Definição Anatômica: Uso de contrastes radiológicos e tomografia para mapear o trajeto, identificar o segmento de origem e verificar a presença de obstrução a jusante (distal).
- Fase 5 – Decisão Cirúrgica Definitiva: Se o fechamento espontâneo não ocorrer após 4 a 6 semanas de manejo clínico otimizado, planeia-se a correção cirúrgica em um “terreno” biológico favorável.
Avaliação Diária dos Dispositivos e da Fístula: Sinais de Alerta
A vigilância diária na enfermaria é a nossa inteligência de campo. O cirurgião deve avaliar os dispositivos com rigor metodológico:
- Efluente da Fístula: Avaliar volume, cor e odor. O aparecimento de bile ou conteúdo gástrico num dreno antes purulento indica uma nova comunicação.
- Proteção da Pele: O suco entérico e pancreático é extremamente cáustico. O uso de bolsas coletoras e pastas de barreira (estomaterapia) é essencial para evitar a evisceração por digestão da parede abdominal.
- Sinais de Alerta (Red Flags):
- Mudança súbita no volume: Parada abrupta de drenagem associada a dor e febre sugere obstrução do trajeto e formação de abcesso.
- Hemorragia “Sentinela”: Sangue vivo no trajeto da fístula pode indicar erosão de vasos mesentéricos pelas enzimas, prenunciando uma hemorragia maciça.
Aplicação na Cirurgia Digestiva: Contexto Brasileiro
No Brasil, autores renomados como Campos, Rasslan e Safatle estabeleceram que o tratamento operatório das fístulas deve ser uma manobra de exceção planejada. Dados brasileiros reforçam que a mortalidade, embora reduzida para patamares de 10% a 20% em centros de excelência, ainda é elevada quando há atraso no suporte nutricional ou controle inadequado da infecção. Em procedimentos como a duodenopancreatectomia (Whipple), a fístula pancreática exige uma tática específica de desvio e proteção vascular, muitas vezes utilizando a somatostatina ou seus análogos para reduzir o débito exócrino e permitir a granulação do leito.
Pontos-Chave
- Paciência Estratégica: Não opere precocemente uma fístula em fase inflamatória (fase de “fricção” máxima). Aguarde a estabilização metabólica.
- Nutrição é Cicatrização: Sem proteínas, não há fechamento. O manejo nutricional é a “munição” do paciente.
- Controle de Sepsis: A fístula não é a vilã; a coleção não drenada é que mata o paciente.
- Anatomia é Destino: Identifique obstruções distais. Uma fístula nunca fechará se houver um obstáculo à frente.
Conclusões Aplicadas à Prática
O manejo das fístulas digestivas pós-operatórias é a prova máxima da resiliência e competência de uma equipe de cirurgia digestiva. Ele exige a fusão entre o conhecimento anatómico profundo e uma disciplina tática inabalável na condução do suporte clínico. O cirurgião que compreende que “operar menos” na fase aguda é, por vezes, a manobra mais heróica, protege a vida do doente. A missão é transformar uma catástrofe pós-operatória numa jornada de reabilitação, onde o bisturi é reservado para o momento da reconstrução definitiva, num organismo capaz de sustentar a cura. Como afirma a literatura clássica, nas fístulas, o cirurgião deve ter a mão de ferro no suporte clínico e a mão de veludo na indicação operatória.
“A fístula digestiva testa a paciência do cirurgião, a resiliência do paciente e a qualidade de todo o sistema de suporte hospitalar.” — Sérgio Rasslan, mestre da cirurgia de urgência e trauma brasileira.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#FistulaDigestiva #CirurgiaDigestiva #ManejoCirurgico #SegurancaDoPaciente #EducacaoMedica
Dieta no Pós-Operatório de Cirurgia Digestiva
Guia Estratégico e Alimentos Proibidos para a Recuperação Tática
Autor: Prof. Dr. Ozimo Gama
Categoria: Cuidados Perioperatórios / Nutrição Clínica / Cirurgia do Aparelho Digestivo
Tempo de Leitura: 14 minutos
Introdução
O sucesso de uma intervenção cirúrgica no aparelho digestivo não se encerra na última sutura ou no grampeamento da anastomose. A fase subsequente, o pós-operatório, exige uma “logística de suprimentos” impecável: a dieta. Historicamente, a conduta cirúrgica era pautada pelo dogma do repouso intestinal absoluto — o infame “nada por via oral” até o retorno dos ruídos hidroaéreos ou a eliminação de flatos. Contudo, a ciência cirúrgica moderna, impulsionada por protocolos como o ERAS (Enhanced Recovery After Surgery) e o brasileiro Projeto ACERTO, promoveu uma quebra de paradigma. Sabemos hoje que a realimentação precoce é uma “manobra tática” essencial para reduzir a resposta endócrino-metabólica ao trauma (REMIT), preservar a barreira mucosa intestinal e acelerar a alta hospitalar. Para o residente e o estudante de medicina, orientar a dieta é prescrever o combustível para a cicatrização e a retomada da “superioridade tática” do organismo do paciente.
Desenvolvimento: A Progressão Fisiológica e o Ciclo de Tolerância
A reintrodução alimentar deve ser vista como uma missão por fases, onde cada etapa é validada pela tolerância do paciente (aplicando o ciclo OODA: Observar a aceitação, Orientar a progressão, Decidir a consistência e Agir na prescrição).
- Fase de Líquidos Claros (Hidratação Tática): Inicia-se nas primeiras 6 a 24 horas. O foco é o teste de motilidade. Inclui água, chais e caldos coados.
- Fase de Líquidos Completos (Aporte Proteico): Introdução de suplementos hiperproteicos para combater a sarcopenia.
- Dieta Pastosa/Branda (Transição de Fluxo): Purês e cozidos de fácil mastigação. É o “reconhecimento do terreno”.
- Dieta Regular (Retorno à Base): Retomada da alimentação sólida, focada em proteínas de alto valor biológico.
Alimentos Proibidos: O que evitar para proteger o “Terreno” em cicatrização
Na estratégia de recuperação, existem “fatores hostis” que podem sabotar a cicatrização das anastomoses ou provocar distensão abdominal, comprometendo a integridade tática da cirurgia. Entender a relação entre a Semente (o procedimento) e o Terreno (o organismo do paciente) é fundamental para o sucesso. Abaixo, listamos os alimentos que devem ser estritamente evitados ou suspensos conforme a fase:
1. Bebidas Gaseificadas (Refrigerantes e Água com Gás)
O dióxido de carbono provoca distensão súbita do lúmen gástrico e intestinal. Em cirurgias com anastomoses recentes, o aumento da pressão intraluminal atua como um estresse mecânico indesejado, podendo agravar o desconforto e retardar o retorno do peristaltismo.
2. Alimentos de Alta Fermentação (FODMAPs e Leguminosas)
Feijão, repolho, brócolis, couve-flor e cebola crua são indutores de gases. A fermentação excessiva no pós-operatório imediato causa cólicas e distensão, dificultando a deambulação e a mecânica respiratória do paciente.
3. Frituras e Gorduras Saturadas
Alimentos gordurosos exigem uma carga biliar e pancreática elevada para a digestão. Em colecistectomias, onde o fluxo biliar é contínuo e sem reservatório, a gordura pode provocar diarreia osmótica e dispepsia severa. Além disso, retardam o esvaziamento gástrico, aumentando o risco de náuseas.
4. Carboidratos Simples e Doces Concentrados
Especialmente críticos em pacientes gastrectomizados. Açúcares refinados, mel e doces de massa podem desencadear a Síndrome de Dumping, caracterizada por tontura, taquicardia e diarreia explosiva devido ao rápido deslocamento osmótico de fluidos para o intestino.
5. Condimentos Irritantes (Pimentas e Molhos Industrializados)
A capsaicina e conservantes químicos agridem a mucosa gastrointestinal em fase de remodelação. A inflamação química da mucosa pode mascarar sinais de alerta e causar desconforto desnecessário.
6. Álcool e Embutidos
O álcool é um agente hepatotóxico e desidratante que interfere na síntese proteica necessária para a cicatrização. Embutidos (salsicha, presunto, salame) contêm níveis elevados de sódio e nitritos, que promovem retenção hídrica e edema de anastomoses.
Aplicação na Cirurgia Digestiva: Especificidades por Procedimento
A tática alimentar deve ser personalizada conforme o “teatro de operações” anatômico:
- Gastrectomias: Fracionamento extremo (6 a 8 refeições). Proibição absoluta: Ingerir líquidos durante as refeições sólidas. Isso acelera o esvaziamento e precipita o Dumping.
- Colecistectomias: Foco em dieta hipogordurosa por pelo menos 15 a 30 dias até a adaptação do esfíncter de Oddi ao fluxo biliar direto.
- Ressecções de Cólon: Evitar fibras insolúveis brutas (cascas de frutas, sementes) nos primeiros dias para não sobrecarregar a área de sutura com volume fecal endurecido.
Pontos-Chave para a Orientação do Paciente
- Mastigação Exaustiva: A digestão mecânica na boca é a primeira linha de defesa contra a sobrecarga das anastomoses.
- Fracionamento: Comer pouco e muitas vezes é o segredo para manter o metabolismo ativo sem gerar distensão.
- Hidratação: Água e chás devem ser consumidos em pequenos goles ao longo do dia, longe das refeições principais.
- Sinais de Alerta: Vômitos, soluços persistentes ou dor abdominal súbita indicam que a progressão da dieta deve ser interrompida (“pausa tática”).
Conclusões Aplicadas
A dieta no pós-operatório de cirurgia digestiva não é um detalhe acessório, mas uma intervenção cirúrgica de caráter biológico. Como cirurgiões e professores, nosso dever é educar o paciente para que ele se torne um aliado tático na sua própria recuperação. Saber o que não comer é tão vital quanto saber o que comer. Ao eliminarmos os alimentos proibidos, estamos blindando o organismo contra complicações mecânicas e funcionais, permitindo que o processo natural de cicatrização ocorra em um ambiente de baixa pressão e inflamação controlada. No “campo de batalha” da recuperação, a disciplina alimentar é a nossa estratégia de elite para garantir uma vitória duradoura contra a doença.
“Que o teu alimento seja o teu único remédio.” — Hipócrates, o pai da medicina. Na cirurgia moderna, a sabedoria reside em saber que a restrição alimentar tática é o caminho mais curto para a liberdade nutricional plena.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#DietaPosOperatória #CirurgiaDigestiva #AlimentosProibidos #ProjetoACERTO #SegurancaDoPaciente
Manobras Cirúrgicas Avançadas no Trauma Abdominal
A Arte da Exposição e o Controle do Caos
Autor: Prof. Dr. Ozimo Gama
Categoria: Cirurgia do Trauma / Técnica Cirúrgica / Anatomia Aplicada
Tempo de Leitura: 18 minutos
Introdução
No “teatro de operações” do trauma abdominal grave, o cirurgião enfrenta o maior de todos os inimigos: o tempo. Quando a cavidade é aberta em uma laparotomia de emergência, a “névoa da guerra” — composta por sangue, coágulos e contaminação entérica — obscurece as lesões vitais. Como ensinam Hirshberg e Mattox no clássico Top Knife, “se você não consegue ver a lesão, você não consegue pará-la”. A cirurgia do trauma não é o lugar para dissecções anatômicas delicadas de livro-texto; é o domínio da exposição tática. O domínio das manobras de rotação visceral e mobilização de órgãos sólidos é o que separa o cirurgião hesitante do estrategista capaz de resgatar um paciente da tríade letal (acidose, hipotermia e coagulopatia). Este artigo aprofunda-se nas manobras fundamentais para o controle de danos e tratamento definitivo dos traumas de órgãos específicos.
Manobras de Exposição e Acesso Vascular
Antes de tratar um órgão específico, o cirurgião deve dominar as grandes manobras de rotação, que permitem o acesso ao retroperitônio e aos grandes vasos.
1. Manobra de Mattox (Rotação Visceral Medial Esquerda)
É a “chave mestra” para o acesso à aorta abdominal suprarrenal e aos vasos renais esquerdos.
- Técnica: Inicia-se com a mobilização do cólon esquerdo (linha de Toldt), estendendo a incisão superiormente, atrás do baço e da cauda do pâncreas. Todo o bloco visceral (cólon, rim, pâncreas e baço) é rotacionado medialmente para a direita.
- Aplicação: Essencial para o controle de sangramentos aórticos próximos ao hiato diafragmático ou lesões da artéria mesentérica superior na sua origem.
2. Manobra de Cattell-Braasch (Rotação Visceral Medial Direita)
Permite a visualização completa do retroperitônio infra e parapancreático.
- Técnica: Realiza-se a mobilização do cólon direito e da base do mesentério do intestino delgado até o ligamento de Treitz. O bloco é rotacionado para a esquerda e para cima.
- Aplicação: Exposição da veia cava inferior (VCI) em toda a sua extensão infra-hepática, dos vasos renais direitos e da aorta infrarrenal.
Manobras Específicas por Órgão
1. Fígado: O Gigante da Hemorragia
O trauma hepático é a principal causa de morte por hemorragia intra-abdominal. As manobras visam o controle do influxo e efluxo sanguíneo.
- Manobra de Pringle: Consiste na oclusão do ligamento hepatoduodenal (veia porta, artéria hepática e via biliar). Se o sangramento persistir após o Pringle, a fonte é provavelmente o efluxo (veias hepáticas ou VCI retro-hepática).
- Mobilização Hepática: Secção dos ligamentos falciforme, coronários e triangulares. Isso permite “trazer o fígado para a linha média”, essencial para suturas em lesões posteriores.
- Packing (Tamponamento): A manobra de controle de danos mais eficaz. Envolve o uso de compressas (“The Big Five”) colocadas entre o fígado e a parede abdominal/diafragma, criando compressão direta sobre a lesão.
2. Baço: Preservação vs. Sacrifício
No trauma esplênico, a decisão é binária: salvar ou retirar.
- Mobilização Esplênica: Através da secção do ligamento esplenorenal. O baço é trazido para a ferida cirúrgica, permitindo a inspeção do hilo e da cauda do pâncreas.
- Técnica: Em pacientes instáveis, a esplenectomia rápida é a conduta tática correta. Suturas esplênicas (esplenorrafias) são reservadas para pacientes estáveis e lesões capsulares simples.
3. Complexo Duodeno-Pâncreas
Devido à sua localização retroperitoneal profunda, as lesões aqui são frequentemente ocultas.
- Manobra de Kocher: Abertura da fáscia de coalescência lateral ao duodeno, permitindo a mobilização da segunda porção do duodeno e da cabeça do pâncreas para a linha média.
- Exposição do Corpo/Cauda: Realizada através da abertura do ligamento gastrocólico (acesso à transcavidade dos epíplons).
A Mentalidade de Controle de Danos
Na prática do cirurgião do aparelho digestivo brasileiro, a aplicação dessas manobras deve seguir a lógica da Cirurgia de Controle de Danos (DCS). No trauma, a técnica deve ser “rápida e suja” (quick and dirty), focada na fisiologia e não na anatomia estética. Por exemplo, em uma lesão pancreática grave com sangramento ativo, a manobra de Kocher extensiva é vital para identificar se há envolvimento da VCI ou da aorta. Se houver contaminação entérica maciça por lesão duodenal, o uso de grampeadores para “fechar as pontas” e realizar o packing é superior a tentativas de reconstruções complexas que consomem tempo e levam à exaustão metabólica.
Pontos-Chave para a Estratégia Operacional
- Exposição Antecipada: Não espere o paciente chocar para realizar uma manobra de Mattox ou Cattell-Braasch. Se há hematoma retroperitoneal em expansão, a manobra deve ser imediata.
- Regra do Pringle: Sempre tente o Pringle primeiro em hemorragias hepáticas; é um teste diagnóstico e terapêutico simultâneo.
- Mobilização Ampla: No trauma, “incisões grandes curam grandes cirurgiões”. Não tente operar através de pequenos acessos; a xifopúbica é a via de regra.
- O Packing é uma Manobra Ativa: O tamponamento deve ser firme e direcionado. Mal executado, ele apenas esconde o sangramento; bem executado, ele salva vidas.
- Respeito aos Grandes Vasos: No retroperitônio, cada milímetro de dissecção exige consciência situacional absoluta das variações anatômicas.
Conclusões Aplicadas à Prática
As manobras cirúrgicas no trauma são ferramentas de Estratégia Operacional. Elas permitem que o cirurgião converta um ambiente de caos absoluto em um cenário de controle relativo. Como Professor de Anatomia, reitero que a técnica manual é inútil sem o mapa mental da anatomia cirúrgica. Dominar a rotação visceral medial e as táticas de tamponamento hepático não é um luxo acadêmico, mas a base da sobrevivência no trauma. Na cirurgia digestiva de urgência, a simplicidade e a rapidez nas manobras de exposição são as maiores virtudes que um cirurgião pode possuir. Lembre-se: no trauma, o cirurgião é o estrategista que decide quando lutar por um órgão e quando recuar para salvar a vida.
“Na cirurgia do politrauma as coisas simples funcionam. Técnicas complexas e manobras sofisticadas permitem belas ilustrações, mas muitas vezes não permitem salvar a vida do paciente.” — Asher Hirshberg e Kenneth Mattox.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#ManobrasCirurgicas #TraumaAbdominal #CirurgiaDeTrauma #TopKnife #AnatomiaCirurgica
Drenos, Sondas e Acessos Vasculares
Sentinelas e Interfaces no Pós-Operatório
Autor: Prof. Dr. Ozimo Gama
Categoria: Cuidados Perioperatórios / Técnica Cirúrgica / Cirurgia do Aparelho Digestivo
Tempo de Leitura: 11 minutos
Introdução
Na prática da cirurgia digestiva de alta complexidade, o cuidado de drenos, sondas e acessos vasculares representa a interface física entre o planejamento tático executado no bloco operatório e a fisiologia do paciente na enfermaria ou UTI. Frequentemente negligenciados, esses dispositivos são, na verdade, sentinelas críticas que fornecem dados em tempo real sobre a homeostase e a integridade das anastomoses. Contudo, vivemos uma mudança de paradigma. Protocolos modernos de aceleração da recuperação, como o ERAS e o brasileiro Projeto ACERTO, têm demonstrado que o uso sistemático e dogmático desses dispositivos pode, em muitos casos, retardar a deambulação e aumentar as taxas de infecção. O cirurgião contemporâneo deve equilibrar a necessidade de monitorização com a premissa de “menos é mais”. Este artigo revisa os fundamentos, as indicações precisas e o manejo desses dispositivos sob uma ótica estratégica e baseada em evidências.
A Trindade da Monitorização e Suporte
A sistematização dos dispositivos cirúrgicos permite uma abordagem organizada para a vigilância do paciente operado.
1. Drenos Cirúrgicos: O “Relator” da Cavidade
O dreno não substitui uma hemostasia rigorosa nem uma anastomose bem feita, mas funciona como um sistema de alerta precoce.
- Drenos de Escoamento (Látex/Penrose): Utilizam a capilaridade. Indicados para coleções superficiais ou espaços contaminados onde se deseja uma comunicação aberta.
- Drenos de Sucção Fechada (Jackson-Pratt/Blake): Operam por pressão negativa. São os preferidos em grandes dissecções (como linfadenectomias D2 ou ressecções pancreáticas), pois permitem a quantificação exata do débito e reduzem o risco de infecção ascendente.
- O “Dreno Falante”: Na cirurgia biliar ou pancreática, a cor do efluente (bilioso, entérico ou “água de lavagem de carne”) é o diagnóstico visual de uma fístula muito antes das alterações laboratoriais.
2. Sondas: Descompressão e Aporte Metabólico
- Sonda Nasogástrica (SNG): O dogma da descompressão gástrica rotineira “até o retorno dos ruídos hidroaéreos” caiu. Atualmente, a SNG é reservada para casos de íleo paralítico prolongado, obstrução intestinal ou no pós-operatório de gdp que evoluiu com gastroparesia prolongada.
- Sondas Nasoenterais e de Ostomia (Gastrostomia/Jejunostomy): São as linhas de suprimento logístico. Em pacientes oncológicos desnutridos, a manutenção do aporte enteral precoce é o fator decisivo para a cicatrização e prevenção de translocação bacteriana.
3. Acessos Vasculares: A Logística de Suporte Vital
- Acessos Periféricos: Suficientes para cirurgias de baixo risco e hidratação de curto prazo.
- Acesso Venoso Central (CVC): Essencial em cirurgias de alta complexidade para monitorização da Pressão Venosa Central (PVC), administração de drogas vasoativas e suporte de Nutrição Parenteral Total (NPT), quando a via enteral está contraindicada.
Avaliação Diária dos Dispositivos: O que avaliar e sinais de alerta
A avaliação diária deve ser metódica, seguindo a lógica da Consciência Situacional tática. O cirurgião deve “ler” o dispositivo como um relatório de inteligência do campo de batalha.
| Dispositivo | O que Avaliar (Checklist) | Sinais de Alerta (Red Flags) |
| Drenos | Volume (curva de débito 24h), coloração, consistência, odor, local da inserção e manutenção do vácuo. | Sangue rutilante (>100ml/h); conteúdo bilioso, entérico ou purulento; perda súbita de vácuo; secreção purulenta no óstio. |
| Sondas | Posicionamento (marcação de profundidade), fixação cutânea, permeabilidade e tolerância à dieta. | Deslocamento/saída acidental; distensão abdominal com sonda “parada”; refluxo de dieta por SNG; dor súbita à infusão. |
| Acessos | Fluxo e refluxo, integridade do curativo, sinais flogísticos no trajeto venoso. | Dor à infusão; edema de membro ipsilateral; febre sem foco aparente; secreção purulenta no sítio de inserção. |
Detalhamento dos Sinais de Alerta
- Hemorragia Sentinela: Um aumento súbito de débito hemático no dreno, mesmo que em volume moderado, pode preceder uma hemorragia maciça (frequentemente por erosão enzimática de vasos no caso de fístulas pancreáticas).
- A “Parada” Suspeita: Um dreno que para de oscilar ou drenar subitamente em um paciente que mantém dor ou distensão sugere obstrução do dispositivo por coágulos ou fibrina, mascarando uma complicação interna.
- Isquemia do Estoma: Em casos de ostomias, a mucosa deve ser avaliada diariamente. Mudança para tons violáceos ou escuros indica sofrimento vascular iminente.
Aplicação na Cirurgia Digestiva
Na cirurgia digestiva brasileira, a racionalização destes dispositivos tem impactado diretamente os indicadores hospitalares. Dados do Projeto ACERTO mostram que a abolição da SNG e do dreno abdominal em colecistectomias e ressecções colorretais eletivas sem intercorrências reduziu o tempo de internação em média 25% a 30%. Entretanto, em cenários de “Teatro de Operações” hostil — como em reoperações por peritonite generalizada ou fístulas de alto débito —, o manejo correto desses dispositivos é o que permite o controle do dano (damage control). Saber posicionar um dreno de Sump para aspiração contínua ou converter uma SNG em sonda de alimentação trans-anastomótica exige um domínio da anatomia e da mecânica de fluidos que é essencial ao cirurgião digestivo.
Pontos-Chave
- Finalidade Específica: Todo dreno ou sonda deve ter uma indicação clara (diagnóstica, descompressiva ou nutritiva). Se você não sabe o que está esperando do dispositivo, ele não deve estar lá.
- Vigilância Qualitativa: O aspecto do conteúdo é tão importante quanto o volume. Mudanças súbitas na cor ou odor são sinais de alerta (red flags).
- Remoção Precoce: A permanência prolongada de drenos e sondas aumenta o risco de infecção hospitalar e formação de fístulas mecânicas.
- Fixação e Mobilidade: Dispositivos mal fixados causam dor e limitam a deambulação precoce, pilar fundamental da recuperação acelerada.
- Acesso Central: Deve ser removido assim que o paciente atinge estabilidade hemodinâmica e tolerância à via oral/enteral, minimizando o risco de sepse relacionada ao cateter.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
Drenos, sondas e acessos não são apenas apêndices do ato operatório; são ferramentas de precisão para a gestão da incerteza. O cirurgião de excelência planeja o posicionamento desses dispositivos ainda na fase de estratégia operacional, antecipando as possíveis complicações que eles deverão denunciar ou tratar. Adoção de protocolos de “desmame” precoce de dispositivos, integrada à vigilância clínica atenta, permite que o paciente cirúrgico digestivo recupere sua autonomia funcional mais rapidamente. No final, o objetivo é que o paciente deixe o hospital com o mínimo de cicatrizes — físicas e sistêmicas — de sua jornada cirúrgica.
“O dreno é um informante; quando ele para de falar, deve ser retirado. Quando ele começa a gritar, o cirurgião deve estar pronto para agir.” — Aforismo clássico da Cirurgia Abdominal.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#ClinicaCirurgica #DrenosESondas #CirurgiaDigestiva #CuidadosPosOperatorios #ProjetoACERTO
Nutrição Perioperatória no Câncer Digestivo
Estratégias para Mitigar Riscos e Otimizar Desfechos Cirúrgicos
Autor: Prof. Dr. Ozimo Gama
Categoria: Nutrição Clínica / Oncologia Cirúrgica / Cirurgia do Aparelho Digestivo
Tempo de Leitura: 12 minutos
Introdução
No âmbito da cirurgia oncológica do aparelho digestivo, a desnutrição não é apenas uma comorbidade; é um fator de risco independente e modificável que impacta diretamente a morbimortalidade operatória. Estimativas globais e dados consolidados apontam que a prevalência de desnutrição em pacientes com câncer varia entre 20% e 85%, atingindo seu ápice justamente nas neoplasias gastrointestinais e hepáticas. No Brasil, onde o Instituto Nacional de Câncer (INCA) estima para o triênio 2023-2025 cerca de 45 mil novos casos de câncer colorretal e 21 mil de câncer gástrico por ano, o cirurgião do aparelho digestivo assume o papel de “arquiteto metabólico”. Compreender que a intervenção nutricional deve começar no momento do diagnóstico — e não apenas no pós-operatório — é fundamental para reduzir taxas de infecção, tempo de internação e custos hospitalares. Este artigo revisa os fundamentos da nutrição perioperatória sob a luz das evidências mais recentes.
Triagem, Sarcopenia e Imunonutrição
1. Triagem e Avaliação Nutricional
A identificação do risco nutricional deve ser universal e precoce. Ferramentas validadas como o MUST (Malnutrition Universal Screening Tool) e o NRS-2002 (Nutritional Risk Screening) são fundamentais para estratificar o paciente. Mais do que a simples perda de peso, devemos atentar para a Sarcopenia (perda de massa e função muscular). Atualmente, a análise da área muscular ao nível da terceira vértebra lombar (L3) em exames de Tomografia Computadorizada é considerada o padrão-ouro para o diagnóstico de miopenia em pacientes oncológicos, permitindo identificar a “obesidade sarcopênica”, onde o excesso de tecido adiposo mascara a depleção muscular proteica.
2. Abreviatura do Jejum e Carga de Carboidratos
O dogma do “NPO após a meia-noite” foi definitivamente substituído por protocolos de aceleração da recuperação, como o ERAS (Enhanced Recovery After Surgery) e o brasileiro Projeto ACERTO. A recomendação atual permite a ingestão de líquidos claros até 2 horas antes da indução anestésica. A oferta de uma bebida rica em carboidratos (maltodextrina a 12,5%) nesse período reduz a resistência insulínica pós-operatória, preserva a massa magra e diminui o desconforto e a ansiedade do paciente, sem aumentar o risco de aspiração broncopulmonar.
3. Imunonutrição
O uso de fórmulas imunomoduladoras — enriquecidas com Arginina, Ácidos Graxos Ômega-3 e Nucleotídeos — tem demonstrado benefícios robustos. A suplementação pré-operatória por 5 a 7 dias em pacientes candidatos a grandes cirurgias digestivas está associada a uma redução significativa nas complicações infecciosas e na deiscência de anastomoses. A arginina, especificamente, atua na melhora da resposta das células T e na síntese de colágeno, essencial para a cicatrização.
Aplicação na Cirurgia Digestiva: O Contexto da Reabilitação
Na prática da cirurgia digestiva de alta complexidade, a nutrição perioperatória é o pilar central da Pré-habilitação. Pacientes com câncer de pâncreas ou esôfago, frequentemente sarcopênicos ao diagnóstico, apresentam melhora drástica nos desfechos quando submetidos a um programa que associa suporte nutricional hiperproteico a exercícios físicos e suporte psicológico antes do ato operatório. A via de escolha deve ser sempre a enteral, preservando a barreira mucosa intestinal e evitando a translocação bacteriana. A Nutrição Parenteral Total (NPT) deve ser reservada para casos de obstrução intestinal completa ou quando a meta calórico-proteica não pode ser atingida por via digestiva em 7 a 10 dias.
Pontos-Chave
- Fator Modificável: A desnutrição é um dos poucos riscos perioperatórios que o cirurgião pode intervir ativamente para melhorar o prognóstico oncológico.
- Triagem Obrigatória: Nenhum paciente oncológico deve ser operado sem uma avaliação de risco nutricional prévia (NRS-2002 ou similar).
- Sarcopenia Oculta: O IMC isolado é insuficiente; a avaliação da massa muscular via imagem (TC em L3) é decisiva para o prognóstico.
- Jejum Moderno: Líquidos claros com carboidratos até 2h antes da cirurgia é o padrão de segurança e conforto metabólico.
- Imunonutrição: Suplementação de 5 a 7 dias pré-operatórios reduz complicações infecciosas em grandes ressecções GI.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A excelência técnica no bloco operatório — a anastomose perfeita ou a linfadenectomia D2 rigorosa — perde eficácia se o terreno biológico do paciente estiver exaurido. O cirurgião do aparelho digestivo moderno deve prescrever nutrição com o mesmo rigor com que planeja a tática operatória. Integrar os protocolos de nutrição perioperatória à rotina clínica não apenas salva vidas, mas devolve ao paciente a capacidade de enfrentar o tratamento oncológico complementar com maior resiliência. A nutrição deixou de ser um cuidado adjuvante para se tornar uma etapa mandatória da estratégia cirúrgica oncológica.
“A nutrição é o braço direito da medicina e o esquerdo da cirurgia; sem ela, o bisturi mais brilhante corta apenas a carne, mas não cura o organismo.” — Adaptado dos princípios de sustentação vital em cirurgia.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#NutricaoPerioperatória #CirurgiaDigestiva #OncologiaCirurgica #ProjetoACERTO #SegurancaDoPaciente
Princípios Estratégicos Militares
Uma Abordagem Operacional Aplicada às Decisões Complexas em Cirurgia Digestiva
Autor: Prof. Dr. Ozimo Gama
Categoria: Gestão de Risco / Técnica Cirúrgica / Educação Médica Continuada
Tempo de Leitura: 15 minutos
Introdução
No campo da cirurgia do aparelho digestivo de alta complexidade, o cirurgião não é apenas um técnico de mãos habilidosas; ele é, essencialmente, um estrategista. A sala de operações, tal como o campo de batalha, é um ambiente de incerteza, pressão temporal e consequências irrevogáveis. A transposição dos princípios da estratégia militar para o planejamento e execução cirúrgica não é uma mera analogia intelectual, mas uma ferramenta de gestão de risco fundamental para a sobrevivência do paciente. Historicamente, a cirurgia e a guerra caminharam juntas. Contudo, a evolução contemporânea exige que o cirurgião digestivo vá além da tática (o ato de costurar ou dissecar) e domine a Arte Operacional. Conforme os ensinamentos de pensadores como Clausewitz, Sun Tzu e Liddell Hart, a vitória — no nosso caso, a cura e a recuperação plena do doente — depende da capacidade de antecipar o “atrito”, gerenciar a “névoa da guerra” anatômica e aplicar a força (técnica cirúrgica) no ponto de maior impacto com o menor dano colateral. Este artigo visa sistematizar como os pilares da estratégia militar podem ser aplicados às decisões complexas na nossa especialidade.
A Filosofia Estratégica no Teatro de Operações Abdominais
A estratégia, conforme definida por Lawrence Freedman, é a “arte de criar poder” para alcançar objetivos em cenários de conflito. Na cirurgia, o conflito se dá contra a patologia, a inflamação e a variabilidade anatômica.
1. Clausewitz e a “Fricção” Cirúrgica
Carl von Clausewitz, em sua obra clássica Da Guerra, introduziu o conceito de fricção: “Tudo na guerra é muito simples, mas a coisa mais simples é difícil”. Na cirurgia digestiva, a fricção é o sangramento inesperado, a falha de um grampeador, a fadiga da equipe ou a descoberta de uma invasão vascular não detectada na imagem. O cirurgião estrategista deve planejar contando com a fricção. Isso significa ter planos de contingência (Planos B e C) prontos. Se a “missão” original (uma duodenopancreatectomia com preservação pilórica) encontrar uma invasão tumoral da veia porta, o cirurgião deve estar taticamente preparado para a reconstrução vascular, e não ser pego de surpresa.
2. Sun Tzu e a Inteligência (Reconhecimento)
“Se você conhece o inimigo e conhece a si mesmo, não precisa temer o resultado de cem batalhas”. A inteligência militar é o equivalente à nossa propedêutica armada. O uso exaustivo de exames de imagem de alta definição (TC e RM com reconstrução 3D) e biomarcadores é o reconhecimento aéreo do terreno. Operar um tumor sem o estadiamento molecular ou anatômico completo é como enviar tropas para um território desconhecido sem mapas: é um convite ao desastre.
3. Liddell Hart e a “Abordagem Indireta”
B. H. Liddell Hart defendia que o objetivo da estratégia é deslocar o inimigo, não atacá-lo onde ele é mais forte. Na cirurgia oncológica digestiva, a Neoadjuvância (quimioterapia ou radioterapia pré-operatória) é a aplicação máxima deste princípio. Ao invés de um ataque direto a um tumor “borderline” ou localmente avançado — o que poderia resultar em uma cirurgia mutilante e de alto risco —, utilizamos a estratégia indireta para reduzir o tumor, facilitando uma ressecção mais segura e preservadora de órgãos (como no GIST ou no câncer de reto).
Aplicação na Cirurgia Digestiva: Casos Táticos
O Planejamento na Oncologia Digestiva
No Brasil, o cenário epidemiológico é desafiador. Segundo dados do INCA (Estimativa 2023-2025), o câncer colorretal é o segundo mais frequente em homens e mulheres (excetuando pele não melanoma), com cerca de 45.630 novos casos anuais. O câncer gástrico segue com mais de 21.000 casos. Perante esses números, o cirurgião atua em uma campanha de longa duração. A estratégia exige que a Linfadenectomia (D2 no estômago, por exemplo) seja executada com precisão militar. Deixar território inimigo (linfonodos comprometidos) para trás é garantir a contraofensiva da doença (recidiva).
A Disciplina da “Visão Crítica de Segurança” (CVS)
Na colecistectomia laparoscópica, a lesão da via biliar é o equivalente a um “fogo amigo”. A aplicação do CVS de Strasberg é um protocolo de disciplina operacional. O cirurgião é obrigado a completar três critérios objetivos (limpeza do triângulo de Calot, separação do leito hepático e identificação das duas únicas estruturas) antes de “disparar” os clipes. É a vitória da doutrina sobre a pressa.
O “Bail-out” como Retirada Estratégica
Na estratégia militar, saber quando recuar para preservar as tropas é um sinal de liderança superior, não de fraqueza. Na cirurgia complexa, quando a anatomia está “congelada” ou a instabilidade hemodinâmica do paciente é crítica, o cirurgião deve optar por procedimentos de saída (bail-out), como a colecistectomia subtotal ou a cirurgia de controle de danos (damage control). O objetivo estratégico é a sobrevivência do paciente, não a conclusão de uma técnica específica a qualquer custo.
Pontos-Chave para a Prática Diária
- Unidade de Comando: O cirurgião principal deve liderar a equipe multidisciplinar de forma clara, garantindo que anestesistas, enfermeiros e auxiliares conheçam os objetivos da missão.
- Simplicidade Operacional: Planos excessivamente complexos falham sob pressão. Busque a tática mais direta e segura para alcançar o objetivo oncológico ou funcional.
- Logística de Retaguarda: Garanta que o “armamento” (grampeadores, energia, fios) e o suporte (Banco de Sangue, UTI) estejam prontos antes do início da operação.
- Consciência Situacional: Mantenha a calma durante a “fricção”. Se o sangramento ocorrer, a primeira manobra é o controle (compressão), seguida da análise tática da solução, nunca o pânico.
- Debriefing (Análise Pós-Ação): Cada cirurgia deve ser revisada pela equipe para identificar falhas táticas e melhorar os processos para o próximo “combate”.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A excelência na cirurgia digestiva de alta complexidade exige uma mudança de mentalidade. Devemos abandonar a visão puramente técnica e abraçar a visão estratégica. O bisturi é apenas o instrumento; a inteligência estratégica é o que dita onde e como ele deve ser usado. Integrar os princípios militares de reconhecimento, disciplina operacional e abordagem indireta permite que o cirurgião navegue com segurança por anatomias distorcidas e biologias agressivas. Como líderes no teatro de operações do abdome, nosso dever é converter a incerteza em resultados previsíveis, garantindo que cada decisão complexa seja fundamentada em princípios que resistiram ao teste do tempo, tanto nos campos de batalha quanto nos centros cirúrgicos.
“Tudo na guerra é simples, mas a coisa mais simples é difícil. As dificuldades se acumulam e acabam produzindo um tipo de atrito que é impensável para quem não presenciou a guerra.” — Carl von Clausewitz. Na cirurgia digestiva, o atrito é a realidade; a estratégia é a nossa única defesa.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#EstrategiaCirurgica #CirurgiaDigestiva #GestaoDeRisco #OncologiaCirurgica #DecisaoMedica
Colecistectomia: O perigo da falsa “Simplicidade”
A cirurgia de retirada da vesícula biliar é a mais comum da especialidade digestiva, mas a percepção de que é um procedimento “simples” esconde riscos catastróficos que exigem uma nova cultura de segurança.
Todos conhecemos alguém que já “tirou a vesícula”. No Brasil, apenas pelo Sistema Único de Saúde (SUS), são realizadas mais de 200 mil colecistectomias anualmente. A introdução da técnica laparoscópica — a cirurgia por vídeo — na década de 1990, trouxe benefícios inquestionáveis: cicatrizes mínimas, menos dor e um retorno quase imediato ao trabalho. No entanto, essa aura de facilidade criou uma armadilha perigosa tanto para pacientes quanto para cirurgiões: a banalização de um procedimento que, em segundos, pode transformar uma patologia benigna em uma tragédia crônica.
O paradoxo é alarmante. Apesar do avanço tecnológico e da vasta experiência acumulada em três décadas, a incidência de lesão da via biliar (LVB) — o dano aos canais que transportam a bile do fígado ao intestino — permanece obstinadamente estável, ocorrendo em cerca de 0,3% a 0,5% dos casos por vídeo. Surpreendentemente, esta taxa é superior à da antiga era da cirurgia aberta. O que a literatura médica qualifica como a “lesão clássica” não é, em regra, fruto de imperícia manual ou falta de conhecimento anatômico, mas sim de uma “ilusão de percepção visual”.
Estudos seminais indicam que 97% das lesões ocorrem porque o cérebro do cirurgião, enganado pela tração e pela inflamação, identifica o ducto biliar principal como se fosse o ducto cístico. É um erro cognitivo, não técnico. Por isso, a colecistectomia não pode ser tratada como um procedimento de “entrada” ou de menor importância. Ela exige o que chamamos de Visão Crítica de Segurança (CVS), uma doutrina sistematizada pelo cirurgião Steven Strasberg que obriga a identificação cabal de estruturas antes de qualquer corte. Sem CVS, não há segurança; há apenas sorte.
A banalização também se manifesta na resistência em adotar as chamadas “manobras de resgate” (bailout). Diante de uma vesícula hostil, com inflamação severa que impede a visão clara da anatomia, o cirurgião prudente deve ter a humildade e a sabedoria de interromper a dissecação clássica. Técnicas como a colecistectomia subtotal — onde parte do órgão é deixada para preservar a via biliar — devem ser vistas como um sinal de maturidade clínica e “sabedoria cirúrgica”, e não como um fracasso. O objetivo final nunca deve ser “tirar a vesícula a qualquer custo”, mas sim “não lesar o paciente”.
O custo humano de uma lesão biliar é imenso. O paciente, outrora saudável, pode tornar-se o que a literatura tragicamente apelida de “aleijado biliar”, enfrentando múltiplas reoperações, internações prolongadas e o risco de cirrose biliar secundária. O impacto médico-legal e o custo para o sistema de saúde são igualmente devastadores.
Felizmente, estamos em uma nova fronteira. O uso de fluorescência com verde de indocianina (ICG), que permite “enxergar” os canais biliares através do tecido, e a integração da Inteligência Artificial como um “segundo par de olhos” no monitor, prometem reduzir a margem para o erro humano. Todavia, a tecnologia sozinha não basta.
É preciso consolidar uma Cultura de Segurança em Colecistectomia (COSIC). Isso envolve desde a documentação fotográfica rigorosa dos passos cirúrgicos até a coragem de pedir ajuda ou converter o procedimento quando a anatomia se torna um enigma. A colecistectomia laparoscópica é uma vitória da medicina moderna, mas sua eficácia depende do reconhecimento de que, sob sua aparente simplicidade, reside uma das operações mais perigosas da cirurgia abdominal. Respeitar essa complexidade é o primeiro passo para proteger a vida de milhares de brasileiros que passam por nossas mesas de operação todos os dias.
Prof. Dr. Ozimo Gama | Cirurgião Digestivo
Planejamento da Cirurgia Digestiva de Alta Complexidade
Adaptação Estratégica das Operações Militares Especiais
Autor: Prof. Dr. Ozimo Gama Categoria: Gestão de Risco / Técnica Cirúrgica / Educação Médica
Tempo de Leitura: 12 minutos
Introdução
No campo da cirurgia do aparelho digestivo de alta complexidade — como em duodenopancreatectomias, ressecções hepáticas maiores ou exenterações pélvicas —, o cirurgião não entra apenas em uma sala de operações; ele adentra um “Teatro de Operações”. A intersecção entre a ciência médica e a estratégia militar não é apenas metafórica; é uma aplicação prática de princípios de elite para mitigar riscos catastróficos. A filosofia das Operações Especiais (SpecOps) foca na obtenção de superioridade relativa em ambientes hostis e incertos. Para o cirurgião digestivo, o abdome “congelado” por múltiplas cirurgias ou por invasão neoplásica é esse ambiente. Este artigo propõe uma transposição da doutrina tática para o planejamento cirúrgico, visando elevar a taxa de sucesso da “missão” e a segurança do paciente.

A Doutrina das Operações Especiais na Cirurgia
A estratégia de operações de elite baseia-se em pilares como Inteligência, Simplicidade, Repetição e Velocidade. Transpostos para a prática cirúrgica de um especialista em Cirurgia do Aparelho Digestivo, esses conceitos transformam a condução do caso:
1. Inteligência (Propedêutica e Reconhecimento)
Em Operações Especiais, nenhuma missão avança sem o reconhecimento profundo do terreno. Na cirurgia digestiva complexa, isso se traduz no uso exaustivo de biomarcadores e exames de imagem de última geração, como a Tomografia Computadorizada (TC) e a Ressonância Magnética (RM) com reconstrução tridimensional. No Brasil, onde o volume de câncer gástrico e colorretal é expressivo, a identificação precisa das relações vasculares e da invasão de órgãos adjacentes é o “reconhecimento aéreo” que define a ressecabilidade.
2. Simplicidade e Tática
A estratégia militar ensina que planos excessivamente complexos falham sob pressão. O planejamento deve ser cirurgicamente simples: identificar as estruturas vitais (pontos de controle) e isolar o alvo (tumor ou processo inflamatório). A técnica de “no-touch” em tumores como o GIST é uma aplicação tática de proteção do perímetro para evitar a sementeira peritoneal.
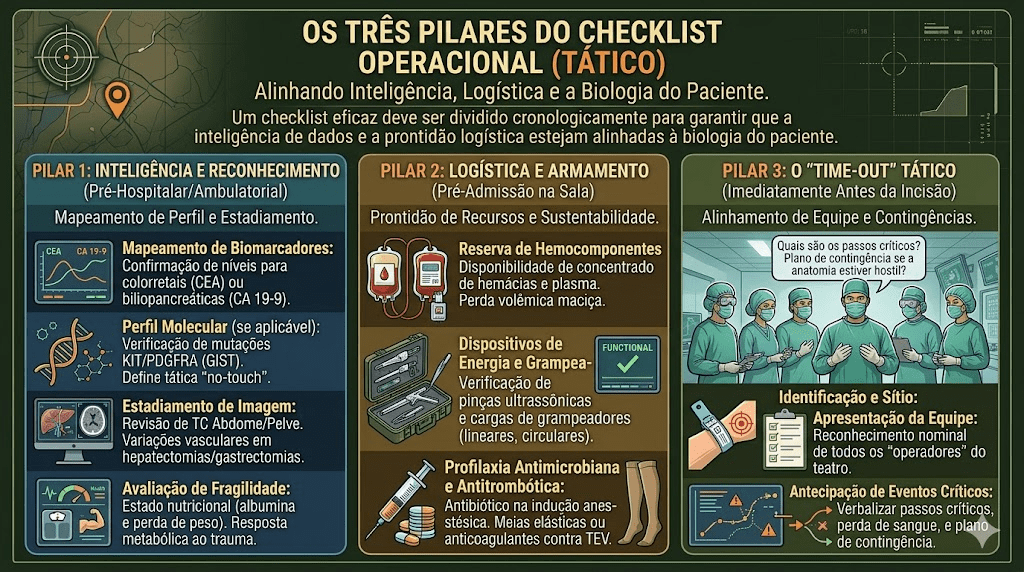
CHECKLIST TÁTICO
Um checklist eficaz deve ser dividido cronologicamente para garantir que a inteligência de dados e a prontidão logística estejam alinhadas à biologia do paciente.
2.1. Inteligência e Reconhecimento (Pré-Hospitalar/Ambulatorial)
- Mapeamento de Biomarcadores: Confirmação dos níveis de CEA para tumores colorretais ou CA 19-9 para lesões biliopancreáticas.
- Perfil Molecular (se aplicável): Verificação de mutações KIT ou PDGFRA em casos de suspeita de GIST para definição de tática de ressecção “no-touch”.
- Estadiamento de Imagem: Revisão da Tomografia de abdome e pelve para identificar variações anatômicas vasculares, cruciais em hepatectomias ou gastrectomias.
- Avaliação de Fragilidade: Checagem do estado nutricional (albumina e perda de peso) para prever a resposta metabólica ao trauma.
2.2. Logística e Armamento (Pré-Admissão na Sala)
- Reserva de Hemocomponentes: Garantia de disponibilidade de concentrado de hemácias e plasma, especialmente em casos com potencial de perda volêmica maciça.
- Dispositivos de Energia e Grampeamento: Verificação da funcionalidade de pinças ultrassônicas e disponibilidade de cargas para grampeadores lineares e circulares.
- Profilaxia Antimicrobiana e Antitrombótica: Confirmação da administração do antibiótico na indução anestésica e início das medidas mecânicas (meias elásticas) ou farmacológicas contra o tromboembolismo venoso.
2.3. O “Time-out” Tático (Imediatamente Antes da Incisão)
- Identificação e Sítio: Confirmação verbal do nome do paciente, procedimento e local da incisão (lateralidade).
- Apresentação da Equipe: Reconhecimento nominal de todos os “operadores” do teatro (cirurgiões, anestesista, instrumentador e circulante).
- Antecipação de Eventos Críticos: O cirurgião deve verbalizar: “Quais são os passos críticos? Qual a perda de sangue esperada? Qual o plano de contingência se a anatomia estiver hostil?”
3. Ensaio e Simulação (O “Sand Table”)
Antes de uma incursão, equipes de elite realizam ensaios exaustivos. O cirurgião moderno deve realizar o “ensaio mental” ou simulação computacional, antecipando cada passo da dissecção e as possíveis contraofensivas da patologia (ex: sangramento massivo inadvertido).
O Plano de Contingência
A aplicação prática dessa estratégia exige que o cirurgião atue com uma mentalidade de comando:
- Superioridade Relativa: Alcançar o controle vascular antes da manipulação da massa tumoral. Em cirurgias hepáticas, a manobra de Pringle é um exemplo de interdição logística temporária para garantir a segurança no terreno principal.
- Gestão da “Névoa da Guerra”: Durante a operação, surgem achados imprevistos (aderências firmes, variações anatômicas). O cirurgião deve possuir a Flexibilidade Tática para alterar a via de acesso ou realizar procedimentos de saída (bail-out), como a colecistectomia subtotal, se a anatomia se tornar inidentificável.
- Estatísticas e Realidade Brasileira: No Brasil, a oncologia cirúrgica digestiva enfrenta o desafio do diagnóstico tardio. Operar pacientes com doenças avançadas exige um planejamento que considere a Logística de Resgate — a capacidade da equipe multidisciplinar e da UTI em manejar a resposta metabólica ao trauma sistêmico.
Pontos-Chave para a Prática Diária
- Unidade de Comando: O cirurgião principal deve liderar a equipe multidisciplinar de forma clara e direta no bloco.
- Checklist Tático: Não é apenas uma burocracia; é a verificação de que todo o “armamento” (grampeadores, energia avançada, fios específicos) está funcional e disponível.
- Consciência Situacional: Avaliar constantemente o tempo cirúrgico e o estado hemodinâmico do paciente, sabendo quando recuar para preservar a vida.
- Análise Pós-Ação: Revisar o vídeo da cirurgia ou o relato cirúrgico detalhado para identificar falhas táticas e melhorar na próxima missão.
Conclusões Aplicadas
A cirurgia digestiva de alta complexidade é um exercício de ética e estratégia. Ao incorporar a disciplina e o planejamento rigoroso das Operações Militares Especiais, o cirurgião deixa de ser um mero executor técnico para se tornar um estrategista da cura. A missão nunca é apenas remover um órgão; a missão é restaurar a homeostase e a dignidade do paciente com a menor agressão possível. Em um país com a diversidade epidemiológica do Brasil, onde o câncer colorretal é o segundo mais comum em mulheres e o terceiro em homens, a precisão tática não é um diferencial — é uma obrigação de sobrevivência.
“A vitória pertence àqueles que mais acreditam nela e a planejam pelo tempo mais longo.” — Napoleão Bonaparte, cujas táticas de organização e logística ainda ecoam na estruturação de grandes serviços cirúrgicos mundiais.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
1º Dia de Pós-Operatório
(PO1): O Mapa de Navegação na Cirurgia Digestiva Complexa
Autor: Prof. Dr. Ozimo Gama
Categoria: Cuidados Perioperatórios / Semiologia Cirúrgica / Cirurgia do Aparelho Digestivo
Tempo de Leitura: 12 minutos
Introdução
O término de uma cirurgia digestiva de alta complexidade — seja uma duodenopancreatectomia, uma gastrectomia total oncológica ou uma hepatectomia maior — não marca o fim da batalha, mas o início de uma nova e crítica fase: a recuperação metabólica e fisiológica. Para o cirurgião, o Primeiro Dia de Pós-Operatório (PO1), geralmente transcorrido em uma Unidade de Terapia Intensiva (UTI), é as “24 horas de ouro”. Neste período, o organismo do paciente está imerso na resposta endócrino-metabólica ao trauma (REMIT). A linha que separa a evolução fisiológica esperada da complicação catastrófica é extremamente tênue. Para o estudante de medicina e o residente de cirurgia, dominar a semiologia cirúrgica do PO1 não significa apenas olhar para exames laboratoriais ou monitores multiparamétricos; significa resgatar a essência da beira do leito. A mão que opera deve ser a mesma mão que examina. Neste artigo, detalharemos o que buscar, o que tocar e como interpretar os sinais de alerta no paciente cirúrgico grave.
1. Avaliação Sistêmica: O Paciente como um Todo
A avaliação do abdome começa muito antes de tocar no abdome do paciente. O exame inicia-se na porta do quarto.
- Estado Neurológico e Nível de Consciência: Um paciente agitado, confuso ou torporoso no PO1 não está simplesmente “sob efeito residual da anestesia”. O delirium hipo ou hiperativo pode ser o primeiro sinal de hipóxia, hipercapnia ou choque (hipoperfusão cerebral). A avaliação do despertar e da orientação (escala de Glasgow) é inegociável.
- Perfusão Periférica e Diurese: A pele fala. Extremidades frias, pegajosas e tempo de enchimento capilar lentificado (> 3 segundos) indicam má perfusão tecidual. Simultaneamente, o débito urinário é a “janela dos rins” para a volemia. A oligúria (< 0,5 ml/kg/h) no PO1 de uma grande cirurgia exige diagnóstico diferencial imediato entre hipovolemia (sangramento/desidratação) e falência renal aguda.
- Sinais Vitais (O Radar do Cirurgião):
- Taquicardia: É o sinal vital mais subestimado. Nunca atribua uma taquicardia (> 100 bpm) apenas à “dor” ou “ansiedade” sem antes descartar as duas maiores ameaças do PO1: Hemorragia e Vazamento Anastomótico Precoce.
- Febre: Temperaturas de até 38°C nas primeiras 24-48h são frequentemente decorrentes da atelectasia pulmonar ou da resposta inflamatória fisiológica ao trauma. Infecções de sítio cirúrgico são raras no PO1. Contudo, febre alta acompanhada de instabilidade hemodinâmica exige descartar lesão inadvertida de alça intestinal ou infecção necrosante.
2. O Exame Físico Abdominal Direcionado
A semiologia do abdome operado difere da semiologia clássica. A dor incisional confunde a avaliação de irritação peritoneal. O cirurgião deve ser sutil e metódico.
- Inspeção: O abdome está distendido? Uma distensão progressiva e tensa no PO1 levanta forte suspeita de hemoperitônio (sangramento interno) ou dilatação gástrica aguda.
- Palpação e Percussão: A palpação deve começar distante da incisão cirúrgica. O objetivo é diferenciar a dor da ferida (esperada) da dor peritonítica profunda (sinal de alarme). A percussão dolorosa (sinal de irritação peritoneal) generalizada em uma cirurgia que deveria ser limpa é um grito de socorro do abdome.
- Ausculta: O íleo adinâmico é fisiológico nas primeiras 24 a 72 horas após grande manipulação entérica. A ausência de ruídos hidroaéreos é esperada, mas a sua presença precoce é um excelente prognóstico.
- Ferida Operatória: Inspecionar o curativo. Um sangramento que encharca o curativo repetidamente nas primeiras horas exige exploração. Observar sinais precoces de isquemia das bordas da ferida ou hematoma expansivo.
3. A Janela da Cavidade: Drenos e Estomas
Os dispositivos deixados no paciente são os relatores da cavidade abdominal. Eles devem ser examinados com rigor obsessivo.
- Drenos Cavitários: Não anote apenas o “volume”, mas a qualidade do efluente.
- Sero-hemático: Esperado no PO1 de grandes dissecções (ex: linfadenectomia).
- Sangue vivo (Hemático): Um débito hemático > 100 a 150 ml/hora no dreno tubular é indicativo de hemorragia ativa (falha na hemostasia, deslizamento de ligadura arterial).
- Bilioso, Entérico ou Purulento: É indicativo de desastre. Uma fístula entérica/biliar no PO1 geralmente significa falha técnica da anastomose ou lesão iatrogênica não reconhecida no intraoperatório.
- Estomas (Colostomia/Ileostomia): A vitalidade do estoma deve ser atestada no PO1. A mucosa deve ser de coloração vermelho-rósea e úmida. Uma coloração violácea, pálida ou enegrecida é diagnóstico de isquemia ou necrose do estoma, exigindo reintervenção cirúrgica de urgência.
4. Sinais de Alerta para Eventos Adversos
O cirurgião do aparelho digestivo deve estar programado para identificar padrões sindrômicos que indicam catástrofes:
- A Síndrome Hemorrágica: Taquicardia progressiva + hipotensão + palidez + oligúria + queda abrupta de Hemoglobina/Hematócrito + dreno com saída de sangue rutilante. Conduta: Ressuscitação volêmica, transfusão maciça e reoperação hemostática imediata.
- A Síndrome Isquêmica Intestinal: Dor abdominal atroz, refratária a doses elevadas de opioides, muitas vezes desproporcional aos achados do exame físico abdominal, associada a acidose metabólica grave (lactato elevado). Comum após revascularizações mesentéricas ou cirurgias com baixo débito prolongado.
- A Síndrome da Deiscência Catastrófica: Instabilidade hemodinâmica súbita, taquicardia inexplicada e abdome em tábua (se não houver analgesia peridural que mascare o quadro).
Pontos-Chave para a Prática Diária do Residente
- A dor nunca é subestimável: Se a dor operatória mudou de padrão, piorou subitamente ou não cede à analgesia potente, presuma uma complicação (isquemia, perfuração, sangramento) até prova em contrário.
- O Dreno fala a verdade: Conheça a localização exata de cada dreno que você deixou na cavidade. Um dreno próximo a uma anastomose pancreática que apresenta débito de “água de lavagem de carne” espessa pode ser o prenúncio de uma fístula.
- O Exame é Seriado: A semiologia no PO1 não é uma “fotografia” tirada no round da manhã; é um “filme”. Avaliar o paciente de manhã, à tarde e à noite é a única forma de perceber a deterioração clínica antes do choque irreversível.
- Laboratório não substitui o dedo: Uma tomografia ou um exame de sangue não substituem a mão do cirurgião no abdome e a reavaliação clínica constante.
Conclusões Aplicadas
A semiologia cirúrgica no primeiro dia de pós-operatório é uma dança complexa entre a ciência da fisiologia e a arte da observação clínica. Na cirurgia digestiva de alta complexidade, o sucesso oncológico e técnico obtido no centro cirúrgico precisa ser consolidado na UTI através da vigilância implacável do cirurgião. Delegar a avaliação do paciente exclusivamente aos intensivistas, eximindo-se de tocar o abdome e checar os drenos, é o primeiro passo para o fracasso. A intuição do cirurgião que operou, baseada numa semiologia apurada, é insubstituível na detecção precoce do evento adverso, permitindo a transição de um “resgate heroico” para um “manejo planejado”.
“Aumente o seu poder de observação… Veja a cor da pele, sinta a pulsação, observe a respiração. Não delegue as suas mãos e os seus olhos para as máquinas. O diagnóstico precoce das complicações cirúrgicas está escrito no rosto e no abdome do paciente, para quem tiver olhos para ler.” — Adaptação dos princípios semiológicos de John B. Murphy, pioneiro da cirurgia abdominal moderna.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#SemiologiaCirurgica #PosOperatorio #CirurgiaDigestiva #CuidadosIntensivos #ResidenciaMedica
Fundamentos da Oncologia Digestiva
Epidemiologia, Marcadores Tumorais e Princípios Cirúrgicos
Por: Prof. Dr. Ozimo Gama
1. Introdução: O Panorama da Oncologia Digestiva
O aumento progressivo da expectativa de vida e o consequente envelhecimento populacional trouxeram consigo um incremento significativo na incidência das neoplasias malignas. Na prática clínica e acadêmica, observamos que, embora os avanços na propedêutica diagnóstica e os sistemas de triagem tenham evoluído, uma parcela considerável de pacientes ainda é diagnosticada em estágios avançados. Esse cenário impõe desafios terapêuticos complexos, elevando a morbimortalidade e os custos para o sistema de saúde. O diagnóstico precoce, idealmente em fases pré-neoplásicas, permanece como o objetivo primordial para a melhoria dos desfechos clínicos. De acordo com os dados mundiais do projeto GLOBOCAN (WHO, 2008), a estimativa global superou os 12 milhões de novos casos de câncer, com mais de 7 milhões de óbitos anuais. No cenário brasileiro, as projeções do Instituto Nacional do Câncer (INCA, 2012) ratificam a magnitude do problema no aparelho digestivo, com destaque para as seguintes incidências:
- Câncer Colorretal: 30.140 casos.
- Câncer de Estômago: 20.090 casos.
- Câncer de Esôfago: 10.420 casos.
2. Desenvolvimento: Biologia Tumoral e Biomarcadores
Os biomarcadores, ou marcadores tumorais, são estruturas moleculares ou teciduais que permitem prever o comportamento biológico de uma neoplasia. No contexto da oncologia digestiva, sua utilidade reside primordialmente na avaliação prognóstica e no seguimento pós-operatório para detecção de recidivas. É imperativo ressaltar que, devido às limitações de sensibilidade e especificidade — especialmente em fases precoces —, a maioria desses marcadores não é indicada para o rastreamento (screening) populacional.
Abaixo, elenco os principais marcadores, seus valores referenciais superiores (conforme Tabela 18.2) e nuances clínicas:
- CEA (Antígeno Carcinoembrionário): Marcador fundamental no carcinoma colorretal. Valor referencial: < 3 ng/mL. Deve-se atentar que níveis elevados podem ser encontrados em tabagistas, o que exige cautela na interpretação.
- AFP (Alfa-fetoproteína): Crucial no diagnóstico do carcinoma hepatocelular. Valor referencial: < 9 U/L. Sua aplicação em screening é aceita apenas em grupos de alto risco, como pacientes com cirrose hepática ou portadores crônicos de Hepatite B e C. Gestação e doenças hepáticas não neoplásicas são causas comuns de falso-positivos.
- CA 19-9: Utilizado na propedêutica de tumores de pâncreas e vias biliares. Valor referencial: < 37 U/mL.
- CA 72-4: Marcador com alta especificidade para o carcinoma gástrico, útil no monitoramento da resposta terapêutica. Valor referencial: < 4 U/mL.
- SCCA (Squamous Cell Carcinoma Antigen): Indicado para o seguimento de carcinomas de células escamosas, notadamente no esôfago e canal anal. Valor referencial: < 1,5 mg/L.
3. GIST e Tumores Neuroendócrinos (TNE)
GIST (Tumor Estromal Gastrointestinal)
O GIST é a neoplasia mesenquimal mais comum do trato digestório, originando-se das células intersticiais de Cajal. Sua patogenia molecular é marcada por mutações nos genes c-kit (proteína KIT/CD117, presente em cerca de 95% dos casos) e PDGFRA (em 5-10% dos casos). O potencial de malignidade e o risco de comportamento agressivo são determinados tecnicamente pelo tamanho tumoral e pelo índice mitótico (Tabela 19.2). O advento do Mesilato de Imatinibe revolucionou o tratamento como terapia-alvo eficaz para ambas as mutações citadas.
TNE (Tumores Neuroendócrinos)
Os TNE constituem um grupo heterogêneo classificado conforme o índice mitótico e a expressão do Ki-67. A diferenciação é vital para o prognóstico:
- G1 e G2: Tumores Neuroendócrinos bem diferenciados.
- G3: Denominados Carcinomas Neuroendócrinos (NEC), caracterizados por comportamento altamente agressivo e Ki-67 > 20%.
A Síndrome Carcinoide (rubor facial, diarreia e valvulopatia) é mediada pela serotonina. O marcador pan-neuroendócrino de eleição é a Cromogranina A, uma proteína presente nos grânulos secretórios das células neuroendócrinas.
4. Aplicação na Cirurgia Digestiva
A ressecção cirúrgica é o pilar do tratamento curativo. O objetivo técnico é a obtenção de margens microscopicamente negativas (Ressecção R0).
No manejo do GIST, a estratégia cirúrgica possui particularidades: a linfadenectomia de rotina não é indicada devido à raridade da disseminação linfática. A técnica deve priorizar a ressecção em bloco com uma margem de segurança de 1 cm, garantindo a integridade da pseudocápsula tumoral para evitar a disseminação peritoneal por ruptura.
Para os Tumores Neuroendócrinos, a conduta é multidisciplinar. Em pacientes com doença avançada e sintomas hormonais intratáveis, a cirurgia citorredutora (debulking) pode ser empregada para controle clínico e ganho de sobrevida.
5. Pontos-Chave para a Prática Médica
Para a formação técnica de residentes e pós-graduandos, os seguintes conceitos são fundamentais:
- Estadiamento TNM: Permanece como o principal determinante prognóstico e guia soberano para indicação de terapias neoadjuvantes e adjuvantes.
- Seguimento Pós-operatório: O CEA é o marcador padrão no seguimento do câncer colorretal, mas sua interpretação deve considerar o status tabágico do paciente.
- Biologia Molecular do GIST: A análise das mutações KIT e PDGFRA é indispensável para o planejamento da terapia biológica com Imatinibe.
- Margens Cirúrgicas: No GIST, a margem R0 com 1 cm de segurança é o parâmetro técnico ideal para evitar recidivas locais.
- Impacto do Estágio na Sobrevida: A disparidade na sobrevida de 5 anos reforça a urgência do diagnóstico precoce. No Câncer de Esôfago, por exemplo, a sobrevida cai drasticamente de 37% em doença localizada para apenas 3% em casos metastáticos (Tabela 17.3).
6. Considerações Finais
A oncologia digestiva moderna exige que o cirurgião domine conhecimentos que transcendem a técnica operatória pura, integrando epidemiologia, biologia molecular e princípios de farmacogenômica. A educação médica continuada e o entendimento rigoroso dos biomarcadores são as ferramentas capazes de mitigar as disparidades nas taxas de sobrevida.
Como bem afirmou Theodor Billroth: “Apenas o homem que está familiarizado com a arte e a ciência do passado é capaz de auxiliar no progresso do futuro.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#OncologiaDigestiva #CirurgiaGeral #EducaçãoMédica #GIST #ResidênciaMédica
Semiologia Cirúrgica do Aparelho Digestivo
A Arte do Diagnóstico Clínico
1. Introdução à Semiologia do Aparelho Digestivo
A semiologia é a pedra angular da prática cirúrgica de excelência. Ela transcende a mera coleta de dados, constituindo-se na harmoniosa composição entre a anamnese — a fase subjetiva em que decodificamos a percepção do paciente — e o exame físico, a fase objetiva na qual o cirurgião exerce sua soberania técnica. Vivemos em uma era de avanços tecnológicos vertiginosos, contudo, o raciocínio clínico permanece insubstituível. Através da semiotécnica apurada, somos capazes de transformar sintomas específicos em hipóteses diagnósticas seguras. Para o cirurgião do aparelho digestivo, a semiologia não é apenas o prelúdio para a propedêutica armada; é o filtro que direciona a solicitação racional de exames complementares e define o diagnóstico diferencial, garantindo que o bisturi nunca preceda o raciocínio clínico apurado.
2. A Anamnese e a História Clínica Detalhada
O diagnóstico clínico diferencial inicia-se na meticulosidade da história clínica. Além da identificação e da queixa principal, o cirurgião deve aprofundar-se na história médica e, crucialmente, na história odontológica. Não negligenciem a investigação de tratamentos prévios: a realização de cirurgias orais, maiores ou menores, pode revelar defeitos ósseos na região bucomaxilofacial ou ausência de elementos dentários que comprometem a primeira fase da digestão. Questionem sobre hábitos de higiene (frequência de escovação e uso de fio dental), pois estes são marcadores indiretos da saúde sistêmica do indivíduo. É imperativo registrar, sob absoluto sigilo profissional, o uso de álcool, fumo e drogas ilícitas. Observem que a via de administração de certas substâncias pode gerar lesões mucosas específicas. Na investigação farmacológica, o uso de Anti-inflamatórios Não Esteroides (AINEs) e Ácido Acetilsalicílico (AAS) deve ser exaustivamente pesquisado. Em pacientes idosos (acima de 65 anos), tais medicamentos elevam exponencialmente o risco de úlceras pépticas complicadas, manifestando-se frequentemente por hemorragias graves ou perfurações sem pródromos exuberantes.
3. Semiologia da Cavidade Oral: A Porta de Entrada
O exame da cavidade oral é o primeiro passo da exploração física. Devemos dominar os recursos semiotécnicos fundamentais:
- Inspeção: Exige iluminação adequada e auxílio de afastadores para avaliar simetria, cor e mensuração.
- Palpação: O tato revela a textura (lisa ou rugosa) e a consistência (mole, fibrosa ou dura).
- Olfação: Embora subestimada, é soberana ao identificar o odor cetônico do diabetes descompensado ou o fétido odor de necrose em carcinomas avançados.
Lesões Fundamentais da Mucosa
Para uma linguagem técnica universal, devemos ser precisos em nossas definições:
- Mancha ou Mácula: Alteração de cor sem alteração de relevo.
- Pápula: Formação sólida elevada de até 5 mm.
- Placa: Formação sólida, plana e elevada, superior a 5 mm.
- Nódulo: Formação sólida, com elevação ou profundidade, de tamanho variado.
- Vesícula e Bolha: Acúmulo de líquido intratecidual (limite de 3 mm para diferenciação).
- Erosão: Perda parcial do tecido epitelial que não ultrapassa a muscularis mucosae.
- Úlcera: Perda total do epitélio com exposição do tecido conjuntivo, frequentemente recoberta por uma membrana fibrinosa amarela ou esbranquiçada.
Atenção Clínica: O cirurgião deve estar alerta para a Leucoplasia, a principal desordem cancerizável, e o Carcinoma de Células Escamosas, que frequentemente se apresenta como uma úlcera de bordas elevadas e endurecidas no assoalho bucal ou bordas laterais da língua.
4. Semiologia do Esôfago: Disfagia e Refluxo
As patologias esofágicas derivam primordialmente de distúrbios na condução do bolo alimentar ou na falha do mecanismo antirrefluxo.
| Tipo de Disfagia | Características Clínicas | Achados Associados & Causas |
| Orofaríngea | Dificuldade inicial; engasgos, tosse, regurgitação nasal. | AVE, Miastenia Gravis, Divertículo de Zenker. |
| Esofágica Obstrutiva | Parada do alimento após deglutição; progressão rápida para líquidos. | Neoplasia (tabagismo/etilismo), Estenose Péptica (pirose crônica). |
| Esofágica Motora | Progressão lenta ou intermitente para sólidos e líquidos. | Acalasia (Chagas), Esclerose Sistêmica (Raynaud/Esclerodactilia). |
Nuance Diagnóstica na DRGE: É vital compreender que sintomas típicos como pirose e regurgitação apresentam elevada sensibilidade (89-95%), porém sua especificidade é baixa (6-38%). Tal fato justifica a necessidade de exames complementares como a pH-metria ou pH-impedanciometria para a confirmação diagnóstica antes de qualquer conduta cirúrgica invasiva.
5. Semiologia do Estômago e Duodeno: A Dor e o “Dia Gástrico”
O raciocínio clínico na úlcera péptica é guiado pela ritmicidade da dor epigástrica em queimação ou “dor de fome”. Observem a pontualidade do “Dia Gástrico”, onde a sequência “dói, come, passa” ocorre em horários clássicos: 10:00h, entre 15:00h e 17:00h, e o despertar noturno entre 01:00h e 03:00h (clocking).
Manobras e Sinais de Alarme
Se o paciente apresenta anorexia, emagrecimento, hematêmese ou história familiar de neoplasia, a investigação deve ser imediata. Ao exame físico do abdome, o cirurgião deve buscar sinais de complicações:
- Rigidez Abdominal (Abdome em Tábua): Indica perfuração com peritonite.
- Sacolejo Gástrico: Realizado movimentando-se lateralmente o abdome do paciente em jejum (>6h) com o ouvido próximo à parede abdominal. A percepção de ruídos hidroaéreos de líquido retido é um sinal inequívoco de obstrução pilórica.
- Peristaltismo Visível: Ondas contráteis na parede abdominal que tentam vencer um obstáculo mecânico distal.
6. O Papel do Helicobacter pylori e Métodos de Diagnóstico
No Brasil, a prevalência do H. pylori na população adulta atinge 65%. Trata-se de um bacilo Gram-negativo classificado pela OMS como Carcinógeno do Grupo I por sua correlação direta com o adenocarcinoma gástrico e o linfoma MALT.
Propedêutica para H. pylori:
- Teste da Urease: Baseia-se na produção da enzima pela bactéria. Atenção: Exige alta densidade bacteriana (10^5 microrganismos) e pode sofrer interferência de hemorragia digestiva ativa, resultando em falsos-negativos.
- Histologia: Permite a visualização direta da bactéria em forma de “S” ou vírgula.
- Teste Respiratório (^{13}C): O padrão-ouro não invasivo para diagnóstico e controle de cura.
- Sorologia (ELISA): Útil em estudos epidemiológicos, mas não distingue infecção ativa de passada.
7. Semiologia do Intestino Delgado e Síndromes de Má Absorção
O intestino delgado é um órgão de adaptação fascinante, medindo de 3,5 a 4 metros in vivo. Sua arquitetura maximiza a absorção através de quatro mecanismos: comprimento tubular, pregas de Kerkring (válvulas coniventes), vilosidades e as microvilosidades (bordadura em escova). Manifestações sistêmicas são fundamentais no diagnóstico diferencial. A presença de artrite ou artralgia associada à diarreia crônica deve nos remeter imediatamente à Doença de Crohn ou à Doença de Whipple. Além disso, o cirurgião deve investigar se processos infecciosos prévios, como tuberculose ou paracoccidioidomicose ganglionar, deixaram sequelas como a linfangiectasia secundária, culminando em perda entérica proteica e má absorção.
8. Conclusão
A semiologia cirúrgica é a base para a solicitação ética e racional de exames como endoscopia, ecoendoscopia e manometria. Dominar a arte do diagnóstico clínico é o que diferencia o verdadeiro cirurgião de um técnico operatório. A precisão diagnóstica nasce da escuta atenta e da observação rigorosa.
——————————————————————————–
“O cirurgião deve ter olhos de águia, coração de leão e mãos de fada.” — Sir John Erichsen
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Semiologia do Abdome Agudo
A Arte do Diagnóstico Clínico na Era da Tecnologia
Caros acadêmicos e colegas cirurgiões, é com o rigor que a ciência exige e o acolhimento que a medicina impõe que iniciamos esta reflexão. No cenário frenético das emergências brasileiras, a dor abdominal não é apenas uma queixa; é um enigma que responde por 5% a 10% de todas as admissões. Embora vivamos a era da imagem (radiologia), é imperativo recordar que, se 30% dos casos desafiam o diagnóstico inicial, 70% são plenamente resolvíveis apenas com as mãos, os olhos e a percepção clínica. Negligenciar a semiologia em prol da tecnologia não é apenas um erro metodológico; é um risco à vida, dada a morbi-mortalidade associada a diagnósticos tardios.
1. A Fisiopatologia da Dor: O Alerta da Lesão Tecidual
Para compreendermos o abdome agudo, devemos interpretar a dor sob a ótica da quebra da homeostase. Observem bem o raciocínio: a dor é um mecanismo de proteção. Quando um estímulo nocivo atinge o organismo, ele ativa nociceptores localizados no mesentério, nas superfícies peritoneais e na mucosa das vísceras. O ponto fundamental aqui é a dualidade do fenômeno. Existe o componente sensorial discriminativo, que nos permite aferir intensidade e localização, e o componente emocional/afetivo, processado pelo sistema límbico. Por que o exame físico deve começar com um aperto de mão e a palpação do pulso? Não apenas para checar a perfusão, mas para o acolhimento. Ao acolher o emocional do paciente, o cirurgião “limpa” o sinal neural, permitindo que o componente sensorial surja com clareza para a anamnese.
Se analisarmos o Homúnculo de Penfield, notaremos que a representação cortical das vísceras é minúscula se comparada à pele. Vejam a lógica: se a representação no córtex é reduzida, o cérebro tem dificuldade em localizar o sinal. É por essa razão fisiopatológica que a dor visceral é vaga e mal definida, enquanto a dor parietal, vastamente representada no homúnculo, é precisa e cortante.
2. A Trindade da Dor: Visceral, Parietal e Referida
A diferenciação dos padrões de dor é o que separa o diagnóstico brilhante da confusão clínica.
- Dor Visceral: Conduzida por Fibras C (lentas), é a dor da distensão ou torção. Como a inervação visceral é tipicamente bilateral e sua origem é embrionária, ela se manifesta na linha média. O Ligamento de Treitz é o nosso marco: dores do intestino anterior (estômago, duodeno, sistema hepatobiliar) surgem no epigástrio; do intestino médio (do Treitz à flexura hepática, incluindo o apêndice) na região periumbilical; e do intestino posterior no hipogástrio.
- Dor Parietal (Somática): Decorre da irritação do peritônio parietal por processos inflamatórios ou químicos (sangue, pus, bile). Diferente da visceral, é conduzida por fibras aferentes somáticas, sendo bem localizada e indicando, quase invariavelmente, gravidade e potencial cirúrgico.
- Dor Referida: Um fenômeno de convergência medular. As fibras aferentes da víscera e as somáticas da pele entram no corno posterior da medula no mesmo nível. O cérebro, confuso, projeta a dor em uma área distante da origem. Recordem-se do Sinal de Kehr (dor no ombro por irritação diafragmática).
Nota de mestre: Considerem a pelve como uma “Muralha Óssea”. Por ser uma estrutura de difícil palpação, a dor referida é, muitas vezes, o único sinal de alerta para patologias pélvicas graves. Nestes casos, o toque retal e vaginal são as únicas janelas para transpor essa muralha.
3. O Método Osleriano e o Exame Físico
Sir William Osler ensinava: “Cultive o seu poder de observação”. O exame não começa na palpação, mas na fácies de dor, na posição antálgica e na sudorese. O paciente com peritonite está imóvel; o que tem cólica, agita-se. O toque médico deve ser o primeiro ato de tratamento. Ao palpar o pulso e sentir uma taquicardia ou um pulso fino, você já está diagnosticando o choque autonômico antes mesmo de olhar o monitor. O acolhimento reduz a agitação, permitindo que você identifique a defesa abdominal (arco reflexo involuntário) ou a irritação peritoneal (percepção cerebral de dor à descompressão).
4. Diagnóstico Sindrômico: O Norte da Conduta
Antes de buscarmos a etiologia exata, devemos classificar o paciente em um dos cinco grandes grupos:
- Inflamatório: Dor insidiosa que se localiza com o tempo (Ex: Apendicite).
- Perfurativo: Dor súbita, em “facada”, com irritação peritoneal franca. Atenção: processos químicos podem ser lentos se bloqueados pelo mesentério.
- Obstrutivo: Dor em cólica, distensão e parada de eliminação de flatos e fezes.
- Vascular (Isquêmico): Caracteriza-se por uma dor desproporcional ao exame físico. Comum em idosos com comorbidades cardiovasculares.
- Hemorrágico: Dor associada a sinais inequívocos de hipovolemia.
5. Armadilhas e o Triunfo da Clínica
Devemos ter vigilância extrema com os “Pacientes de Alto Risco”: idosos, imunodeprimidos, gestantes e, sobretudo, o paciente que retorna. Qualquer indivíduo que volta à unidade com a mesma queixa é, por definição, de altíssimo risco. Lembrem-se: os exames são complementares. A “rotina de abdome agudo” solicitada sem critério é uma muleta para quem não soube examinar. Como dizemos na cirurgia: “Ausência de evidência não é evidência de ausência.”
6. A Regra de Ouro
O diagnóstico de um abdome agudo deve ser um filme, não uma fotografia. Se o diagnóstico não estiver claro, a observação seriada é o caminho. O prontuário deve ser o registro fiel desse cuidado, contendo a história de vida daquela dor. Encerro com a máxima de Sir Zachary Cope, que deve ecoar em cada plantão:
“Qualquer dor abdominal que perdure por mais de 6 horas em paciente previamente saudável é de importância cirúrgica até prova em contrário.”
O diagnóstico precoce é o que define a fronteira entre a vida e o óbito. Honrem a semiologia.
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Extra | Epônimos Semiológicos Abdominais
A
- Sinal de Aaron
Dor ou sensação de desconforto no epigástrio/precordial provocada por pressão contínua no ponto de McBurney; classicamente ligado à apendicite.
Descrito por: Charles Dettie Aaron — norte-americano — 1913. - Sinal de Alders
Na dor parietal/aderencial, o ponto doloroso tende a permanecer fixo quando a paciente muda de posição; útil no diferencial de dor abdominal na gestação.
Descrito por: Nicholas Alders — húngaro de origem, naturalizado britânico — 1951.
B
- Sinal de Ballance
Macicez fixa no flanco esquerdo e macicez móvel no flanco direito; sugere ruptura esplênica com hemoperitônio.
Descrito por: Charles Alfred Ballance — britânico/inglês — 1898. - Sinal de Bassler
Dor à compressão do ponto apendicular por trás da linha médio-axilar; descrito em contexto de apendicite crônica.
Descrito por: Anthony Bassler — norte-americano — 1913. - Sinal de Bastedo
Dor em FID desencadeada por insuflação do cólon; empregado na investigação de apendicite.
Descrito por: Walter Bastedo — canadense radicado nos EUA / canadense-americano — 1910. - Sinal de Blumberg
Dor à descompressão brusca; indica irritação peritoneal.
Descrito por: Jacob Moritz Blumberg — alemão — 1907. - Sinal de Boas
Hiperestesia subescapular direita; clássico em colecistite aguda.
Descrito por: Ismar Isidor Boas — alemão — 1894. - Sinal de Bryant
Equimose azulada escrotal por rastreamento de sangue retroperitoneal/hemoperitônio.
Descrito por: John Henry Bryant — britânico/inglês — 1903. - Sinal de Bryan
Dor em FID à tração do útero ou da vagina em gestante; descrito em apendicite na gravidez.
Descrito por: R. Bryan — norte-americano — 1955.
C
- Sinal de Carnett
A dor piora ou não se altera com contração da parede abdominal, favorecendo origem na parede abdominal.
Descrito por: John Berton Carnett — norte-americano — 1926. - Sinal de Chutro
Desvio umbilical para a direita por contratura antálgica; descrito em apendicite aguda.
Descrito por: Pedro Chutro — argentino — 1912. - Sinal do obturador de Cope
Dor hipogástrica/FID com rotação interna da coxa fletida, sugerindo apêndice pélvico inflamado.
Descrito por: Zachary Cope — britânico/inglês — 1919. - Sinal do psoas de Cope
Dor à extensão passiva da coxa direita, associada a apendicite retrocecal.
Descrito por: Zachary Cope — britânico/inglês — 1921. - Sinal de Courvoisier–Terrier
Vesícula biliar palpável, aumentada e indolor em paciente ictérico; sugere obstrução biliar maligna mais que litíase crônica.
Descrito por: Ludwig Georg Courvoisier — suíço — 1890; antecedente histórico associado a Louis-Félix Terrier — francês — 1889. - Sinal de Cullen
Equimose periumbilical, típica de hemorragia intra/retroperitoneal, incluindo pancreatite hemorrágica e gravidez ectópica rota.
Descrito por: Thomas Stephen Cullen — canadense — 1918.
D
- Sinal de Dance
“Vazio” à palpação na fossa ilíaca direita; clássico em intussuscepção.
Descrito por: Jean-Baptiste Hippolyte Dance — francês — 1826. - Sinal de Danforth
Dor no ombro à inspiração por irritação diafragmática; associado a hemoperitônio, especialmente em gravidez ectópica rota.
Descrito por: William C. Danforth — norte-americano — década de 1920. - Sinal de Dunphy
A tosse exacerba a dor abdominal, sugerindo irritação peritoneal localizada.
Descrito por: geralmente atribuído a John Englebert Dunphy — norte-americano — 1953; a atribuição histórica não é totalmente uniforme nas fontes secundárias.
F
- Sinal de Fothergill
Massa da parede abdominal que permanece palpável com contração do reto e não cruza a linha média; típico de hematoma da bainha do reto.
Descrito por: William Edward Fothergill — britânico/inglês — 1926. - Sinal de Fox
Equimose abaixo do ligamento inguinal; indica hemorragia retroperitoneal.
Descrito por: John Adrian Fox — britânico/inglês — 1966.
G
- Sinal de Grey Turner
Equimose em flancos, típica de hemorragia retroperitoneal, sobretudo na pancreatite hemorrágica.
Descrito por: George Grey Turner — britânico/inglês — descrito em 1919 e publicado em 1920.
H
- Sinal de Howship–Romberg
Dor irradiada para a face medial da coxa por compressão do nervo obturatório; clássico de hérnia obturatória.
Descrito por: John Howship — britânico/inglês — 1840; complementado por Moritz Heinrich Romberg — alemão — 1847/1848.
J
- Sinal de Jobert
Desaparecimento da macicez hepática à percussão, sugerindo pneumoperitônio.
Descrito por: Antoine Joseph Jobert de Lamballe — francês — tradicionalmente atribuído à década de 1830; a data primária exata não ficou inequívoca nas fontes abertas consultadas.
K
- Sinal de Kehr
Dor referida no ombro esquerdo por irritação diafragmática, típica de ruptura esplênica ou hemoperitônio.
Descrito por: Hans Kehr — alemão — epônimo consolidado entre o fim do século XIX e início do XX.
L
- Sinal de Lapinsky
Dor em FID agravada por compressão local enquanto o paciente eleva o membro inferior direito estendido; associado à apendicite retrocecal.
Descrito por: Michael Lapinsky — russo — tradicionalmente situado no fim do século XIX; a data exata não foi confirmada com segurança nas fontes abertas consultadas. - Sinal de Lennander
Diferença entre temperatura retal e axilar, usada historicamente em processos inflamatórios abdominais, incluindo apendicite.
Descrito por: Karl Gustaf Lennander — sueco — fim do século XIX. - Sinal de Lockwood
Sensação de gorgolejo ou “borbulhamento” à palpação na FID em apendicite.
Descrito por: Charles Barrett Lockwood — britânico/inglês — epônimo difundido em publicação de 1932, baseada em seu ensino clínico prévio.
M
- Sinal de Mallet-Guy
Dor profunda no hipocôndrio esquerdo/ângulo costovertebral esquerdo, associada à pancreatite crônica.
Descrito por: Pierre Mallet-Guy — francês — 1943. - Sinal de Markle
Dor provocada pela queda sobre os calcanhares (“heel drop test”), sugerindo irritação peritoneal.
Descrito por: George B. Markle IV — norte-americano — 1973. - Sinal de Mayo-Robson
Dor à palpação no ângulo costovertebral esquerdo/“ponto de Mayo-Robson”, associado a doença pancreática.
Descrito por: Arthur William Mayo-Robson — britânico/inglês — início do século XX. - Sinal de McBurney
Dor máxima no ponto de McBurney, típica de apendicite aguda.
Descrito por: Charles Heber McBurney — norte-americano — 1889. - Sinal de Moynihan
Dor/hipersensibilidade no ponto cístico ou sob o rebordo costal direito, em contexto de colecistite.
Descrito por: Berkeley George Andrew Moynihan — britânico/inglês — 1905. - Sinal de Murphy
Interrupção inspiratória por dor à palpação do ponto cístico; clássico em colecistite aguda.
Descrito por: John Benjamin Murphy — norte-americano — 1903.
P
- Sinal de Perman
Dor em FID desencadeada por manobras compressivas à esquerda; historicamente, antecipa o que depois se difundiu como “Rovsing”.
Descrito por: Emil Samuel Perman — sueco — 1904.
R
- Sinal de Rovsing
Hoje descrito como dor em FID à palpação/compressão do lado esquerdo; usado no diagnóstico de apendicite aguda, embora a descrição original de 1907 fosse mecanicamente mais específica.
Descrito por: Niels Thorkild Rovsing — dinamarquês — 1907. - Sinal de Rosenstein / Sitkovskiy
Dor em FID ao decúbito lateral esquerdo; clássico em apendicite.
Descrito por: Piotr Porfiryevich Sitkovskiy — russo — 1922; associação histórica com Paul Rosenstein — alemão.
S
- Triângulo de Sherren
Área triangular de hiperestesia cutânea na FID, descrita na apendicite aguda.
Descrito por: James Sherren — britânico — 1903.
T
- Sinal de Torres Homem
Dor intensa à percussão digital na projeção hepática; classicamente associado a abscesso hepático.
Descrito por: João Vicente Torres Homem — brasileiro — século XIX; a data primária exata não foi localizada de forma confiável nas fontes abertas consultadas.
V
- Sinal de Volkovich–Kocher
Migração da dor do epigástrio/periumbilical para a FID, típica da apendicite aguda.
Descrito por: Nikolay Markianovich Volkovich — ucraniano — epônimo consolidado no início do século XX; frequentemente associado também à tradição clínica de Theodor Kocher — suíço. - Sinal de Voskresensky
Dor em FID durante deslizamento rápido da mão sobre a parede abdominal (“sinal da camisa”); descrito em apendicite.
Descrito por: Vladimir Mikhailovich Voskresensky — russo — 1940.
Proteção Médica contra Fiscalizações Invasivas e Sensacionalistas
No atual cenário das unidades de urgência e emergência, observa-se uma crescente e indevida ingerência de agentes externos que, sob o pretexto do escrutínio midiático, submetem o corpo clínico — em especial os profissionais em início de carreira — a constrangimentos e intimidações. Tais condutas, frequentemente pautadas pelo sensacionalismo digital, não apenas desestabilizam o ato médico, como violam preceitos basilares da Bioética e do Direito, notadamente o sigilo profissional e a dignidade do paciente. Diante desse panorama de vulnerabilidade técnica e jurídica, o presente Decálogo de Proteção Profissional Médica emerge como um instrumento essencial de salvaguarda, transpondo a dogmática jurídica para a práxis cotidiana. Este guia oferece diretrizes objetivas de conduta e interlocução, permitindo ao médico exercer sua autonomia com o rigor técnico, a polidez e a segurança jurídica que a complexidade do ambiente hospitalar exige.

I. Manutenção da Postura Ética e Prevenção do Conflito O médico deve manter o controle emocional, adotando postura polida e técnica. É imperativo evitar qualquer embate físico ou agressão verbal com os invasores, neutralizando o objetivo sensacionalista da ação.
- Fundamentação Jurídica e Ética: Código de Ética Médica (Resolução CFM nº 2.217/2018), Capítulo I, Inciso II; Art. 5º, inciso X, da Constituição Federal.
- O que fazer e o que falar: Respire fundo, cruze os braços ou mantenha as mãos visíveis (evitando qualquer gesto que possa ser interpretado como agressão). Não tente tomar o celular ou a câmera do invasor.
- Fala: “Boa noite/dia. Por favor, peço que mantenham a calma e baixem o tom de voz. Estamos em um ambiente hospitalar de respeito e cuidado.”
II. Exigência de Identificação e Recusa Expressa de Filmagem O profissional deve solicitar a identificação da autoridade e declarar, em bom tom (para que fique registrado na própria gravação), que não autoriza o uso, captura ou veiculação de sua imagem e voz.
- Fundamentação Jurídica e Ética: Art. 5º, inciso X, da Constituição Federal; Art. 20 do Código Civil.
- O que fazer e o que falar
- Ação: Olhe diretamente para a pessoa (e não para a câmera). Adote um tom de voz firme, porém sem gritar.
- Fala: “Sou o médico plantonista responsável neste momento. Por gentileza, identifiquem-se. Deixo expressamente claro, e peço que fique registrado em sua gravação, que não autorizo a captura, o uso ou a veiculação da minha imagem e da minha voz.”
III. Acionamento Imediato da Cadeia de Comando Institucional A gestão do fluxo de pessoas não é atribuição do médico assistente. Deve-se delegar a um membro da equipe (enfermagem/técnicos) o acionamento urgente da Segurança Patrimonial, da Chefia de Plantão e do Diretor Técnico da instituição.
- Fundamentação Jurídica e Ética: Resolução CFM nº 2.147/2016.
- O que fazer e o que falar
- Ação: Vire-se calmamente para um colega da equipe multidisciplinar e delegue a função, sem dar as costas totalmente aos invasores.
- Fala: “Enfermeiro(a) [Nome], por favor, acione imediatamente a segurança do hospital, a chefia de plantão e comunique o Diretor Técnico de que temos pessoas não autorizadas e com câmeras no setor.”
IV. Isolamento Físico e Proteção da Intimidade do Paciente A prioridade máxima é salvaguardar o paciente em atendimento. O médico deve fechar portas, fechar cortinas e posicionar-se fisicamente na entrada de consultórios ou enfermarias, bloqueando o acesso visual das câmeras aos enfermos.
- Fundamentação Jurídica e Ética: Art. 5º, inciso X, da Constituição Federal; Art. 73 do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Caminhe de forma decidida até a porta do consultório ou enfermaria, feche-a e coloque-se à frente dela como um obstáculo físico pacífico.
- Fala: “Os senhores não podem entrar aqui. Temos pacientes desnudos e vulneráveis em atendimento. Exijo que respeitem a privacidade e a intimidade dos enfermos.”
V. Evocação Expressa do Sigilo Médico e Profissional Diante da tentativa de invasão em áreas assistenciais, o médico deve declarar que está sob o imperativo legal do sigilo e que a presença de terceiros não autorizados munidos de câmeras configura quebra indireta de confidencialidade.
- Fundamentação Jurídica e Ética: Art. 154 do Código Penal Brasileiro; Art. 73, parágrafo único, do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Ignore perguntas médicas ou clínicas sobre qualquer paciente (“por que fulano está esperando?”, “o que aquele paciente tem?”).
- Fala: “Como médico, sou regido pela lei do sigilo absoluto. Não responderei a perguntas sobre casos clínicos e não permitirei filmagens nesta área, pois isso configura crime de quebra de confidencialidade médica.”
VI. Negativa Peremptória de Acesso a Prontuários Médicos O profissional deve negar a entrega, leitura ou auditoria de prontuários por parte de parlamentares ou fiscais não vinculados aos Conselhos de Medicina, informando que o documento pertence exclusivamente ao paciente.
- Fundamentação Jurídica e Ética: Resolução CFM nº 1.638/2002; Art. 89 do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Feche pastas de prontuários físicos ou bloqueie a tela do computador/sistema eletrônico.
- Fala: “O prontuário é um documento legal e de propriedade exclusiva do paciente. Sem a autorização expressa e por escrito dele, ou sem ordem judicial, os senhores não terão acesso a nenhum documento médico.”
VII. Esclarecimento dos Limites da Competência Fiscalizatória O médico deve esclarecer à autoridade que o exercício de “poder de polícia” parlamentar se restringe à infraestrutura e à administração pública, não abrangendo a fiscalização do ato médico ou da conduta ética, que possuem foro específico.
- Fundamentação Jurídica e Ética: Lei nº 3.268/1957.
- O que fazer e o que falar:
- Ação: Mantenha a compostura caso o político tente dar “ordens” ou fazer “ameaças” usando o cargo.
- Fala: “Reconheço o seu cargo, mas o senhor tem competência para fiscalizar a estrutura administrativa. A fiscalização do ato médico e da conduta profissional é prerrogativa exclusiva do Conselho Regional de Medicina (CRM). Por favor, dirija suas demandas ao Diretor da unidade.”
VIII. Suspensão do Atendimento sob Coação (Garantia de Condições Dignas) Caso as câmeras adentrem o recinto de atendimento e os invasores se recusem a sair, o médico deve suspender temporariamente o ato médico — desde que não seja caso de urgência/emergência — informando que só retornará quando a privacidade for restabelecida.
- Fundamentação Jurídica e Ética: Código de Ética Médica, Capítulo II, Inciso IV (Direitos dos Médicos).
- O que fazer e o que falar:
- Ação: Caso invadam a sala de forma ríspida, interrompa a consulta (se não houver risco de morte), sente-se na cadeira, afaste-se do paciente e aguarde em silêncio a retirada dos invasores pela segurança.
- Fala: “Diante da invasão e da coação com câmeras, informo que não possuo condições dignas e éticas para exercer a medicina neste momento. Retomarei o atendimento imediatamente após os senhores se retirarem e a segurança do paciente for garantida.”
IX. Registro Documental Exaustivo do Incidente Tão logo a situação seja normalizada, o médico deve registrar o fato detalhadamente no Livro de Ocorrências do plantão. Caso a invasão tenha prejudicado ou atrasado o atendimento de algum paciente, tal interferência externa deve constar no prontuário do mesmo.
- Fundamentação Jurídica e Ética: Art. 87 do Código de Ética Médica; Art. 299 do Código Penal.
- O que fazer e o que falar:
- Ação: Escreva de forma objetiva, descrevendo fatos (sem adjetivos emocionais). Cite os nomes dos invasores, a recusa em parar de filmar e os horários exatos.
- Fala (orientação mental e à equipe): “Precisamos registrar tudo. Vamos anotar o nome do vereador/político, o horário exato da invasão, o que foi dito e as paralisações geradas no atendimento para colocar no livro do plantão agora mesmo.”
X. Acionamento das Autoridades Policiais e Desagravo Institucional Ao término do plantão, é fundamental registrar Boletim de Ocorrência Policial e enviar comunicação oficial (Ofício) ao Diretor Técnico e ao Conselho Regional de Medicina (CRM) relatando o constrangimento.
- Fundamentação Jurídica e Ética: Decreto-Lei nº 3.688/1941, Art. 42; Art. 150 do Código Penal; Código de Ética Médica, Capítulo II, Inciso V.
- O que fazer e o que falar:
- Ação: Dirija-se a uma delegacia (ou delegacia virtual) e, nos dias subsequentes, contrate um advogado e/ou assessoria jurídica institucional e/ou do sindicato médico.
- Fala (na Delegacia / no Ofício): “Desejo registrar um Boletim de Ocorrência por constrangimento ilegal, perturbação do ambiente de trabalho e quebra tentada de sigilo médico contra o invasor. Solicitarei também, via ofício, o desagravo público junto ao CRM.”
A nossa missão máxima será sempre a defesa da saúde e da dignidade do doente. Que as orientações sirvam para garantir que nenhum profissional permita a usurpação do seu local de trabalho, assegurando que a nobre arte da Medicina seja sempre exercida em condições de absoluta segurança, privacidade e respeito mútuo.
Prof. Dr. Ozimo Gama
Cuidados Perioperatórios na Cirurgia Laparoscópica
Otimizando a Fisiologia e Acelerando a Recuperação no Aparelho Digestivo
Autor: Prof. Dr. Ozimo Gama Categoria: Cuidados Perioperatórios / Cirurgia Laparoscópica / Cirurgia do Aparelho Digestivo Tempo de Leitura: 10 minutos
Introdução
A cirurgia laparoscópica revolucionou a especialidade do aparelho digestivo ao atenuar o trauma cirúrgico, reduzir a resposta inflamatória sistêmica e proporcionar uma recuperação estética e funcional superior. No entanto, para o estudante de medicina, o residente de cirurgia geral e o pós-graduando, é imperativo compreender que incisões milimétricas não isentam o paciente da resposta endócrino-metabólica ao trauma. No Brasil, o Sistema Único de Saúde (SUS) realiza centenas de milhares de procedimentos videolaparoscópicos anualmente, desde colecistectomias até gastrectomias oncológicas complexas. Para maximizar os benefícios da via minimamente invasiva, a excelência no centro cirúrgico deve ser invariavelmente acompanhada por um manejo perioperatório de vanguarda. A transição dos cuidados dogmáticos do passado para protocolos modernos baseados em evidências — como o ERAS (Enhanced Recovery After Surgery) e o Projeto ACERTO (Aceleração da Recuperação Total Pós-Operatória), amplamente adotado e validado em território nacional — é o que define o cirurgião contemporâneo de alta performance.
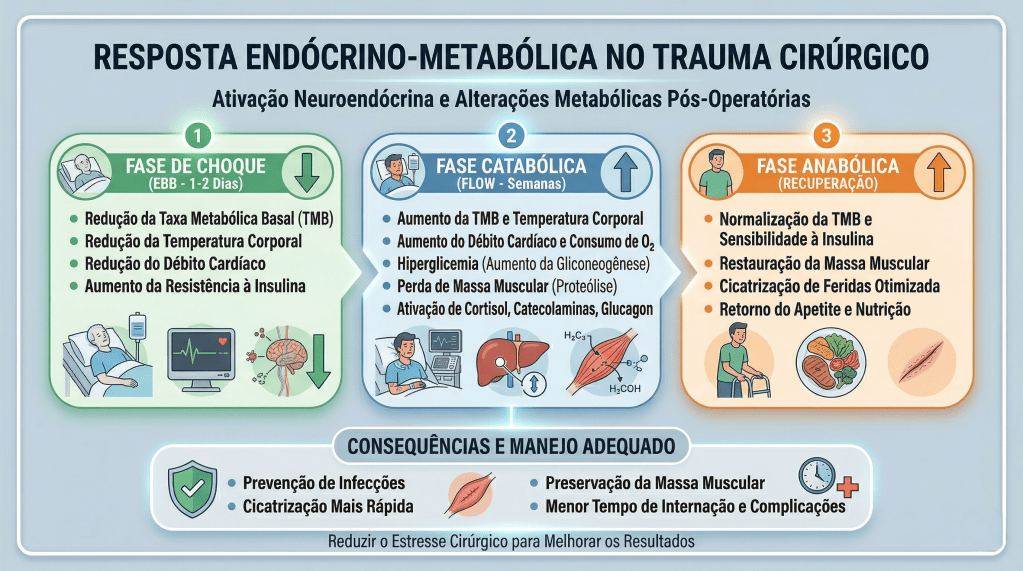
A Fisiologia do Pneumoperitônio e a Quebra de Paradigmas
O manejo perioperatório na laparoscopia exige um profundo respeito pelas alterações fisiológicas induzidas pelo pneumoperitônio com dióxido de carbono (CO2). A insuflação abdominal eleva a pressão intra-abdominal (usualmente entre 12 e 15 mmHg), acarretando compressão da veia cava inferior, diminuição do retorno venoso, aumento da resistência vascular sistêmica e elevação das pressões das vias aéreas com risco de hipercapnia. A equipe cirúrgico-anestésica deve atuar em sintonia para mitigar estes efeitos.
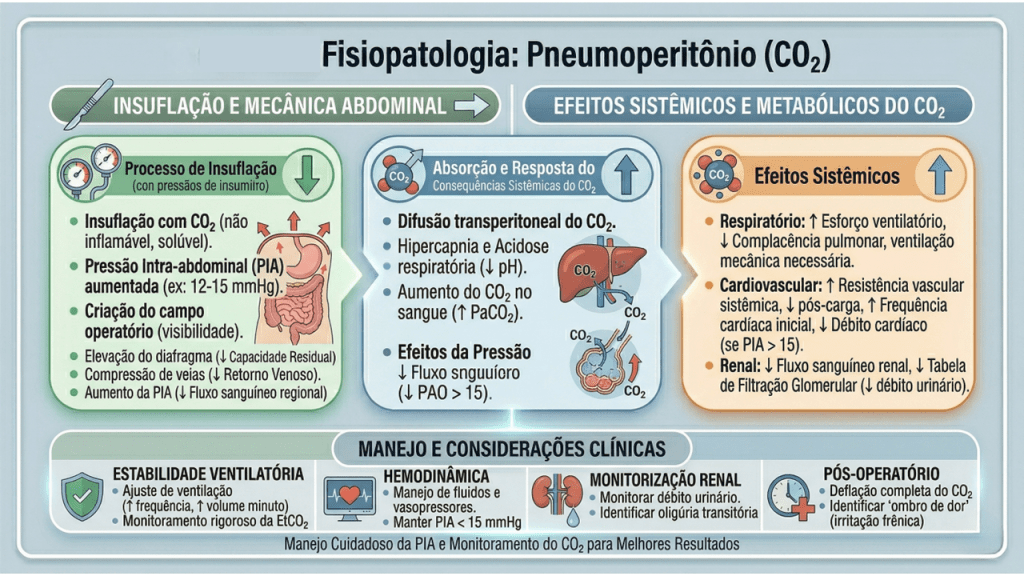
Sob a ótica dos protocolos de aceleração da recuperação, o manejo perioperatório é dividido em três fases cruciais, onde dogmas seculares foram cientificamente derrubados:
- Pré-Operatório (O Fim do Jejum Prolongado): O jejum absoluto de 8 a 12 horas está proscrito. Evidências robustas, corroboradas por dados do Projeto ACERTO no Brasil, demonstram que a oferta de líquidos claros enriquecidos com carboidratos até 2 horas antes da indução anestésica reduz a resistência insulínica pós-operatória, minimiza a perda de massa magra e diminui a ansiedade do paciente.
- Intraoperatório (A Terapia Hídrica Guiada por Metas): A hiper-hidratação venosa, outrora comum, resulta em edema esplâncnico e retardo do esvaziamento gástrico. A fluidoterapia deve ser restritiva ou guiada por metas (avaliando a variação do volume sistólico), garantindo perfusão tecidual sem encharcar o paciente. A analgesia deve ser preemptiva e multimodal, poupadora de opioides.
- Pós-Operatório (Mobilização e Realimentação): O repouso no leito prolongado favorece o tromboembolismo venoso (TEV) e a atelectasia pulmonar. O paciente deve ser encorajado a deambular no mesmo dia da operação.
Aplicação na Cirurgia Digestiva
Na cirurgia do trato gastrointestinal, a aplicação destes cuidados perioperatórios altera drasticamente os desfechos e o tempo de internação:
- Preparo Mecânico do Cólon (PMC): Em cirurgias colorretais laparoscópicas, o uso rotineiro do PMC foi abandonado por grande parte das diretrizes, pois causa desidratação, distúrbios hidroeletrolíticos e não reduz as taxas de deiscência anastomótica ou infecção de sítio cirúrgico (exceto quando associado a antibióticos orais específicos em cirurgias de reto baixo).
- Sondas e Drenos: A utilização profilática de sondas nasogástricas (SNG) e drenos cavitários não encontra mais respaldo na literatura para procedimentos eletivos sem complicações. A SNG prolonga o íleo adinâmico e predispõe a infecções respiratórias. O dreno deve ser uma exceção fundamentada, não a regra.
- Realimentação Precoce: O íleo pós-operatório é inerente à manipulação das alças, mas a dieta por via oral iniciada precocemente (muitas vezes nas primeiras 24 horas) estimula o reflexo gastrocólico, protege a barreira mucosa intestinal contra a translocação bacteriana e acelera a alta hospitalar. Estatísticas brasileiras apontam que a realimentação precoce em ressecções intestinais laparoscópicas reduz o tempo de internação em até 2 a 3 dias.
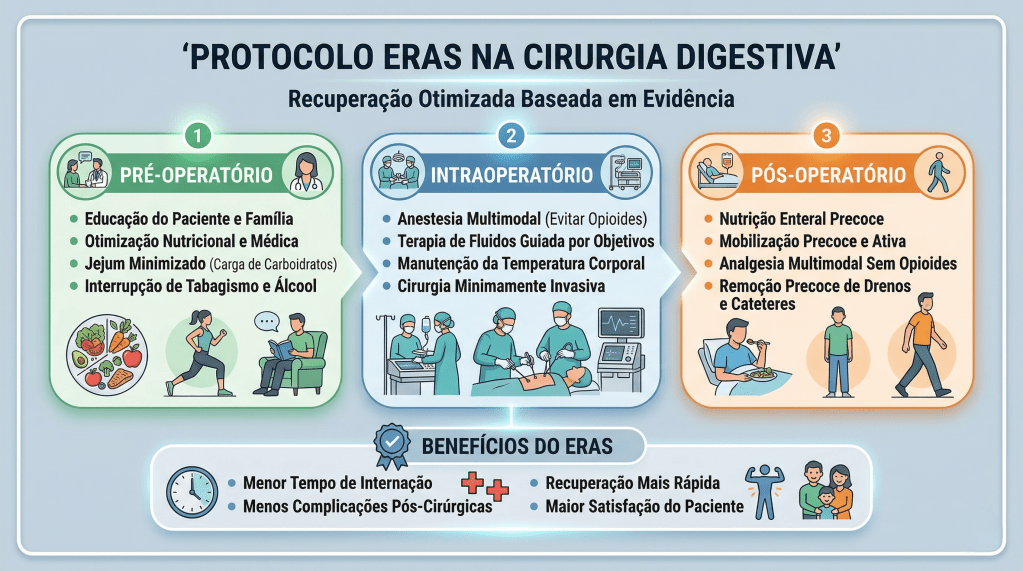
Pontos-Chave
- Avaliação Fisiológica: O pneumoperitônio exige monitorização hemodinâmica rigorosa devido às alterações cardiocirculatórias e ventilatórias.
- Jejum Abreviado: Utilização de maltodextrina a 12,5% até 2 horas antes da cirurgia é uma prática segura e metabolicamente protetora.
- Analgesia Opioid-Sparing: Uso de infiltração dos portais com anestésicos locais, anti-inflamatórios e bloqueios regionais (ex: TAP block) para evitar os efeitos eméticos e obstipantes dos opioides.
- Tromboprofilaxia: O uso de meias elásticas de compressão graduada, compressores pneumáticos intermitentes e heparina de baixo peso molecular é mandatório, dada a estase venosa promovida pela pressão intra-abdominal elevada.
- Racionalização de Dispositivos: Evitar o uso sistemático e prolongado de sondas vesicais de demora (SVD), SNG e drenos cavitários.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A destreza na dissecção laparoscópica e a precisão das anastomoses intracavitárias perdem parte de seu brilho se o paciente for submetido a um estresse fisiológico não gerenciado. O cirurgião do aparelho digestivo moderno deve compreender que o seu papel transcende a mecânica operatória; ele é o arquiteto da recuperação do paciente. Adoção de protocolos de cuidados perioperatórios como o ACERTO exige uma mudança de cultura institucional e o alinhamento de toda a equipe multidisciplinar (cirurgião, anestesiologista, enfermagem, nutrição e fisioterapia). Ao unirmos a mínima agressão tecidual da videolaparoscopia à máxima otimização metabólica do perioperatório, entregamos não apenas uma cirurgia segura, mas a restituição rápida e plena da qualidade de vida do nosso paciente.
“O principal objetivo da cirurgia moderna não é apenas curar a doença anatômica, mas suprimir a resposta ao trauma cirúrgico, proporcionando uma recuperação sem dor e sem risco.” — Henrik Kehlet, cirurgião dinamarquês e idealizador do protocolo ERAS.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Fístula Pancreática Pós-Operatória
Estratégias de Prevenção, Diagnóstico Precoce e Manejo Terapêutico na Prática Cirúrgica
Autor: Prof. Dr. Ozimo Gama
Categoria: Cirurgia Hepatopancreatobiliar (HPB) / Oncologia Cirúrgica / Complicações Pós-Operatórias
Tempo de Leitura: 11 minutos
Introdução
A duodenopancreatectomia (DP), ou cirurgia de Whipple, representa o ápice da complexidade técnica na cirurgia do aparelho digestivo. Indicada primariamente para neoplasias periampulares, tumores da cabeça do pâncreas e colangiocarcinomas distais, este procedimento viu sua taxa de mortalidade reduzir-se drasticamente em centros de alto volume nas últimas décadas (atualmente abaixo de 3% a 5%). Contudo, a morbidade perioperatória permanece teimosamente elevada, oscilando entre 30% e 50%. O verdadeiro “calcanhar de Aquiles” da cirurgia de Whipple é a Fístula Pancreática Pós-Operatória (POPF – Postoperative Pancreatic Fistula), originada na falha de cicatrização da anastomose pancreatojejunal ou pancreatogástrica. Para o estudante de medicina, o residente em cirurgia geral e o pós-graduando em cirurgia digestiva, dominar a fisiopatologia, a identificação precoce e o fluxograma de tratamento da POPF é o que difere a recuperação oncológica de um desfecho fatal.
Fisiopatologia e a Nova Definição (ISGPS 2016)
Historicamente, a literatura cirúrgica era confusa quanto à definição de uma fístula pancreática, dificultando a comparação entre os serviços. Em 2016, o International Study Group of Pancreatic Surgery (ISGPS) atualizou e padronizou esta definição, mudando o paradigma do diagnóstico.
O antigo “Grau A” foi abolido da categoria de fístula verdadeira e passou a ser denominado apenas como Vazamento Bioquímico (Biochemical Leak). Para que uma POPF seja clinicamente diagnosticada (Graus B e C), exige-se a presença de líquido no dreno (ou coleção intra-abdominal) com nível de amilase superior a 3 vezes o limite superior da normalidade sérica, associado a uma mudança obrigatória no curso clínico do paciente (necessidade de jejum prolongado, antibioticoterapia específica, manutenção do dreno por mais de 3 semanas, drenagem percutânea ou reoperação).
Estatísticas e o Cenário Brasileiro: No Brasil, dados do Sistema Único de Saúde (DATASUS) e registros de serviços multicêntricos de cirurgia HPB demonstram que a incidência de POPF clinicamente relevante em grandes centros de referência nacionais varia de 10% a 20%. Essa incidência robusta reforça a premissa de que a cirurgia pancreática deve ser estritamente centralizada em hospitais de alto volume, onde a equipe multidisciplinar está plenamente apta a “resgatar” o paciente da complicação (failure to rescue).
Aplicação na Cirurgia Digestiva
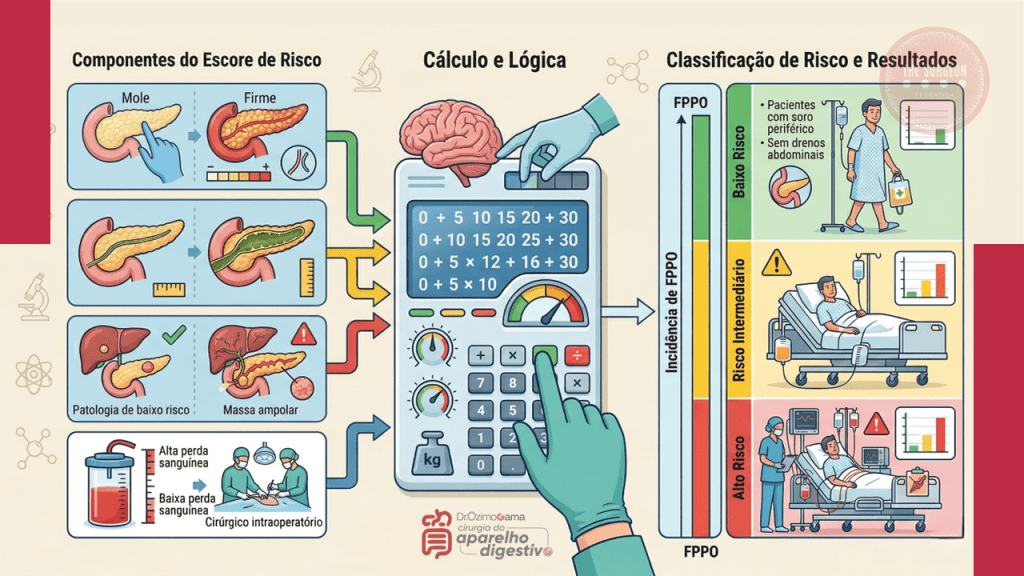
1. Prevenção e Avaliação de Risco Intraoperatório
A prevenção da POPF não começa na síntese, mas na avaliação tátil e visual da glândula. O Fistula Risk Score (FRS) é uma ferramenta validada que estratifica o risco do paciente.
- Fatores de Alto Risco: Uma glândula pancreática de consistência amolecida (infiltração gordurosa) associada a um ducto pancreático principal fino (< 3 mm) compõe a “tempestade perfeita” para o rasgo capsular e a deiscência da sutura. Pacientes com IMC elevado e patologias como o adenocarcinoma de ampola de Vater (que não fibrosam a glândula previamente) têm risco muito maior do que aqueles com pancreatite crônica.
- Tática Cirúrgica: A escolha entre anastomose ducto-mucosa e a técnica de invaginação (dunking) deve respeitar a anatomia do coto e a familiaridade técnica do cirurgião. O uso de fios monofilamentares e agulhas delicadas é imperativo. Estratégias adjuvantes, como a colocação de stents pancreáticos (internos ou externos) para desviar a secreção enzimática da linha de sutura, são valiosas em ductos exíguos. O uso profilático de análogos da somatostatina (octreotida) não é rotina universal, sendo reservado por muitos serviços apenas para o grupo de risco extremo.
2. Diagnóstico Precoce no Dia a Dia da Enfermaria
O diagnóstico da fístula pancreática exige um cirurgião com altíssimo índice de suspeição clínica. O suco pancreático ativado é extremamente cáustico e desencadeia uma resposta inflamatória sistêmica rápida.
- Sinais de Alarme: A taquicardia inexplicada no 3º ou 4º dia de pós-operatório (DPO) é frequentemente o primeiro sinal biológico de vazamento, precedendo a febre, a leucocitose e a dor peritonítica. O retardo do esvaziamento gástrico (gastroparesia) é quase sempre secundário à coleção inflamatória peripancreática adjacente ao estômago.
- O Teste do 3º Dia: A dosagem rotineira da amilase no fluido do dreno no 3º DPO dita o ritmo. Níveis baixos indicam a remoção precoce do dreno (evitando a infecção ascendente e a fístula mecânica induzida pelo próprio dreno). Níveis altos em um paciente com piora clínica impõem a realização imediata de uma Tomografia Computadorizada (TC) de abdome com duplo contraste para mapear coleções fluidas não drenadas.
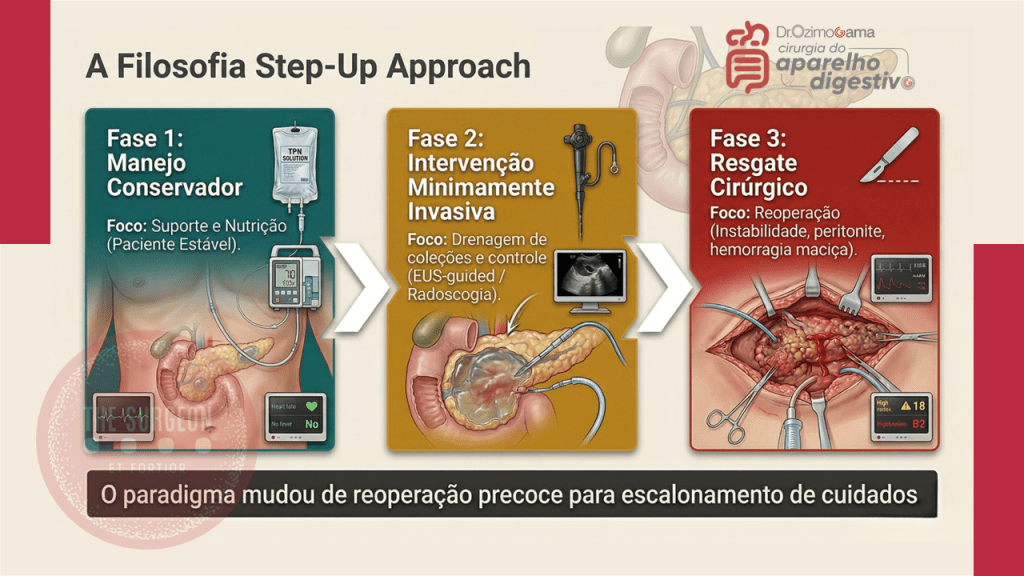
3. A Abordagem Step-Up
A conduta perante uma fístula estabelecida segue um raciocínio de escalonamento (step-up approach):
- Fístula Grau B (Tratamento Clínico/Intervencionista): O pilar é o manejo conservador. Envolve o reposicionamento do dreno cirúrgico ou a inserção de novos drenos percutâneos guiados por TC ou ultrassonografia (Radiologia Intervencionista). Associa-se antibioticoterapia de amplo espectro (focada em translocação entérica), inibidores de bomba de prótons, e rigoroso suporte nutricional (preferencialmente nutrição enteral pós-pilórica ou nutrição parenteral total se o trânsito estiver inviável).
- Fístula Grau C (A Catástrofe e o Resgate Cirúrgico): Representa a falência do tratamento clínico, caracterizada por instabilidade hemodinâmica, disfunção de múltiplos órgãos, sepse não controlada ou hemorragia pós-operatória (decorrente da erosão da artéria gastroduodenal ou hepática pelas enzimas pancreáticas ativadas). A reoperação de urgência é mandatória. Consiste em lavagem ampla da cavidade, múltiplas drenagens calibrosas e hemostasia. Em cenários de desintegração total da anastomose, a tática extrema de salvamento é a totalização da pancreatectomia (remoção do coto pancreático remanescente).
Pontos-Chave
- Definição Atualizada: Vazamento bioquímico não altera conduta. A fístula verdadeira (POPF Grau B/C) exige amilase > 3x o valor sérico no dreno associada a mudança terapêutica (ISGPS 2016).
- Consistência Glandular: Pâncreas mole e ducto fino (<3mm) requerem alerta máximo e técnica cirúrgica de precisão microscópica.
- Vigilância Clínica: Taquicardia isolada no 3º DPO nunca é “normal”; é o principal prenúncio de coleção intracavitária.
- Remoção de Drenos: A prática de manter drenos profiláticos por longos períodos em pacientes sem evidência de vazamento (Amilase no 3º DPO baixa) aumenta a taxa de complicações infecciosas.
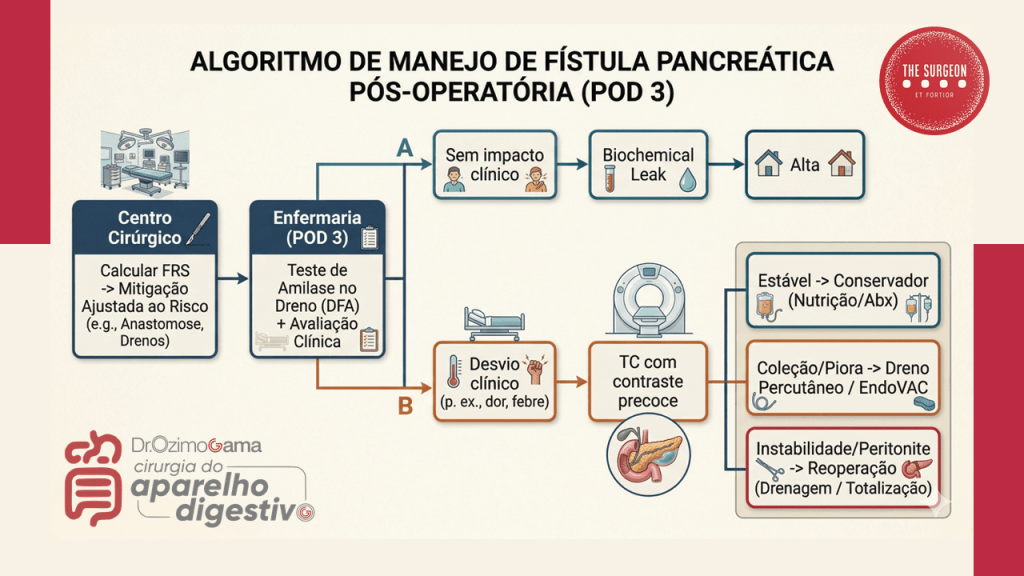
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A excelência na execução de uma duodenopancreatectomia não isenta o cirurgião do risco de uma fístula pancreática. O vazamento enzimático é uma realidade inerente à biologia do pâncreas amolecido. Portanto, a verdadeira assinatura de um cirurgião do aparelho digestivo diferenciado não é apenas a técnica de anastomose que ele emprega, mas a sua implacável vigilância no pós-operatório. Compreender o momento exato de acionar a radiologia intervencionista e ter a coragem cirúrgica para reoperar um paciente em deterioração clínica (aplicando adequadamente a filosofia do step-up approach) são as atitudes éticas e técnicas que reduzem a mortalidade. Na cirurgia pancreática, o diagnóstico precoce é o único antídoto contra o desastre.
“A cirurgia pancreática é um exercício contínuo de humildade; o pâncreas é um órgão que não perdoa a manipulação rude, o desrespeito anatômico e a anastomose imprecisa.” — Adaptado dos ensinamentos imortais de Allen O. Whipple, pioneiro da duodenopancreatectomia.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Prevenção Jurídica na Cirurgia Digestiva
As bases das Boas Práticas Jurídicas para o Cirurgião Moderno
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
Introdução
A prática da cirurgia do aparelho digestivo evoluiu exponencialmente com o advento da cirurgia minimamente invasiva e da robótica. Contudo, essa evolução técnica foi acompanhada por um fenômeno igualmente complexo: a crescente judicialização da medicina. Dados recentes do Conselho Nacional de Justiça (CNJ) apontam um aumento alarmante nas ações judiciais envolvendo supostos erros médicos no Brasil, sendo as especialidades cirúrgicas (incluindo a cirurgia bariátrica e a cirurgia geral) alvos frequentes. Para o estudante de medicina, o residente e o cirurgião já estabelecido, a excelência técnica no centro cirúrgico não é mais suficiente. A proteção jurídica e a gestão de riscos tornaram-se competências inegociáveis. Este artigo traduz as melhores evidências em compliance e prevenção de responsabilidade profissional para a nossa realidade clínica e cirúrgica.
Os Pilares da Proteção Jurídica
A literatura jurídica e de gestão de risco em saúde não prescreve uma “fórmula mágica”, mas estabelece padrões consistentes de governança. A defesa do cirurgião baseia-se na proatividade, ou seja, em ações tomadas antes que o dano ou o litígio ocorram. A mitigação ativa de riscos envolve desde a estruturação de fluxos de atendimento até o amparo em apólices de seguros, garantindo que o profissional exerça sua vocação com autonomia, segurança e paz de espírito.
Práticas Fundamentais
Como aplicamos esses conceitos abstratos na rotina de um serviço de cirurgia digestiva? Abaixo, detalho as boas práticas essenciais baseadas em evidências:
1. Documentação Inatacável e o TCLE
O prontuário médico é a principal (e muitas vezes a única) testemunha de defesa do cirurgião. A documentação deve ser clara, tempestiva e exaustiva. Na cirurgia digestiva, isso se traduz em:
- Descrição Cirúrgica Detalhada: Não economize palavras ao descrever aderências severas, variações anatômicas do trato biliar ou a qualidade dos tecidos ao realizar uma anastomose intestinal.
- Termo de Consentimento Livre e Esclarecido (TCLE): Um TCLE genérico não tem valor jurídico. Ele deve ser individualizado. Vai operar um câncer gástrico? O paciente deve estar ciente (e assinar) sobre os riscos de fístula do coto duodenal, necessidade de CTI e mortalidade associada. O diálogo prévio bem documentado anula a alegação de “falta de informação”.
2. Adesão à Lex Artis e aos Protocolos Clínicos
Agir de acordo com a Lex Artis significa atuar conforme as práticas médicas consagradas e atualizadas. A adesão a diretrizes técnicas (Guidelines) de sociedades como o CBCD, SBCBM ou protocolos internacionais (como o ERAS – Enhanced Recovery After Surgery) é a forma mais eficaz de afastar a culpa (negligência, imprudência ou imperícia).
- Exemplo prático: O uso sistemático do Checklist de Cirurgia Segura da OMS previne a catástrofe médico-legal da cirurgia em paciente ou local errado, além de garantir a profilaxia antimicrobiana no tempo correto.
3. A Comunicação como Escudo Protetor
A ampla maioria dos processos por erro médico nasce de uma quebra na relação médico-paciente. Quando ocorre uma complicação grave — como uma deiscência de anastomose após uma retossigmoidectomia —, a postura do cirurgião dita o desfecho legal. A comunicação proativa, empática, transparente e não conflituosa com o paciente e seus familiares reduz drasticamente o ímpeto litigioso. Abandonar o paciente aos cuidados exclusivos do médico plantonista da UTI após uma complicação é o caminho mais rápido para um tribunal.
4. Governança de Dados e a LGPD
A Lei Geral de Proteção de Dados (LGPD) transformou a gestão de informações médicas. Os controles técnicos de privacidade são mandatórios.
- Cuidado com imagens: Fotografar peças cirúrgicas (como vesículas, tumores) ou exames de imagem e compartilhar em grupos de WhatsApp, mesmo que com intenção didática, sem a anonimização estrita ou o consentimento expresso do paciente, constitui grave infração ética e legal.
5. Seguro de Responsabilidade Profissional e Assessoria Especializada
Operar sem um seguro de responsabilidade civil profissional é expor o patrimônio pessoal a um risco imensurável. É vital compreender as coberturas da apólice, especialmente a cláusula de tail coverage (cobertura de cauda), que garante proteção contra processos instaurados anos após a ocorrência do fato gerador. Além disso, ao primeiro sinal de notificação administrativa (CRM) ou citação judicial, a prática de agir sem o suporte de advogados especialistas em Direito Médico é temerária. A confidencialidade e a estratégia da relação advogado-cliente são irrenunciáveis.
Pontos-Chave para a Prática Diária
- O Prontuário Médico não é apenas uma obrigação burocrática; é a sua armadura legal.
- TCLE é um processo de comunicação, não apenas um pedaço de papel a ser assinado na porta do bloco cirúrgico.
- Aderir estritamente aos Guidelines e Protocolos estabelece um padrão de cuidado defensável.
- Nunca subestime o poder de uma Relação Médico-Paciente baseada na confiança, empatia e presença contínua, especialmente nas adversidades.
Conclusões Aplicadas
A complexidade inerente à cirurgia do aparelho digestivo nos expõe diariamente a riscos biológicos imponderáveis. Fístulas, sangramentos e infecções podem ocorrer mesmo quando a técnica cirúrgica é executada com perfeição. O que diferencia a complicação inerente do ato cirúrgico (o risco aceitável) do ilícito civil ou ético é, fundamentalmente, a nossa conduta preventiva, a clareza da nossa documentação e o rigor ético no trato com o paciente. A proteção jurídica não deve gerar uma “medicina defensiva” prejudicial ao doente (que gera exames e intervenções desnecessárias), mas sim uma medicina preventiva, baseada em compliance, transparência e excelência documental.
“As complicações fazem parte da cirurgia, mas o abandono do paciente e a negligência na documentação são escolhas exclusivas do cirurgião. A verdadeira destreza opera tanto com o bisturi quanto com a caneta.” — Adaptado de preceitos de grandes mestres da cirurgia contemporânea.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Evolução da Medicina Operacional
Lições Eternas para a Logística de Saúde Militar
Prof. Dr. Ozimo Gama
1. O Elo entre o Passado e a Prontidão Operacional
A Medicina Operacional transcende a mera assistência de retaguarda; ela se consolida como um multiplicador de poder de combate indispensável para a manutenção do efetivo combatente. No cenário de defesa contemporâneo, a preservação da integridade física e psíquica da tropa é o fiel da balança que sustenta a viabilidade de campanhas em ambientes de conectividade limitada e logística hostil, como a Região Amazônica, ou em missões sob a égide de organismos internacionais. Aprender com a história militar não é um exercício diletante, mas um imperativo para a Prontidão Operacional. O objetivo é garantir que o apoio de saúde seja resiliente o suficiente para operar sob o atrito do combate, assegurando a “Manutenção do Poder de Combate” — princípio basilar para que o soldado retorne à linha de fogo com a celeridade que a manobra exige. Embora as tecnologias cinéticas evoluam, os princípios de salvar vidas sob fogo permanecem como constantes doutrinárias desde a Antiguidade.
2. Das Cruzadas ao Campo de Batalha Primitivo: O Nascimento do Cuidado sob Fogo
Os conflitos da Idade Média, notadamente as Cruzadas, forçaram a Medicina a migrar do conforto clerical para a brutalidade das intervenções cirúrgicas de campanha. Um exemplo emblemático da “Medicina Operacional” rudimentar ocorreu em agosto de 1097, quando o Duque Godofredo de Bouillon, ao enfrentar um urso selvagem próximo a Antioquia, feriu gravemente a própria perna com sua espada. O sangramento arterial massivo exigiu que os médicos da época agissem minuto a minuto sob extrema pressão. Utilizando técnicas de cauterização com ferro em brasa — inspiradas nos ensinamentos de Albucasis — os cirurgiões estancaram a hemorragia, permitindo que o líder sobrevivesse para retomar o comando meses depois. A organização dos hospitais pela Ordem de São João (Hospitaleiros) em Jerusalém e Acre representou o embrião da triagem e da higiene militar. Estes “xenodóquios” separavam pacientes por diagnóstico, demonstrando que a gestão de leitos e a separação de feridos traumáticos de doentes infectocontagiosos eram vitais para a sustentabilidade da força.
Inteligência Técnica Imediata e Sensível (ITIS): O episódio de Godofredo de Bouillon ilustra que o sucesso do apoio de saúde depende da capacidade de decisão técnica sob estresse. Para o oficial moderno, a lição é clara: sem intervenção imediata no ponto de ferimento, a taxa de retorno ao serviço é nula. A sustentabilidade das campanhas de longo prazo nas Cruzadas só foi possível devido a essa estruturação de cuidados que, embora rudimentar, já visava a preservação do componente humano como recurso estratégico.

3. A Revolução de Dominique Jean Larrey: O Surgimento da Logística Moderna
O Barão Dominique Jean Larrey, cirurgião-chefe da Guarda Imperial de Napoleão, é legitimamente o “pai da medicina operativa moderna”. Sua visão transformou o apoio de saúde de um serviço passivo na Zona de Retaguarda (ZRE) para uma força de intervenção proativa na vanguarda.
- Inovação das Ambulances Volantes: Larrey revolucionou a Evacuação de Feridos (Evoz) ao criar ambulâncias leves que retiravam os feridos ainda sob o fogo inimigo, combatendo a “inatividade administrativa” que abandonava soldados no campo.
- Cirurgia de Urgência e Técnica: Defensor do débridement précoce (debridamento precoce) e das feridas laissées ouvertes (deixadas abertas) para prevenir a gangrena gasosa, Larrey era dotado de uma velocidade técnica prodigiosa, capaz de realizar a desarticulação de um ombro em apenas dois minutos — perícia vital em um Posto de Socorro (PS) operando sem anestesia moderna.
- Ética e Neutralidade: Precursor da Cruz Vermelha, Larrey estabeleceu o “Hôpital destiné à l’ennemi” em Valladolid, tratando prisioneiros espanhóis e ingleses com a mesma dignidade que os franceses.
Napoleão reconheceu sua importância ao declarar: “O homem mais honesto que já conheci… uma imagem do verdadeiro homem de bem”.
ITIS: : A mobilidade de Larrey alterou o moral da tropa (segurança psicológica). No Exército Brasileiro, essa herança reflete-se na busca pela “Hora de Ouro”. O legado de Larrey ensina que o oficial médico deve ser um “chirurgien combattant de l’avant”, integrado à manobra e não apenas um espectador na retaguarda.

4. Século XIX à Era Contemporânea: Da Enfermagem de Nightingale à Evacuação Aeromédica
A Guerra da Crimeia trouxe Florence Nightingale e a revolução da sanidade, mas foi a inovação técnica de Antonius Mathijsen, em 1851, com o gesso (Plaster of Paris), que transformou a logística de saúde. A estabilização eficaz de fraturas permitiu que a Cadeia de Evacuação se tornasse mais resiliente e longa; fraturas estáveis reduziam o risco de embolia gordurosa e trauma secundário durante o transporte, permitindo fluxos logísticos mais audazes. No século XX, o advento dos Mobile Army Surgical Hospitals (MASH) e do helicóptero na Coreia e Vietnã redefiniu a velocidade do apoio. Atualmente, o protocolo Tactical Combat Casualty Care (TCCC) padroniza intervenções salvadoras de vida no ponto de ferimento, focando na tríade: controle de hemorragia, manejo de via aérea e prevenção de hipotermia.
ITIS”: A evolução tecnológica, da tração animal ao Medevac, exige uma coordenação logística impecável de insumos críticos, como sangue e oxigênio. A lição para o planejamento atual é que a tecnologia só é eficaz se houver uma rede de suprimento integrada que acompanhe a velocidade do transporte.

5. O Cenário Brasileiro: Doutrina e o Papel das Forças Armadas
No Brasil, a doutrina é balizada pelo manual MD42-M-04 (Logística de Saúde em Operações Conjuntas), que estabelece os Níveis de Apoio e a interoperabilidade entre o Exército, a Marinha e a Força Aérea. O Corpo de Fuzileiros Navais (CFN), através do seu Batalhão Logístico (BtlLogFuzNav), opera unidades de saúde móveis com alta capacidade expedicionária, aplicando os princípios de Larrey em ambientes litorâneos e ribeirinhos. Na Amazônia, o planejamento exige o uso intensivo de Inteligência Médica (conforme a doutrina NATO AJMEDP-3). Isso significa mais do que tratar doenças; implica mapear a “capacidade hospitalar local” em municípios remotos e monitorar “vetores endêmicos” (como a malária e a leishmaniose). Sem a Inteligência Médica para antecipar ameaças sanitárias, uma força pode ser dizimada antes mesmo do primeiro contato cinético com o inimigo.
ITIS”: A Inteligência Médica é uma ferramenta de antecipação estratégica. Em operações na selva, conhecer o terreno e o perfil epidemiológico local permite ao oficial de saúde dimensionar a Cadeia de Evacuação de forma a garantir que a distância não seja uma sentença de morte para o combatente.

6. Planejamento Logístico Atual
- A Proximidade do Apoio (O Legado do PS): A estabilização deve ocorrer no Nível 1 de apoio, o mais próximo possível da linha de contato, para maximizar a sobrevivência e o retorno à linha de fogo.
- Mobilidade e Evoz: O sistema de saúde deve ser tão ágil quanto a força de manobra. O apoio estático é apoio vulnerável.
- Triagem como Decisão Ética e Tática: Em situações de baixas em massa (MASCAL), a triagem rápida e imparcial é a única ferramenta capaz de otimizar recursos limitados.
- Inteligência Médica (AJMEDP-3): Antecipar ameaças ambientais e mapear a infraestrutura civil local é essencial para a resiliência logística em teatros de operações isolados.
- Unicidade da Cadeia Logística: Do suprimento de sangue à evacuação aeromédica, a logística de saúde deve ser tratada como um sistema unitário e ininterrupto.
7. A Missão Continua
A Medicina Operacional é uma ciência de adaptação, coragem e sacrifício. Do cirurgião que operava sob o frio da Berezina ao oficial médico que planeja o apoio em um Pelotão Especial de Fronteira na Amazônia, o compromisso com a vida do combatente permanece o mesmo. A logística de saúde é o sistema circulatório da força; seu sucesso garante que o Brasil disponha de militares prontos, resilientes e protegidos.
Leitura Recomendada
- BRASIL. Ministério da Defesa. MD42-M-04: Logística de Saúde em Operações Conjuntas. Brasília: MD, 2015.
- GABRIEL, R. A.; METZ, K. S. A History of Military Medicine. New York: Greenwood Press, 1992.
- LARREY, Dominique Jean. Mémoires de chirurgie militarie et campagnes. Baltimore: Joseph Cushing, 1814.
- MCCALLUM, Jack E. Military Medicine: From Ancient Times to the 21st Century. Santa Barbara: ABC-CLIO, 2008.
- MITCHELL, Piers D. Trauma and Surgery in the Crusades. London: University of London, 2002.
- ORGANIZAÇÃO DO TRATADO DO ATLÂNTICO NORTE (NATO). AJMEDP-3: Allied Joint Medical Doctrine for Medical Intelligence. Brussels: NATO Standardization Office, 2015.
- SOUBIRAN, A. Le Baron Larrey: Chirurgien de Napoléon. Paris: Fayard, 1966.
- VAYRE, P.; FERRANDIS, J. J. Dominique Larrey (1766-1842), Chirurgien militaire – Baron d’Empire. e-mémoires de l’Académie Nationale de Chirurgie, v. 3, n. 1, p. 37-46, 2004.
Comunicação Cirúrgica
Padrões, Tensões e Implicações para a Segurança do Paciente na Cirurgia Digestiva
Introdução
A comunicação cirúrgica constitui elemento fundamental da prática operatória contemporânea. Mais do que mera troca de informações técnicas, ela representa o mecanismo central de coordenação relacional entre cirurgião, anestesista, instrumentador, circulante e demais membros da equipe. Na cirurgia digestiva — campo marcado por procedimentos de alta complexidade, como gastrectomias, hepatectomias e colectomias — a qualidade da comunicação influencia diretamente os desfechos clínicos, a formação de residentes e a cultura de segurança do paciente.
Estudos observacionais e revisões sistemáticas recentes demonstram que falhas comunicativas contribuem para mais de 70% dos eventos sentinelas em saúde, conforme dados da The Joint Commission. Na sala de operação, análises de 421 interações revelaram falhas em aproximadamente 30% das trocas entre membros da equipe, gerando ineficiência, tensão e risco aumentado ao paciente (Dingley et al., 2008). Neste artigo examinaremos algunmas evidências científicas sobre o tema, com ênfase em três investigações paradigmáticas, visando oferecer subsídios educativos para cirurgiões digestivos e equipes multidisciplinares.
Padrões de Comunicação Observados em Equipes Cirúrgicas
Pesquisa etnográfica conduzida por Tørring et al. (2019), que acompanhou 39 equipes cirúrgicas (85 profissionais) durante substituições de quadril e joelho — procedimentos com paralelos diretos à cirurgia digestiva —, identificou quatro padrões distintos de interação:
- Comunicação proativa e intuitiva: presente em cirurgias complexas, caracterizada por compartilhamento de objetivos, respeito mútuo e antecipação de problemas.
- Comunicação silenciosa e comum: típica de procedimentos rotineiros, marcada por instruções breves e atmosfera de familiaridade.
- Comunicação desatenta e ambígua: marcada por foco individual, tom ambíguo e, por vezes, ironia ou desrespeito explícito.
- Comunicação contraditória e altamente dinâmica: oscilações rápidas entre respeito e rispidez, levando muitos profissionais ao silêncio estratégico.
Esses padrões ilustram que a comunicação eficaz transcende palavras: envolve linguagem corporal, tom de voz e alinhamento de expectativas coletivas.
Locais de Tensão Comunicativa e Seus Efeitos Formativos
Lingard et al. (2002), em estudo com 128 horas de observação em 35 procedimentos cirúrgicos, mapearam cinco temas recorrentes de tensão: tempo, segurança/esterilidade, recursos (equipamentos), papéis da equipe e controle da situação. Tais tensões surgem com frequência entre cirurgião sênior e equipe de enfermagem, impactando diretamente os novatos. Os residentes e estudantes reagem predominantemente de duas formas: imitação (mimicry) do estilo autoritário ou retirada (withdrawal), silenciando-se. Ambas as respostas perpetuam ciclos de conflito interprofissional em vez de promover resolução colaborativa. Na cirurgia digestiva, onde o tempo de isquemia, sangramento e integridade anastomótica são críticos, tais dinâmicas podem comprometer a qualidade assistencial e o aprendizado.
Aspectos Emocionais e o Clima da Sala Cirúrgica
Revisão de escopo realizada por Lee et al. (2023), que analisou 10 estudos qualitativos e quantitativos, reforça que o centro cirúrgico é ambiente emocionalmente carregado. O tom e a atitude do líder — geralmente o cirurgião — definem o “clima” da sala. Cultura hierárquica e estresse inibem o speaking up, especialmente entre enfermeiros e residentes. Quando o cirurgião manifesta frustração por meio de gritos ou culpas, instalam-se medo, redução da moral e falhas comunicativas que elevam o risco de eventos adversos. A revisão destaca que o controle emocional não é aspecto acessório, mas competência clínica essencial. Em cirurgias digestivas de grande porte, onde a pressão temporal e a complexidade técnica são elevadas, a regulação emocional do cirurgião torna-se fator protetor da segurança do paciente.
Implicações Práticas para a Cirurgia Digestiva Atual
A integração dos achados demonstra que competência técnica isolada é insuficiente. O cirurgião digestivo contemporâneo deve cultivar inteligência emocional e habilidade retórica para fomentar coordenação relacional. Estratégias como briefing pré-operatório, debriefing estruturado e treinamento em comunicação não-técnica (CRM — Crew Resource Management) reduzem significativamente as falhas identificadas nos estudos citados. Na prática diária, reconhecer os padrões descritos por Tørring, mitigar tensões mapeadas por Lingard e gerir o clima emocional destacado por Lee constitui caminho direto para melhores desfechos, menor burnout da equipe e formação mais robusta de novos cirurgiões.
Conclusão
A comunicação cirúrgica não é mero instrumento operacional; é o alicerce da segurança do paciente e da excelência assistencial na cirurgia digestiva. Os estudos analisados evidenciam que padrões comunicativos saudáveis, gestão de tensões e controle emocional reduzem riscos, otimizam processos e fortalecem o desenvolvimento profissional. Investir nesse domínio representa avanço científico e ético irrenunciável.
“A cirurgia não é um desempenho individual, mas um trabalho de equipe.” Sir Berkeley Moynihan (1865-1936)
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Prof. Dr. Ozimo Gama
Cirurgião do Aparelho Digestivo
Editor Científico – The Surgeon: A Cirurgia Digestiva Atual
Engenharia de Prompt na Educação Médica
Dissecando a Nova Fronteira Tecnológica na Cirurgia Digestiva
1. A Evolução do Estetoscópio ao Algoritmo
A prática médica contemporânea atravessa uma metamorfose digital que exige mais do que a simples adaptação: exige o domínio da técnica. A ascensão dos Modelos de Linguagem Gerativa (GLMs), como o ChatGPT e o Google Bard, não representa apenas a disponibilidade de novos bancos de dados, mas sim uma mudança sísmica na síntese do conhecimento clínico. Neste contexto, a Engenharia de Prompt emerge como a competência estratégica definitiva para o médico do século XXI. Conforme definido por Heston e Khun (2023), trata-se de uma abordagem sistemática de comunicação com IAs para obter resultados de alta precisão. Não se trata de uma conversa informal, mas de uma instrumentação precisa da inteligência artificial.
Para compreendermos o “salto qualitativo” desta ferramenta, basta observar a evolução técnica: o GPT-1 (2018) operava com 117 milhões de parâmetros; o GPT-4 (2023) é estimado em 1 trilhão. Essa expansão colossal mudou o paradigma da simples busca por informação (o modelo “indexador”) para a síntese complexa de conhecimento. Para o cirurgião, isso significa que a IA deixou de ser um dicionário para se tornar um consultor de lógica, exigindo que a instrução dada pelo médico seja tão precisa quanto o plano de clivagem em uma dissecção oncológica.
Esta transição da intuição para a estrutura técnica é o que separa o uso recreativo da IA da sua aplicação acadêmica e clínica de alto nível.
2. A Anatomia de um Prompt de Alta Precisão: Estrutura e Componentes
Na sala de operação, a improvisação é o terreno do erro; na Engenharia de Prompt, a vagueza é a raiz da alucinação. Estruturar uma entrada para a IA não é meramente fazer uma pergunta, mas sim redigir um protocolo de intenção. Podemos comparar a precisão de um prompt bem construído ao rigor técnico necessário em uma colecistectomia: cada componente tem seu “tempo cirúrgico” e função específica.
Para garantir resultados consistentes, um prompt estruturado deve conter quatro pilares fundamentais:
- Contexto: A descrição de quem está perguntando (ex: “Sou um preceptor de residência em cirurgia digestiva”).
- Solicitação Geral: O objetivo macro da interação (ex: “Necessito de uma revisão de condutas”).
- Persona/Papel: Como a IA deve se comportar (ex: “Atue como um Professor Titular de Gastroenterologia Cirúrgica”).
- Formato de Saída: A estrutura final do dado (ex: “Gere uma lista de verificação,” “tabela comparativa,” ou “resumo em tópicos”).
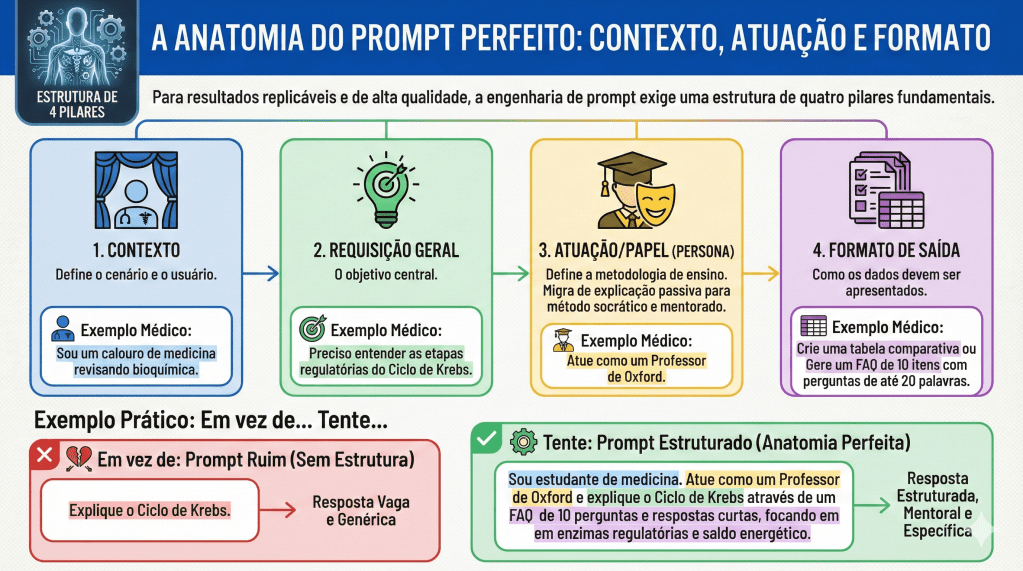
Comparativo de Eficiência: Prompts Vagos vs. Alta Precisão
| Tipo de Prompt | Exemplo Vago | Exemplo de Alta Precisão (Padrão Ouro) |
| Objetivo | Informação Geral | FAQ de Estadiamento Acadêmico |
| Entrada | “Fale sobre câncer gástrico.” | “Como Professor Doutor de Cirurgia, forneça um FAQ de 10 itens sobre o estadiamento do adenocarcinoma gástrico para residentes do R3. Foque no TNM 8ª edição, critérios de ressecabilidade e manejo perioperatório do paciente bariátrico. Entregue em formato de tabela.” |
| Resultado | Texto genérico e superficial. | Guia técnico, hierarquizado e pronto para discussão em round clínico. |
Essa estrutura permite que a IA realize uma triagem interna em seu vasto banco de parâmetros, ajustando o nível de complexidade para a realidade do interlocutor, seja ele um estudante de graduação ou um cirurgião experiente.
3. Níveis de Complexidade: Do Zero-Shot à Decomposição Lógica
No cotidiano acadêmico, a sofisticação da resposta da IA é diretamente proporcional à técnica de prompting utilizada. Podemos escalonar a interação em níveis que desafiam a capacidade de processamento do modelo:
- Zero-Shot vs. Few-Shot: O Zero-Shot é uma solicitação sem exemplos prévios. O Few-Shot é o fornecimento de modelos de comportamento dentro do prompt (ex: “Crie um caso clínico de isquemia mesentérica seguindo rigorosamente este modelo: [Exemplo]”).
- Hierarquia de Lawton (Níveis 1 a 4): Evoluímos de perguntas simples (Nível 1) até o Nível 4, definido como a desconstrução de solicitações complexas em componentes lógicos (Chain of Thought).
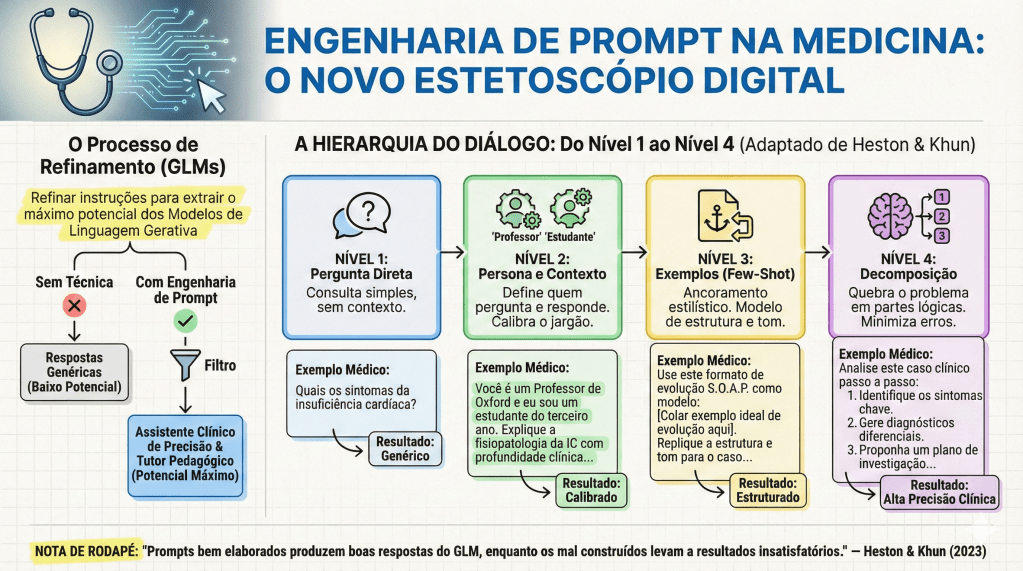
O impacto clínico mais relevante desta última técnica, ativada pelo comando “Pense passo a passo” (Think step by step), é a mitigação de erros catastróficos. Um exemplo clássico do texto de Heston e Khun (2023) ilustra isso: ao perguntar sobre um malabarista que tem 16 bolas, sendo que metade são bolas de golfe e metade das bolas de golfe são azuis, o GPT-3.0 pode falhar se responder instantaneamente. Ao ser instruído a pensar passo a passo, a IA decompõe o problema: total (16) -> metade são golfe (8) -> metade das de golfe são azuis (4). No ambiente cirúrgico, essa lógica é o que previne a iatrogenia em cálculos farmacológicos complexos ou em lógicas de estadiamento multinível.
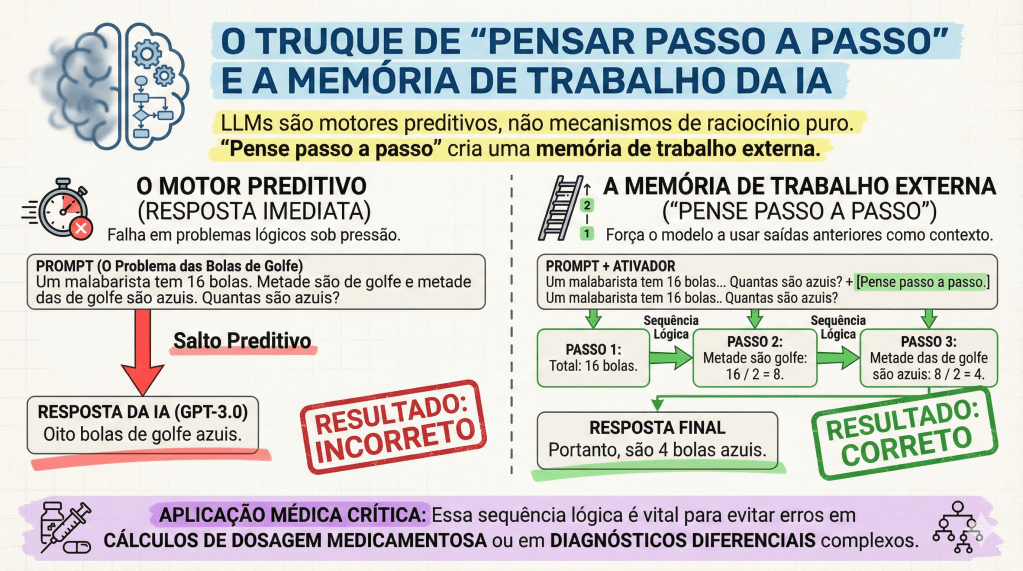
A eficácia do aprendizado, portanto, não reside em receber a resposta pronta, mas em observar o “raciocínio” da máquina, permitindo ao estudante identificar falhas lógicas e consolidar o conhecimento.
4. Aplicação no Ensino-Aprendizado e a Realidade Brasileira (Enare/Enamed)
No cenário brasileiro, marcado pela altíssima competitividade do Enare (Exame Nacional de Residência) e do Enamed, a IA deve ser encarada como um “cérebro periférico”. A densidade do conteúdo cirúrgico exige ferramentas que otimizem a densidade temporal do estudo.
Aplicações Práticas para o Estudante de Alto Desempenho:
- Mnemônicos Cirúrgicos: Criação de fórmulas personalizadas para critérios de gravidade em pancreatite aguda ou escalas de Alvarado.
- Simulação de Pacientes Realistas: Uso de GLMs para simular “Virtual Patients”, permitindo que o estudante treine a anamnese antes do contato real (conforme princípios da AAMC).
- Bancos de Questões De Novo: Geração de simulados inéditos baseados nos exames anteriores do MEC.
A realidade curricular está mudando drasticamente. Segundo dados do Curriculum SCOPE Survey (AAMC), houve um crescimento acentuado na inserção de IA nos currículos das escolas médicas entre 2023 e 2024. No Brasil, essa tendência impacta diretamente a competitividade: o candidato que domina a Engenharia de Prompt processa evidências científicas com uma agilidade que os métodos tradicionais de leitura passiva não conseguem acompanhar. Contudo, este potencial exige uma advertência: a tecnologia deve fortalecer a mente, não atrofiar o raciocínio clínico original.
5. Riscos, Alucinações e a Ética na Medicina Digital
A precisão cirúrgica exige o reconhecimento dos riscos. O maior perigo dos GLMs são as “alucinações”, onde a IA gera informações falsas com um tom de autoridade inquestionável, incluindo referências bibliográficas totalmente inventadas no formato Vancouver.
Checklist de Segurança de Dados e Integridade:
- Verificação de Citações: Jamais utilize uma referência gerada sem conferir o DOI ou a base primária.
- Privacidade: É imperativo nunca inserir dados identificáveis de pacientes reais em prompts de IA.
- Senso Crítico: O cirurgião nunca deve abdicar de sua responsabilidade ética em favor do algoritmo.
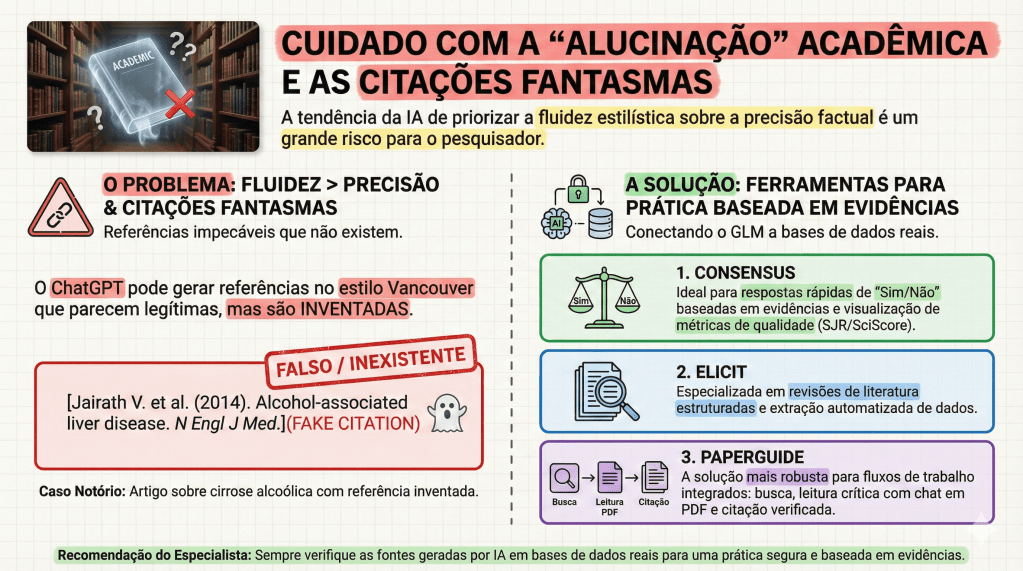
Para o fluxo de trabalho do pesquisador, é vital diferenciar as ferramentas. Modelos de chat puro (como o ChatGPT) são excelentes para síntese, mas para revisões sistemáticas, deve-se integrar ferramentas de evidência real como Elicit e Consensus. Especial destaque deve ser dado ao Paperguide: uma ferramenta “all-in-one” que, além de busca semântica, oferece suporte a múltiplos idiomas — uma vantagem estratégica para o estudante brasileiro que precisa transitar entre a literatura internacional e a prática local. O uso institucional da IA deve, portanto, ser pautado por uma cultura de ética inegociável.
6. Conclusões e Perspectivas Futuras
A Engenharia de Prompt deixará de ser um diferencial para se tornar um requisito básico, tão essencial ao cirurgião digestivo quanto o domínio da estatística básica ou do inglês técnico. Ela é a ponte entre a explosão de dados médicos e a aplicação prática à beira do leito. A mensagem central é clara: a IA deve ser utilizada para fortalecer a compreensão humana, mitigando a falha e ampliando o cuidado, sem jamais criar dependência ou o que chamamos de “pensamento fraco”. O futuro da nossa especialidade pertence àqueles que souberem interrogar a realidade — e a inteligência artificial — com a maior clareza e precisão.
#EngenhariaDePrompt #EducacaoMedica #CirurgiaDigestiva #IAnaMedicina #ResidenciaMedica
——————————————————————————–
“O sucesso da educação médica não reside apenas na transferência de fatos, mas na formação de uma mente capaz de interrogar a realidade com precisão.” — Adaptado dos princípios de William Osler
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
A Cirurgia Digestiva Atual
Autoridade Cirúrgica
A Prerrogativa da Autoridade Cirúrgica sob a Égide da Incerteza Fiscal: Uma Reflexão sobre a Evolução do Prestígio e a Prática Contemporânea
Por: Prof. Dr. Ozimo Gama
O Conceito de Autoridade e a Evolução do Prestígio Médico
O prestígio da profissão médica, historicamente alicerçado em um modelo de prática liberal e em um paternalismo inquestionável, atravessa uma metamorfose estrutural profunda. Sob a égide da contemporaneidade, o cenário de autonomia plena foi substituído por um ambiente de pressões administrativas e incerteza fiscal. Como salientou o Dr. Jesse M. Ehrenfeld, ex-presidente da American Medical Association (AMA), a transição das práticas independentes para os sistemas corporativos é emblemática do estresse econômico enfrentado pela classe. Urge salientar que essa evolução é indissociável da realidade financeira da formação médica. Enquanto na década de 1980 — como recorda o Dr. Weigel — o custo total de quatro anos de medicina girava em torno de US32.000, o cirurgião hodierno egressa com um endividamento médio de US 246.659, podendo ultrapassar os US$ 300.000 em instituições privadas. Concomitantemente, a “Autoridade Cirúrgica” deve ser ressignificada: ela não representa mero poder hierárquico, mas uma responsabilidade derivada da expertise técnica e de um contrato de confiança renovado, essencial para a subsistência ética da especialidade.
Desafios Econômicos e a Realidade da Prática Médica
A análise da medicina atual revela uma insatisfação latente quanto à valorização profissional. Segundo o Medscape Physician Compensation Report 2025, a remuneração média subiu apenas 2,9%, acompanhando parcamente a inflação, enquanto 52% dos médicos manifestam não se sentirem compensados de forma justa. A disparidade entre gerações é notória: em 1985, a vasta maioria dos médicos era proprietária de seu próprio negócio, gerindo equipes e processos. Hoje, o cirurgião é, majoritariamente, um elo em grandes sistemas de saúde, onde a autonomia administrativa é mitigada por métricas corporativas. Os principais vetores de estresse e erosão do prestígio incluem:
• Incerteza Fiscal e Econômica: Oscilações que dificultam o planejamento de carreira a longo prazo.
• Cortes Estatutários de Pagamento: Reduções em sistemas de reembolso (como o Medicare) que não acompanham a complexidade dos atos.
• Burocracia Administrativa e Custos Operacionais: Uma carga intrusiva que desvia o cirurgião de sua atividade fim, exacerbada pelo aumento constante dos custos de manutenção das práticas.
A Autoridade na Cirurgia do Aparelho Digestivo: Expertise e Autonomia
Dentro da hierarquia nosocomial, a Cirurgia do Aparelho Digestivo preserva um “clout” (influência política e social) diferenciado. No entanto, é necessário compreender que esse poder político, como observa Barnett, está profundamente enraizado em um sistema de reembolso não baseado estritamente na saúde da população, mas na valorização de procedimentos de alta complexidade que geram receita crítica para os sistemas hospitalares. A fundamentação da autoridade do cirurgião repousa sobre sua “expertise esotérica”. Jenkins, em suas pesquisas, demonstrou que cirurgiões que operam neonatos de 500g ou realizam duodenopancreatectomias complexas desfrutam de um nível de confiança pública superior. Diferente do pediatra geral, que muitas vezes enfrenta questionamentos constantes sobre condutas rotineiras — como a segurança de vacinas —, o cirurgião digestivo lida com situações de risco vital onde as decisões são técnicas e imediatas. Essa especificidade confere maior autonomia organizacional e confiança do paciente, resultando em um paradoxo interessante: embora o cirurgião trabalhe mais horas e em maior intensidade, os índices de burnout tendem a ser menores do que em especialidades menos procedimentais. A valorização social e a percepção de utilidade crítica atuam como um escudo psicossocial para o especialista.
A Nova Configuração do Contrato Médico-Paciente
O declínio do modelo “Marcus Welby” de paternalismo médico deu lugar a uma era de consumismo em saúde e ceticismo científico, exacerbada pela democratização desenfreada da informação via internet. O contrato médico, outrora um pacto sagrado entre indivíduos, foi reescrito pela burocracia e pelo big business. A autoridade contemporânea deve, portanto, acolher a diversidade e a inclusão como pilares de sua legitimidade. O prestígio moderno não se mede mais pela deferência cega, mas pela busca de equilíbrio, sustentabilidade e autopreservação do profissional.
Aplicação na Prática e Pontos-Chave
No cenário brasileiro, o desafio é análogo. O endividamento do recém-formado em instituições privadas pode variar entre R500.000 e R 800.000, criando uma pressão financeira imediata. Com aproximadamente 5.000 especialistas titulados em Cirurgia do Aparelho Digestivo, enfrentamos uma severa saturação nos grandes centros, o que demanda uma gestão de carreira mais estratégica e técnica.
Pontos-Chave para o Residente e Pós-Graduando:
• Expertise Técnica: O domínio de procedimentos complexos e de alto risco é o fundamento de sua soberania clínica e influência hospitalar.
• Gestão da Autonomia: É imperativo aprender a navegar em sistemas corporativos preservando a integridade da decisão médica frente às pressões administrativas.
• Relação de Confiança: Substitua o paternalismo obsoleto por uma transparência baseada na erudição técnica, gerenciando as expectativas do paciente-consumidor.
• Sustentabilidade da Carreira: A longevidade profissional depende de uma prática que valorize a autopreservação e o equilíbrio ético-financeiro.
Em última análise, a missão do cirurgião permanece como um chamado de serviço. Devemos retomar a visão de Weigel, sentindo-nos como “pastores de nosso rebanho”, responsáveis diretos por vidas que nos são confiadas em momentos de extrema vulnerabilidade. A medicina evoluiu, e o prestígio agora exige mais do que títulos; exige resiliência e adaptação. Como preconizou Theodor Billroth, mestre da cirurgia digestiva:
“O cirurgião deve ter o coração de um leão e as mãos de uma dama.”
A autoridade reside, precisamente, na virtude de equilibrar a força da decisão técnica com a delicadeza do compromisso ético.
The Surgeon – A Cirurgia Digestiva Atual – Et Fortior
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#CirurgiaDigestiva #ResidenciaMedica #AutoridadeCirurgica #EducaçãoMédica #TheSurgeon
Estilo e Brio Cirúrgico
A Ciência da Comunicação Não Verbal na Liderança Cirúrgica
1. A Ontologia do Brio no Bloco Operatório
No teatro de alta complexidade que caracteriza a cirurgia do aparelho digestivo, a excelência técnica, embora imperativa, é apenas o estrato basal da competência. O que verdadeiramente distingue o mestre do técnico é o “Surgical Panache” — ou brio cirúrgico. Longe de ser um adereço estético, o brio é uma competência transformadora fundamentada na ciência da comunicação não verbal. A literatura contemporânea em liderança cirúrgica e comportamento organizacional sublinha que os seres humanos possuem um imperativo biológico para processar mensagens não verbais com extrema celeridade, realizando julgamentos sociais eficientes antes mesmo da primeira incisão. A eficácia de um líder depende da congruência: o alinhamento absoluto entre o rigor do discurso técnico e a presença executiva. É fundamental desmistificar a interpretação vulgar do modelo de Mehrabian: embora as palavras sejam o veículo das instruções técnicas, o canal não verbal domina 93% do clima emocional e da percepção de confiança da equipe. Sem essa harmonia, a confiança organizacional se dissolve, e a comunicação ineficaz manifesta-se não como um erro acessório, mas como uma complicação clínica gravíssima.
2. A Dualidade da Liderança: Receptividade e Formidabilidade
A mobilização do contingente humano em procedimentos complexos exige o domínio da teoria dos sinais de status, conforme a estrutura de Keating. O cirurgião deve projetar uma imagem binária que ative simultaneamente dois sistemas motivacionais na equipe:
• Receptividade (Calor e Atratividade): Sinaliza abertura e convite à colaboração (sistema de approach). É a vertente que garante a segurança psicológica, permitindo que um instrumentador ou anestesista comunique intercorrências sem temor de retaliação.
• Formidabilidade (Competência e Poder): Transmite agência, autoridade e domínio técnico (sistema de avoidance). É a projeção de distância profissional necessária para manter a hierarquia e o rigor em momentos críticos.
O carisma cirúrgico reside na capacidade de transitar entre esses sinais duplos. Um líder puramente formidável silencia a equipe; um líder exclusivamente receptivo compromete o tempo operatório. A maestria está em ser, simultaneamente, um porto seguro e uma autoridade inquestionável.
3. Kinésica e Oculésica: A Arquitetura da Autoridade e Espaço
A taxonomia dos sinais não verbais oferece diretrizes práticas para o refinamento da postura operatória:
• Kinésica (Postura e Gestual): A adoção de posturas expansivas comunica controle. De acordo com o estudo de Holler e Beatie, o uso de gestos intencionais e fluidos aumenta o valor da mensagem falada em 60%. Deve-se eliminar comportamentos nervosos (fidgeting), que corroem a credibilidade.
• Oculésica (O Olhar): O olhar é o regulador supremo da interação. Aplico rigorosamente a regra de manter contato visual durante 50% do tempo ao falar e 70% ao ouvir. Isso estabelece persuasão e engajamento, permitindo “ler o ar” da sala.
• Vocalização (Vocalics): A gestão paralinguística é vital. Em uma intercorrência hemorrágica durante uma duodenopancreatectomia, o cirurgião deve contra-atacar a sobrecarga do sistema nervoso simpático da equipe através de uma voz de tom grave, volume controlado e ritmo pausado. A calma do líder deve ser auditível para ser contagiante.
4. Aplicação na Cirurgia Digestiva e a Realidade Brasileira
A segurança do paciente é o objetivo teleológico de toda comunicação. Estatísticas globais revelam que 86% das falhas no ambiente de trabalho são atribuídas à comunicação ineficaz. Na cirurgia do aparelho digestivo, onde procedimentos como esofagectomias demandam sincronia absoluta, esse dado é alarmante. No contexto brasileiro, classificado como uma cultura de alto contexto (High-Context), a habilidade de interpretar pistas sutis e nuanças não verbais é ainda mais vital do que em ambientes anglo-saxões ou germânicos. No Brasil, a liderança é exercida através do relacionamento e da percepção de brio; o silêncio de um assistente pode significar uma dúvida técnica que o cirurgião deve ser capaz de decifrar visualmente. Ademais, a educação médica continuada em 2026 exige o domínio da “presença digital”. Em boards clínicos virtuais, a Chronemics (gestão do tempo) e a pontualidade tornaram-se indicadores de prioridade organizacional. O enquadramento da câmera deve ser preciso, mantendo o olhar na lente e as mãos visíveis para preservar a transparência e a autoridade virtual.
5. Pontos-Chave para a Prática do Cirurgião Digestivo
Para os alunos, residentes e pós-graduandos sob minha orientação, estabeleço as seguintes prerrogativas:
1. Domínio do Quadro: “Seja dono da sala”. Use posturas expansivas e movimentos deliberados para sinalizar que o ambiente está sob controle técnico e emocional.
2. Estratégia Dual: Não busque ser apenas amado ou apenas temido. Seja formidável na competência e receptivo na colaboração.
3. Monitoramento Ativo: Realize a “análise muda” de suas performances. Assista a vídeos de suas cirurgias sem áudio para identificar vícios gestuais e falhas de congruência.
4. Inteligência Cultural: Adapte sua comunicação ao estrato da equipe. Em culturas de alto contexto, o brio cirúrgico é o que preenche as lacunas do que não foi dito, mas precisa ser compreendido.
6. O Cirurgião como Líder Corporificado
O brio não é um dom estático, mas uma disciplina dinâmica refinada pela autoconsciência e pelo rigor. Projetar uma presença física autêntica, confiante e empática é o diferencial definitivo para a liderança de alto impacto. O cirurgião que negligencia sua comunicação não verbal é apenas um executor de técnicas; o cirurgião que domina seu brio é um comandante de destinos, capaz de tratar a falha de comunicação com o mesmo rigor científico com que trata uma fístula anastomótica.
——————————————————————————–
“O cirurgião deve ter mãos de dama, olhos de águia e coração de leão; contudo, é a sua presença que confere à equipe a têmpera necessária para a vitória sobre a patologia.” — Baseado no legado de William Stewart Halsted

Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
The Surgeon A Cirurgia Digestiva Atual Et Fortior
#CirurgiaDigestiva #BrioCirurgico #LiderançaMedica #EducacaoMedica #TheSurgeon
Segurança em Foco
A Evolução da Cultura de Segurança e o Desafio da Burocratização na Cirurgia do Aparelho Digestivo
1. A Gênese do Risco na Medicina e Cirurgia
A gênese da prática cirúrgica moderna está intrinsecamente vinculada a uma audácia que, aos olhos contemporâneos, beira a temeridade. Em seus primórdios, a medicina não dispunha de comitês de ética, protocolos de conduta ou salvaguardas administrativas; o progresso era esculpido em tempo real, frequentemente à beira do leito e sob um custo humano excruciante. A introdução de tecnologias disruptivas como o éter e o clorofórmio exemplifica essa tensão dialética: embora tenham viabilizado a revolução do ato operatório, enfrentaram resistências ferrenhas sob o pretexto de serem substâncias perigosas e incontroláveis. Esse padrão de rejeição institucional repetiu-se com Ignaz Semmelweis, cuja demonstração empírica da eficácia da higiene das mãos foi ignorada por desafiar identidades profissionais e normas vigentes. Aquela era, magistralmente retratada na série The Knick, exibe uma medicina criativa e heróica, porém caótica e, por vezes, deletéria. Aprendemos, por meio de um processo histórico doloroso, que a inovação desmedida gera danos, mas reconhecemos que, naquele período de obscuridade técnica, a assunção do risco era o único vetor possível para o avanço da ciência.
2. O Surgimento da Segurança como Bússola Moral
Como resposta necessária ao caos da era heróica, a segurança emergiu como a bússola moral da medicina do século XX. A transição para a era da padronização e dos protocolos buscou transformar a arte cirúrgica em uma ciência de alta confiabilidade. O advento do Checklist Cirúrgico consolidou-se como o marco definitivo desta evolução, mitigando erros catastróficos, como intervenções em locais incorretos, e reduzindo drasticamente as complicações evitáveis. A segurança estabeleceu-se como nossa âncora ética sob o aforismo Primum non nocere. Contudo, observamos hoje que este preceito tem sido distorcido: em vez de guiar a prudência clínica, é frequentemente invocado para justificar a inação diante de riscos calculados e necessários. A medicina não deve ser a busca utópica pela anulação do risco a qualquer custo, mas sim o sopesamento rigoroso entre danos e benefícios, sempre pautado pela soberania dos valores do paciente.
3. Quando a Segurança se Torna Burocracia
Assistimos, na atualidade, a uma patologia sistêmica de ordem administrativa: a “fossilização” da segurança em estruturas burocráticas estáticas. O que deveria proteger o paciente transmutou-se em mecanismos de proteção institucional e conformidade legal. Um sintoma alarmante dessa realidade foi o caso de um residente sênior norte-americano que, a seis meses de sua graduação, jamais havia realizado uma drenagem de tórax de forma autônoma — um reflexo direto de sistemas que priorizam a ausência de intercorrências administrativas em detrimento da formação de competência técnica real. No cenário brasileiro, essa estagnação é agravada por infraestruturas hospitalares rígidas e prontuários eletrônicos (EHRs) deficientes, que impõem as seguintes consequências:
• Dreno de Intelecto: Residentes de Cirurgia do Aparelho Digestivo e de Terapia Intensiva gastam, em média, 50% de sua jornada diária em tarefas clericais e documentação redundante, sacrificando o raciocínio clínico e o treinamento em técnica cirúrgica.
• Engessamento da Pesquisa: Processos de governança para estudos de baixo risco podem levar meses ou anos para aprovação, priorizando a prevenção de riscos hipotéticos sobre o aprendizado derivado de pacientes reais.
• Desvio de Finalidade: A documentação clínica passou a servir mais ao faturamento e à auditoria do que à comunicação entre a equipe e ao cuidado direto.
4. O “Stress Test” da Inteligência Artificial (IA) na Prática Cirúrgica
A Inteligência Artificial atua hoje como um teste de estresse que revela as falhas de governança do nosso sistema de saúde. A barreira para a adoção da IA não é de ordem técnica, mas sim uma incapacidade de adaptação das instâncias decisórias. O paradoxo é flagrante: algoritmos de IA são prontamente implementados quando o objetivo é otimizar códigos de faturamento e captura de receita. No entanto, ferramentas de IA como escribas clínicos, que reduzem a carga documental, ou sistemas de suporte ao raciocínio clínico e triagem de imagens, enfrentam barreiras instransponíveis sob o pretexto da privacidade — ainda que sejam mais seguros do que os “atalhos” improvisados por cirurgiões em seus dispositivos pessoais para contornar sistemas hospitalares obsoletos. A resistência atual é uma falha de governança disfarçada de segurança do paciente.
5. Aplicação Prática na Cirurgia Digestiva
Para o cirurgião do aparelho digestivo em formação, o conceito de “Custo do Atraso” deve ser central. Estatísticas demonstram que, em média, transcorrem 14 anos para que uma evidência científica consolidada chegue efetivamente ao leito do paciente. Para um residente que inicia sua trajetória hoje, esse hiato significa que ele poderá estar na metade de sua carreira antes de dominar as inovações que já estão disponíveis agora. É imperativo compreender que a inação não é um porto seguro; ela possui consequências invisíveis, mas mensuráveis. A espera por uma segurança absoluta e estática é, em si, uma forma de dano. Na residência médica e na pós-graduação, a inovação tecnológica deve ser integrada não como um acessório, mas como um pilar da segurança do paciente e da eficiência clínica.
6. Pontos-Chave para o Residente e Pós-Graduando
1. A segurança deve ser adaptativa, não estática: Protocolos que não evoluem com a tecnologia tornam-se obstáculos ao exercício da excelência cirúrgica.
2. Inação também gera dano: O perigo invisível da espera e o atraso na adoção de novas técnicas são formas reais de prejuízo ao paciente.
3. O cirurgião como avaliador e tradutor: O cirurgião não é um usuário passivo, mas um filtro crítico que absorve incertezas para que o sistema de saúde possa aprender e evoluir com segurança.
7. Perspectivas Futuras
A segurança que não evolui torna-se, inexoravelmente, insegura. O desafio da Cirurgia Digestiva contemporânea é resgatar a coragem intelectual de nossos predecessores, temperada pelo rigor científico atual. Devemos atuar como early adopters responsáveis, integrando novas ferramentas de forma monitorada e contínua. A tradição, que outrora serviu para proteger o enfermo, não pode ser reduzida a um mecanismo que protege as instituições contra a mudança. Somente através de uma cultura de aprendizado contínuo e da superação da burocracia fóssil poderemos assegurar que a próxima geração de cirurgiões esteja plenamente capacitada para os desafios de sua era.
Como bem asseverou Lord Lister, o mestre da antissepsia: “O sucesso de uma operação depende da atenção cuidadosa a cada detalhe.” Que essa atenção aos detalhes transcenda o campo operatório e alcance a gestão crítica da tecnologia e dos sistemas que regem nossa profissão. O zelo pelo paciente exige, agora mais do que nunca, zelo pela evolução do sistema.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
The Surgeon A Cirurgia Digestiva Atual Et Fortior
#CirurgiaDigestiva #SegurancaDoPaciente #EducacaoMedica #IAnaMedicina #ResidenciaMedica
A Revolução da Inteligência Artificial no Aprendizado e na Pesquisa em Cirurgia do Aparelho Digestivo
1. O Novo Paradigma da Educação Médica
A evolução da Inteligência Artificial (IA) no seio da medicina contemporânea transcendeu a mera aplicação subsidiária em diagnósticos por imagem para consolidar-se como um pilar epistemológico na pesquisa e no aprendizado. O cirurgião moderno defronta-se com um desafio multifacetado, por vezes sísifo: a imperiosa necessidade de conciliar uma extenuante carga operatória com a produção científica de alto impacto e a gestão de um volume oceânico de literatura especializada. Neste cenário, a Inteligência Artificial Generativa (GenAI) e os Modelos de Linguagem de Grande Escala (LLMs) emergem não como substitutos do intelecto, mas como catalisadores de um fluxo de trabalho otimizado. É fundamental compreendermos que a maestria cirúrgica hoje exige, além da destreza manual, a competência digital para navegar nesta nova fronteira tecnológica com idoneidade e rigor acadêmico.
2. Ferramentas de IA e Categorização Funcional
A fundamentação científica para a adoção dessas tecnologias repousa em evidências recentes e robustas. O estudo seminal de Valencia-Coronel et al. (2026) realizou uma avaliação qualitativa rigorosa de 43 plataformas de IA, fornecendo um arcabouço para a seleção criteriosa de ferramentas no pipeline da pesquisa cirúrgica.
Dentre as categorias funcionais identificadas, destacam-se:
• Mecanismos de Busca Acadêmica: Superando os buscadores genéricos, plataformas como Consensus AI, Elicit e EvidenceHunt focam na síntese de evidências baseadas em revisão por pares. Ressalto a relevância do Manus AI, projetado especificamente para lidar com tópicos cirúrgicos complexos e desfechos heterogêneos, auxiliando inclusive na estruturação de protocolos e tabelas prontas para publicação.
• Interação com Documentos: Ferramentas como AskYourPDF e ChatPDF permitem um diálogo heurístico com manuscritos complexos, facilitando a extração de dados técnicos e a análise metodológica profunda.
• Análise de Literatura: A visualização de redes de citações através de algoritmos (ex: Connected Papers e Litmaps) permite mapear a trajetória de um tema e identificar conexões semânticas fundamentais.
• Assistentes de Escrita e Síntese: Modelos como ChatGPT (em sua versão GPT-4) e ClaudeAI auxiliam na estruturação de rascunhos. Para sínteses profundas, o modo “Deep Research” do Perplexity AI destaca-se por realizar varreduras exaustivas em múltiplos repositórios. No design, o Napkin AI converte conceitos complexos em diagramas técnicos precisos.
O ganho de eficiência é estatisticamente substancial. Conforme demonstrado no estudo de Trad et al. (2024), a aplicação de fluxos baseados em LLM reduziu o tempo de triagem em revisões sistemáticas de 564,4 para 25,5 horas. Mais relevante que a celeridade é a precisão: o estudo reportou um índice de falso-negativo de 0% e uma especificidade de 99,6%, superando o crivo humano em termos de consistência.
3. Aplicação Prática na Cirurgia do Aparelho Digestivo
Para o residente ou o pós-graduando em Cirurgia do Aparelho Digestivo, a integração destas ferramentas deve ser metódica. Imaginemos o preparo para um Journal Club: o acadêmico pode utilizar o Consensus AI para identificar o estado da arte sobre uma técnica de anastomose, empregar o Litmaps para visualizar a rede de citações do ensaio clínico seminal e, finalmente, interpelar a metodologia do artigo através do ChatPDF para identificar possíveis vieses.
O fluxo de trabalho idealizado integra ciclicamente: (1) Busca em mecanismos especializados; (2) Interação conversacional com documentos para extração de dados; (3) Análise de redes de citações para contextualização histórica; (4) Escrita assistida por modelos generativos e (5) Design gráfico para comunicação visual dos resultados.
4. O Papel da Supervisão Humana
Embora a tecnologia seja fascinante, o rigor acadêmico nos impõe cautela. Devemos atentar para o conceito de “débito cognitivo”. Pesquisas conduzidas por Kosmyna et al. (2024) revelam que a dependência excessiva e exclusiva de IAs reduz o engajamento neural e a retenção de conteúdo. Em contrapartida, a sequência “cérebro primeiro, IA depois” — em que a conceituação inicial e o esboço partem do cirurgião — resulta em um aumento de 50% a 200% na conectividade neural através de bandas de EEG, preservando a capacidade de recordação e o pensamento crítico.
No âmbito ético, as diretrizes de entidades como ICMJE, COPE e WAME são peremptórias:
1. Veto à Autoria: Sistemas de IA não possuem personalidade jurídica ou intelectual para figurar como autores.
2. Transparência Radical: Todo uso de GenAI deve ser declarado explicitamente na seção de Métodos ou Agradecimentos.
3. Auditoria e Registros: Seguindo as orientações da JAMA Network, o pesquisador deve manter um registro de todos os prompts (comandos) relevantes utilizados, para fins de auditoria ética e científica.
A IA jamais substituirá o julgamento clínico soberano ou a responsabilidade ética do cirurgião sobre o dado publicado.
BOX DE DESTAQUE: ESSENCIAL PARA A PRÁTICA ACADÊMICA
• Eficiência Paradigmática: Redução drástica no tempo de triagem bibliográfica com precisão cirúrgica (0% de falso-negativos).
• Rigor Científico: Obrigatoriedade de verificação manual das referências para mitigar “alucinações” do modelo.
• Metodologia “Brain-First”: Priorizar a reflexão intelectual antes da assistência tecnológica para evitar o débito cognitivo.
• Ética e Transparência: Registro de prompts e declaração formal do uso de ferramentas conforme diretrizes internacionais.
6. Conclusões Aplicadas à Prática
A Inteligência Artificial é um catalisador de produtividade sem precedentes na história da medicina. Contudo, o cirurgião deve permanecer como o árbitro final da precisão científica. O futuro da educação médica continuada no Brasil exige o desenvolvimento de habilidades digitais que permitam ao médico não apenas consumir tecnologia, mas governá-la sob a égide do rigor científico. Dominar estas ferramentas é, hoje, um requisito para a excelência acadêmica e clínica.
“O cirurgião que conhece apenas a técnica é um artesão; o que domina a ciência por trás dela é um mestre. A IA é nossa nova ferramenta de maestria.”
Gostou? ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
——————————————————————————–
Palavras-chave: Inteligência Artificial na Cirurgia, Educação Médica, Cirurgia do Aparelho Digestivo, Pesquisa Cirúrgica, LLMs na Medicina, Residência Médica.
Hashtags: #CirurgiaDigestiva #InteligenciaArtificial #MedicinaAcademica #TheSurgeon #EducacaoMedica
PROF. DR. OZIMO GAMA
The Surgeon
A Cirurgia Digestiva Atual
Et Fortior
A Origem da Cirurgia Abdominal
A História da Primeira Laparotomia de Sucesso

1.0 Um Olhar Sobre o Abismo do Passado Cirúrgico
Hoje, um cirurgião pode remover um grande tumor ovariano em um procedimento seguro, utilizando um eletrocautério para uma cirurgia sem sangue e clipes de plástico para ligar os vasos. É um feito notável, mas que se torna assombroso quando contrastado com a brutal realidade cirúrgica de 1809. Naquele mundo, o interior do abdômen era um santuário inviolável. O que aconteceu na cidade fronteiriça de Danville, Kentucky, não foi uma evolução, mas uma metamorfose. Foi, nas palavras de Shakespeare, “a sea change into something rich and strange” — uma transformação profunda e maravilhosa.
Voltemos a esse tempo. Não existia anestesia — o éter só seria usado 33 anos depois. Os princípios da antissepsia de Joseph Lister só surgiriam em 1865. Transfusões de sangue eram algo para mais de um século no futuro, e a penicilina de Alexander Fleming só seria descoberta em 1928. Qualquer tentativa de abrir a cavidade abdominal era considerada uma sentença de morte.
Foi neste cenário desolador que um cirurgião visionário, Dr. Ephraim McDowell, e uma paciente de coragem extraordinária, Jane Todd Crawford, se uniram para realizar o impossível. Juntos, eles conduziram um “experimento” que não apenas salvou uma vida, mas fundou a cirurgia abdominal. Esta é a crônica desse evento marcante — uma lição fundamental de inovação, diagnóstico e coragem para todos os estudantes e cirurgiões contemporâneos.
2.0 A Crônica de um Milagre Cirúrgico
2.1 O Cenário Cirúrgico do Século XIX: O Dogma da Peritonite Fatal
No início do século XIX, a comunidade médica, especialmente a europeia, operava sob um dogma inabalável: qualquer violação da cavidade peritoneal resultaria invariavelmente em peritonite e morte. Cirurgiões renomados, como o anatomista escocês William Hunter, consideravam a remoção de ovários uma prática incurável e perigosa. Desafiar essa crença não exigia uma evolução do pensamento, mas uma completa revolução. Apesar do medo paralisante, a ideia já havia sido aventada. John Bell, professor de McDowell e um dos cirurgiões mais famosos da Europa, havia falado sobre a possibilidade teórica de tal operação em suas aulas, embora nunca tenha ousado tentar realizá-la. O abismo entre a teoria e a prática era, até então, intransponível.
2.2 Os Protagonistas: Um Cirurgião Visionário e uma Paciente de Coragem Extraordinária
Dr. Ephraim McDowell: Nascido em 1771, McDowell era um cirurgião competente e respeitado na fronteira americana. Sua formação incluiu um aprendizado com o Dr. Alexander Humphreys e estudos na Universidade de Edimburgo, onde foi influenciado por John Bell. Ao retornar para Danville, estabeleceu uma reputação por realizar cirurgias complexas para a época, como a remoção de cálculos vesicais de 23 pacientes sem uma única fatalidade. Um desses pacientes foi um jovem chamado James K. Polk, que mais tarde se tornaria o 11º presidente dos Estados Unidos.

Jane Todd Crawford: Nascida em 1763, Jane era uma mulher de 45 anos, mãe de quatro filhos, que vivia em uma cabana de toras a 60 milhas (quase 100 km) de Danville. Por meses, ela sofreu com um inchaço abdominal persistente e enorme. Seus médicos locais, perplexos, diagnosticaram-na erroneamente como estando em uma gravidez gemelar que já havia passado do tempo. Sua coragem e fortitude seriam postas à prova de uma forma que ninguém poderia imaginar.
2.3 O Diagnóstico e a Decisão Audaciosa
Em 13 de dezembro de 1809, McDowell cavalgou até a casa de Crawford. Seu relato do encontro demonstra uma impressionante acuidade clínica. Ele escreveu:
“Ao exame, per vaginam, não encontrei nada no útero, o que me induziu à conclusão de que deveria ser um ovarium aumentado.”
Sem nenhuma tecnologia, apenas com seu conhecimento de anatomia e o exame físico, McDowell descartou a gravidez e diagnosticou corretamente um tumor ovariano gigante. O passo seguinte foi ainda mais notável. Em um dos primeiros exemplos de consentimento informado, ele explicou a Crawford sua perigosa situação e que a única chance, uma operação, seria um “experimento” nunca antes realizado com sucesso. Ele prometeu tentar se ela estivesse disposta a ir até sua casa em Danville.
A resposta de Jane Todd Crawford foi um ato de coragem monumental. Durante o rigoroso inverno do Kentucky, ela viajou as 60 milhas a cavalo, uma jornada de vários dias, equilibrando o enorme tumor sobre a sela de seu cavalo para se submeter a um procedimento que muito provavelmente a mataria.
2.4 A Operação: Um Marco na História em 25 de Dezembro de 1809
No dia de Natal de 1809, em um quarto no andar de cima da casa de McDowell, a cirurgia foi realizada. Não havia anestesia; a mesa de operação era uma mesa comum de madeira. Para se acalmar durante o procedimento de 25 minutos, Jane Todd Crawford recitava salmos e cantava hinos. A cena, conforme descrita por McDowell, desafia a imaginação moderna. Ele realizou uma incisão de 9 polegadas no lado esquerdo do abdômen, realizando a primeira laparotomia eletiva da história. Com a súbita liberação da pressão, os intestinos “correram para fora sobre a mesa”. Preenchido pelo tumor, o abdômen não podia contê-los. As alças intestinais, segundo o próprio McDowell, “permaneceram de fora por cerca de vinte minutos e, sendo dia de Natal, ficaram tão frias” que ele precisou lavá-las com água morna antes de recolocá-las. Em meio a esse caos controlado, ele passou uma ligadura forte na trompa de Falópio, incisou o tumor, removeu 15 libras de “substância gelatinosa suja” e, em seguida, o saco tumoral de 7,5 libras. Finalmente, fechou a incisão com sutura interrompida, deixando a ponta da ligadura para fora. A recuperação de Crawford foi tão milagrosa quanto a cirurgia. Cinco dias depois, McDowell a encontrou “fazendo sua cama”. Em 25 dias, ela estava saudável o suficiente para fazer a viagem de 60 milhas de volta para casa, novamente a cavalo. Jane Todd Crawford viveu por mais 33 anos, superando seu cirurgião em 12 anos.

2.5 Publicação e Reconhecimento Tardio
Com modéstia, McDowell só publicou seu feito em 1817, no Eclectic Repertory and Analytical Review, após realizar com sucesso mais duas operações semelhantes. A reação da comunidade médica europeia foi de ceticismo e desprezo. James Johnson, editor do influente London Medico-Chirurgical Review, expressou sua incredulidade com um desdém mordaz: “Credat Judoeus, non ego” (Conte isso a outra pessoa, não a mim). Anos depois, ao relutantemente aceitar o feito, sua retratação revelou o preconceito geográfico e racial que McDowell enfrentava: “…todas as mulheres operadas no Kentucky, exceto uma, eram negras, e como essas pessoas suportam cortes com quase, se não total, impunidade como cães e coelhos, nossa admiração diminui…”. O reconhecimento de McDowell como o “Pai da Cirurgia Abdominal” veio postumamente, em grande parte devido aos esforços do Dr. Samuel D. Gross.
3.0 Aplicação e Legado para a Cirurgia Digestiva
Qual a importância deste evento para nós, cirurgiões do aparelho digestivo? A resposta é simples e profunda: tudo. O feito de McDowell foi a refutação prática e definitiva do dogma de que a cavidade peritoneal era intocável. Sua coragem não criou apenas um novo procedimento; ela criou a possibilidade de toda a nossa especialidade. A cirurgia de estômago, fígado, pâncreas e intestino simplesmente não existiria sem aquele primeiro passo revolucionário. É crucial também clarificar a terminologia. O procedimento não foi uma “ovariotomia” — termo que tecnicamente significa apenas incisar o ovário. O que McDowell realizou foi uma laparotomia (a abertura da cavidade abdominal) com uma salpingo-ooforectomia (a extração do ovário e da trompa de Falópio). Usar o termo impreciso diminui a magnitude de seu feito, que não foi apenas remover um órgão, mas sim invadir e manejar com sucesso o santuário proibido do abdômen. A ousadia de McDowell ressoa em sua própria voz, vinte anos depois do evento, em uma carta de 1829:
“Portanto, parece-me uma mera bobagem sobre o perigo da inflamação peritoneal tão falada pela maioria dos cirurgiões.”
4.0 Pontos-Chave para a Prática do Cirurgião Digestivo
A história de McDowell e Crawford oferece lições atemporais para a prática médica moderna:
- Inovação Contra o Dogma: A medicina avança quando profissionais questionam verdades estabelecidas. McDowell transcendeu o “padrão de cuidado” de sua época, confiando em seu julgamento clínico e conhecimento anatômico para fazer o que era considerado impossível.
- A Primazia do Diagnóstico Clínico: A habilidade de McDowell em realizar um diagnóstico preciso baseado unicamente no exame físico e no raciocínio clínico é uma lição poderosa. É um lembrete contundente de que, na ausência de tecnologia, a verdadeira excelência reside no raciocínio anatômico e na sensibilidade do exame clínico — habilidades que jamais devem ser atrofiadas.
- A Aliança Cirurgião-Paciente: O sucesso da empreitada dependeu de uma confiança mútua extraordinária. O consentimento informado de Crawford, mesmo em sua forma mais rudimentar, foi o pilar que sustentou a realização de um procedimento de altíssimo risco.
- Coragem e Responsabilidade: McDowell demonstrou a coragem necessária para ser o primeiro, assumindo total responsabilidade pelo resultado. Ele sabia que, em caso de falha, o peso do fracasso recairia inteiramente sobre ele.
5.0 O Legado Imortal de McDowell e Crawford
A cirurgia realizada em um quarto de uma cidade fronteiriça não foi um avanço evolutivo; foi um salto revolucionário. Nasceu da visão de um homem e da coragem de uma mulher, que juntos mudaram o curso da história da medicina. Não foi o produto de uma “escola” cirúrgica ou de anos de pesquisa, mas um ato de genialidade e audácia que surgiu de novo. Com sua genialidade, Ephraim McDowell devolveu a vida a Jane Todd Crawford; ela, em troca, garantiu-lhe a imortalidade. O que eles realizaram juntos foi, de fato, o início de uma nova era para a medicina.
#HistóriaDaMedicina #CirurgiaAbdominal #Laparotomia #EphraimMcDowell #CirurgiaDigestiva
Anatomia Cirúrgica da Região Inguinal
1.0 Introdução
A anatomia da região inguinal é notoriamente complexa, representando um desafio constante até mesmo para os cirurgiões mais experientes. Como afirmado de forma sucinta por Skandalakis et al. no início de seu trabalho seminal, a anatomia da região inguinal é “enigmática e confusa” (enigmatic and confusing). Este artigo tem como objetivo desmistificar essa complexidade, focando nas estruturas anatômicas cruciais e em suas relações, a fim de fornecer um guia claro e prático para estudantes de medicina, residentes e cirurgiões que atuam no reparo de hérnias inguinais. Uma compreensão precisa desta topografia não é apenas um exercício acadêmico, mas a fundação para a excelência cirúrgica.
2.0 Desvendando as Estruturas Anatômicas
2.1 As Paredes do Canal Inguinal
O canal inguinal é uma passagem oblíqua através das camadas inferiores da parede abdominal. Suas paredes são formadas por diversas estruturas musculoaponeuróticas, como visto na ilustração anatômica. Os limites são:
• Parede Anterior: Formada pela aponeurose do músculo oblíquo externo e, em sua porção lateral, reforçada pelo músculo oblíquo interno.
• Parede Posterior: Considerada a mais importante do ponto de vista anatômico e cirúrgico, é composta primariamente pela fusão da aponeurose do músculo transverso do abdome com a fáscia transversal. Em 23-25% dos indivíduos, é formada apenas pela fáscia transversal.
• Parede Superior (Teto): Composta pelas fibras arqueadas dos músculos oblíquo interno e transverso do abdome e suas aponeuroses.
• Parede Inferior (Assoalho): Constituída pelo ligamento inguinal e, mais medialmente, pelo ligamento lacunar.
A reconstrução desta parede posterior, especialmente quando deficiente, é o objetivo central de qualquer herniorrafia inguinal bem-sucedida.
2.2 As Camadas Musculoaponeuróticas Fundamentais
A parede abdominal é composta por três camadas de músculos planos. De superficial para profundo, temos o oblíquo externo, o oblíquo interno e o transverso do abdome. Embora todos sejam importantes, o músculo transverso do abdome assume um papel de destaque. De acordo com McVay, ele é a camada mais importante da parede abdominal. A integridade deste músculo e sua aponeurose é o que, em última análise, previne a formação de hérnias. Consequentemente, a mobilização e o uso da aponeurose deste músculo são manobras essenciais para um reparo anatômico e duradouro.
2.3 A Crucial Fáscia Transversal
A fáscia transversal é a lâmina de tecido conjuntivo que reveste a superfície interna da musculatura da cavidade abdominal. Ela é uma estrutura fundamental, contribuindo para a formação da fáscia espermática interna no funículo espermático e, como já mencionado, compondo a parede posterior do canal inguinal. Sua integridade é vital para a contenção visceral. Portanto, a dissecção e o reparo desta fáscia não são opcionais, mas sim o alicerce fundamental de uma herniorrafia sem tensão.
2.4 O “Mito” do Tendão Conjunto
O termo “tendão conjunto” é amplamente utilizado, mas sua existência anatômica como uma fusão distinta das fibras inferiores do oblíquo interno e do transverso do abdome é extremamente rara. A evidência anatômica mostra que essa configuração é mais uma exceção do que uma regra.
• Hollinshead considerou sua existência em apenas 5% dos corpos.
• Condon relatou uma incidência de apenas 3%.
• McVay acreditava que o “tendão conjunto” era um artefato de dissecação.
Por essa razão, o conceito de “área conjunta” (conjoined area) é um termo mais preciso e clinicamente relevante. Esta área refere-se à região medial da parede posterior, que inclui estruturas como o ligamento de Henle, a aponeurose do transverso do abdome e a borda lateral da bainha do reto, que são de fato utilizadas no reparo de hérnias.
2.5 Pontos de Referência Cirúrgicos Essenciais
A navegação segura na região inguinal exige o reconhecimento de marcos anatômicos chave:
• Triângulo de Hesselbach: É o local onde ocorre a maioria das hérnias inguinais diretas. Suas bordas, na definição moderna, são: os vasos epigástricos inferiores (borda superior), a bainha do músculo reto abdominal (borda medial) e o ligamento inguinal (borda lateroinferior).
• Ligamento de Cooper: Estrutura robusta formada pelo periósteo do ramo superior do púbis. É reforçado por múltiplas camadas, incluindo contribuições da fáscia transversal, do trato iliopúbico e da aponeurose do transverso do abdome. É um ponto de ancoragem fundamental em diversas técnicas de herniorrafia, como a de McVay.
• Trato Iliopúbico: Uma banda aponeurótica densa que se estende do arco iliopectíneo até o ramo superior do púbis, formando a margem inferior da camada musculoaponeurótica profunda. Condon o encontrou presente em 98% das dissecações. É uma estrutura profunda, paralela ao ligamento inguinal (que é mais superficial), e um marco essencial em reparos laparoscópicos e abertos.
3.0 Aplicação na Cirurgia Digestiva
3.1 Fisiologia e Mecanismos de Contenção da Hérnia
A integridade do canal inguinal normal é mantida por dois mecanismos fisiológicos dinâmicos:
1. Ação Esfincteriana: A fáscia transversal forma um “sling” (alça) ao redor do anel inguinal interno. A contração do músculo transverso do abdome tensiona essa alça, fechando o anel sob a borda muscular do oblíquo interno.
2. Ação de Obturador (“Shutter Action”): Durante a contração da musculatura abdominal (por exemplo, ao tossir), o arco aponeurótico do músculo transverso do abdome se move em direção ao trato iliopúbico e ao ligamento inguinal, reforçando e obliterando qualquer fraqueza na parede posterior.
3.2 Implicações Diretas na Herniorrafia
O conhecimento anatômico se traduz diretamente na estratégia cirúrgica. Como mencionado, a parede posterior é anatomicamente deficiente em 23-25% dos indivíduos, carecendo do suporte da aponeurose do músculo transverso. Nesses casos, a fáscia transversal torna-se a única barreira contra a herniação direta. O objetivo de uma herniorrafia eficaz não é apenas fechar um defeito, mas reconstruir a dinâmica da parede posterior. A compreensão precisa de quais estruturas estão presentes, quais são deficientes e quais podem ser utilizadas para um reparo sólido e sem tensão é o que diferencia um procedimento bem-sucedido de uma recidiva.
4.0 Pontos-Chave
1. A Complexidade é Real: A anatomia inguinal é intrincada e repleta de controvérsias terminológicas. Um estudo contínuo é necessário.
2. O Transverso do Abdome é Rei: A integridade do músculo transverso do abdome e sua aponeurose é o fator mais crítico na fisiopatologia e prevenção das hérnias inguinais.
3. O Tendão Conjunto é Raro: O “tendão conjunto” clássico é um achado raro (3-5%). O termo “área conjunta” é mais apropriado para descrever as estruturas utilizadas cirurgicamente.
4. A Parede Posterior é a Chave: O sucesso do reparo de uma hérnia inguinal depende fundamentalmente da reconstrução de uma parede posterior forte e sem tensão, utilizando as estruturas anatômicas corretas.
5. Conheça Seus Marcos: O Ligamento de Cooper, o Trato Iliopúbico e o Triângulo de Hesselbach são referências anatômicas indispensáveis para o cirurgião realizar um procedimento seguro e eficaz.
5.0 Conclusões Aplicadas à Prática Clínica
Apesar de séculos de estudo e de incontáveis procedimentos cirúrgicos, a região inguinal permanece um desafio anatômico. As variações individuais e as controvérsias na nomenclatura exigem do cirurgião mais do que a memorização de diagramas; exigem um conhecimento profundo e funcional. Esta compreensão detalhada da anatomia é a ferramenta mais poderosa que possuímos para avaliar defeitos herniários, escolher a técnica de reparo mais adequada e, por fim, garantir resultados cirúrgicos superiores e duradouros para nossos pacientes.
#AnatomiaCirurgica #HerniaInguinal #CirurgiaGeral #ResidenciaCirurgia #AparelhoDigestivo
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Conteúdo Extra
Lista Completa de Epônimos Anatômicos da Região Inguinal
Esta lista abrange as estruturas clássicas nomeadas em homenagem a anatomistas e cirurgiões, fundamentais para a compreensão cirúrgica da região inguiocrural.
1. Ligamentos e Estruturas Fibrosas
Os elementos estruturais que formam as paredes e o assoalho da região.
- Ligamento de Poupart
- Nome Moderno: Ligamento Inguinal.
- Quem foi: François Poupart (cirurgião francês, séc. XVII).
- Descrição: A borda inferior enrolada da aponeurose do músculo oblíquo externo. Estende-se da EIAS ao tubérculo púbico, formando o “assoalho” do canal inguinal.
- Ligamento de Gimbernat
- Nome Moderno: Ligamento Lacunar.
- Quem foi: Antonio de Gimbernat y Arbós (cirurgião espanhol, séc. XVIII).
- Descrição: Extensão posterior e triangular do ligamento de Poupart que se fixa no osso púbico. Forma a borda medial cortante do anel femoral.
- Ligamento de Cooper
- Nome Moderno: Ligamento Pectíneo.
- Quem foi: Astley Cooper (cirurgião inglês, séc. XIX).
- Descrição: Espessamento forte do periósteo (membrana do osso) ao longo do ramo superior do púbis. É o principal ponto de ancoragem óssea para telas em cirurgias complexas.
- Trato de Thomson
- Nome Moderno: Trato Iliopúbico.
- Quem foi: James Thomson (anatomista escocês, séc. XIX).
- Descrição: Espessamento da fáscia transversal profundo e paralelo ao ligamento inguinal. É o divisor profundo entre o canal inguinal (acima) e o canal femoral (abaixo).
- Ligamento de Henle
- Nome Moderno: Expansão lateral da bainha do músculo reto abdominal (frequentemente associado à foice inguinal/tendão conjunto).
- Quem foi: Friedrich Gustav Jakob Henle (anatomista e patologista alemão, séc. XIX).
- Descrição: Uma estrutura aponeurótica na parede posterior do canal inguinal, logo na borda do músculo reto abdominal. Ele forma o limite medial do Trígono de Hesselbach. Sua presença é variável; quando forte, protege contra hérnias diretas; quando ausente ou fraco, predispõe a elas.
- Ligamento de Colles (ou Ligamento Reflexo)
- Nome Moderno: Ligamento Inguinal Reflexo.
- Quem foi: Abraham Colles (cirurgião e anatomista irlandês, séc. XIX).
- Descrição: É uma camada de fibras derivadas da aponeurose do oblíquo externo do lado oposto, que cruzam a linha média e se inserem na crista púbica e na linha pectínea do lado afetado. Ele passa atrás do anel inguinal superficial e na frente do tendão conjunto, reforçando a porção medial do canal inguinal.
2. Fáscias e Camadas
As camadas de cobertura da parede abdominal anterior.
- Fáscia de Scarpa
- Nome Moderno: Lâmina membranácea da tela subcutânea.
- Quem foi: Antonio Scarpa (anatomista italiano, séc. XVIII/XIX).
- Descrição: A camada membranosa mais profunda da gordura abaixo da pele.
- Fáscia de Camper
- Nome Moderno: Lâmina adiposa da tela subcutânea.
- Quem foi: Petrus Camper (anatomista holandês, séc. XVIII).
- Descrição: A camada de gordura superficial e fofa logo abaixo da pele.
- Fáscia de Colles
- Nome Moderno: Fáscia membranácea do períneo.
- Quem foi: Abraham Colles (cirurgião irlandês, séc. XIX).
- Descrição: A continuação da Fáscia de Scarpa na região do períneo e escroto/lábios.
3. Espaços e Áreas Definidas
Marcos geográficos críticos para cirurgia e diagnóstico.
- Trígono de Hesselbach
- Nome Moderno: Trígono Inguinal.
- Quem foi: Franz Kaspar Hesselbach (anatomista alemão, séc. XIX).
- Descrição: A área triangular na parede posterior da virilha onde ocorrem as hérnias diretas. Delimitado pelos vasos epigástricos inferiores, a borda do músculo reto (Ligamento de Henle) e o ligamento inguinal.
- Orifício de Fruchaud
- Nome Moderno: Orifício Miopectíneo.
- Quem foi: Henri Fruchaud (cirurgião francês, séc. XX).
- Descrição: O “grande buraco” conceitual na parede abdominal que engloba todas as potenciais hérnias da virilha (direta, indireta e femoral), que deve ser coberto pela tela.
- Espaço de Bogros
- Nome Moderno: Espaço Retroinguinal (pré-peritoneal lateral).
- Quem foi: Jean-Annet de Bogros (anatomista francês, séc. XIX).
- Descrição: O espaço virtual atrás da fáscia transversal e à frente do peritônio, onde as telas são colocadas nas cirurgias laparoscópicas (TEP/TAPP).
- Espaço de Retzius
- Nome Moderno: Espaço Retropúbico (pré-vesical).
- Quem foi: Anders Retzius (anatomista sueco, séc. XIX).
- Descrição: O espaço atrás do osso púbico e à frente da bexiga, medial ao espaço de Bogros.
4. Linfonodos e Vasos
- Gânglio de Cloquet (ou Rosenmüller)
- Nome Moderno: Linfonodo inguinal profundo proximal.
- Quem foram: Jules Germain Cloquet (francês) e Johann Christian Rosenmüller (alemão).
- Descrição: O linfonodo localizado dentro do canal femoral, que pode simular uma hérnia se estiver inflamado.
- Corona Mortis (Termo clássico, não epônimo de pessoa)
- Tradução: Coroa da Morte.
- Descrição: Uma variação vascular perigosa (conexão entre vasos ilíacos externos e internos) que passa sobre o osso púbico, com risco de hemorragia grave se lesada durante a cirurgia.
Hemorragia Digestiva
Hemorragia Gastrointestinal: Visão do Cirurgião
1.0 Introdução
A hemorragia gastrointestinal (HGI) representa uma das principais emergências médicas e cirúrgicas, demandando diagnóstico rápido e manejo assertivo. Classificamos a HGI com base em sua origem anatômica em relação ao ligamento de Treitz. A Hemorragia Digestiva Alta (HDA), proximal a este ligamento, é a mais comum, com uma incidência, no contexto dos Estados Unidos, de 67 casos por 100.000 habitantes. A Hemorragia Digestiva Baixa (HDB), originada distalmente ao ligamento, apresenta uma incidência de 36 casos por 100.000 habitantes, conforme dados do mesmo país. O manejo eficaz desses quadros exige uma abordagem sistemática e multidisciplinar, na qual o cirurgião ocupa uma posição de destaque, sendo peça-chave na condução dos casos refratários ao tratamento clínico e endoscópico.
2.0 Avaliação e Diagnóstico
2.1 Apresentação Clínica e Avaliação Inicial
A apresentação clínica da HGI varia conforme a localização e a intensidade do sangramento.
- HDA: Os sinais clássicos são a hematêmese (vômito com sangue vivo ou com aspecto de “borra de café”) e a melena (fezes enegrecidas, de odor fétido), que pode manifestar-se com perdas sanguíneas de apenas 50 mL.
- HDB: O principal sinal é a hematoquezia (eliminação de sangue vivo ou marrom nas fezes). É crucial ressaltar que a hematoquezia pode ocorrer em 10-15% dos casos de hemorragia digestiva alta maciça, um sinal de sangramento de grande volume e trânsito acelerado, geralmente associado à instabilidade hemodinâmica.
- Avaliação de Severidade: A anamnese e o exame físico são primordiais para determinar a estabilidade hemodinâmica do paciente. A perda de volemia correlaciona-se diretamente com as alterações nos sinais vitais:
- Perda < 15% (<750 mL): Sinais vitais podem estar normais.
- Perda de 15-30% (750-1500 mL): Taquicardia (FC > 100 bpm) e débito urinário reduzido (≤ 30 mL/h).
- Perda de 30-40% (1500-2000 mL): Hipotensão, taquicardia acentuada (FC > 120 bpm) e confusão mental.
- Perda > 40% (>2000 mL): Hipotensão grave, taquicardia (FC > 140 bpm), taquipneia e letargia, com débito urinário insignificante.
- Exames Laboratoriais: A avaliação laboratorial inicial deve incluir hemograma completo, tipagem sanguínea e provas de coagulação, além de avaliação das funções hepática e renal. Uma relação entre a Ureia e a Creatinina sérica (em inglês, Blood Urea Nitrogen ou BUN, resultando na razão BUN/Creatinina) ≥ 30 é um forte preditor de hemorragia digestiva alta.
2.2 Abordagem Diagnóstica por Localização
- Hemorragia Digestiva Alta (HDA):
- Causas Principais: Doença ulcerosa péptica, varizes esofágicas, esofagite, gastrite, lesão de Dieulafoy, lacerações de Mallory-Weiss e neoplasias.
- Diagnóstico: A endoscopia digestiva alta é a modalidade diagnóstica e terapêutica de eleição, devendo ser realizada preferencialmente nas primeiras 24 horas. É imperativo notar que, na suspeita de sangramento por varizes esofágicas, este intervalo deve ser encurtado para 12 horas. Os achados são estratificados pela Classificação de Forrest, que correlaciona o aspecto da lesão (sangramento arterial ativo [Ia], sangramento babante [Ib], vaso visível não sangrante [IIa], coágulo aderido [IIb]) com o risco de ressangramento, orientando a terapêutica.
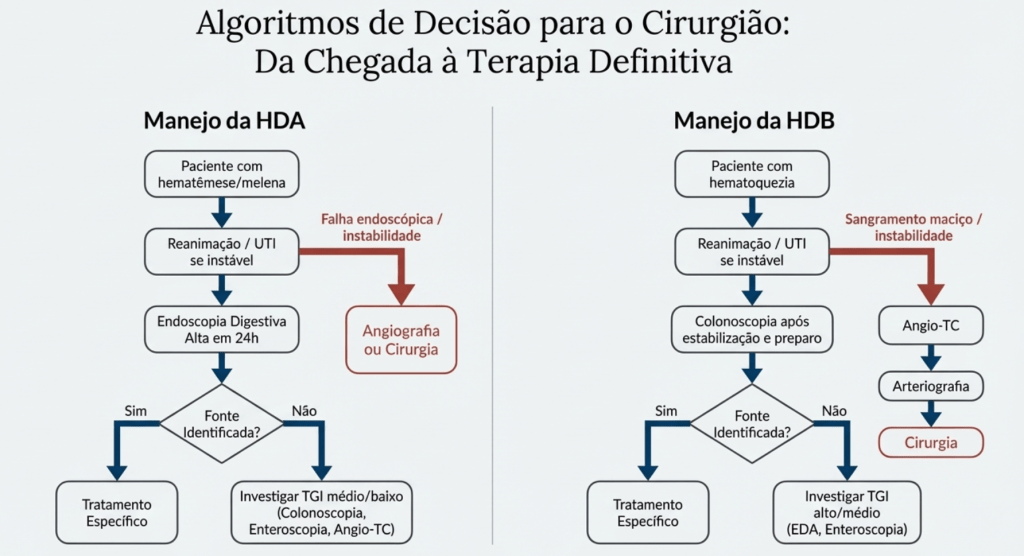
- Hemorragia Digestiva Baixa (HDB):
- Causas Principais: Doença diverticular, angiodisplasia, colites (infecciosa, isquêmica), doença inflamatória intestinal e neoplasias.
- Diagnóstico: Em pacientes estáveis, a colonoscopia é o exame de escolha. Para pacientes instáveis com sangramento ativo e maciço, a angiotomografia computadorizada (Angio-TC) e a arteriografia são fundamentais. A Angio-TC pode detectar sangramentos a uma taxa de 0.3-0.5 mL/min, enquanto a arteriografia requer uma taxa de 0.5-1.0 mL/min, permitindo, contudo, a embolização terapêutica. Em casos de melena com endoscopia alta negativa, a colonoscopia é frequentemente indicada para investigar lesões no cólon direito, que podem apresentar-se de forma atípica.
3.0 Aplicação na Cirurgia Digestiva: O Papel do Cirurgião
A intervenção cirúrgica é, na maioria das vezes, uma terapia de resgate na HGI, reservada para cenários específicos e bem definidos.
3.1 Indicações para Intervenção Cirúrgica
As indicações para uma cirurgia de urgência incluem:
- Falha do tratamento endoscópico ou angiográfico em controlar a hemorragia.
- Instabilidade hemodinâmica persistente, a despeito de ressuscitação volêmica adequada.
- Ressangramento recorrente ou persistente.
- Necessidade de transfusão maciça (para HDB, definida como > 4 unidades de concentrado de hemácias em 24 horas).
- Condições associadas que requerem abordagem cirúrgica, como perfuração ou malignidade.
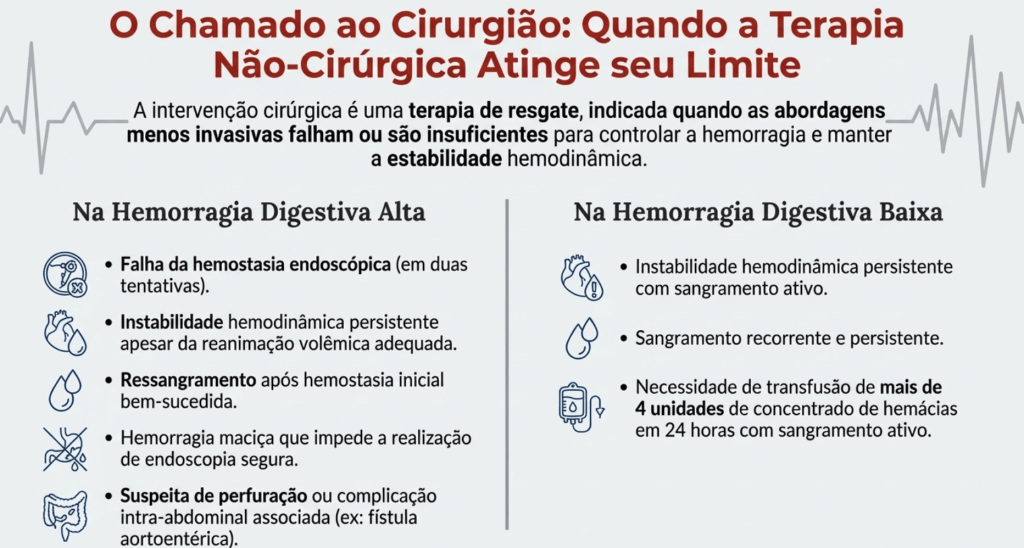
3.2 Estratégias Cirúrgicas na Hemorragia Digestiva Alta
- Úlcera Duodenal: A abordagem cirúrgica clássica para a úlcera péptica duodenal sangrante consiste em duodenotomia anterior, ligadura direta do vaso sangrante (geralmente a artéria gastroduodenal) com pontos em “U, seguida de uma piloroplastia tipo Heineke-Mikulicz. Em pacientes estáveis, a vagotomia troncular pode ser associada para tratar a doença ulcerosa de base.
- Úlcera Gástrica: O tratamento de eleição é a ressecção da úlcera, seja por gastrectomia em cunha ou por uma gastrectomia distal (ex: Billroth I/II). A excisão é imperativa para análise histopatológica, dado que 4-5% das úlceras com aparência benigna podem, na verdade, ser malignas.
- Varizes Esofágicas: A cirurgia é uma terapia de resgate. As opções incluem shunts portossistêmicos (seletivos ou não seletivos) para descompressão portal ou procedimentos de desvascularização (ex: procedimento de Sugiura) em casos selecionados.
- Lesão de Mallory-Weiss: A necessidade de tratamento cirúrgico é rara, consistindo na sutura da laceração esofágica por via laparoscópica ou aberta.
3.3 Estratégias Cirúrgicas na Hemorragia Digestiva Baixa
A localização pré-operatória precisa do sítio de sangramento é o fator mais crítico para o sucesso cirúrgico na HDB.
- Quando o ponto de sangramento é identificado, a ressecção segmentar do cólon é o procedimento de escolha.
- Em pacientes instáveis com sangramento maciço de fonte não localizada, a colectomia subtotal com ileostomia terminal é o procedimento indicado. Esta abordagem está associada a taxas de morbidade de aproximadamente 37% e de mortalidade entre 11% e 33%.
4.0 Pontos-Chave para a Prática Cirúrgica
- A avaliação inicial focada na estabilidade hemodinâmica dita o ritmo da investigação e do tratamento.
- A endoscopia digestiva alta é o pilar diagnóstico e terapêutico na HDA, e a Classificação de Forrest é essencial para guiar a conduta pós-endoscópica.
- A cirurgia na HGI é predominantemente uma terapia de resgate, indicada após falha das modalidades menos invasivas ou em casos de instabilidade hemodinâmica refratária.
- A localização pré-operatória do sítio de sangramento na HDB é fundamental para minimizar a morbimortalidade, sendo a colectomia subtotal reservada para emergências com fonte desconhecida.

5.0 Conclusões
O manejo da hemorragia gastrointestinal é um desafio complexo que exige uma abordagem multidisciplinar e bem coordenada. Embora a cirurgia seja frequentemente uma terapia de resgate, o cirurgião deve ser envolvido precocemente na avaliação de pacientes graves. Um profundo conhecimento das opções diagnósticas e terapêuticas, tanto cirúrgicas quanto não cirúrgicas, é indispensável para garantir a tomada de decisão correta e o melhor desfecho para o paciente.
“A ligadura direta das varizes esofágicas representa a abordagem mais fundamental para controlar uma hemorragia que ameaça a vida.” – Inspirado nos trabalhos de George Crile Jr., 1950.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#HemorragiaGastrointestinal #CirurgiaDigestiva #CirurgiaGeral #HemorragiaDigestiva #Medicina
Além do Bisturi: A Filosofia Cirúrgica de René Leriche e a Revolução Fisiológica
Na história da medicina, existem cirurgiões famosos por suas mãos ágeis e outros lembrados por suas mentes inquietas. René Leriche (1879-1955) pertence, indubitavelmente, à segunda categoria. Embora fosse um operador exímio, seu verdadeiro bisturi era o intelecto. Autor da obra seminal “La chirurgie, discipline de la connaissance” (A cirurgia, disciplina do conhecimento), Leriche não via a sala de operações apenas como um local de reparo mecânico, mas como um laboratório vivo de fisiologia. Para ele, operar era interrogar a vida. Neste artigo, revisitamos a filosofia deste gigante francês e como ele transformou a cirurgia de um ofício anatômico para uma ciência biológica.
Da Anatomia Estática à Fisiologia Dinâmica
Até o início do século XX, a cirurgia era predominantemente “anatômica”. O cirurgião era visto como um encanador de luxo: removia-se o que estava podre, costurava-se o que estava rasgado, drenava-se o que estava infectado. O foco era a lesão estrutural visível. Leriche desafiou esse paradigma. Ele argumentava que a lesão anatômica era frequentemente o fim da doença, não o começo. A verdadeira patologia residia no distúrbio funcional invisível que precedia a lesão.
“A doença é, antes de tudo, um drama funcional que se desenrola no silêncio dos tecidos.”
Essa visão deu origem ao conceito de Cirurgia Fisiológica. O objetivo não deveria ser apenas a extirpação do órgão doente (uma “confissão de fracasso”, segundo ele), mas a restauração da função normal, muitas vezes através da manipulação do sistema nervoso autônomo e da vascularização.
A Cirurgia como “Vivissecção Terapêutica”
Leriche elevou o status epistemológico da cirurgia. Ele afirmava que o cirurgião possuía um privilégio que o anatomista e o patologista não tinham: o acesso à patologia viva. Enquanto a anatomia patológica estuda as “cinzas do incêndio” (o tecido morto), a cirurgia observa o “fogo” (a doença em evolução). Portanto, a cirurgia deveria ser uma “disciplina do conhecimento”, onde cada intervenção serve para entender a fisiopatologia humana. Para Leriche, um cirurgião que não raciocina fisiologicamente é apenas um técnico manual, fadado à obsolescência.
A Dor como Doença (La Douleur-Maladie)
Talvez a contribuição mais humanística de Leriche tenha sido sua guerra contra a dor. Rompendo com a tradição estoica que via a dor como um mecanismo de defesa útil ou um aviso benéfico, Leriche a classificou como um erro biológico. Ele cunhou o termo “A Dor-Doença”. Na visão dele, a dor crônica não protege; ela destrói. Ela altera a personalidade, esgota as reservas vitais e cria um ciclo vicioso simpático que perpetua o sofrimento. Suas pesquisas sobre a causalgia e o papel do sistema simpático na dor abriram as portas para a moderna medicina da dor e a neuromodulação. Foi neste contexto que ele nos legou sua definição poética e precisa de saúde:
“A saúde é a vida no silêncio dos órgãos.”
O Legado de Leriche na Cirurgia Moderna
Por que ler René Leriche em pleno século XXI? Porque a cirurgia moderna é, em essência, a realização do sonho de Leriche. Quando realizamos uma cirurgia metabólica para tratar diabetes (e não apenas para reduzir o estômago), estamos praticando a cirurgia fisiológica de Leriche. Quando utilizamos bloqueios nervosos ou estimulação medular, estamos seguindo seus passos no controle da dor. Quando buscamos a preservação de órgãos em vez da amputação, honramos sua filosofia. René Leriche nos ensinou que a técnica é indispensável, mas é a fisiologia que deve guiar a mão do cirurgião. O virtuosismo técnico sem base científica é perigoso; a ciência sem técnica é impotente. O cirurgião completo, portanto, é aquele que opera com as mãos, mas corta com a mente.

Gostou desta reflexão histórica? Acompanhe o The Surgeon para mais conteúdos sobre a arte, a ciência e a filosofia da cirurgia.
A Dualidade da Humanização Cirúrgica
Era da Inteligência Artificial e da Robótica: Desafios e Perspectivas
Por Prof. Dr. Ozimo Gama
Coordenador do Curso de Medicina da Universidade Federal do Maranhão
O Cenário Atual e a Necessidade do “Toque Humano”
A prática da cirurgia do aparelho digestivo vive um momento de paradoxo fascinante. Por um lado, testemunhamos avanços tecnológicos sem precedentes, com a cirurgia robótica e a inteligência artificial (IA) redefinindo os limites do possível. Por outro, enfrentamos uma crise silenciosa na relação médico-paciente. No Brasil, segundo dados do DATASUS e do Conselho Federal de Medicina, realizamos milhões de procedimentos cirúrgicos anuais pelo Sistema Único de Saúde (SUS), onde a alta demanda muitas vezes colide com a necessidade de um atendimento individualizado.
A “humanização” não é apenas um conceito abstrato ou uma “soft skill” desejável; é um imperativo clínico. Estudos epidemiológicos demonstram que pacientes que estabelecem uma relação de confiança sólida com seus cirurgiões apresentam melhor adesão ao tratamento pós-operatório, menores índices de litígios médicos e, subjetivamente, uma percepção de dor reduzida.
O desafio contemporâneo, como bem alertava o Prof. Alcino Lázaro da Silva, é evitar que o “fascínio das máquinas e dos botões” transforme o jovem cirurgião em um “sectário ou robô”. Precisamos integrar a tecnologia sem permitir que ela congele nossos sentimentos ou desqualifique a essência da nossa profissão: o cuidado com o ser humano.
Tecnologia, Ontoética e Controvérsias
A Inteligência Artificial e a Despersonalização
A IA chegou para auxiliar na tomada de decisão, predizendo complicações e otimizando fluxos em centros cirúrgicos. No entanto, existe o risco real da “atrofia da empatia”. Quando confiamos excessivamente em algoritmos para o prognóstico, corremos o risco de tratar o paciente como um conjunto de dados biométricos, e não como uma biografia. A tecnologia deve ser uma ferramenta de meio, nunca de fim. O cirurgião que se esconde atrás da tela do computador perde a oportunidade de exercer a “Ontoética” — a ética do ser, o respeito intrínseco à vida humana que transcende a técnica.
Aspectos Médico-Legais e o Consentimento
A humanização tem implicações jurídicas diretas. O Prof. Milton Glezer destaca que o paciente tem o direito de recusar tratamento (salvo risco iminente de morte) e de ter acesso irrestrito ao seu prontuário. No Brasil, a judicialização da medicina tem crescido exponencialmente. Uma parte significativa desses processos não decorre de erro técnico (imperícia), mas de falha na comunicação. O Consentimento Livre e Esclarecido não deve ser apenas um formulário burocrático assinado na admissão. Ele deve ser um processo contínuo de diálogo, onde o cirurgião traduz o “tecniquês” para a realidade do doente, respeitando sua autonomia e dignidade.
Controvérsias Filosóficas
A controvérsia reside na formação médica atual. Os currículos de graduação e residência muitas vezes privilegiam a técnica em detrimento da humanística. Criamos excelentes técnicos capazes de realizar gastrectomias complexas, mas que, por vezes, falham em comunicar um diagnóstico de câncer gástrico com a compaixão necessária. A escola médica deve ensinar que o doente prefere, acima do tecnocrata, o amigo; acima do executor de procedimentos, o médico que orienta e acolhe.
Aplicação Prática na Cirurgia do Aparelho Digestivo
Na nossa especialidade, a humanização possui facetas muito específicas e impactantes:
- Ostomias e Imagem Corporal: Ao indicar uma colostomia definitiva, por exemplo, o cirurgião digestivo não está apenas alterando a anatomia, mas a autoimagem e a vida social do paciente. A abordagem humanizada exige preparar este paciente psicologicamente, envolvendo a equipe multidisciplinar e a família antes mesmo do ato cirúrgico.
- Oncologia Cirúrgica: O diagnóstico de neoplasias do trato gastrointestinal carrega um estigma de mortalidade. A forma como a notícia é dada (protocolo SPIKES, por exemplo) é tão crucial quanto a linfadenectomia realizada.
- Cirurgia Bariátrica: O paciente obeso muitas vezes carrega anos de preconceito, inclusive dentro do sistema de saúde. A humanização aqui significa enxergar a obesidade como doença crônica, despindo-se de julgamentos morais e oferecendo um suporte que vai além da perda de peso.
Pontos-Chave para a Prática Diária
Para o residente e o jovem cirurgião, a humanização se traduz em atos concretos:
- Olho no Olho: A tecnologia (computadores, tablets) não deve ser uma barreira física durante a consulta. Sente-se, olhe para o paciente e escute ativamente antes de examinar.
- Identificação Clara: Como preconizado nas diretrizes hospitalares, apresente-se, diga sua função e certifique-se de que o paciente sabe quem é o responsável pelo seu tratamento (titularidade médica).
- Gestão da Expectativa: Explique riscos e benefícios reais. A frustração do paciente muitas vezes nasce de uma expectativa irrealista não corrigida pelo cirurgião no pré-operatório.
- O “Não” Terapêutico: Saber a hora de não operar (obstinação terapêutica) em casos de carcinomatose ou doenças terminais é, talvez, a maior prova de humanidade e ética cirúrgica.
Perspectivas Futuras
O futuro da Cirurgia do Aparelho Digestivo não será definido apenas pela precisão dos robôs ou pela acurácia dos algoritmos de IA, mas pela nossa capacidade de manter a essência humana em um ambiente cada vez mais digital. A “Ontoética” deve guiar nossas mãos tanto quanto a anatomia.
Precisamos formar cirurgiões “híbridos”: exímios operadores da tecnologia, mas profundos conhecedores da alma humana. Como discutido nos textos bases, o hospital deve buscar trazer esperança, e o médico deve lembrar que, ao tratar um doente, está lidando com uma unidade indivisível de medos, histórias e sentimentos. A tecnologia passa, a máquina obsoleta é trocada, mas o impacto de um atendimento humano perdura na memória do paciente e de seus familiares.
#CirurgiaDigestiva #HumanizaçãoMédica #Bioética #EducaçãoMédica #OncologiaCirurgica
“O único meio de combater a praga da impessoalidade e da frieza na medicina moderna é cultivar as humanidades no médico, pois o segredo do cuidado com o paciente é importar-se com o paciente.” — Adaptado de Francis Peabody (embora clínico, sua máxima é a pedra angular para a humanização cirúrgica moderna).
Gostou? ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat Online em nossa DM do Instagram.
Princípios Fundamentais da Oncologia Cirúrgica Digestiva
Uma Abordagem Contemporânea e Baseada em Evidências
O Cenário Atual do Câncer Digestivo no Brasil
A Cirurgia do Aparelho Digestivo vive um momento de transformação sem precedentes. Não somos mais apenas “técnicos de ressecção”, mas parte integrante de uma complexa engrenagem multidisciplinar. A relevância deste tema é sublinhada pelos dados epidemiológicos alarmantes. Se no passado nos baseávamos em estimativas modestas, hoje a realidade é desafiadora: segundo a Estimativa 2023-2025 do Instituto Nacional de Câncer (INCA), esperam-se 704 mil casos novos de câncer por ano no Brasil.
Destaque-se que as neoplasias do trato gastrointestinal ocupam posições cimeiras. O câncer colorretal figura como o segundo mais incidente em mulheres e homens na maioria das regiões, com cerca de 45 mil novos casos anuais, seguido de perto pelo câncer de estômago (21 mil casos) e esôfago. Estes números não são apenas estatísticas; representam uma demanda crescente por cirurgiões oncológicos altamente qualificados, capazes de compreender não apenas a anatomia, mas a biologia tumoral.
A Biologia como Norte da Técnica Cirúrgica
Fisiopatologia e Disseminação
O entendimento clássico da cirurgia oncológica, herdado dos princípios de William Halsted no final do século XIX, baseava-se na premissa de que o câncer era uma doença puramente local que se disseminava centrifugamente. Embora a radicalidade (ressecção em bloco) permaneça um pilar, hoje compreendemos a doença como sistêmica desde fases precoces em muitos casos.
A disseminação ocorre por três vias principais que o cirurgião deve dominar:
- Linfática: Predominante em carcinomas (ex: adenocarcinoma gástrico e cólon).
- Hematogênica: Preferencial em sarcomas e carcinomas avançados (fígado e pulmões como sítios-alvo).
- Transcelômica (Peritoneal): Comum em neoplasias gástricas T3/T4, ovário e apêndice, exigindo estratégias específicas como a peritoniectomia.
O Princípio da Radicalidade e Margens (R0)
O objetivo primário da cirurgia oncológica curativa é a ressecção R0 (ausência de doença residual macroscópica e microscópica). A cirurgia R1 (doença microscópica residual) ou R2 (macroscópica) impacta drasticamente o prognóstico.
- Ressecção em Bloco: O tumor nunca deve ser violado. A peça deve ser removida envolta por tecido saudável, respeitando as fáscias anatômicas e os pedículos vasculares na sua origem.
- Linfadenectomia: Não serve apenas para estadiamento, mas tem papel terapêutico. No câncer gástrico, por exemplo, a linfadenectomia D2 é o padrão-ouro em centros especializados, associada a menor recidiva locorregional.
Neoadjuvância vs. Adjuvância
A decisão entre operar primeiro (upfront surgery) ou indicar terapia neoadjuvante é um dos grandes debates atuais.
- Vantagens da Neoadjuvância: Tratamento precoce de micrometástases, redução do tumor (downstaging) facilitando a ressecção R0 e teste in vivo da quimiossensibilidade. É o padrão atual para câncer de esôfago localmente avançado e câncer de reto médio/baixo.
- Vantagens da Adjuvância: Baseada no estadiamento patológico preciso (pTNM), evitando tratamento excessivo em estádios precoces.
Aplicação Prática na Cirurgia Digestiva
A prática moderna exige que o cirurgião diferencie dois conceitos cruciais frequentemente confundidos: Ressecabilidade e Operabilidade.
- Ressecabilidade: É uma característica do tumor (relação com estruturas vitais).
- Operabilidade: É uma característica do paciente (reserva funcional, comorbidades, status performance). Um tumor pode ser ressecável, mas o paciente inoperável.
O Papel da Citorredução e HIPEC
Para a carcinomatose peritoneal, historicamente considerada uma condição terminal, houve uma mudança de paradigma. Em neoplasias selecionadas (como pseudomixoma peritoneal, mesotelioma e alguns casos de câncer colorretal), a combinação de Cirurgia de Citorredução (Peritoniectomia) com Quimioterapia Intraperitoneal Hipertérmica (HIPEC) tem oferecido sobrevida em longo prazo, transformando uma doença fatal em uma condição crônica tratável.
Planejamento Multidisciplinar
O cirurgião oncológico não atua isolado. A discussão em Tumor Boards é mandatória. A indicação cirúrgica deve considerar a biologia molecular (ex: status do gene APC em colorretal, superexpressão de HER2 em gástrico) e a resposta a terapias sistêmicas.
Pontos-Chave para a Prática Cirúrgica
- Estadiamento Preciso: Nunca leve um paciente à sala sem um estadiamento completo. A laparoscopia diagnóstica é fundamental em tumores gástricos e pancreáticos para evitar laparotomias desnecessárias em casos de carcinomatose oculta.
- Margens Cirúrgicas: A margem circunferencial (radial) no câncer de reto e a margem proximal no câncer gástrico e esofágico são preditores independentes de sobrevida.
- Manuseio da Peça (“No-touch technique”): Evite a manipulação direta do tumor. A ligadura vascular prévia e a mobilização cuidadosa previnem a embolização tumoral intraoperatória.
- Documentação: O relatório cirúrgico deve detalhar as cadeias linfáticas dissecadas e as estruturas preservadas ou ressecadas, orientando o patologista e o oncologista clínico.
Perspectivas Futuras
A cirurgia digestiva na sua área de atuação oncológica evoluiu de amputações extensas para procedimentos de precisão, muitas vezes minimamente invasivos (laparoscópicos ou robóticos), sem perder a radicalidade oncológica. O futuro aponta para uma integração ainda maior com a biologia molecular e a imunoterapia. O cirurgião do futuro deverá ser, antes de tudo, um oncologista que opera: alguém que entende que o bisturi é apenas uma das armas, e que saber quando não operar é tão vital quanto a técnica operatória refinada.
Como nos ensinou o pai da cirurgia oncológica moderna:
“O cirurgião deve ser o médico do paciente oncológico, e não apenas o técnico que remove o tumor.” — William Stewart Halsted
Hashtags
#CirurgiaDigestiva #OncologiaCirurgica #EducaçãoMédica #ResidenciaCirurgia #CancerDigestivo
Gostou? ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat Online em nossa DM do Instagram.
Efeito Cascata do Transplante Hepático
Introdução
O transplante hepático é universalmente reconhecido como o “padrão-ouro” para o tratamento de doenças hepáticas terminais. No entanto, o impacto deste procedimento transcende a substituição de um órgão doente. O sucesso fenomenal do transplante nas últimas cinco décadas produziu o que chamamos de ripple effect (efeito cascata) sobre toda a cirurgia geral e, especificamente, sobre a cirurgia hepatobiliar. Muitos dos princípios anatômicos, refinamentos técnicos e bases científicas que hoje aplicamos rotineiramente em hepatectomias regradas e cirurgias de trauma foram desenvolvidos ou aperfeiçoados nas salas de transplante. O objetivo desta exposição é dissecar como essa “escola” transformou a nossa prática diária, convertendo procedimentos outrora considerados de risco proibitivo em operações seguras e eficazes.
Desenvolvimento: Anatomia e Fisiologia Aplicadas
A base de qualquer cirurgia hepática segura é o domínio absoluto da anatomia e da fisiologia. O transplante nos forçou a olhar para o fígado não apenas como uma massa parenquimatosa, mas como uma estrutura segmentar com variações vasculares frequentes.
1. O Novo Mapa Anatômico
A experiência com doadores vivos e hepatectomias em cadáveres nos ensinou que a anatomia “de livro” é a exceção, não a regra.
- Variações Arteriais: Estudos clássicos de grandes centros transplantadores, como a série da UCLA, demonstram que aproximadamente 24% dos fígados possuem anomalias arteriais significativas. As mais comuns incluem a artéria hepática direita acessória ou substituída (originada da artéria mesentérica superior) e a esquerda (originada da artéria gástrica esquerda). O cirurgião que ignora essas variantes durante uma duodenopancreatectomia ou gastrectomia corre o risco de desvascularizar o fígado.
- Variações Biliares: A trifurcação do ducto hepático comum ocorre em cerca de 12% dos casos. O reconhecimento dessas nuances é vital para evitar estenoses e fístulas biliares, as complicações mais temidas no pós-operatório.
2. Regeneração e Isquemia
O transplante impulsionou a pesquisa sobre a capacidade regenerativa do fígado. O conceito de síndrome small-for-size (insuficiência hepática pós-resecção por remanescente pequeno) migrou do transplante intervivos para a oncologia. Hoje, calculamos com precisão o volume do fígado remanescente antes de grandes ressecções tumorais, utilizando estratégias como a embolização prévia da veia porta para hipertrofiar o lobo que ficará no paciente — uma aplicação direta do conhecimento de regeneração hepática. Além disso, o manuseio da lesão de isquemia-reperfusão evoluiu. Técnicas de precondicionamento isquêmico (clampeamento intermitente) permitem que realizemos ressecções complexas com menor perda sanguínea e menor dano hepatocelular.
Aplicação na Cirurgia Digestiva Geral e Oncológica
A transferência de tecnologia do transplante para a cirurgia digestiva geral é evidente em três pilares principais:
1. Ressecções Hepáticas Complexas e Preservação Caval
Antigamente, tumores no lobo caudado ou que envolviam a veia cava retro-hepática eram considerados irressecáveis. A técnica de “piggyback” (preservação da veia cava inferior do receptor durante o transplante) ensinou aos cirurgiões oncológicos como dissecar o fígado da veia cava com segurança, permitindo a ressecção de tumores centrais e posteriores com margens livres.
2. Controle Vascular no Trauma
O cirurgião de trauma moderno utiliza manobras de exclusão vascular total (clampeamento da porta e da veia cava supra e infra-hepática) para reparar lesões venosas complexas em fígados traumatizados. Esta é uma manobra derivada diretamente da hepatectomia do receptor no transplante. Em pacientes estáveis, isso permite reparos exangues; em instáveis, técnicas de damage control com shunts portocavais temporários podem ser salvadoras.
3. Cirurgia Ex Situ
Para casos extremos de tumores invadindo a confluência cavo-hepática, a técnica de hepatectomia total, seguida de perfusão fria do órgão na bancada (bench surgery), ressecção do tumor ex vivo e reimplante do fígado (autotransplante), é a fronteira final da cirurgia hepatobiliar, tornada possível apenas pelo domínio das técnicas de preservação de órgãos.
Cenário Brasileiro: Uma Potência Mundial 🇧🇷
É fundamental contextualizar nossa realidade. O Brasil possui o maior sistema público de transplantes do mundo.
- Segundo dados recentes da Associação Brasileira de Transplante de Órgãos (ABTO) e do Ministério da Saúde, o Brasil realiza mais de 2.000 transplantes hepáticos anualmente, posicionando-se consistentemente entre as três nações com maior número absoluto de procedimentos no mundo.
- Essa estatística não é apenas um número; ela representa um volume crítico de treinamento. Residentes brasileiros em centros de excelência têm uma exposição prática à anatomia hepática complexa superior à de muitos países desenvolvidos. O “Cirurgião SUS” é, por necessidade e oportunidade, um especialista em variações anatômicas e manuseio de situações complexas.
Pontos-Chave para o Cirurgião em Formação
- Identificação Pré-operatória: Sempre investigue variações arteriais (ex: artéria hepática direita vindo da mesentérica) em exames de imagem antes de qualquer cirurgia do andar supramesocólico.
- Manobras de Exclusão: Familiarize-se com a Manobra de Pringle e a exclusão vascular total; elas são suas ferramentas de segurança em sangramentos maciços.
- Dissecção Hilar: A técnica de baixar a placa hilar e dissecar as estruturas glissonianas extra-hepáticas é mais segura e oncológica do que a dissecção intraparenquimatosa cega.
- Interdisciplinaridade: A cirurgia moderna não é um ato solitário. Radiologia intervencionista, hepatologia e terapia intensiva são extensões do braço do cirurgião.
Conclusão
O transplante hepático não deve ser visto pelos estudantes e residentes apenas como uma subespecialidade de nicho, mas como a “Universidade da Cirurgia Abdominal”. As lições aprendidas com a preservação de órgãos, a dissecção meticulosa de vasos de calibre milimétrico e o manejo fisiológico do paciente hepatopata elevaram o padrão técnico de toda a cirurgia digestiva. Dominar esses conceitos é o que diferencia o operador técnico do verdadeiro cirurgião cientista.
“A história da medicina é que o que era inconcebível ontem, e apenas alcançável hoje, muitas vezes torna-se rotina amanhã.” — Thomas Starzl (Pioneiro do Transplante Hepático)
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags: #CirurgiaHepatobiliar #TransplanteHepatico #EducacaoMedica #ResidenciaCirurgia #CirurgiaDigestiva
A Importância da Competência em Endoscopia Flexível na Formação e Prática do Cirurgião do Aparelho Digestivo
Introdução
A endoscopia digestiva flexível transcende a sua definição clássica de método diagnóstico. Na contemporaneidade, ela se consolida como uma extensão essencial da propedêutica e da terapêutica cirúrgica. Historicamente, é imperativo recordar que a endoscopia é um campo desbravado por cirurgiões. Grandes marcos, como a polipectomia colônica (Dr. Hiromi Shinya), a ligadura elástica de varizes (Dr. Goffredo Steigman) e a gastrostomia percutânea (Dr. Jeffrey Ponsky), foram estabelecidos por mentes cirúrgicas que vislumbravam além da incisão convencional. No entanto, observa-se um fenômeno preocupante na formação médica atual: a delegação progressiva dessa competência quase exclusivamente à gastroenterologia clínica e aos Endoscopistas. Este artigo, direcionado a estudantes, residentes e pós-graduandos, visa reafirmar a premissa de que a habilidade endoscópica não é opcional, mas intrínseca à prática do cirurgião do aparelho digestivo de excelência. Discutiremos as bases técnicas, a aplicabilidade clínica e a necessidade premente de retomar o protagonismo nesta área.
Desenvolvimento: O Cirurgião e a Habilidade Endoscópica
A Curva de Aprendizado e a Visão Cirúrgica
A competência em endoscopia flexível depende da fusão entre conhecimento cognitivo e habilidade técnica (psicomotora). O cirurgião, habituado à manipulação tecidual e à anatomia tridimensional (seja por via aberta ou laparoscópica), possui, intrinsecamente, uma coordenação olho-mão refinada. Estudos demonstram que cirurgiões tendem a apresentar uma curva de aprendizado acelerada para procedimentos endoscópicos devido à sua familiaridade com a anatomia topográfica e patológica, permitindo uma transição fluida entre a visão bidimensional da tela e a realidade espacial do órgão.
O Cenário Brasileiro e a Demografia Médica
No contexto do Brasil, um país de dimensões continentais, a distribuição de especialistas é heterogênea. Segundo dados da Demografia Médica no Brasil (CFM/USP, 2023), a concentração de gastroenterologistas clínicos | endoscopistas é significativamente menor e mais centralizada em grandes capitais do que a de cirurgiões gerais. O cirurgião geral, muitas vezes, é o único provedor de saúde especializado em regiões remotas. Portanto, capacitar o cirurgião em endoscopia não é apenas uma questão de reserva de mercado, mas uma estratégia de saúde pública para garantir o rastreamento de neoplasias (como o câncer colorretal) e o atendimento de urgências (Hemorragia Digestiva Alta) em todo o território nacional.
Do “Cirurgião de Resgate” ao “Cirurgião Estrategista”
Tradicionalmente, o cirurgião é chamado apenas quando ocorre uma complicação endoscópica, como perfuração ou sangramento incontrolável. Este paradigma do “cirurgião de resgate” deve evoluir para o de “cirurgião estrategista”. Ao dominar a endoscopia, o profissional torna-se o capitão do navio terapêutico, capaz de indicar, executar e, se necessário, converter procedimentos com total autonomia e continuidade do cuidado (Continuity of Care), otimizando desfechos e minimizando a fragmentação do tratamento.
Aplicação na Cirurgia Digestiva
A integração da endoscopia na rotina cirúrgica pode ser categorizada em três momentos cruciais:
1. Endoscopia Pré-Operatória: O Planejamento
Na cirurgia oncológica gástrica e esofágica, a endoscopia realizada pelo próprio cirurgião permite uma avaliação precisa das margens tumorais, da extensão para a cárdia ou piloro, influenciando diretamente a decisão entre uma gastrectomia total ou subtotal. Na cirurgia bariátrica, a identificação prévia de hérnias hiatais, esofagites severas ou patologias gástricas altera o algoritmo de tratamento, contraindicando, por exemplo, uma gastrectomia vertical (Sleeve) em favor de um bypass gástrico em Y de Roux.
2. Endoscopia Intraoperatória: A Segurança
O uso transoperatório é uma ferramenta de segurança inestimável. Exemplos clássicos incluem:
- Cardiomiotomia de Heller: Confirmação da adequação da miotomia e teste de vazamento (air leak test) para exclusão de perfurações mucosas.
- Exploração de Vias Biliares: O uso do coledocoscópio flexível permite a visualização direta e a certeza da remoção completa de cálculos, superior à colangiografia em casos complexos.
- Localização de Lesões: Em cirurgias colorretais laparoscópicas, a tatuagem prévia pode ser imprecisa; a colonoscopia intraoperatória garante a ressecção do segmento correto.
3. Endoscopia Pós-Operatória e Manejo de Complicações
O cirurgião que realiza a endoscopia está melhor posicionado para tratar as complicações de seus próprios procedimentos ou de seus pares. O arsenal terapêutico inclui:
- Dilatação com balão de estenoses de anastomoses (gástricas ou colorretais).
- Tratamento de fístulas e deiscências pós-Sleeve ou Bypass com uso de próteses autoexpansíveis ou clips endoscópicos.
- Hemostasia de linhas de sutura sangrantes.
A Era da Cirurgia Sem Cicatrizes (NOTES e Endoterapia)
Caminhamos para a era da intervenção minimamente invasiva máxima. Procedimentos como o POEM (Peroral Endoscopic Myotomy) substituem a miotomia laparoscópica; o Endoscopic Sleeve Gastroplasty (ESG) surge como alternativa à cirurgia bariátrica convencional. O cirurgião do futuro deve dominar estas técnicas para oferecer o tratamento “estado da arte” aos seus pacientes.
Pontos-Chave para o Residente e o Cirurgião Jovem
- Necessidade de Treinamento: As sociedades cirúrgicas (como o CBC e a CBCD no Brasil) têm enfatizado a importância de currículos de residência que contemplem carga horária prática em endoscopia.
- Visão Integral: O domínio da luz luminal (endoscopia) e da cavidade peritoneal (laparoscopia/robótica) cria um cirurgião completo.
- Autonomia: A capacidade de diagnosticar, estadiar, tratar e resolver complicações sem depender de terceiros é o ápice da eficiência clínica.

Conclusão
A endoscopia flexível não é uma especialidade à parte, mas uma ferramenta cirúrgica, tal qual o bisturi ou a pinça laparoscópica. Para o cirurgião do aparelho digestivo, delegar essa competência é abdicar de uma parte fundamental da sua herança histórica e do seu futuro profissional. A formação médica deve encorajar a retomada deste espaço, garantindo que as novas gerações de cirurgiões sejam proficientes tanto na ciência da cirurgia quanto na arte da endoscopia. Como mentores e instituições de ensino, nosso dever é prover o acesso e a estrutura para que essa integração seja a norma, e não a exceção.
“Nenhum homem pode ser um grande cirurgião sem ser também um grande médico; e o inverso é igualmente verdadeiro no que tange ao diagnóstico e compreensão da fisiopatologia.” > — Adaptado de Theodor Billroth, pai da cirurgia gástrica moderna, refletindo a necessidade do conhecimento integral.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags: #CirurgiaDigestiva #EndoscopiaTerapeutica #ResidenciaMedica #CirurgiaGeral #EducacaoMedica
Epônimos da Anatomia Abdominal: A Linguagem Clássica da Prática Cirúrgica
Resumo Apesar dos esforços contínuos da Federative International Programme on Anatomical Terminologies (FIPAT) para padronizar a nomenclatura médica através da Terminologia Anatômica, a linguagem operatória permanece, em essência, histórica. Este artigo revisa os epônimos fundamentais da anatomia abdominal, correlacionando a definição topográfica precisa com a aplicação prática na cirurgia do aparelho digestivo, demonstrando que o domínio destes termos é imperativo para a segurança cirúrgica e a comunicação clínica eficaz.
Introdução
A anatomia topográfica é o alicerce da cirurgia. Contudo, existe uma dicotomia persistente entre a taxonomia descritiva moderna e a tradição oral dos centros cirúrgicos. Enquanto a academia privilegia termos locativos e funcionais, a prática diária — da passagem de plantão à descrição operatória — é dominada por epônimos. Estes nomes não são meros vestígios históricos; funcionam como “atalhos cognitivos” que evocam, simultaneamente, uma localização, uma relação anatômica complexa e, frequentemente, uma manobra cirúrgica específica. Este estudo revisa os principais epônimos do abdome, dissecando sua anatomia estrutural e sua relevância crítica na cirurgia geral e digestiva.
1. Parede Abdominal e Região Inguinal
A compreensão da estratigrafia da parede abdominal é o primeiro passo para o acesso seguro à cavidade peritoneal e para o reparo eficaz dos defeitos herniários.
Estratigrafia Subcutânea e Fascial
- Fáscia de Camper
- Anatomia: Camada superficial, de espessura variável e consistência adiposa, do tecido subcutâneo da parede abdominal anterior.
- Relevância Cirúrgica: A sua aproximação é frequentemente negligenciada, mas o manejo adequado do espaço morto nesta camada é vital na prevenção de seromas e infecções de sítio cirúrgico superficiais em laparotomias.
- Fáscia de Scarpa
- Anatomia: Camada membranosa profunda do tecido subcutâneo, contendo fibras elásticas amarelas, que se funde com a fáscia lata da coxa inferiormente e continua como Fáscia de Colles no períneo.
- Relevância Cirúrgica: Estrutura de fixação essencial no fechamento da parede abdominal. A sutura desta camada reduz a tensão sobre a pele e melhora o resultado estético da cicatriz. Devido à sua continuidade perineal, orienta a disseminação de extravasamentos de urina (fraturas de uretra) ou infecções necrotizantes (Síndrome de Fournier).
- Fáscia Inominada de Gallaudet
- Anatomia: A fáscia profunda aderida intimamente à aponeurose do músculo oblíquo externo e bainha do reto.
- Relevância Cirúrgica: Define o plano de dissecção “limpo” sobre a aponeurose durante o reparo de hérnias ou a confecção de retalhos miocutâneos.
Anatomia Inguinal e o Assoalho Pélvico
- Ligamento de Poupart (Ligamento Inguinal)
- Anatomia: A borda inferior espessa e recorvada da aponeurose do oblíquo externo, estendendo-se da Espinha Ilíaca Antero-Superior (EIAS) ao tubérculo púbico.
- Relevância Cirúrgica: O marco anatômico absoluto que diferencia hérnias inguinais (acima) de femorais (abaixo) e serve como âncora inferior para reparos teciduais clássicos (técnica de Bassini) e fixação de telas (técnica de Lichtenstein).
- Ligamento de Gimbernat (Ligamento Lacunar)
- Anatomia: Uma extensão triangular das fibras do ligamento inguinal que se reflete posteriormente e se insere na linha pectínea.
- Relevância Cirúrgica: Forma a borda medial do anel femoral. Em casos de hérnia femoral encarcerada, este ligamento é frequentemente a estrutura constritora que deve ser incisada (com cautela devido à presença eventual da Corona Mortis vascular) para redução do conteúdo.
- Ligamento de Cooper (Ligamento Pectíneo)
- Anatomia: Espessamento do periósteo e fáscia ao longo da crista pectínea (pecten do púbis), posterior ao ligamento inguinal.
- Relevância Cirúrgica: Estrutura de ancoragem robusta utilizada na técnica de McVay e ponto de fixação crítico para telas em reparos laparoscópicos (TAPP/TEP), prevenindo a recidiva direta.
- Triângulo de Hesselbach
- Anatomia: Delimitado inferiormente pelo ligamento inguinal, lateralmente pelos vasos epigástricos inferiores e medialmente pela borda lateral do músculo reto abdominal.
- Relevância Cirúrgica: Local anatômico das hérnias inguinais diretas, resultantes da fraqueza da fáscia transversal, sem passagem pelo anel inguinal profundo.
- Orifício Miopectíneo de Fruchaud
- Anatomia: Área de fraqueza da parede abdominal inferior que engloba tanto a região inguinal quanto a femoral, delimitada pelo músculo oblíquo interno (superior), reto abdominal (medial), iliopsoas (lateral) e osso púbico (inferior).
- Relevância Cirúrgica: Conceito fundamental para a cirurgia moderna de hérnia. Estabelece que o tratamento definitivo deve cobrir toda esta área com material protético (tela) para prevenir todos os tipos de hérnias da virilha, princípio base das abordagens pré-peritoneais (Stoppa, TEP, TAPP).
2. Peritônio e Topografia Gastrointestinal
A semiologia abdominal e o acesso cirúrgico baseiam-se em projeções de superfície e recessos cavitários.
- Linha de Monro-Richter e Ponto de McBurney
- Anatomia: Linha traçada da EIAS à cicatriz umbilical. O Ponto de McBurney localiza-se na união do terço lateral com os dois terços mediais desta linha.
- Relevância Cirúrgica: Projeção clássica da base do apêndice cecal. Guia a incisão oblíqua (McBurney) para apendicectomias abertas e é o ponto de máxima dor à descompressão na apendicite aguda.
- Ponto de Lanz
- Anatomia: Ponto na junção do terço direito com o terço médio da linha bi-ilíaca.
- Relevância Cirúrgica: Representa a projeção variável do ápice de um apêndice pélvico longo, importante no diagnóstico diferencial de patologias anexiais em mulheres.
- Ponto de Murphy
- Anatomia: Ponto situado abaixo do rebordo costal direito, na linha hemiclavicular (borda lateral do reto abdominal).
- Relevância Cirúrgica: Local de palpação do fundo da vesícula biliar. A interrupção súbita da inspiração à palpação profunda (Sinal de Murphy) é altamente sugestiva de colecistite aguda.
- Espaços Peritoneais (Morison, Douglas, Proust, Winslow)
- Bolsa de Morison (Recesso Hepatorrenal): Espaço virtual entre o fígado e o rim direito. É o local mais dependente do abdome superior em decúbito dorsal; local primário de acúmulo de líquido livre (sangue/pus) detectável pelo FAST em trauma.
- Fundo de Saco de Douglas (Retouterino): Ponto mais declive da cavidade peritoneal em mulheres. Local de acúmulo de líquido e de implantes metastáticos palpáveis ao toque retal/vaginal (Prateleira de Blumer).
- Espaço de Proust (Retovesical): Correspondente masculino ao de Douglas, entre o reto e a bexiga/próstata. Relevante na dissecção oncológica do reto baixo.
- Forame de Winslow (Forame Epiploico): Comunicação entre a grande cavidade e a retrocavidade dos epíplons (bolsa omental). Clinicamente, é a via de acesso para a Manobra de Pringle (clampeamento do pedículo hepático para controle de hemorragia) e local potencial para hérnias internas.
3. Trato Digestório e Glândulas Anexas
Nesta região, os epônimos descrevem marcos críticos para ressecções oncológicas e reconstruções.
- Ângulo de His
- Anatomia: Ângulo agudo formado entre o esôfago abdominal e o fundo gástrico.
- Relevância Cirúrgica: Componente anatômico crucial do mecanismo valvular anti-refluxo. A restauração ou acentuação deste ângulo é o objetivo central das fundoplicaturas (Nissen, Toupet) no tratamento da DRGE.
- Espaço de Traube e Triângulo de Labbé
- Anatomia: Áreas de projeção gástrica na parede toracoabdominal.
- Relevância Cirúrgica: O Espaço de Traube (timpânico à percussão) torna-se maciço em casos de esplenomegalia ou derrame pleural. O Triângulo de Labbé é a área de contato do estômago com a parede abdominal anterior, local seguro para gastrostomias percutâneas ou cirúrgicas.
- Ligamento de Treitz
- Anatomia: Músculo suspensor do duodeno, marcando a transição duodeno-jejunal (ângulo de Treitz).
- Relevância Cirúrgica: Divisor anatômico e clínico entre hemorragia digestiva alta e baixa. Marco fundamental para a mobilização do intestino delgado e identificação da primeira alça jejunal em reconstruções (ex: Y de Roux).
- Vias Biliares: Calot, Heister, Vater, Oddi
- Triângulo de Calot (Cisto-Hepático): Delimitado pelo ducto cístico, ducto hepático comum e borda inferior do fígado. Contém a artéria cística e, frequentemente, o linfonodo de Mascagni. Sua dissecção meticulosa para obter a “Visão Crítica de Segurança” (Critical View of Safety) é mandatória para prevenir lesões iatrogênicas da via biliar principal na colecistectomia.
- Válvulas de Heister: Pregas espirais na mucosa do ducto cístico que podem dificultar a cateterização durante a colangiografia intraoperatória.
- Ampola de Vater e Esfíncter de Oddi: A confluência biliopancreática e seu aparelho esfincteriano. Alvo terapêutico nas papilotomias endoscópicas (CPRE) para tratamento de coledocolitíase ou pancreatite biliar.
- Ductos Pancreáticos (Wirsung e Santorini)
- Anatomia: Wirsung é o ducto principal; Santorini é o acessório.
- Relevância Cirúrgica: A variante Pancreas Divisum (falha na fusão dos ductos) drena a maior parte do pâncreas pelo ducto menor (Santorini), sendo causa de pancreatites recorrentes inexplicadas.
- Marcos Hepáticos (Glisson e Cantlie)
- Cápsula de Glisson: Bainha de tecido conjuntivo que envolve o fígado e a tríade portal intra-hepática. Importante na manobra de Pringle e nas hepatectomias.
- Linha de Cantlie: Linha imaginária que divide o fígado funcionalmente em lobos direito e esquerdo, estendendo-se do leito da vesícula biliar à veia cava inferior. Base para hepatectomias anatômicas maiores.
4. Vascularização Abdominal e Colateralização
O conhecimento das arcadas vasculares é determinante para a viabilidade de anastomoses intestinais.
- Tripus Halleri (Tronco Celíaco)
- Anatomia: Trifurcação da aorta abdominal em artéria gástrica esquerda, esplênica e hepática comum.
- Relevância Cirúrgica: O controle vascular proximal em cirurgias de trauma, gastrectomias e pancreatectomias. Variações anatômicas são frequentes e devem ser antecipadas em planejamentos pré-operatórios.
- Arcada de Riolan e Artéria Marginal de Drummond
- Anatomia: Sistemas de anastomose entre a artéria cólica média (ramo da mesentérica superior) e a cólica esquerda (ramo da mesentérica inferior).
- Relevância Cirúrgica: Garantem a irrigação do cólon, especialmente na flexura esplênica (ponto crítico de Griffiths). A preservação ou a integridade funcional destas arcadas é vital para evitar isquemia do coto colônico em retossigmoidectomias e cirurgias de aneurisma de aorta.
- Veias de Sappey e Retzius
- Anatomia: Veias acessórias do sistema porta. As de Sappey correm no ligamento falciforme; as de Retzius são anastomoses retroperitoneais.
- Relevância Cirúrgica: Em pacientes com hipertensão portal, estas veias dilatam-se dramaticamente. As veias de Sappey podem causar hemorragia maciça na incisão supraumbilical. A dissecção do retroperitônio em cirróticos pode encontrar o “sangramento em lençol” das veias de Retzius, de difícil controle.
5. Retroperitônio e Rins
A anatomia dos planos fasciais renais é a chave para a cirurgia oncológica retroperitoneal radical.
- Fáscia de Gerota (e suas lâminas Toldt e Zuckerkandl)
- Anatomia: A fáscia renal envolve o rim e a adrenal em um compartimento fechado. A lâmina anterior é frequentemente associada à Fáscia de Toldt (no contexto da fusão com o mesocólon), e a posterior à Fáscia de Zuckerkandl.
- Relevância Cirúrgica: A “Fáscia de Toldt” (ou linha de Toldt) representa o plano de clivagem avascular embrionário entre o mesocólon e o retroperitônio. A dissecção neste plano permite a mobilização incruenta do cólon direito e esquerdo, essencial em colectomias oncológicas laparoscópicas e abertas. A violação da Fáscia de Gerota propriamente dita é indicativa de invasão tumoral renal ou necessária para nefrectomias radicais.
Conclusão
O domínio dos epônimos da anatomia abdominal transcende o exercício de erudição histórica; constitui uma ferramenta de precisão técnica. Falar em “Triângulo de Calot” ou “Fáscia de Toldt” evoca, instantaneamente, uma estratégia cirúrgica e um plano de segurança. Para o cirurgião do aparelho digestivo, a fluência nesta linguagem clássica é tão fundamental quanto a destreza manual, permitindo a integração segura entre o conhecimento anatômico estático e a dinâmica do ato operatório.
Referência-Base: ten Donkelaar HJ, Quartu M, Kachlík D. An Illustrated Guide to Anatomical Eponyms. Springer, 2025.
Índice Biográfico dos Epônimos Citados
Abaixo listam-se os dados biográficos dos anatomistas e cirurgiões cujos epônimos foram discutidos neste artigo.
- Blumer, George (Reino Unido/EUA, 1858–1940)
- Calot, Jean-François (França, 1861–1944)
- Camper, Pieter (Holanda, 1722–1789)
- Cantlie, James (Escócia, 1851–1926)
- Colles, Abraham (Irlanda, 1773–1843)
- Cooper, Astley Paston (Reino Unido, 1768–1841)
- Douglas, James (Escócia, 1675–1742)
- Drummond, Hamilton (Reino Unido, 1882–1925)
- Fruchaud, Henri (França, 1894–1960)
- Gallaudet, Bern Budd (EUA, 1860–1934)
- Gerota, Dimitrie (Romênia, 1867–1939)
- Gimbernat, Antoni de (Espanha, 1734–1816)
- Glisson, Francis (Reino Unido, 1597–1677)
- Griffiths, Joseph (País de Gales, 1863–1945)
- Haller, Albrecht von (Suíça, 1708–1777)
- Heister, Lorenz (Alemanha, 1683–1758)
- Hesselbach, Franz Kaspar (Alemanha, 1759–1816)
- His, Wilhelm Jr. (Suíça, 1863–1934)
- Labbé, Léon (França, 1832–1916)
- Lanz, Otto (Suíça, 1865–1935)
- Mascagni, Paolo (Itália, 1755–1815)
- McBurney, Charles (EUA, 1845–1913)
- Monro, Alexander III (Escócia, 1773–1859)
- Morison, James Rutherford (Reino Unido, 1853–1939)
- Murphy, John Benjamin (EUA, 1857–1916)
- Oddi, Ruggero (Itália, 1864–1913)
- Poupart, François (França, 1661–1709)
- Pringle, James Hogarth (Escócia, 1863–1941)
- Proust, Robert (França, 1873–1935)
- Retzius, Anders (Suécia, 1796–1860)
- Richter, August Gottlieb (Alemanha, 1742–1812)
- Riolan, Jean (o Jovem) (França, 1580–1657)
- Santorini, Giovanni Domenico (Itália, 1681–1737)
- Sappey, Marie Philibert Constant (França, 1810–1896)
- Scarpa, Antonio (Itália, 1752–1832)
- Toldt, Carl (Áustria, 1840–1920)
- Traube, Ludwig (Alemanha, 1818–1876)
- Treitz, Václav (República Tcheca/Áustria, 1819–1872)
- Vater, Abraham (Alemanha, 1684–1751)
- Winslow, Jacob Benignus (Dinamarca/França, 1669–1760)
- Wirsung, Johann Georg (Alemanha, 1589–1643)
- Zuckerkandl, Emil (Áustria, 1849–1910)
Lifelong Learning na Cirurgia: como a Aprendizagem Autorregulada Define o Cirurgião que Você Vai Ser
Introdução
A cirurgia é uma das áreas da medicina que mais muda ao longo do tempo. Técnicas, tecnologias, diretrizes e condutas são revisadas continuamente. Ninguém termina a residência “pronto para tudo”. O que diferencia o cirurgião que se mantém competente e atualizado ao longo da carreira não é apenas o que aprendeu na formação inicial, mas a capacidade de continuar aprendendo de forma ativa, intencional e estruturada.
Esse é o núcleo do conceito de Lifelong Learning: o compromisso de adquirir, revisar e integrar novos conhecimentos desde o primeiro dia de faculdade até o último dia de atividade profissional. E, na prática, o que sustenta isso é um conjunto de habilidades chamado aprendizagem autorregulada.
O problema: bons alunos, maus aprendizes
Grande parte dos estudantes que chegam à residência é formada por “altos desempenhos acadêmicos”. Mas muitos:
- atribuem sucesso e fracasso quase sempre ao professor, ao serviço ou ao tipo de prova;
- não conseguem descrever com clareza como estudam;
- acreditam que aprender é algo que “acontece com eles”, não algo que podem controlar.
Esse modelo funciona em um ambiente escolar tradicional, com provas previsíveis e conteúdo delimitado. Em cirurgia, não. No centro cirúrgico, na UTI ou no pronto-socorro, o cirurgião depende de outra coisa: da capacidade de identificar o que não sabe, de estudar com foco e de ajustar a própria prática a partir de resultados reais.
É aqui que entra a aprendizagem autorregulada.
O que é aprendizagem autorregulada?
Aprendizagem autorregulada é o conjunto de hábitos, estratégias e atitudes que fazem o aluno assumir o controle do próprio processo de aprendizagem.
Um aprendiz autorregulado:
- pensa sobre como aprende (metacognição);
- acredita que é capaz de melhorar com esforço e estratégia (autoeficácia realista);
- organiza o ambiente, o tempo e os recursos para aprender melhor (comportamento ativo).
Na prática, isso aparece em três dimensões:
1. Metacognitiva
- Define objetivos de aprendizado (“quero entender critérios de indicação de neoadjuvância no pâncreas”, “quero melhorar decisão em vesícula difícil”).
- Planeja como chegar lá (o que ler, que casos observar, que vídeos rever).
- Monitora se está, de fato, avançando.
- Se autoavalia com honestidade ao final.
2. Motivacional
- Liga esforço a desempenho.
- Não se vê como “bom” ou “ruim”, mas como alguém em processo de desenvolvimento.
- Usa erros como feedback, não como sentença.
3. Comportamental
- Seleciona ativamente casos, plantões e oportunidades que trazem aprendizado.
- Busca ajuda, feedback e coaching quando necessário.
- Usa estratégias de estudo estruturadas, não apenas leitura passiva.
O ciclo da aprendizagem autorregulada
Você pode enxergar esse processo como um ciclo contínuo:
- Planejamento (forethought)
- O que quero aprender?
- Por que isso é importante agora?
- Quanto tempo vou dedicar? Com que materiais?
- Execução (performance)
- Implementar o plano (leitura, vídeo, simulação, prática supervisionada).
- Monitorar em tempo real: estou entendendo? estou apenas decorando? estou aplicando?
- Reflexão (self-reflection)
- O que funcionou? O que não funcionou?
- O problema foi falta de esforço, estratégia inadequada, falta de recurso ou algo fora do meu controle?
- O que vou manter, o que vou mudar no próximo ciclo?
Quem atribui tudo a “azar”, “caso difícil”, “erro do serviço” sai mais fraco do caso.
Quem atribui a fatores ajustáveis (estratégia, preparação, decisão) sai mais forte, mesmo depois de um erro.
Ferramentas práticas para residentes e cirurgiões
1. Autoavaliação de como você aprende
Não é apenas “sou bom ou ruim”, mas:
- eu planejo o que estudar ou vou “apagando incêndio”?
- eu mudo de estratégia quando não entendo um tema?
- eu reviso os casos difíceis depois do plantão?
- eu procuro ativamente feedback objetivo sobre minha performance?
Transformar isso em rotina escrita (um caderno, um app, uma planilha) ajuda a tirar a aprendizagem do improviso e colocá-la em modo profissional.
2. Coaching cirúrgico
Coaching não é “mais uma aula”; é uma conversa estruturada para:
- definir objetivos de melhoria claros (ex.: decisão de conversão; planejamento de colecistectomia difícil; comunicação com a equipe);
- identificar pontos cegos (o que você não está vendo sobre a própria prática);
- desenhar um plano concreto de treinamento e estudo.
Ferramentas de vídeo-coaching (assistir a uma operação sua com um colega experiente e revisar decisões, tempos e manobras) têm efeito duplo: refinam a técnica e amplificam a sua metacognição.
3. Leitura inteligente: saindo do “sublinhar tudo”
Um exemplo prático é a estratégia SQ3R para capítulos e diretrizes:
- Survey (percurso) – passar rapidamente pelos subtítulos, tabelas, figuras.
- Question (perguntas) – transformar subtítulos em perguntas (“quando indicar intervalo apendicectomia?”, “como manejar abscesso apendicular?”).
- Read (leitura) – ler com foco em responder suas próprias perguntas.
- Recall (recordar) – fechar o texto e anotar o que lembra de cabeça.
- Review (revisar) – conferir no texto e corrigir lacunas.
É mais trabalhoso do que reler passivamente, mas a retenção é muito maior — e é isso que interessa na prática cirúrgica.
Aplicando isso na formação cirúrgica
Na rotina de um serviço de cirurgia, aprendizagem autorregulada se traduz em ações muito concretas:
- Antes do plantão: definir 1–2 objetivos de aprendizado (por exemplo, “revisar escore de Alvarado e conduta em apendicite complicada”).
- Durante o plantão: escolher conscientemente 1–2 casos para estudar em profundidade depois.
- Após a cirurgia: registrar rapidamente:
- o que foi bem,
- o que não foi,
- o que você precisa estudar para a próxima situação semelhante.
No nível do serviço, vale estimular:
- discussão de M&M com foco em análise de processo, não apenas em “culpa”;
- preceptores que verbalizam seu raciocínio e seus próprios erros;
- metas claras por ano de residência (o que se espera que o R1, R2, R3 saiba de fato).
Pontos-chave para o cirurgião que quer ser lifelong learner
- Assuma o comando do próprio aprendizado – ninguém fará isso por você.
- Planeje o estudo como planeja uma cirurgia – com objetivo, estratégia e checagem.
- Use erro e desconforto como combustível, não como fonte de paralisia.
- Busque feedback específico, não elogios genéricos.
- Padronize suas estratégias de leitura e revisão, fuja do improviso.
Conclusão
Formar um cirurgião tecnicamente competente é obrigatório.
Formar um cirurgião capaz de continuar aprendendo, se adaptando e se avaliando ao longo da vida é o verdadeiro diferencial.
Lifelong learning não é um slogan bonito de documento institucional.
É uma competência prática, treinável, que começa na residência, mas precisa acompanhar cada decisão, cada leitura, cada caso difícil.
Quanto mais cedo você organizar o próprio processo de aprender, mais preparado estará para os desafios que ainda nem existem hoje – mas que certamente farão parte da cirurgia de amanhã.
Hashtags (SEO)
#LifelongLearningEmCirurgia #AprendizagemAutorregulada #FormaçãoDoCirurgião #EducaçãoMédicaContinuada #ResidênciaEmCirurgia
Gostou❔ Deixe seu comentário ✍️, compartilhe com seus colegas e mande sua dúvida pelo 💬 Chat On-line em nossa DM no Instagram.
COMPARAÇÃO ENTRE AS TÉCNICAS DE CORREÇÃO DA LESÃO INADVERTIDA DA VIA BILIAR: RAFIA PRIMÁRIA, COLEDOCODUODENOSTOMIA E HEPATICOJEJUNOSTOMIA
1. Introdução
A lesão inadvertida da via biliar (LVB) é a complicação mais grave da colecistectomia. A escolha da técnica reconstrutiva influencia diretamente:
- morbidade imediata,
- risco de estenose tardia,
- necessidade de reoperações,
- qualidade de vida do paciente.
As três técnicas clássicas de reparo são: rafia primária, coledocoduodenostomia (CDD) e hepaticojejunostomia (HJ). Cada uma tem indicações específicas, benefícios, riscos e performance diferente no longo prazo.
2. RAFIA PRIMÁRIA
1. Conceito
Sutura primária do ducto biliar lesado, podendo ou não incluir T-tube (Kehr).
2. Indicações Reais (muito restritas)
- Lesões pequenas (<3 mm).
- Lesões laterais (tipo Strasberg D).
- Lesões reconhecidas imediatamente, antes de isquemia ductal.
- Sem perda de ducto.
- Paciente estável e equipe experiente em HPB.
3. Vantagens
- Técnica simples, rápida.
- Preserva a anatomia original.
- Não exige anastomose complexa.
4. Desvantagens / Problemas Clássicos
- Alto risco de estenose tardia (20–40%).
- Risco de falha se há trauma térmico ou perda segmentar.
- Depende de tecido viável, o que raramente ocorre após clipagem ou eletrocoagulação.
- Necessita drenagem adequada (Kehr), aumento de morbidade.
5. Resultados na Literatura
- Bons resultados em lesões muito precoces e laterais.
- Em perdas ductais ou transecções completas, a falha é a regra.
→ Por isso, seu uso caiu drasticamente e é considerado excepcional.
3. COLEDOCODUODENOSTOMIA (CDD)
1. Conceito
Anastomose término-lateral entre ducto biliar comum e duodeno.
2. Indicações Clássicas
- Estenoses baixas do colédoco (tipos B ou C).
- Lesões distais de fácil acesso.
- Situações especiais: idade avançada, comorbidades, impossibilidade técnica de HJ, cirurgias prévias complexas.
3. Vantagens
- Técnica relativamente simples.
- Tempo cirúrgico menor que HJ.
- Acesso endoscópico pós-operatório facilitado.
4. Desvantagens
- Maior risco de refluxo enterobiliar.
- Colangite recorrente (10–25%).
- Maior incidência de estenose em comparação à HJ a longo prazo.
- Evitada em lesões altas (porta hepatis).
5. Resultados
- Boa taxa de permeabilidade inicial.
- Menor durabilidade em pacientes jovens.
- Taxa de reoperação superior à HJ em seguimento >5 anos.
→ Técnica válida em casos selecionados, mas não é padrão-ouro.
4. HEPATICOJEJUNOSTOMIA (HJ) – PADRÃO-OURO
1. Conceito
Anastomose término-lateral do ducto hepático (ou confluência) com alça jejunal em Roux-en-Y.
2. Indicações
- Transecções completas (Strasberg E1–E5).
- Lesões proximais ou com perda de ducto.
- Lesões tardias diagnosticadas em estenose.
- Reconstrução definitiva após falha de outros métodos.
- Lesão térmica ou isquemia ductal.
3. Vantagens
- Melhor durabilidade entre as técnicas.
- Menor risco de estenose tardia (5–15%).
- Melhor vascularização do ducto.
- Evita refluxo duodenal.
4. Desvantagens
- Cirurgia complexa, exige centro especializado.
- Risco de fístula biliar inicial.
- Acesso endoscópico pós-operatório difícil.
5. Resultados
- Padrão-ouro mundial em lesões significativas.
- Sucesso >85–90% em centros de referência.
- Menor risco de reoperação tardia.
→ É a técnica mais segura e duradoura para a maioria das lesões importantes.
5. TABELA COMPARATIVA
| Técnica | Indicação | Vantagens | Desvantagens | Êxito a longo prazo |
|---|---|---|---|---|
| Rafia primária | Lesões laterais pequenas, precoces | Preserva anatomia | Alta estenose | 60–80% |
| CDD | Lesões distais, baixa lesão | Técnica simples, acesso endoscópico fácil | Refluxo, colangite, mais estenose | 65–85% |
| HJ (Roux-en-Y) | Transecções, perdas ductais, lesões proximais | Melhor durabilidade e segurança | Cirurgia longa e complexa | 85–95% |
6. Avaliação crítica da literatura
- Timing importa mais que técnica
– Reconstrução precoce (<72h) tem melhores resultados se feita em centro especializado.
– Reconstruções tardias (6–12 semanas) podem reduzir edema e inflamação, facilitando HJ. - Presença de isquemia do ducto
– Torna a rafia primária praticamente contraindicada.
– Favorece HJ como opção definitiva. - Perícia cirúrgica
– Estudos multicêntricos mostram falha recorrente quando cirurgiões não HPB tentam reparo primário. - ODM (outcomes dependem do centro)
– HJ feita por especialistas tem estenose <10%.
– HJ feita em hospital não especializado chega a 30–40% de falha.
7. Conclusões práticas
1. RAFIA PRIMÁRIA
Reservada apenas para lesões pequenas, laterais e reconhecidas imediatamente. Deve ser exceção.
2. COLEDOCODUODENOSTOMIA
Útil em lesões baixas e pacientes idosos ou debilitados. Boa opção quando HJ é desproporcional ao risco.
3. HEPATICOJEJUNOSTOMIA – Padrão-ouro
Melhor técnica na imensa maioria das lesões graves. Deve ser realizada em centros de referência HPB.
Regra de ouro:
Transecção ductal = HJ em centro especializado.
ASPECTOS MÉDICO-LEGAIS DA LESÃO INADVERTIDA DA VIA BILIAR
- Membro Titular do Colégio Brasileiro de Cirurgiões
- Membro Titular do Colégio Brasileiro de Cirurgia Digestiva
1. Introdução
A lesão inadvertida da via biliar (LVB) é a complicação com maior impacto clínico, emocional e jurídico da colecistectomia. Em muitos países, é uma das principais causas de processos por erro médico em cirurgia geral. Do ponto de vista médico-legal, o ponto central não é a existência da lesão em si, mas a forma como o cirurgião:
- Indicou a cirurgia;
- Conduziu o procedimento (técnica, CVS, bailouts);
- Reconheceu e tratou a lesão;
- Documentou e comunicou o evento ao paciente e à família.
Este texto aborda, em linguagem direta, os principais aspectos médico-legais que o cirurgião geral | cirurgião do aparelho digestivo precisa dominar frente a uma suspeita de lesão iatrogênica da via biliar principal.

2. Lesão de via biliar ≠ erro médico automático
Juridicamente, lesão de via biliar é, em princípio, um evento de risco inerente ao procedimento, sobretudo na colecistectomia laparoscópica, reconhecido em diretrizes nacionais e internacionais.
Em termos de responsabilidade profissional, o que será avaliado é se houve:
- Indicação adequada da cirurgia;
- Técnica compatível com o padrão atual (CVS, uso de bailouts, conversão quando necessário);
- Diligência no reconhecimento precoce da lesão;
- Conduta correta após o dano (referência, reconstrução, suporte);
- Transparência na comunicação.
Ou seja: não é a complicação que gera responsabilidade, e sim a condução inadequada antes, durante ou depois do evento.
3. Consentimento informado
Do ponto de vista pericial, o consentimento é peça-chave:
- A colecistectomia deve ser apresentada como procedimento com:
- Risco de sangramento, infecção, fístula biliar, lesão de via biliar e necessidade de reoperação.
- O termo deve ser:
- Claro, objetivo, datado, assinado pelo paciente (ou responsável) e pela equipe.
- Ideal:
- Anotação no prontuário reforçando que os riscos foram explicados verbalmente.
Em muitos litígios, a ausência de menção à possibilidade de lesão de via biliar no consentimento é usada como argumento de falha na informação, mesmo quando a técnica foi correta.
4. Padrão técnico esperado (CVS, bailouts e conversão)
Peritos costumam avaliar:
- Se houve tentativa documentada de obter o Critical View of Safety;
- Se o cirurgião reconheceu a “vesícula difícil” e utilizou manobras de bailout (subtotal, fundo–primeiro, conversão, abandono);
- Se a insistência em dissecar um triângulo de Calot obliterado foi temerária.
Alguns pontos práticos com peso médico-legal:
- Não obter CVS e mesmo assim clipar e seccionar estruturas é quase sempre visto como conduta imprudente.
- Não converter ou não chamar ajuda em cirurgias claramente difíceis pode ser interpretado como negligência.
- O uso de colangiografia intraoperatória em caso de dúvida anatômica é bem visto pericialmente, mesmo que não obrigatório.
5. Reconhecimento e manejo da lesão intraoperatória
Do ponto de vista jurídico, lesão reconhecida e tratada intraoperatória gera cenário muito mais favorável ao cirurgião do que lesão ignorada e diagnosticada tardiamente com peritonite biliar ou sepse.
Boas práticas com impacto médico-legal:
- Se houver suspeita de lesão maior:
- Registrar no ato operatório que houve dificuldade anatômica e suspeita de dano.
- Se a equipe não tiver expertise em reconstrução, não improvisar; encaminhar para centro de referência.
- Drenos adequados e exames de imagem precoces (TC, colangioRM, CPRE) em pós-operatório duvidoso demonstram diligência.
A omissão em investigar icterícia, febre ou saída de bile por dreno no pós-operatório é frequentemente qualificada como negligência em perícias.
6. Documentação operatória
O relatório cirúrgico é uma das peças centrais em processos médico-legais. Deve conter:
- Indicação da cirurgia (cólica biliar, colecistite aguda, etc.);
- Achados intraoperatórios (inflamação, fibrose, “vesícula difícil”);
- Descrição da técnica:
- Tentativa de obter CVS;
- Uso de colangiografia intraoperatória, quando feita;
- Manobras de bailout, conversão, subtotal, etc.;
- Quaisquer intercorrências (sangramento, suspeita de lesão, necessidade de sutura em via biliar, etc.).
A ausência de descrição detalhada geralmente é interpretada contra o cirurgião, pois abre espaço para a presunção de que padrões técnicos não foram seguidos.
7. Comunicação com o paciente e a família
A forma como o cirurgião comunica a complicação é relevante:
- Negar ou minimizar o evento, ou culpar exclusivamente “o organismo do paciente”, costuma agravar o conflito.
- O recomendado é:
- Explicar com clareza o que ocorreu;
- Deixar claro que a complicação está sendo manejada com todos os recursos disponíveis;
- Documentar o conteúdo da conversa no prontuário.
Transparência e empatia costumam reduzir a judicialização. O oposto também é verdadeiro.
8. Encaminhamento a centros de referência
Outra questão avaliada em perícia é quando e para onde o paciente foi encaminhado:
- Lesões complexas (Strasberg E, perda de segmento biliar, comprometimento vascular) não devem ser reparadas por equipes sem experiência em reconstrução biliodigestiva.
- Reconstruções malsucedidas em série, feitas em hospitais sem expertise, frequentemente são interpretadas como imprudência e imperícia.
O encaminhamento precoce para centro com cirurgião HPB experiente demonstra boa prática e costuma ter peso favorável em eventual demanda judicial.
9. Papel das diretrizes e literatura
Em perícia, é comum a comparação da conduta com:
- Diretrizes societárias (CBCD, WSES, SAGES, EAES etc.);
- Protocolos hospitalares;
- “Estado da arte” à época do procedimento (por exemplo, reconhecimento do CVS como padrão-ouro).
Se o cirurgião seguiu recomendações amplamente aceitas, é difícil sustentar que houve erro grosseiro, mesmo diante de complicação grave.
10. Dano, nexo causal e responsabilidade
Em termos médico-legais, três elementos precisam estar presentes para configurar responsabilidade civil:
- Ato ou omissão culposa (técnica inadequada, falta de diligência, ausência de informação).
- Dano (lesão de via biliar, perda de parte do fígado, invalidez, óbito).
- Nexo causal entre a conduta e o dano.
Exemplo:
– Lesão biliar reconhecida, adequadamente reparada em centro de referência, com boa evolução → pode ser entendida como complicação aceitável.
– Lesão ignorada, sem investigação, evoluindo para sepse e transplante → há forte argumento de falha na assistência.
11. Estratégias de prevenção médico-legal
Em linhas simples:
- Indique bem (indicação precisa e registrada).
- Faça CVS sempre que possível e saiba abandonar a dissecção perigosa.
- Use bailouts (subtotal, fundo–primeiro, conversão) quando necessário; não insista em anatomia impossível.
- Documente tudo (relatório detalhado, fotos/vídeos, evolução diária).
- Investigue sinais de complicação sem demora.
- Encaminhe cedo lesões complexas a centros de referência.
- Comunique com clareza e registre a conversa.
Essas medidas não apenas reduzem o risco de processo, mas principalmente melhoram o cuidado ao paciente, que é o objetivo central.
“O objetivo técnico principal na Colecistectomia, não é a retirada da vesícula biliar, mas proteger a via biliar principal.” Prof. Dr. Ozimo Gama
Hashtags (SEO)
#LesãoDeViaBiliar #ResponsabilidadeMédica #CirurgiaDigestiva #MedicinaLegal #CriticalViewOfSafety
QUANDO INDICAR A RADIOLOGIA INTERVENCIONISTA NAS LESÕES DAS VIAS BILIARES
1. Introdução
Lesões da via biliar (LVB) – sobretudo pós-colecistectomia, cirurgias hepáticas e transplante – são causas relevantes de morbidade. O manejo moderno é obrigatoriamente multidisciplinar, envolvendo cirurgia, endoscopia e radiologia intervencionista (RI).
A pergunta prática do cirurgião é:
“Em que momento eu devo chamar a radiologia intervencionista?”
A seguir, um guia objetivo, organizado por cenários clínicos.
2. Papel da Radiologia Intervencionista nas LVB
De forma geral, a RI é indicada para:
- Drenar: coleções biliares ou abscessos (biloma, coleções perihepáticas).
- Desobstruir: drenagem biliar percutânea (PTBD) em obstrução ou estenose.
- Modelar/Tratar estenoses: dilatação com balão e/ou stent em estenoses benignas ou neoplásicas.
- Tratar complicações vasculares associadas: pseudoaneurisma de artéria hepática, hemobilia, sangramento.
- Criar acesso para procedimentos combinados (rendezvous com endoscopia).
RI raramente é “primeira linha isolada”, mas é frequentemente essencial como ponte ou complemento de endoscopia/cirurgia.
3. Situações agudas: quando indicar RI?
3.1. Biloma e coleções biliares pós-operatórias
Indicar drenagem percutânea guiada por US/TC quando:
- Coleção sintomática (dor, febre, instabilidade).
- Coleção ≥ 3–5 cm ou progressiva em exames seriados.
- Sinais de sepse ou disfunção orgânica.
- Fístula biliar externa com grande débito associada a coleção interna.
Nesses casos, a drenagem percutânea:
- Controla sepse rapidamente.
- Permite análise do líquido (bile x pus).
- Facilita avaliação posterior por CPRE/PTBD.
3.2. Fístula biliar com anatomia difícil ou falha da CPRE
A CPRE é primeira linha para a maioria das fístulas.
A RI entra quando:
- CPRE falhou (papila inacessível, canulação impossível).
- Anatomia alterada (gastrectomia, bypass gástrico em Y-de-Roux, anastomose biliodigestiva alta).
- Fístula alta (hepático comum/proximal) com difícil acesso endoscópico.
Nesses casos, indica-se drenagem biliar percutânea trans-hepática (PTBD):
- Dreno interno–externo ou externo, desviando o fluxo e reduzindo pressão intraductal.
- Frequentemente associada à drenagem de coleções.
3.3. Hemobilia e pseudoaneurisma de artéria hepática
Lesões combinadas artério-biliares (p. ex. pós-clipagem, lesão térmica) podem cursar com:
- Hemobilia (TRIÁDE: dor abdominal, icterícia, hematêmese/melena).
- Sangramento intra-abdominal.
Indicação imediata de RI:
- Angiografia diagnóstica + embolização seletiva da artéria lesada.
- Evita reoperação em pedículo hepático inflamado e de alto risco.
Cirurgião deve suspeitar e acionar RI em:
- Sangramento digestivo tardio em paciente com LVB.
- Queda de Hb sem foco óbvio + drenagem biliar sanguinolenta.
4. Situações tardias: estenoses e reconstruções
4.1. Estenoses benignas pós-cirúrgicas (hepáticojejunostomia / LVB reparada)
A RI é especialmente útil em pacientes com anatomia não acessível por CPRE:
- Hepáticojejunostomia em Y-de-Roux.
- Reconstruções biliares altas.
Indicações típicas:
- Icterícia colestática / colangite tardia.
- Estenose diagnosticada em colangiorressonância ou PTC.
Técnicas:
- Acesso percutâneo ao sistema biliar.
- Dilatação com balão da estenose.
- Posicionamento de cateteres de calibração ou stents.
- Sessões seriadas ao longo de meses.
Em estritos pós-hepáticojejunostomia, a combinação RI + cirurgia é muitas vezes a melhor estratégia (tratamento híbrido em casos recidivantes).
4.2. Obstrução biliar maligna (colangiocarcinoma, metástases hiliares, pâncreas)
Embora aqui não haja “lesão iatrogênica”, na prática HPB o raciocínio é o mesmo:
Indicar PTBD quando:
- ERCP falhou ou não é possível.
- Obstrução hilar complexa (Bismuth III/IV).
- Necessidade de drenagem seletiva de segmentos específicos antes de grande hepatectomia.
É uma área clássica da RI, mas o cirurgião deve lembrar que lesão biliar complexa iatrogênica com estenose alta se comporta de forma semelhante.
5. Lesões de via biliar complexas: como integrar cirurgia, endoscopia e RI?
5.1. Lesão parcial do ducto biliar comum com fístula (Strasberg D)
- Primeira linha: CPRE + stent.
- Se CPRE falha → PTBD + drenagem de coleções por RI.
- Reconstrução cirúrgica planejada se não houver fechamento após suporte combinado.
5.2. Secção completa do ducto com perda de segmento (Strasberg E)
- RI tem função adjuvante, não curativa.
- Indicações:
- Drenagem de biloma/coleções.
- PTBD para controle de icterícia e sepse.
- Objetivo: estabilizar o paciente e planejar hepáticojejunostomia definitiva em centro de referência.
5.3. Fístulas de pequeno ducto (Luschka, coto cístico) com biloma
- Drenagem percutânea se coleção significativa.
- CPRE para reduzir pressão e favorecer cicatrização.
- RI é especialmente útil se o paciente estiver séptico ou longe de centro endoscópico.
6. Critérios práticos para o cirurgião saber “quando chamar a RI”
Em linguagem direta:
Chame Radiologia Intervencionista quando houver:
- Coleção intra-abdominal (biloma/abscesso) que:
- seja volumosa, sintomática ou séptica;
- não possa ser drenada cirurgicamente com baixo risco.
- Falha ou impossibilidade de CPRE em:
- fístulas biliares;
- obstrução/estenose biliar;
- anatomia alterada (Y-de-Roux, gastrectomia).
- Obstrução ou estenose alta (hilar, hepático comum proximal) onde:
- a drenagem seletiva de segmentos é desejada;
- o acesso endoscópico é tecnicamente desfavorável.
- Suspeita de complicação vascular associada à LVB:
- hemobilia, pseudoaneurisma de artéria hepática, sangramento sem foco claro.
- Paciente de alto risco cirúrgico, em que:
- a drenagem percutânea (biloma ou biliar) pode estabilizar o quadro até uma reconstrução eletiva.
7. Conclusão
A radiologia intervencionista é hoje pilar central no manejo das lesões de via biliar, especialmente:
- como ponte segura entre o evento agudo e a reconstrução definitiva;
- como alternativa em anatomias complexas ou inacessíveis à endoscopia;
- como ferramenta essencial no tratamento de complicações vasculares associadas.
Para o cirurgião HPB e digestivo, o ponto-chave não é “se” deve envolver a RI, mas em que momento:
Quanto mais precoce a integração cirurgião–endoscopista–radiologista, menor a chance de reoperações desnecessárias, falhas terapêuticas e perda definitiva da via biliar.
Hashtags (SEO)
#LesãoDeViaBiliar #RadiologiaIntervencionista #CirurgiaHepatobiliar #Biloma #DrenagemPercutanea
TRATAMENTO ENDOSCÓPICO DAS FÍSTULAS BILIARES PÓS-COLECISTECTOMIA: RESULTADOS, TÉCNICAS E AVALIAÇÃO CRÍTICA DA LITERATURA
Introdução
As fístulas biliares são uma complicação relativamente comum após colecistectomia, sobretudo nos casos difíceis, inflamatórios ou com lesão térmica inadvertida no ducto cístico. Embora a maioria das fístulas provenientes do coto cístico ou de pequenos ramos do ducto hepático direito sejam de baixo débito, seu manejo inadequado pode evoluir para sepse, coleções biliares ou necessidade de reoperação.
O tratamento endoscópico por CPRE (colangiopancreatografia retrógrada endoscópica) tornou-se o padrão-ouro para o manejo inicial dessas fístulas, com elevadas taxas de sucesso, baixa morbidade e impacto significativo na resolução precoce das complicações.
Este artigo revisa criticamente as técnicas endoscópicas disponíveis, seus resultados e as principais evidências da literatura.
Classificação das Fístulas Biliares Pós-Colecistectomia
As fístulas podem se originar de:
- Coto do ducto cístico – mais comum e com excelente resposta à CPRE.
- Ducto hepático direito acessório (ducto de Luschka).
- Lesão térmica do ducto hepático comum ou ducto biliar comum (DBC) – frequentemente associada a estenose.
- Lesões de alto débito (>300 mL/dia) – pior prognóstico, maior chance de lesão maior.
As fístulas tipo I (bile leak leve) respondem quase sempre ao tratamento endoscópico.
As fístulas tipo II, III e IV (maior lesão ductal) podem necessitar cirurgia reconstrutiva.
Racional do Tratamento Endoscópico
A CPRE atua em dois pilares fisiopatológicos:
1. Redução da pressão no ducto biliar
- Realizada por esfinterotomia e/ou stents biliares.
- Diminui o fluxo de bile para o local da fístula, favorecendo cicatrização.
2. Desvio do fluxo biliar
- O stent oferece um caminho de menor resistência.
- A fístula deixa de ser a via preferencial de drenagem.
Com isso, >90% das fístulas simples fecham em poucos dias.
Técnicas Endoscópicas
1. Esfinterotomia isolada
- Indicações: fístulas leves, baixo débito.
- Vantagem: técnica simples, baixo custo.
- Limitação: menor eficácia comparada ao uso de stent.
Taxas de sucesso relatadas: 70–85%.
2. Colocação de stent biliar plástico
- Técnica mais utilizada.
- Stents de 7 a 10 Fr, posicionados transpapilares.
- Associado ou não à esfinterotomia.
Taxas de sucesso: 85–95%
(Fechamento geralmente em 3–6 semanas.)
3. Stents totalmente cobertos (SEMS)
- Indicações:
- Fístulas persistentes após stent plástico.
- Estenose concomitante.
- Lesão térmica do hepático comum.
Taxas de sucesso: 90–100% em séries selecionadas.
Limitações: custo e risco de migração.
4. Retirada de cálculos associados
- Coledocolitíase residual pode perpetuar pressão intraductal.
- Papilotomia + extração de cálculos é essencial nesses casos.
5. Drenagem guiada por EUS
- Usada em casos complexos, falha da CPRE ou anatomia difícil.
- Promissora, mas ainda com dados limitados.
Resultados da Literatura
A maior parte dos estudos é formada por séries retrospectivas, mas os achados são consistentes:
• Sucesso global da CPRE no manejo de fístulas pós-colecistectomia: 85–95%
(meta-análises mostram média entre 88–92%)
• Tempo médio para fechamento da fístula: 2 a 6 semanas
• Principais fatores associados à falha:
- Estenose concomitante não tratada
- Lesão mais proximal (lesões tipo C/D de Strasberg)
- Fístulas de alto débito com lesão ductal maior
- Perfurações térmicas extensas
Complicações da CPRE
- Pancreatite pós-CPRE: 5–10%
- Sangramento: 1–3%
- Perfuração: <1%
Apesar disso, o risco é muito inferior ao de uma reoperação precoce.
Avaliação Crítica da Evidência
1. Estudos predominantemente retrospectivos
- Faltam ensaios clínicos randomizados comparando métodos endoscópicos.
- A heterogeneidade das classificações dificulta comparações.
2. Ausência de padronização universal
- Alguns centros utilizam stent + esfinterotomia como rotina.
- Outros reservam stent para casos complexos.
3. Papel crescente dos SEMS cobertos
- Cada vez mais estudados, principalmente para fístulas persistentes.
- Necessidade de mais estudos comparativos.
4. O dilema das lesões complexas
- Lesões de alto grau (como BDI tipo E de Strasberg) não devem ser tratadas exclusivamente por via endoscópica.
- A CPRE funciona como ponte para cirurgia de reconstrução (hepáticojejunostomia).
5. Importância da drenagem percutânea combinada
- Em casos com coleções biliares, a drenagem percutânea associada à CPRE acelera a resolução.
Quando o tratamento endoscópico falha?
A falha deve ser suspeitada quando:
- Vazamento persiste por >8 semanas.
- Débito permanece alto apesar do stent.
- Haja estenose proximal não tratada.
- Evidência de lesão de via biliar maior.
Nesses casos, a cirurgia reconstrutiva (hepáticojejunostomia) deve ser indicada.
Conclusão
A CPRE é o tratamento de primeira linha para as fístulas biliares pós-colecistectomia, oferecendo taxas de sucesso superiores a 85–95% nas fístulas simples e bom desempenho mesmo em cenários moderadamente complexos.
A literatura demonstra de forma consistente que:
- A endoscopia reduz morbidade
- Evita reoperações desnecessárias
- Promove fechamento rápido da fístula
- É custo-efetiva e amplamente disponível
No entanto, a interpretação crítica mostra que ainda faltam estudos prospectivos e padronização universal das técnicas. Para fístulas complexas ou quando associadas a estenose ductal significativa, a abordagem multidisciplinar é essencial.
A integração entre endoscopia, radiologia intervencionista e cirurgia HPB continua sendo a chave para reduzir complicações e preservar a via biliar.
Hashtags (SEO)
#FistulaBiliar #CPRE #CirurgiaDigestiva #Colecistectomia #LesãoDeViaBiliar
INTELIGÊNCIA ARTIFICIAL NA COLECISTECTOMIA LAPAROSCÓPICA: A NOVA FRONTEIRA DA SEGURANÇA CIRÚRGICA
A colecistectomia laparoscópica (LC) é um dos procedimentos mais realizados no mundo, mas ainda carrega um risco significativo de lesão de via biliar (BDI – bile duct injury), especialmente em situações inflamatórias ou anatômicas complexas. Nas últimas décadas, o Critical View of Safety (CVS) se consolidou como o padrão-ouro para evitar BDI, porém estudos mostram que sua identificação é altamente subjetiva e frequentemente superestimada pelos cirurgiões.
A inteligência artificial (IA), especialmente por meio de deep learning e visão computacional aplicada a vídeos cirúrgicos, surge como uma ferramenta promissora para padronizar, reconhecer e validar o CVS em tempo real, além de mapear estruturas anatômicas e zonas seguras e de risco durante a LC.
Este artigo resume de forma clara e aplicada o estado da arte da IA na colecistectomia.
IA e o Critical View of Safety (CVS)
Mesmo com o uso disseminado do CVS, a taxa de BDI permanece maior na LC do que na técnica aberta. Estudos revelam um dado alarmante: a maioria dos cirurgiões acredita ter alcançado o CVS, mas uma análise independente mostra o oposto.
- Em um estudo com 1108 LCs, cirurgiões relataram CVS em 80% dos casos.
- Revisores externos encontraram CVS verdadeiro em apenas 10,8%.
- Em todos os casos com BDI, o CVS não havia sido obtido.
A IA foi então proposta como forma de reduzir a subjetividade e melhorar a precisão do reconhecimento anatômico.
Modelos de IA para reconhecimento do CVS
Os primeiros modelos usaram milhares de imagens anotadas por cirurgiões experientes:
- Um deep neural network treinado com 201 vídeos e 2854 imagens alcançou:
- Acurácia: 71,9%
- Precisão: 71,4%
Outro estudo mais recente, com mais de 71 mil imagens, alcançou:
- Precisão: 0,97
- Acurácia: 0,83
Resultados muito superiores aos obtidos por humanos revisando apenas imagens estáticas.
IA para áreas seguras e zonas de risco (Go/No-Go)
O modelo GoNoGoNet, um dos mais influentes, usou semantic segmentation para marcar:
- Go zone — área segura de dissecção
- No-Go zone — região perigosa, associada a BDI
Resultados:
- Acurácia superior a 90% na maioria das estruturas.
- Cirurgiões que avaliaram vídeos com o suporte da IA corrigiram suas anotações em até 26,9%, sendo 70% correções que aumentavam a segurança.
Estudos subsequentes mostraram que:
- Vídeos com BDI tinham 33,6% mais interações em zonas No-Go.
- A IA demonstrou maior sensibilidade para regiões de risco do que o olho humano.
IA para reconhecimento de estruturas anatômicas
Modelos recentes conseguem identificar automaticamente:
- ducto cístico
- ducto biliar extra-hepático
- sulco de Rouvière
- margem inferior do segmento 4 (S4)
Esses pontos são cruciais na prevenção de BDI.
Em um estudo experimental:
- Cirurgiões mudaram sua interpretação anatômica em 25–30% dos casos após ver o vídeo anotado pela IA.
- A maioria considerou as correções mais seguras.
Em outro estudo, o modelo YOLOv3 foi integrado ao sistema laparoscópico da sala cirúrgica:
- Latência de apenas 0,09 s
- Reconhecimento de estruturas em 92% das etapas da LC
Esse é o primeiro passo real para um “copiloto cirúrgico” intraoperatório.
IA na identificação das fases cirúrgicas
Modelos baseados em redes temporais (MS-TCN, ResNet50, Cholec80) alcançaram:
- Acurácia de 78% a 91% na identificação das fases da LC
- Melhor desempenho nas etapas críticas:
- dissecção do triângulo de Calot
- clipagem
- liberação do leito da vesícula
Entretanto, eventos adversos diminuem o desempenho:
- Sem complicações: ~90%
- Com perfuração da vesícula: 87%
- Com grande vazamento biliar: 77%
Limitações atuais da IA na LC
As principais barreiras identificadas nos estudos:
1. Anotações humanas inconsistentes
• Pequeno número de cirurgiões anotadores
• Variabilidade inter-observador
• Falta de padronização global
2. Datasets pequenos e monocêntricos
• Reduz a generalização dos modelos
• Dificulta adaptação para diferentes câmeras, óticas e técnicas
3. Dificuldade com casos complexos
• Inflamação severa
• Cirurgias com fibrose, gordura densa ou sangramento
• Mudança de ângulo da ótica
4. Desafios práticos no intraoperatório
• Flicker de ROIs
• Distração visual
• Confiabilidade limitada em visão ampliada ou com fumaça
5. Falta de integração nativa
Apenas um estudo conectou a IA diretamente ao sistema laparoscópico (EndoALPHA).
Perspectivas Futuras
- Sistemas dual-AI integrados ao console laparoscópico
– Reconhecimento simultâneo de estruturas + fases cirúrgicas. - ROI dinâmico com “tiles” inteligentes, reduzindo flicker e adaptando-se a limites anatômicos.
- Combinação IA + ICG, ampliando reconhecimento além das capacidades isoladas de cada método.
- Incorporação ao treinamento cirúrgico e simulação, especialmente para residentes.
- Criação de guideline global para anotação, validação e uso clínico da IA.
- Suporte intraoperatório em tempo real, semelhante a um copiloto de aviação.
Conclusão
A inteligência artificial aplicada à colecistectomia laparoscópica está avançando rapidamente, com resultados promissores para:
- melhorar a identificação anatômica
- reforçar zonas de segurança
- padronizar o Critical View of Safety
- apoiar o treinamento e reduzir o risco de lesões de via biliar
Embora nenhum sistema esteja pronto para substituição completa do julgamento cirúrgico, o conjunto de evidências sugere que a IA será, em breve, parte essencial da estratégia global para aumentar a segurança da LC.
Como toda tecnologia emergente, o sucesso dependerá de:
- validação multicêntrica
- integração prática ao workflow operatório
- aceitação e liderança dos cirurgiões
A IA não substituirá o cirurgião.
Mas cirurgiões que usam IA substituirão os que não usam.
INTELIGÊNCIA ARTIFICIAL E CIRURGIA: O QUE O CIRURGIÃO PRECISA SABER HOJE
Introdução
A inteligência artificial (IA) deixou de ser um conceito futurista e entrou definitivamente na prática cirúrgica. Longe das versões “fortes” retratadas em filmes, as aplicações atuais são formas de IA estreita, projetadas para executar tarefas específicas com elevado desempenho. Na cirurgia, essas ferramentas já atuam na estratificação de risco, análise intraoperatória, previsão de desfechos e processamento de prontuários, com evidências crescentes de impacto na segurança, eficiência e precisão das decisões clínicas.
Este artigo apresenta, de forma didática e objetiva, as principais aplicações da IA ao ato cirúrgico moderno, suas limitações e o papel do cirurgião nessa transição tecnológica.
1. Fundamentos: o que de fato é Inteligência Artificial?
A IA é o campo que estuda algoritmos capazes de executar funções cognitivas. É composta por subáreas, sendo as mais relevantes para a cirurgia:
1.1 Machine Learning (ML)
Sistemas que aprendem padrões a partir de dados estruturados.
• Supervisionado: aprende com dados previamente rotulados.
• Não supervisionado: identifica padrões sem rótulos.
• Reforço: aprendizado por tentativa e erro.
1.2 Redes Neurais e Deep Learning
Capazes de extrair automaticamente características de dados complexos como imagens e vídeos.
• Especialmente úteis em laparoscopia, endoscopia e robótica.
1.3 Computer Vision (CV)
Permite que máquinas “vejam” e interpretem imagens cirúrgicas.
• Base da análise intraoperatória em tempo real.
1.4 Processamento de Linguagem Natural (NLP)
Interpreta texto livre, facilitando mineração de prontuários, relatórios operatórios e predição de risco.
2. Aplicações atuais na cirurgia
2.1 Predição pré-operatória de risco
Ferramentas como POTTER, MySurgeryRisk e modelos de aprendizado profundo superam calculadoras tradicionais (ASA, RCRI, NSQIP), oferecendo:
• Previsão personalizada de mortalidade e complicações.
• Integração em tempo real com prontuários eletrônicos.
• Aprendizado contínuo com novos dados.
Esses modelos já apresentam C-statistics superiores às ferramentas tradicionais, mostrando melhor acurácia na previsão de desfechos.
2.2 Análise intraoperatória por vídeo
Uma das fronteiras mais promissoras. A IA já consegue:
• Reconhecer fases cirúrgicas com >85% de acurácia em colecistectomias, gastrectomias e colectomias.
• Identificar estruturas anatômicas críticas.
• Estimar tempo cirúrgico restante.
• Detectar “near misses” e eventos de risco antes que causem dano.
Estudos mostram que até 70% das cirurgias apresentam um “near miss”. A IA pode atuar como um “copiloto” cirúrgico, alertando o cirurgião nesses momentos.
2.3 Suporte ao treinamento e avaliação de habilidades
A análise automatizada de vídeo já permite:
• Avaliar habilidades com base em padrões de movimento.
• Comparar a performance do cirurgião com especialistas (“global surgical consciousness”).
• Reduzir subjetividade na avaliação de residentes.
2.4 Integração com prontuário eletrônico (EMR)
Com NLP, a IA pode:
• Detectar infecções precocemente.
• Extrair automaticamente complicações de textos livres.
• Identificar pacientes de alto risco.
• Correlacionar decisões intraoperatórias a desfechos.
3. Limitações e riscos
Apesar do entusiasmo, a IA em cirurgia ainda é limitada:
• Modelos podem superajustar (overfitting) e não generalizar bem.
• Dados cirúrgicos são altamente variáveis e ruidosos.
• Lacunas éticas e jurídicas permanecem (privacidade de vídeo, responsabilidade por erros).
• Necessidade de padronização: cirurgiões diferem até na definição de fases cirúrgicas.
Portanto, o uso clínico deve ser crítico, criterioso e apoiado em validação robusta.
4. O papel do cirurgião na era da IA
A introdução da IA não substituirá o cirurgião — mas exigirá um profissional capaz de integrar tecnologia com julgamento clínico. O cirurgião deve:
• Entender princípios básicos de IA.
• Conhecer limitações dos modelos.
• Participar da criação de bases de dados cirúrgicas.
• Atuar na validação clínica de algoritmos.
• Liderar discussões éticas e regulatórias.
Como afirmam Hashimoto e col. (2020), a participação ativa da comunidade cirúrgica é essencial para garantir interoperabilidade, segurança e sustentabilidade dessas tecnologias.
Conclusão
A IA está transformando a cirurgia em múltiplas frentes — do risco pré-operatório ao suporte intraoperatório e análise pós-operatória. A integração com sistemas de visão computacional, prontuários eletrônicos e mecanismos de aprendizado contínuo aponta para um futuro onde o cirurgião terá ferramentas mais precisas para decisões mais seguras.
O desafio atual não é se a IA entrará no centro cirúrgico, mas como garantiremos que essa entrada ocorra de forma ética, segura e cientificamente validada.
CUSTO E VALOR NA HERNIOPLASTIA INGUINAL: O QUE REALMENTE IMPORTA PARA O PACIENTE E PARA O SISTEMA DE SAÚDE
Introdução
A discussão sobre “qual é a melhor técnica de hernioplastia inguinal” não pode ser feita apenas com base em recidiva ou complicações.
No cenário atual de restrição de recursos e busca por eficiência, o valor cirúrgico importa tanto quanto o desfecho clínico.
Segundo o modelo clássico de Michael Porter, Valor = Qualidade / Custo.
Quando os resultados clínicos entre técnicas são equivalentes, quem determina o valor final é o custo total envolvido — variável e fixo.
O estudo analisado comparou 100 procedimentos consecutivos em cada modalidade:
- Hernioplastia aberta
- Laparoscópica
- Robótica
A seguir, o que realmente importa quando comparamos custo, eficiência e valor em cada abordagem.
1. Entendendo Custos: Variáveis × Fixos
1.1. Custos variáveis (por caso)
Incluem:
- Materiais descartáveis
- Mão de obra direta
- Overhead variável por procedimento
Resultados:
- Laparoscópica: 1,02× o custo da aberta (diferença não significativa).
- Robótica: 2,11× o custo da aberta e 2,06× o custo da laparoscópica (P < 0,001).
Ou seja: a laparoscopia custa praticamente o mesmo que a aberta, mas a robótica mais que dobra o custo.
1.2. Custos fixos (capital + manutenção)
Incluem:
- Torre laparoscópica
- Robô cirúrgico
- Contratos de manutenção
- Amortização por caso
Quando esses custos entram na conta:
- Laparoscópica: 1,03× o custo da aberta
- Robótica: 3,18× o custo da aberta
A robótica triplica o custo total da operação.
2. O Impacto no Valor Cirúrgico
Se assumirmos que as taxas de recidiva são equivalentes (o que os dados atuais sustentam para as três técnicas), então:
- Laparoscopia → reduz o valor do procedimento em apenas 2–3%, o que é compensado por:
- menos dor
- retorno mais rápido às atividades
- melhor satisfação do paciente
Com isso, a laparoscopia entrega mais valor ao paciente do que a via aberta.
- Robótica → reduz o valor em:
- 53% quando analisado apenas custo variável
- 69% quando incluídos custo fixo + manutenção
O principal fator destruidor de valor é o custo incremental da tecnologia, sem ganho proporcional em desfecho.
3. O Que Isso Significa na Prática?
A. Hernioplastia aberta
- Custo menor
– Pior recuperação e mais dor em muitos casos
= Valor intermediário
B. Hernioplastia laparoscópica
- Mesma taxa de recidiva
- Melhores resultados relatados pelo paciente
- Custos semelhantes à aberta
= Maior valor
C. Hernioplastia robótica
- Conforto operatório (para o cirurgião)
- Ergonomia superior
– Sem benefícios clínicos comprovados ao paciente
– 3× mais cara
= Menor valor global
4. Conclusão: Qual Técnica Oferece Mais Valor?
Com base na melhor evidência disponível:
- A laparoscopia é a técnica com melhor relação custo-benefício, entregando mais valor ao paciente.
- A via aberta permanece adequada em cenários de baixo recurso ou limitação técnica.
- A robótica, apesar de tecnologicamente avançada, não aumenta o valor cirúrgico, pois aumenta custos sem melhorar resultados.
Em uma era de medicina baseada em valor, mais tecnologia não significa melhor cuidado — especialmente quando o resultado final esperado (baixa recidiva) é o mesmo.
Hashtags (SEO)
#HerniaInguinal #CirurgiaDigestiva #Laparoscopia #CirurgiaRobótica #MedicinaBaseadaEmValor
BAILOUT MANEUVERS NA COLECISTECTOMIA LAPAROSCÓPICA: O QUE FAZER QUANDO A DISSECÇÃO SE TORNA PERIGOSA
Introdução
Embora a colecistectomia laparoscópica seja um procedimento rotineiro e de baixo risco, a chamada “vesícula difícil” representa um dos cenários mais desafiadores da cirurgia digestiva. Inflamação intensa, distorção anatômica e aderências densas podem transformar um procedimento simples em uma operação com potencial de causar lesões biliares e vasculares graves, com impacto direto na morbimortalidade do paciente.
Diante desse cenário, é essencial que todo cirurgião domine as Bailout Maneuvers — manobras de resgate seguras e bem estabelecidas para interromper a progressão de uma dissecção perigosa e evitar complicações catastróficas.
Reconhecendo o Momento de Parar: “Red Flags” Intraoperatórios
Muitas lesões graves ocorrem não por falta de habilidade técnica, mas por falha na percepção do risco. Alguns sinais de alerta devem fazer o cirurgião pausar, reavaliar e reorientar a dissecção:
- Artéria “cística” incomumente calibrosa (possível artéria hepática).
- Pulsação intensa atrás do suposto ducto cístico (pode ser ducto hepático comum).
- Clip grande incapaz de ocluir o ducto (sugere lesão do DBC ou DHC).
- Presença de bile no campo sem perfuração da vesícula.
Perceber esses red flags pode evitar até 18% das lesões vasculares e biliares relatadas em séries grandes.
Quando o Critical View of Safety Não é Possível
Há situações em que a inflamação é tão intensa que não existe plano anatômico seguro para liberar o Triângulo de Calot. Nesses casos, insistir é o caminho mais rápido para uma lesão maior.
O cirurgião deve reconhecer o limite técnico e aplicar uma manobra de resgate apropriada.
Principais Bailout Maneuvers
1. Abortar o Procedimento
A decisão mais segura em casos extremos:
- Campo tomado por fibrose, necrose ou tecido friável.
- Vesícula não identificável.
- Distinção anatômica impossível mesmo com auxílio.
Condutas subsequentes:
- Manter antibióticos.
- Considerar colecistostomia percutânea.
- Planejar colecistectomia intervalar após 8–12 semanas.
Apesar de não resolver a doença naquele momento, salva o paciente de lesões irreversíveis.
2. Fundus-First Cholecystectomy (Técnica Fundo-Primeiro)
Indicada quando o Calot é impenetrável.
Técnica:
- Iniciar a dissecção no fundo da vesícula.
- Avançar cuidadosamente em direção ao infundíbulo.
- Manter o plano “na parede da vesícula” para evitar o pedículo portal.
Pontos de atenção:
- Em inflamação severa, o cystic plate pode estar espessado e encurtado.
- A proximidade com o pedículo hepático aumenta conforme a inflamação destrói planos naturais.
Estudos mostram que a técnica fundo-primeiro reduz a necessidade de subtotal e pode diminuir o tempo de internação.
3. Subtotal Cholecystectomy (Colecistectomia Subtotal)
Opção preferível quando a anatomia permanece obscura após fundus-first.
Técnica geral:
- Abrir a vesícula.
- Aspirar conteúdo.
- Ressecar o máximo da parede possível.
- Ablacionar a mucosa remanescente.
Existem duas variantes:
A. Reconstituting (Reconstituinte)
- O remanescente é fechado com sutura.
- Menor risco de fístula biliar.
- Maior risco de vesícula residual sintomática.
B. Fenestrating (Fenestrante)
- O remanescente permanece aberto.
- Maior chance de fístula, mas geralmente autolimitada.
- Menor risco de doença residual.
Fechamento do cístico, quando indicado, deve ser interno, pois o fechamento externo pode ocluir o DHC/DBC.
4. Conversão para Cirurgia Aberta
Historicamente considerada a principal manobra de resgate, hoje sabemos que:
- A conversão não garante segurança.
- O risco de lesão biliar aumenta até 100 vezes em conversões não planejadas (NSQIP).
- A anatomia pode permanecer obscura mesmo no campo aberto.
Ainda é válida quando:
- Há sangramento importante.
- Há perda completa dos planos anatômicos.
- Há necessidade de controle manual do pedículo hepático.
Entretanto, em muitos casos, uma subtotal laparoscópica é mais segura do que uma conversão difícil.
Como Escolher a Manobra Correta?
A lógica de decisão deve considerar:
- Visibilidade anatômica (CVS alcançável ou não).
- Grau de inflamação.
- Experiência do cirurgião.
- Risco iminente de lesão maior.
- Disponibilidade de outro cirurgião para segunda opinião.
Regra prática:
Se você não consegue obter o Critical View of Safety e não está absolutamente certo do que está dissecando, é hora de um bailout.
Conclusão
A vesícula difícil exige maturidade cirúrgica, capacidade de reconhecer limites e domínio das manobras de resgate. Insistir em uma dissecção perigosa é receita para desastre. Saber abandonar, retroceder ou mudar a estratégia é parte fundamental da segurança do paciente e da preservação da via biliar.
A Evolução dos Traumas de Guerra: De Termópilas aos Drones na Ucrânia
A progressão dos traumas de guerra espelha a interseção entre inovações bélicas, avanços médicos e sistemas logísticos. Desde a Batalha de Termópilas em 480 a.C., símbolo da guerra organizada antiga, até o conflito atual na Ucrânia, os ferimentos evoluíram de penetrações de baixa energia para explosões complexas de alta energia. Este artigo examina a biomecânica dos traumas – como forças mecânicas interagem com tecidos humanos –, junto com causas de morte, taxas de mortalidade e medidas de tratamento ao longo de períodos históricos chave. Baseado em análises históricas, registros médicos militares e relatórios recentes, destaca uma tendência de redução na mortalidade por meio de inovações, mas temperada por ameaças emergentes como a tecnologia de drones.
A ERA ANTIGA: TERMÓPILAS E FERIMENTOS PRIMITIVOS
Nas guerras antigas, como as Greco-Persas incluindo Termópilas, o combate dependia de armas corpo a corpo como lanças, espadas, flechas e pedras. Biomecanicamente, esses causavam ferimentos penetrantes de baixa velocidade, com transferência de energia cinética limitada a lacerações diretas e cavitação ao longo do trajeto do projétil. Por exemplo, golpes de lança criavam cavidades permanentes no peito ou abdômen, lesando órgãos vitais com expansão temporária mínima devido à baixa velocidade (geralmente abaixo de 300 m/s). Forças contundentes de pedras ou escudos provocavam fraturas compressivas ou tórax instável, onde segmentos de costelas se soltavam, prejudicando a respiração por movimento paradoxal.
As causas de morte eram predominantemente falha orgânica imediata ou exsanguinação por rupturas vasculares, com infecções secundárias agravando os sobreviventes. Proxies históricos, como descrições na Ilíada de Homero (ligada a contextos semelhantes à Guerra de Troia), indicam que 63% dos traumas torácicos resultavam de lanças, levando a letalidade rápida por hemotórax ou pneumotórax. Taxas de mortalidade para ferimentos torácicos graves aproximavam-se de 70%, com 89% classificados como médios a graves; fatalidades gerais no campo de batalha eram altas, pois muitos feridos ficavam sem tratamento em meio ao combate.
Os tratamentos eram primitivos, dependendo de aplicações herbais, banhos quentes e ataduras básicas por figuras como Machaon nos textos antigos. Princípios hipocráticos enfatizavam a drenagem de pus, mas sem antissepsia, a gangrena era comum. Não havia evacuação organizada, com cuidados limitados a guerreiros de elite, destacando a alta letalidade da era por biomecânica não gerenciada, como sangramento descontrolado.
PRIMEIRA GUERRA MUNDIAL: A DESTRUIÇÃO INDUSTRIALIZADA
A Primeira Guerra Mundial (1914-1918) introduziu a destruição mecanizada por metralhadoras, projéteis de artilharia e rifles de alta velocidade (acima de 600 m/s). Biomecanicamente, esses criavam danos teciduais extensos por projéteis instáveis e fragmentação, formando grandes cavidades temporárias (até 10-15 cm de diâmetro) que esticavam tecidos elásticos como músculos, causando necrose tardia, enquanto órgãos inelásticos como o fígado se estilhaçavam. Ondas de explosão de projéteis adicionavam barotrauma primário, comprimindo estruturas cheias de ar como pulmões e levando a contusões pulmonares.
Infecções e doenças superavam ferimentos de combate como causas de morte, com 63.000 fatalidades por doenças nos EUA versus 51.000 por batalhas. Hemorragia e choque de fraturas compostas eram principais matadores relacionados a ferimentos, exacerbados pela contaminação das trincheiras. A mortalidade por ferimentos de batalha era de aproximadamente 21% (53.402 mortes de 257.404 casos), com fraturas de fêmur inicialmente em 80% de fatalidade antes de intervenções. Doenças como tifo e gripe ceifaram milhões globalmente, alterando a proporção doença-para-batalha para 2:1 em algumas forças.
Os tratamentos avançaram com sistemas de triagem priorizando cuidados salvadores, desenvolvidos por médicos franceses. Estações de Limpeza de Feridos forneciam debridamento, irrigação com solução Carrel-Dakin (à base de hipoclorito) e transfusões de sangue iniciais (introduzidas em 1916, embora arriscadas). Taxas de amputação eram de 12%, com mortalidade caindo para 5% pós-procedimento via técnicas estéreis e talas como o método Thomas. Evacuação por ambulâncias motorizadas reduziu atrasos, marcando uma virada do cuidado ad hoc antigo.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Antiga (Termópilas) | Penetração de baixa velocidade; laceração direta | Falha orgânica, infecção | ~70% para torácicos graves | Remédios herbais, ataduras |
| Primeira Guerra Mundial | Cavitação de alta velocidade; barotrauma por explosão | Infecção, doença, hemorragia | 21% geral por ferimentos | Debridamento, irrigação, transfusões iniciais |
SEGUNDA GUERRA MUNDIAL: REFINAMENTOS EM EVACUAÇÃO E ANTIMICROBIANOS
A Segunda Guerra Mundial (1939-1945) viu uso contínuo de explosivos e armas de fogo, com biomecânica enfatizando ferimentos secundários por fragmentos causando politrauma (ex.: 70-75% envolvimento de extremidades). Cargas moldadas em armas antitanque produziam jatos de alta velocidade, levando a spallation (fragmentação interna) e inalação de fumos tóxicos. Traumas contundentes terciários por deslocamentos adicionavam fraturas e síndrome compartimental.
Hemorragia e infecção permaneciam causas primárias, mas mortalidade por doenças caiu para uma proporção de 0,1:1 com vacinas. Mortalidade geral por ferimentos era de 3,3%, com abdominais em 21%. Queimaduras de veículos blindados afetavam 35% das baixas em contextos navais, complicando com lesões inalatórias.
Tratamentos incluíam fechamento primário atrasado (sucesso de 93%), debridamento e penicilina (produzida em massa até 1945), cortando taxas de infecção. Transfusões de sangue e plasma tornaram-se rotineiras, com envios aéreos garantindo suprimento. Cadeias de evacuação – de postos de socorro a hospitais de campo – levavam em média 12-15 horas, usando amputações circulares e gaze com vaselina. Sobrevivência para feridos atingiu 50%, acima dos 4% da Primeira Guerra.
GUERRA DA COREIA: MOBILIDADE E INOVAÇÕES VASCULARES
A Guerra da Coreia (1950-1953) apresentava ferimentos semelhantes de alta energia, mas o frio amplificava riscos de congelamento. Biomecanicamente, fragmentos causavam fraturas cominutivas, com sobrepressão por explosão levando a barotrauma em terrenos confinados. Causas de morte mudaram para hemorragia gerenciada, com doenças em proporção de 0,2:1. Mortalidade por ferimentos caiu para 2,4%, abdominais para 12%. Gangrena gasosa foi quase eliminada por antibióticos.
Hospitais Cirúrgicos Móveis do Exército (MASH) permitiam cuidados em 3-12 horas via helicópteros. Tratamentos incluíam reparos vasculares (taxa de amputação de 49,6% na Segunda Guerra para 20,5%), profilaxia com penicilina-estreptomicina e gesso funcional. Injeções de procaína tratavam congelamento, destacando adaptações ambientais.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Segunda Guerra Mundial | Fragmentação, spallation; toxicidade inalatória | Hemorragia, infecção | 3,3% geral | Penicilina, transfusões de sangue, fechamento atrasado |
| Guerra da Coreia | Explosões cominutivas; compressão por congelamento | Hemorragia gerenciada, infecção reduzida | 2,4% geral | Evacuação por helicóptero, cirurgia vascular, antibióticos |
GUERRA DO VIETNÃ: RESPOSTA RÁPIDA EM GUERRA DE SELVA
O Vietnã (1955-1975) envolvia balas de alta velocidade e minas, criando politrauma contaminado com balas instáveis (ex.: fragmentação do M-16 após 12 cm de penetração). Biomecânica incluía cavitação profunda por armadilhas, levando a perda tecidual massiva e infecções secundárias. Choque hipovolêmico e infecções causavam a maioria das mortes, com doenças em 0,2:1. Mortalidade hospitalar era de 2,6%, abdominais de 4,5%; transportes rápidos inflavam números ao salvar quem antes era considerado morto em ação.
Evacuações por helicóptero (1-6 horas) e lavagem pulsátil para debridamento eram chave. Antibióticos lidavam com resistências (ex.: Pseudomonas), com reparos venosos preservando membros. Registros do Wound Data and Munitions Effectiveness Team (WDMET) informavam cuidados, reduzindo falha renal para 0,17%.
GUERRA DA UCRÂNIA: TRAUMAS IMPULSIONADOS POR DRONES
Desde 2022, drones revolucionaram os traumas, entregando explosivos com precisão. Biomecanicamente, drones FPV causam explosões quaternárias: barotrauma primário em pulmões/GI, shrapnel secundário (81% dos ferimentos), deslocamentos terciários e queimaduras por cargas termobáricas. Shrapnel cria múltiplas cavidades pequenas, com queimaduras e remoção tecidual semelhantes a IEDs, mas em escalas menores. Explosões repetidas de baixo nível elevam biomarcadores para danos crônicos, adicionando tensão psicológica por zumbidos constantes e vigilância.
Sangramento por danos arteriais é a causa primária de morte, com drones responsáveis por até 80% das baixas militares e 27-30% das mortes civis em 2025 (ex.: 38 fatalidades civis por drones de curto alcance em janeiro). Mortalidade civil geral por drones: 395 mortos, 2.635 feridos (2022-2025). Relatórios indicam que drones causam 70-80% das mortes e ferimentos no campo, com 75% das lesões vindas deles em análises recentes, e evacuações atrasadas elevando morbidade por uso prolongado de torniquetes.
Tratamentos incluem torniquetes para hemorragia, debridamento, shunts vasculares e fechamento assistido a vácuo. Amputações são comuns para lesões em membros, com termografia auxiliando reconstrução. Desafios incluem drones alvejando médicos, atrasando cuidados e elevando mortalidade. Equipes Cirúrgicas Avançadas adaptam modelos da Segunda Guerra-Coreia, mas a complexidade demanda inovação contínua, como treinamento em resiliência para fadiga de batalha e uso de ultrassom portátil. A onipresença de drones cria “zonas de exclusão tecnológica”, complicando operações médicas e sugerindo robótica para evacuações.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Vietnã | Fragmentação instável; explosões por minas | Choque, infecção | 2,6% hospitalar | Lavagem pulsátil, reparos venosos |
| Ucrânia (2022-) | Cavitação por shrapnel de drones; queimaduras termobáricas | Sangramento, politrauma | Até 80% das baixas relacionadas a drones | Torniquetes, retalhos, termografia |
Em conclusão, os traumas de guerra evoluíram de penetrações diretas não gerenciadas para explosões multifacetadas, com mortalidade declinando por avanços logísticos e farmacológicos. No entanto, drones na Ucrânia sinalizam um possível ressurgimento na complexidade, necessitando pesquisa em biomecânica adaptativa e tratamentos. Como visto em lições de conflitos passados, a inovação médica pode mitigar esses desafios, mas requer investimento global para proteger tanto militares quanto civis em guerras futuras.
Principais Pontos sobre a Evolução dos Traumas de Guerra
- A história dos ferimentos de guerra reflete avanços em armamentos e medicina: de ferimentos penetrantes de baixa energia em batalhas antigas, como Termópilas, para traumas complexos de alta energia causados por explosivos e drones modernos, com mortalidade caindo de mais de 70% em ferimentos graves antigos para menos de 3% em conflitos do século XX, graças a evacuações rápidas e antibióticos.
- Causas de morte evoluíram: hemorragia e infecções dominavam antigamente, mas explosões e drones na Ucrânia adicionam danos por ondas de choque, queimaduras e traumas psicológicos, com drones responsáveis por até 80% das baixas em alguns relatórios recentes.
- Taxas de mortalidade diminuíram historicamente, mas guerras modernas como a da Ucrânia podem inverter isso devido a atrasos em evacuações e ataques a instalações médicas; evidências sugerem que drones causam mais baixas civis do que qualquer outra arma em 2025.
- Tratamentos progrediram de remédios herbais para cirurgias de controle de danos e uso de torniquetes, com inovações como drones de evacuação sendo exploradas para mitigar riscos atuais.
Biomecânica dos Traumas Pesquisas indicam que ferimentos antigos envolviam lacerações diretas com cavidades permanentes mínimas, enquanto armas modernas criam cavidades temporárias extensas, danificando tecidos elásticos e causando necrose. Na Ucrânia, drones FPV geram fragmentos múltiplos e barotrauma, amplificando lesões em órgãos.
Causas de Morte e Taxas de Mortalidade Hemorragia permanece a principal causa, mas infecções caíram com antibióticos. Na Primeira Guerra Mundial, mortalidade por ferimentos era de cerca de 21%, caindo para 2,4% na Guerra da Coreia. Na Ucrânia, drones causam 70-80% das mortes e ferimentos no campo de batalha, com atrasos em cuidados elevando morbidade.
Medidas de Tratamento Avanços incluem debridamento precoce, transfusões e vacinas. Na era moderna, ênfase em treinamento de resiliência para estresse por drones e uso de ultrassom portátil para detecção precoce. Recomendações incluem integrar controle de estresse operacional em frentes avançadas.
A Doutrina da Visão Crítica de Segurança (CVS) na Colecistectomia Laparoscópica
Fundamentos, Técnica e a Era da Inteligência Artificial
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 11 minutos)
Introdução
A colecistectomia laparoscópica é, indiscutivelmente, uma das intervenções cirúrgicas mais realizadas no planeta. Contudo, apesar de sua consolidação como um procedimento seguro, as lesões iatrogênicas da via biliar principal continuam a assombrar os blocos operatórios, configurando-se como uma das complicações mais temidas, onerosas e medicolegais da cirurgia moderna. Com o advento da laparoscopia no final dos anos 1980, observou-se um pico alarmante na incidência dessas lesões. Em resposta a esta crise global de segurança, o Dr. Steven Strasberg propôs, em 1995, a Visão Crítica de Segurança (CVS – Critical View of Safety). Mais do que uma simples manobra, o CVS transformou-se no dogma central da cirurgia biliar. Este artigo disseca o racional anatômico, a sistematização técnica rigorosa e as aplicações contemporâneas do CVS na prática do cirurgião do aparelho digestivo.
A Patogenia da Lesão e o Racional Anatômico
Para prevenir um erro, é imperativo compreender a sua gênese. A literatura clássica é taxativa: a esmagadora maioria das lesões biliares graves não ocorre por imperícia motora, mas por um erro de percepção visual conhecido como “Misidentification” (identificação errônea). A armadilha mais letal é a técnica da “Visão Infundibular”. Em cenários de inflamação aguda, fibrose ou tração inadequada, o ducto colédoco ou o ducto hepático comum alinham-se paralelamente à vesícula, mimetizando o ducto cístico. O cirurgião se convence de forma ilusória e perigosa de que “tem certeza de estar vendo o cístico”. Essa confiança excessiva, aliada à dissecção apressada, resulta na transecção trágica da via biliar principal. O CVS foi desenhado cirurgicamente para aniquilar esta ilusão de ótica.
Os Três Critérios Obrigatórios do CVS
O CVS não é uma suposição; é um método de prova anatômica objetiva. Para declarar que o CVS foi atingido, todos os três critérios abaixo devem ser cumpridos simultaneamente antes do disparo de qualquer clipe ou grampeador:
- Dissecção Completa do Triângulo Hepatocístico: Toda a gordura e o tecido fibroso devem ser removidos do triângulo formado pelo ducto cístico, ducto hepático comum e borda hepática. A exposição deve ser visualizada nas faces anterior e posterior. Atenção: o ducto hepático comum não precisa (e não deve) ser dissecado ativamente, mas o triângulo deve estar limpo.
- Separação Parcial da Vesícula do Leito Hepático: O terço inferior da vesícula biliar (infundíbulo) deve ser completamente descolado da placa cística (leito hepático). Este passo, muitas vezes negligenciado, é o que garante que as estruturas dissecadas estão efetivamente a entrar na vesícula, e não a passar por ela.
- Apenas Duas Estruturas Adentrando a Vesícula: Exclusivamente um ducto (ducto cístico) e uma artéria (artéria cística) devem ser vistos penetrando a vesícula. Ambos devem estar completamente isolados e identificáveis circunferencialmente (visão de 360 graus).
Robustez, Saídas e a Era Moderna
A eficácia do CVS é incontestável. Em grandes séries publicadas, como a de Yegiyants et al. (3.042 casos) e Avgerinos et al. (998 casos) em que o CVS foi estritamente aplicado, a taxa de lesão biliar grave foi de zero.
Cenários Adversos e Técnicas de Saída (Bail-Out Procedures)
O verdadeiro valor do CVS revela-se na dificuldade. Quando há inflamação severa, colecistite crônica esclerosante ou um Triângulo de Calot “obliterado” (frozen Calot), atingir o CVS torna-se perigoso ou impossível.
Para o cirurgião maduro, a incapacidade de completar o CVS não é um obstáculo a ser forçado, mas um alarme biológico de perigo iminente. Nestes casos, impõe-se a adoção imediata de técnicas de saída:
- Colecistectomia Subtotal (Fenestrada ou Reconstituída).
- Conversão precoce para cirurgia aberta.
- Realização de Colangiografia Intraoperatória.
Documentação Médico-Legal
A diretriz cirúrgica contemporânea exige que o CVS seja documentado. O cirurgião deve registrar uma fotografia ou um breve vídeo intraoperatório que demonstre inequivocamente a exposição anterior e posterior das estruturas isoladas. A inclusão desta prova no prontuário médico eletrônico protege o paciente contra complicações e o cirurgião em contextos litigiosos.
Cirurgia Robótica e Inteligência Artificial (IA)
Os princípios de Strasberg são universais e imutáveis, independentemente do instrumental. Na Cirurgia Robótica, a visão 3D ampliada de altíssima definição e a estabilidade das pinças facilitam o cumprimento escrupuloso dos três critérios. As perspetivas futuras são ainda mais fascinantes: algoritmos de Inteligência Artificial (IA) e visão computacional estão a ser treinados para reconhecer e validar os critérios do CVS em tempo real no monitor cirúrgico, funcionando como um checklist virtual que impede a clipagem até que o software confirme a segurança anatômica.
Pontos-Chave para a Prática Diária
- Prova, não Suposição: O CVS transforma um julgamento subjetivo (“parece o cístico”) numa evidência anatômica irrefutável.
- O Risco da Visão Infundibular: Confundir um infundíbulo dilatado com o ducto cístico é a principal via para a lesão do colédoco.
- Descolar para Comprovar: Não basta limpar o triângulo de Calot; a separação do terço inferior da vesícula do leito hepático (Critério 2) é essencial.
- Pausa Obrigatória: A identificação das duas únicas estruturas deve ser um “momento estático”, validado verbalmente por toda a equipe antes da transecção.
Conclusões Aplicadas
O Critical View of Safety transcendeu a classificação de “técnica cirúrgica” para se consolidar como o padrão-ouro e a obrigação ética universal na colecistectomia laparoscópica. A sua adoção obrigatória nos programas de residência médica aniquila a cultura do achismo visual, substituindo-a pelo rigor científico. Ao reconhecer que o principal inimigo do cirurgião biliar é a sua própria mente (na forma de ilusão de ótica), o CVS fornece a armadura necessária para proteger o doente de morbidades devastadoras. Na cirurgia do aparelho digestivo, a paciência não é apenas uma virtude; é uma técnica de salvamento.A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.”
“A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.” — Dr. Steven Strasberg, cirurgião e idealizador da Visão Crítica de Segurança.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.