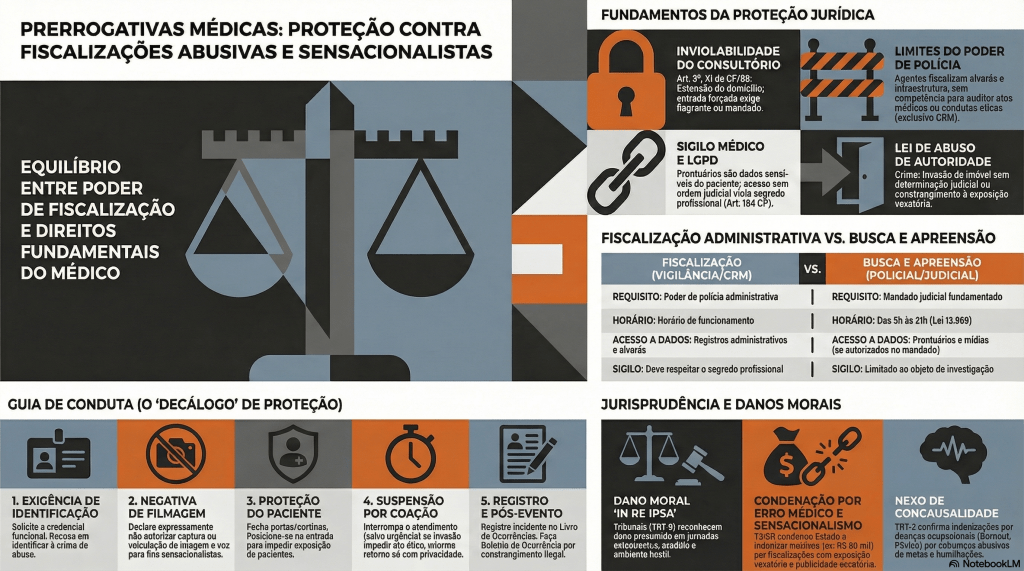
Proteção Médica contra Fiscalizações Invasivas e Sensacionalistas
No atual cenário das unidades de urgência e emergência, observa-se uma crescente e indevida ingerência de agentes externos que, sob o pretexto do escrutínio midiático, submetem o corpo clínico — em especial os profissionais em início de carreira — a constrangimentos e intimidações. Tais condutas, frequentemente pautadas pelo sensacionalismo digital, não apenas desestabilizam o ato médico, como violam preceitos basilares da Bioética e do Direito, notadamente o sigilo profissional e a dignidade do paciente. Diante desse panorama de vulnerabilidade técnica e jurídica, o presente Decálogo de Proteção Profissional Médica emerge como um instrumento essencial de salvaguarda, transpondo a dogmática jurídica para a práxis cotidiana. Este guia oferece diretrizes objetivas de conduta e interlocução, permitindo ao médico exercer sua autonomia com o rigor técnico, a polidez e a segurança jurídica que a complexidade do ambiente hospitalar exige.

I. Manutenção da Postura Ética e Prevenção do Conflito O médico deve manter o controle emocional, adotando postura polida e técnica. É imperativo evitar qualquer embate físico ou agressão verbal com os invasores, neutralizando o objetivo sensacionalista da ação.
- Fundamentação Jurídica e Ética: Código de Ética Médica (Resolução CFM nº 2.217/2018), Capítulo I, Inciso II; Art. 5º, inciso X, da Constituição Federal.
- O que fazer e o que falar: Respire fundo, cruze os braços ou mantenha as mãos visíveis (evitando qualquer gesto que possa ser interpretado como agressão). Não tente tomar o celular ou a câmera do invasor.
- Fala: “Boa noite/dia. Por favor, peço que mantenham a calma e baixem o tom de voz. Estamos em um ambiente hospitalar de respeito e cuidado.”
II. Exigência de Identificação e Recusa Expressa de Filmagem O profissional deve solicitar a identificação da autoridade e declarar, em bom tom (para que fique registrado na própria gravação), que não autoriza o uso, captura ou veiculação de sua imagem e voz.
- Fundamentação Jurídica e Ética: Art. 5º, inciso X, da Constituição Federal; Art. 20 do Código Civil.
- O que fazer e o que falar
- Ação: Olhe diretamente para a pessoa (e não para a câmera). Adote um tom de voz firme, porém sem gritar.
- Fala: “Sou o médico plantonista responsável neste momento. Por gentileza, identifiquem-se. Deixo expressamente claro, e peço que fique registrado em sua gravação, que não autorizo a captura, o uso ou a veiculação da minha imagem e da minha voz.”
III. Acionamento Imediato da Cadeia de Comando Institucional A gestão do fluxo de pessoas não é atribuição do médico assistente. Deve-se delegar a um membro da equipe (enfermagem/técnicos) o acionamento urgente da Segurança Patrimonial, da Chefia de Plantão e do Diretor Técnico da instituição.
- Fundamentação Jurídica e Ética: Resolução CFM nº 2.147/2016.
- O que fazer e o que falar
- Ação: Vire-se calmamente para um colega da equipe multidisciplinar e delegue a função, sem dar as costas totalmente aos invasores.
- Fala: “Enfermeiro(a) [Nome], por favor, acione imediatamente a segurança do hospital, a chefia de plantão e comunique o Diretor Técnico de que temos pessoas não autorizadas e com câmeras no setor.”
IV. Isolamento Físico e Proteção da Intimidade do Paciente A prioridade máxima é salvaguardar o paciente em atendimento. O médico deve fechar portas, fechar cortinas e posicionar-se fisicamente na entrada de consultórios ou enfermarias, bloqueando o acesso visual das câmeras aos enfermos.
- Fundamentação Jurídica e Ética: Art. 5º, inciso X, da Constituição Federal; Art. 73 do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Caminhe de forma decidida até a porta do consultório ou enfermaria, feche-a e coloque-se à frente dela como um obstáculo físico pacífico.
- Fala: “Os senhores não podem entrar aqui. Temos pacientes desnudos e vulneráveis em atendimento. Exijo que respeitem a privacidade e a intimidade dos enfermos.”
V. Evocação Expressa do Sigilo Médico e Profissional Diante da tentativa de invasão em áreas assistenciais, o médico deve declarar que está sob o imperativo legal do sigilo e que a presença de terceiros não autorizados munidos de câmeras configura quebra indireta de confidencialidade.
- Fundamentação Jurídica e Ética: Art. 154 do Código Penal Brasileiro; Art. 73, parágrafo único, do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Ignore perguntas médicas ou clínicas sobre qualquer paciente (“por que fulano está esperando?”, “o que aquele paciente tem?”).
- Fala: “Como médico, sou regido pela lei do sigilo absoluto. Não responderei a perguntas sobre casos clínicos e não permitirei filmagens nesta área, pois isso configura crime de quebra de confidencialidade médica.”
VI. Negativa Peremptória de Acesso a Prontuários Médicos O profissional deve negar a entrega, leitura ou auditoria de prontuários por parte de parlamentares ou fiscais não vinculados aos Conselhos de Medicina, informando que o documento pertence exclusivamente ao paciente.
- Fundamentação Jurídica e Ética: Resolução CFM nº 1.638/2002; Art. 89 do Código de Ética Médica.
- O que fazer e o que falar:
- Ação: Feche pastas de prontuários físicos ou bloqueie a tela do computador/sistema eletrônico.
- Fala: “O prontuário é um documento legal e de propriedade exclusiva do paciente. Sem a autorização expressa e por escrito dele, ou sem ordem judicial, os senhores não terão acesso a nenhum documento médico.”
VII. Esclarecimento dos Limites da Competência Fiscalizatória O médico deve esclarecer à autoridade que o exercício de “poder de polícia” parlamentar se restringe à infraestrutura e à administração pública, não abrangendo a fiscalização do ato médico ou da conduta ética, que possuem foro específico.
- Fundamentação Jurídica e Ética: Lei nº 3.268/1957.
- O que fazer e o que falar:
- Ação: Mantenha a compostura caso o político tente dar “ordens” ou fazer “ameaças” usando o cargo.
- Fala: “Reconheço o seu cargo, mas o senhor tem competência para fiscalizar a estrutura administrativa. A fiscalização do ato médico e da conduta profissional é prerrogativa exclusiva do Conselho Regional de Medicina (CRM). Por favor, dirija suas demandas ao Diretor da unidade.”
VIII. Suspensão do Atendimento sob Coação (Garantia de Condições Dignas) Caso as câmeras adentrem o recinto de atendimento e os invasores se recusem a sair, o médico deve suspender temporariamente o ato médico — desde que não seja caso de urgência/emergência — informando que só retornará quando a privacidade for restabelecida.
- Fundamentação Jurídica e Ética: Código de Ética Médica, Capítulo II, Inciso IV (Direitos dos Médicos).
- O que fazer e o que falar:
- Ação: Caso invadam a sala de forma ríspida, interrompa a consulta (se não houver risco de morte), sente-se na cadeira, afaste-se do paciente e aguarde em silêncio a retirada dos invasores pela segurança.
- Fala: “Diante da invasão e da coação com câmeras, informo que não possuo condições dignas e éticas para exercer a medicina neste momento. Retomarei o atendimento imediatamente após os senhores se retirarem e a segurança do paciente for garantida.”
IX. Registro Documental Exaustivo do Incidente Tão logo a situação seja normalizada, o médico deve registrar o fato detalhadamente no Livro de Ocorrências do plantão. Caso a invasão tenha prejudicado ou atrasado o atendimento de algum paciente, tal interferência externa deve constar no prontuário do mesmo.
- Fundamentação Jurídica e Ética: Art. 87 do Código de Ética Médica; Art. 299 do Código Penal.
- O que fazer e o que falar:
- Ação: Escreva de forma objetiva, descrevendo fatos (sem adjetivos emocionais). Cite os nomes dos invasores, a recusa em parar de filmar e os horários exatos.
- Fala (orientação mental e à equipe): “Precisamos registrar tudo. Vamos anotar o nome do vereador/político, o horário exato da invasão, o que foi dito e as paralisações geradas no atendimento para colocar no livro do plantão agora mesmo.”
X. Acionamento das Autoridades Policiais e Desagravo Institucional Ao término do plantão, é fundamental registrar Boletim de Ocorrência Policial e enviar comunicação oficial (Ofício) ao Diretor Técnico e ao Conselho Regional de Medicina (CRM) relatando o constrangimento.
- Fundamentação Jurídica e Ética: Decreto-Lei nº 3.688/1941, Art. 42; Art. 150 do Código Penal; Código de Ética Médica, Capítulo II, Inciso V.
- O que fazer e o que falar:
- Ação: Dirija-se a uma delegacia (ou delegacia virtual) e, nos dias subsequentes, contrate um advogado e/ou assessoria jurídica institucional e/ou do sindicato médico.
- Fala (na Delegacia / no Ofício): “Desejo registrar um Boletim de Ocorrência por constrangimento ilegal, perturbação do ambiente de trabalho e quebra tentada de sigilo médico contra o invasor. Solicitarei também, via ofício, o desagravo público junto ao CRM.”
A nossa missão máxima será sempre a defesa da saúde e da dignidade do doente. Que as orientações sirvam para garantir que nenhum profissional permita a usurpação do seu local de trabalho, assegurando que a nobre arte da Medicina seja sempre exercida em condições de absoluta segurança, privacidade e respeito mútuo.
Prof. Dr. Ozimo Gama
Princípios Fundamentais da Oncologia Cirúrgica Digestiva
Uma Abordagem Contemporânea e Baseada em Evidências
O Cenário Atual do Câncer Digestivo no Brasil
A Cirurgia do Aparelho Digestivo vive um momento de transformação sem precedentes. Não somos mais apenas “técnicos de ressecção”, mas parte integrante de uma complexa engrenagem multidisciplinar. A relevância deste tema é sublinhada pelos dados epidemiológicos alarmantes. Se no passado nos baseávamos em estimativas modestas, hoje a realidade é desafiadora: segundo a Estimativa 2023-2025 do Instituto Nacional de Câncer (INCA), esperam-se 704 mil casos novos de câncer por ano no Brasil.
Destaque-se que as neoplasias do trato gastrointestinal ocupam posições cimeiras. O câncer colorretal figura como o segundo mais incidente em mulheres e homens na maioria das regiões, com cerca de 45 mil novos casos anuais, seguido de perto pelo câncer de estômago (21 mil casos) e esôfago. Estes números não são apenas estatísticas; representam uma demanda crescente por cirurgiões oncológicos altamente qualificados, capazes de compreender não apenas a anatomia, mas a biologia tumoral.
A Biologia como Norte da Técnica Cirúrgica
Fisiopatologia e Disseminação
O entendimento clássico da cirurgia oncológica, herdado dos princípios de William Halsted no final do século XIX, baseava-se na premissa de que o câncer era uma doença puramente local que se disseminava centrifugamente. Embora a radicalidade (ressecção em bloco) permaneça um pilar, hoje compreendemos a doença como sistêmica desde fases precoces em muitos casos.
A disseminação ocorre por três vias principais que o cirurgião deve dominar:
- Linfática: Predominante em carcinomas (ex: adenocarcinoma gástrico e cólon).
- Hematogênica: Preferencial em sarcomas e carcinomas avançados (fígado e pulmões como sítios-alvo).
- Transcelômica (Peritoneal): Comum em neoplasias gástricas T3/T4, ovário e apêndice, exigindo estratégias específicas como a peritoniectomia.
O Princípio da Radicalidade e Margens (R0)
O objetivo primário da cirurgia oncológica curativa é a ressecção R0 (ausência de doença residual macroscópica e microscópica). A cirurgia R1 (doença microscópica residual) ou R2 (macroscópica) impacta drasticamente o prognóstico.
- Ressecção em Bloco: O tumor nunca deve ser violado. A peça deve ser removida envolta por tecido saudável, respeitando as fáscias anatômicas e os pedículos vasculares na sua origem.
- Linfadenectomia: Não serve apenas para estadiamento, mas tem papel terapêutico. No câncer gástrico, por exemplo, a linfadenectomia D2 é o padrão-ouro em centros especializados, associada a menor recidiva locorregional.
Neoadjuvância vs. Adjuvância
A decisão entre operar primeiro (upfront surgery) ou indicar terapia neoadjuvante é um dos grandes debates atuais.
- Vantagens da Neoadjuvância: Tratamento precoce de micrometástases, redução do tumor (downstaging) facilitando a ressecção R0 e teste in vivo da quimiossensibilidade. É o padrão atual para câncer de esôfago localmente avançado e câncer de reto médio/baixo.
- Vantagens da Adjuvância: Baseada no estadiamento patológico preciso (pTNM), evitando tratamento excessivo em estádios precoces.
Aplicação Prática na Cirurgia Digestiva
A prática moderna exige que o cirurgião diferencie dois conceitos cruciais frequentemente confundidos: Ressecabilidade e Operabilidade.
- Ressecabilidade: É uma característica do tumor (relação com estruturas vitais).
- Operabilidade: É uma característica do paciente (reserva funcional, comorbidades, status performance). Um tumor pode ser ressecável, mas o paciente inoperável.
O Papel da Citorredução e HIPEC
Para a carcinomatose peritoneal, historicamente considerada uma condição terminal, houve uma mudança de paradigma. Em neoplasias selecionadas (como pseudomixoma peritoneal, mesotelioma e alguns casos de câncer colorretal), a combinação de Cirurgia de Citorredução (Peritoniectomia) com Quimioterapia Intraperitoneal Hipertérmica (HIPEC) tem oferecido sobrevida em longo prazo, transformando uma doença fatal em uma condição crônica tratável.
Planejamento Multidisciplinar
O cirurgião oncológico não atua isolado. A discussão em Tumor Boards é mandatória. A indicação cirúrgica deve considerar a biologia molecular (ex: status do gene APC em colorretal, superexpressão de HER2 em gástrico) e a resposta a terapias sistêmicas.
Pontos-Chave para a Prática Cirúrgica
- Estadiamento Preciso: Nunca leve um paciente à sala sem um estadiamento completo. A laparoscopia diagnóstica é fundamental em tumores gástricos e pancreáticos para evitar laparotomias desnecessárias em casos de carcinomatose oculta.
- Margens Cirúrgicas: A margem circunferencial (radial) no câncer de reto e a margem proximal no câncer gástrico e esofágico são preditores independentes de sobrevida.
- Manuseio da Peça (“No-touch technique”): Evite a manipulação direta do tumor. A ligadura vascular prévia e a mobilização cuidadosa previnem a embolização tumoral intraoperatória.
- Documentação: O relatório cirúrgico deve detalhar as cadeias linfáticas dissecadas e as estruturas preservadas ou ressecadas, orientando o patologista e o oncologista clínico.
Perspectivas Futuras
A cirurgia digestiva na sua área de atuação oncológica evoluiu de amputações extensas para procedimentos de precisão, muitas vezes minimamente invasivos (laparoscópicos ou robóticos), sem perder a radicalidade oncológica. O futuro aponta para uma integração ainda maior com a biologia molecular e a imunoterapia. O cirurgião do futuro deverá ser, antes de tudo, um oncologista que opera: alguém que entende que o bisturi é apenas uma das armas, e que saber quando não operar é tão vital quanto a técnica operatória refinada.
Como nos ensinou o pai da cirurgia oncológica moderna:
“O cirurgião deve ser o médico do paciente oncológico, e não apenas o técnico que remove o tumor.” — William Stewart Halsted
Hashtags
#CirurgiaDigestiva #OncologiaCirurgica #EducaçãoMédica #ResidenciaCirurgia #CancerDigestivo
Gostou? ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat Online em nossa DM do Instagram.
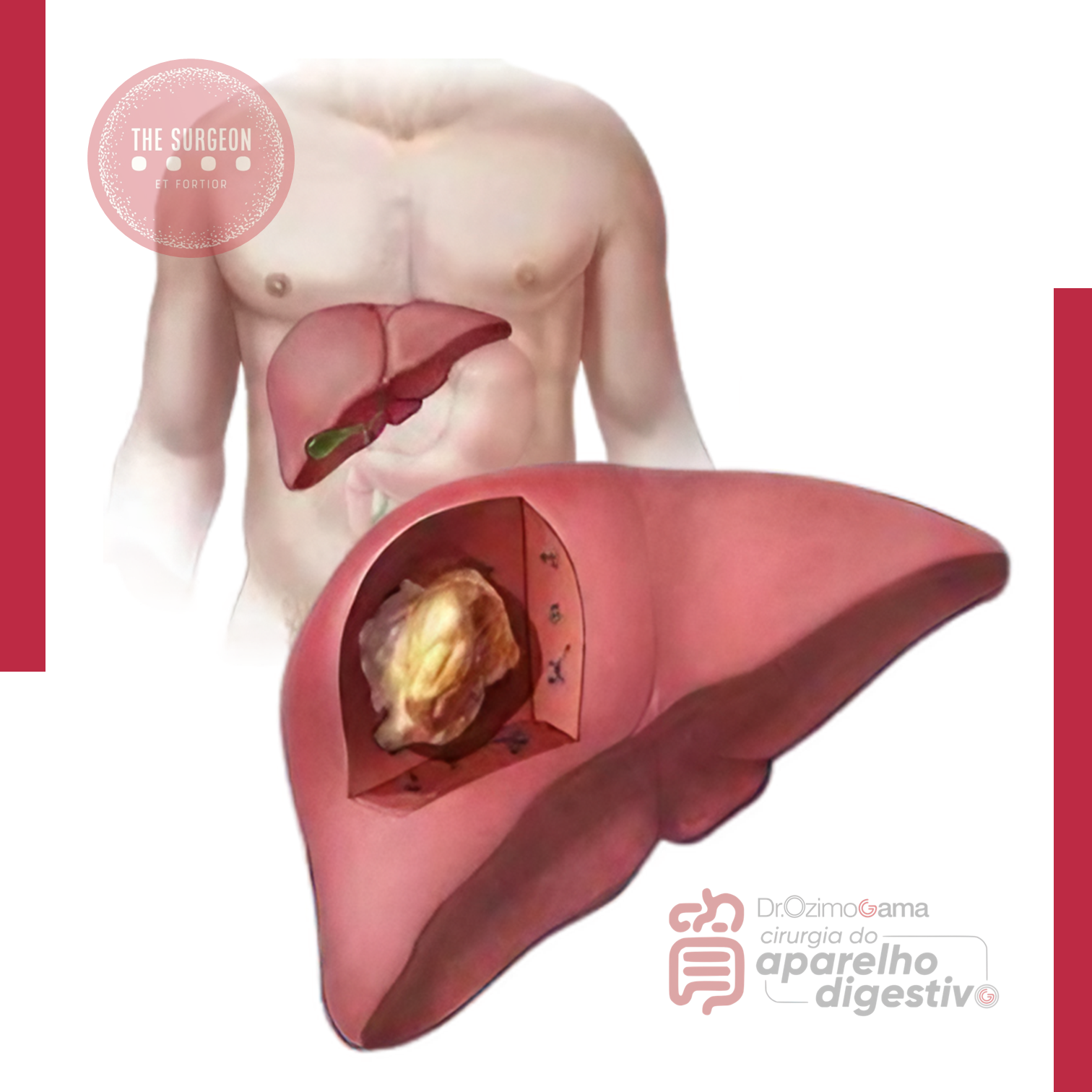
Efeito Cascata do Transplante Hepático
Introdução
O transplante hepático é universalmente reconhecido como o “padrão-ouro” para o tratamento de doenças hepáticas terminais. No entanto, o impacto deste procedimento transcende a substituição de um órgão doente. O sucesso fenomenal do transplante nas últimas cinco décadas produziu o que chamamos de ripple effect (efeito cascata) sobre toda a cirurgia geral e, especificamente, sobre a cirurgia hepatobiliar. Muitos dos princípios anatômicos, refinamentos técnicos e bases científicas que hoje aplicamos rotineiramente em hepatectomias regradas e cirurgias de trauma foram desenvolvidos ou aperfeiçoados nas salas de transplante. O objetivo desta exposição é dissecar como essa “escola” transformou a nossa prática diária, convertendo procedimentos outrora considerados de risco proibitivo em operações seguras e eficazes.
Desenvolvimento: Anatomia e Fisiologia Aplicadas
A base de qualquer cirurgia hepática segura é o domínio absoluto da anatomia e da fisiologia. O transplante nos forçou a olhar para o fígado não apenas como uma massa parenquimatosa, mas como uma estrutura segmentar com variações vasculares frequentes.
1. O Novo Mapa Anatômico
A experiência com doadores vivos e hepatectomias em cadáveres nos ensinou que a anatomia “de livro” é a exceção, não a regra.
- Variações Arteriais: Estudos clássicos de grandes centros transplantadores, como a série da UCLA, demonstram que aproximadamente 24% dos fígados possuem anomalias arteriais significativas. As mais comuns incluem a artéria hepática direita acessória ou substituída (originada da artéria mesentérica superior) e a esquerda (originada da artéria gástrica esquerda). O cirurgião que ignora essas variantes durante uma duodenopancreatectomia ou gastrectomia corre o risco de desvascularizar o fígado.
- Variações Biliares: A trifurcação do ducto hepático comum ocorre em cerca de 12% dos casos. O reconhecimento dessas nuances é vital para evitar estenoses e fístulas biliares, as complicações mais temidas no pós-operatório.
2. Regeneração e Isquemia
O transplante impulsionou a pesquisa sobre a capacidade regenerativa do fígado. O conceito de síndrome small-for-size (insuficiência hepática pós-resecção por remanescente pequeno) migrou do transplante intervivos para a oncologia. Hoje, calculamos com precisão o volume do fígado remanescente antes de grandes ressecções tumorais, utilizando estratégias como a embolização prévia da veia porta para hipertrofiar o lobo que ficará no paciente — uma aplicação direta do conhecimento de regeneração hepática. Além disso, o manuseio da lesão de isquemia-reperfusão evoluiu. Técnicas de precondicionamento isquêmico (clampeamento intermitente) permitem que realizemos ressecções complexas com menor perda sanguínea e menor dano hepatocelular.
Aplicação na Cirurgia Digestiva Geral e Oncológica
A transferência de tecnologia do transplante para a cirurgia digestiva geral é evidente em três pilares principais:
1. Ressecções Hepáticas Complexas e Preservação Caval
Antigamente, tumores no lobo caudado ou que envolviam a veia cava retro-hepática eram considerados irressecáveis. A técnica de “piggyback” (preservação da veia cava inferior do receptor durante o transplante) ensinou aos cirurgiões oncológicos como dissecar o fígado da veia cava com segurança, permitindo a ressecção de tumores centrais e posteriores com margens livres.
2. Controle Vascular no Trauma
O cirurgião de trauma moderno utiliza manobras de exclusão vascular total (clampeamento da porta e da veia cava supra e infra-hepática) para reparar lesões venosas complexas em fígados traumatizados. Esta é uma manobra derivada diretamente da hepatectomia do receptor no transplante. Em pacientes estáveis, isso permite reparos exangues; em instáveis, técnicas de damage control com shunts portocavais temporários podem ser salvadoras.
3. Cirurgia Ex Situ
Para casos extremos de tumores invadindo a confluência cavo-hepática, a técnica de hepatectomia total, seguida de perfusão fria do órgão na bancada (bench surgery), ressecção do tumor ex vivo e reimplante do fígado (autotransplante), é a fronteira final da cirurgia hepatobiliar, tornada possível apenas pelo domínio das técnicas de preservação de órgãos.
Cenário Brasileiro: Uma Potência Mundial 🇧🇷
É fundamental contextualizar nossa realidade. O Brasil possui o maior sistema público de transplantes do mundo.
- Segundo dados recentes da Associação Brasileira de Transplante de Órgãos (ABTO) e do Ministério da Saúde, o Brasil realiza mais de 2.000 transplantes hepáticos anualmente, posicionando-se consistentemente entre as três nações com maior número absoluto de procedimentos no mundo.
- Essa estatística não é apenas um número; ela representa um volume crítico de treinamento. Residentes brasileiros em centros de excelência têm uma exposição prática à anatomia hepática complexa superior à de muitos países desenvolvidos. O “Cirurgião SUS” é, por necessidade e oportunidade, um especialista em variações anatômicas e manuseio de situações complexas.
Pontos-Chave para o Cirurgião em Formação
- Identificação Pré-operatória: Sempre investigue variações arteriais (ex: artéria hepática direita vindo da mesentérica) em exames de imagem antes de qualquer cirurgia do andar supramesocólico.
- Manobras de Exclusão: Familiarize-se com a Manobra de Pringle e a exclusão vascular total; elas são suas ferramentas de segurança em sangramentos maciços.
- Dissecção Hilar: A técnica de baixar a placa hilar e dissecar as estruturas glissonianas extra-hepáticas é mais segura e oncológica do que a dissecção intraparenquimatosa cega.
- Interdisciplinaridade: A cirurgia moderna não é um ato solitário. Radiologia intervencionista, hepatologia e terapia intensiva são extensões do braço do cirurgião.
Conclusão
O transplante hepático não deve ser visto pelos estudantes e residentes apenas como uma subespecialidade de nicho, mas como a “Universidade da Cirurgia Abdominal”. As lições aprendidas com a preservação de órgãos, a dissecção meticulosa de vasos de calibre milimétrico e o manejo fisiológico do paciente hepatopata elevaram o padrão técnico de toda a cirurgia digestiva. Dominar esses conceitos é o que diferencia o operador técnico do verdadeiro cirurgião cientista.
“A história da medicina é que o que era inconcebível ontem, e apenas alcançável hoje, muitas vezes torna-se rotina amanhã.” — Thomas Starzl (Pioneiro do Transplante Hepático)
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags: #CirurgiaHepatobiliar #TransplanteHepatico #EducacaoMedica #ResidenciaCirurgia #CirurgiaDigestiva
A Importância da Competência em Endoscopia Flexível na Formação e Prática do Cirurgião do Aparelho Digestivo
Introdução
A endoscopia digestiva flexível transcende a sua definição clássica de método diagnóstico. Na contemporaneidade, ela se consolida como uma extensão essencial da propedêutica e da terapêutica cirúrgica. Historicamente, é imperativo recordar que a endoscopia é um campo desbravado por cirurgiões. Grandes marcos, como a polipectomia colônica (Dr. Hiromi Shinya), a ligadura elástica de varizes (Dr. Goffredo Steigman) e a gastrostomia percutânea (Dr. Jeffrey Ponsky), foram estabelecidos por mentes cirúrgicas que vislumbravam além da incisão convencional. No entanto, observa-se um fenômeno preocupante na formação médica atual: a delegação progressiva dessa competência quase exclusivamente à gastroenterologia clínica e aos Endoscopistas. Este artigo, direcionado a estudantes, residentes e pós-graduandos, visa reafirmar a premissa de que a habilidade endoscópica não é opcional, mas intrínseca à prática do cirurgião do aparelho digestivo de excelência. Discutiremos as bases técnicas, a aplicabilidade clínica e a necessidade premente de retomar o protagonismo nesta área.
Desenvolvimento: O Cirurgião e a Habilidade Endoscópica
A Curva de Aprendizado e a Visão Cirúrgica
A competência em endoscopia flexível depende da fusão entre conhecimento cognitivo e habilidade técnica (psicomotora). O cirurgião, habituado à manipulação tecidual e à anatomia tridimensional (seja por via aberta ou laparoscópica), possui, intrinsecamente, uma coordenação olho-mão refinada. Estudos demonstram que cirurgiões tendem a apresentar uma curva de aprendizado acelerada para procedimentos endoscópicos devido à sua familiaridade com a anatomia topográfica e patológica, permitindo uma transição fluida entre a visão bidimensional da tela e a realidade espacial do órgão.
O Cenário Brasileiro e a Demografia Médica
No contexto do Brasil, um país de dimensões continentais, a distribuição de especialistas é heterogênea. Segundo dados da Demografia Médica no Brasil (CFM/USP, 2023), a concentração de gastroenterologistas clínicos | endoscopistas é significativamente menor e mais centralizada em grandes capitais do que a de cirurgiões gerais. O cirurgião geral, muitas vezes, é o único provedor de saúde especializado em regiões remotas. Portanto, capacitar o cirurgião em endoscopia não é apenas uma questão de reserva de mercado, mas uma estratégia de saúde pública para garantir o rastreamento de neoplasias (como o câncer colorretal) e o atendimento de urgências (Hemorragia Digestiva Alta) em todo o território nacional.
Do “Cirurgião de Resgate” ao “Cirurgião Estrategista”
Tradicionalmente, o cirurgião é chamado apenas quando ocorre uma complicação endoscópica, como perfuração ou sangramento incontrolável. Este paradigma do “cirurgião de resgate” deve evoluir para o de “cirurgião estrategista”. Ao dominar a endoscopia, o profissional torna-se o capitão do navio terapêutico, capaz de indicar, executar e, se necessário, converter procedimentos com total autonomia e continuidade do cuidado (Continuity of Care), otimizando desfechos e minimizando a fragmentação do tratamento.
Aplicação na Cirurgia Digestiva
A integração da endoscopia na rotina cirúrgica pode ser categorizada em três momentos cruciais:
1. Endoscopia Pré-Operatória: O Planejamento
Na cirurgia oncológica gástrica e esofágica, a endoscopia realizada pelo próprio cirurgião permite uma avaliação precisa das margens tumorais, da extensão para a cárdia ou piloro, influenciando diretamente a decisão entre uma gastrectomia total ou subtotal. Na cirurgia bariátrica, a identificação prévia de hérnias hiatais, esofagites severas ou patologias gástricas altera o algoritmo de tratamento, contraindicando, por exemplo, uma gastrectomia vertical (Sleeve) em favor de um bypass gástrico em Y de Roux.
2. Endoscopia Intraoperatória: A Segurança
O uso transoperatório é uma ferramenta de segurança inestimável. Exemplos clássicos incluem:
- Cardiomiotomia de Heller: Confirmação da adequação da miotomia e teste de vazamento (air leak test) para exclusão de perfurações mucosas.
- Exploração de Vias Biliares: O uso do coledocoscópio flexível permite a visualização direta e a certeza da remoção completa de cálculos, superior à colangiografia em casos complexos.
- Localização de Lesões: Em cirurgias colorretais laparoscópicas, a tatuagem prévia pode ser imprecisa; a colonoscopia intraoperatória garante a ressecção do segmento correto.
3. Endoscopia Pós-Operatória e Manejo de Complicações
O cirurgião que realiza a endoscopia está melhor posicionado para tratar as complicações de seus próprios procedimentos ou de seus pares. O arsenal terapêutico inclui:
- Dilatação com balão de estenoses de anastomoses (gástricas ou colorretais).
- Tratamento de fístulas e deiscências pós-Sleeve ou Bypass com uso de próteses autoexpansíveis ou clips endoscópicos.
- Hemostasia de linhas de sutura sangrantes.
A Era da Cirurgia Sem Cicatrizes (NOTES e Endoterapia)
Caminhamos para a era da intervenção minimamente invasiva máxima. Procedimentos como o POEM (Peroral Endoscopic Myotomy) substituem a miotomia laparoscópica; o Endoscopic Sleeve Gastroplasty (ESG) surge como alternativa à cirurgia bariátrica convencional. O cirurgião do futuro deve dominar estas técnicas para oferecer o tratamento “estado da arte” aos seus pacientes.
Pontos-Chave para o Residente e o Cirurgião Jovem
- Necessidade de Treinamento: As sociedades cirúrgicas (como o CBC e a CBCD no Brasil) têm enfatizado a importância de currículos de residência que contemplem carga horária prática em endoscopia.
- Visão Integral: O domínio da luz luminal (endoscopia) e da cavidade peritoneal (laparoscopia/robótica) cria um cirurgião completo.
- Autonomia: A capacidade de diagnosticar, estadiar, tratar e resolver complicações sem depender de terceiros é o ápice da eficiência clínica.

Conclusão
A endoscopia flexível não é uma especialidade à parte, mas uma ferramenta cirúrgica, tal qual o bisturi ou a pinça laparoscópica. Para o cirurgião do aparelho digestivo, delegar essa competência é abdicar de uma parte fundamental da sua herança histórica e do seu futuro profissional. A formação médica deve encorajar a retomada deste espaço, garantindo que as novas gerações de cirurgiões sejam proficientes tanto na ciência da cirurgia quanto na arte da endoscopia. Como mentores e instituições de ensino, nosso dever é prover o acesso e a estrutura para que essa integração seja a norma, e não a exceção.
“Nenhum homem pode ser um grande cirurgião sem ser também um grande médico; e o inverso é igualmente verdadeiro no que tange ao diagnóstico e compreensão da fisiopatologia.” > — Adaptado de Theodor Billroth, pai da cirurgia gástrica moderna, refletindo a necessidade do conhecimento integral.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags: #CirurgiaDigestiva #EndoscopiaTerapeutica #ResidenciaMedica #CirurgiaGeral #EducacaoMedica
Epônimos da Anatomia Abdominal: A Linguagem Clássica da Prática Cirúrgica
Resumo Apesar dos esforços contínuos da Federative International Programme on Anatomical Terminologies (FIPAT) para padronizar a nomenclatura médica através da Terminologia Anatômica, a linguagem operatória permanece, em essência, histórica. Este artigo revisa os epônimos fundamentais da anatomia abdominal, correlacionando a definição topográfica precisa com a aplicação prática na cirurgia do aparelho digestivo, demonstrando que o domínio destes termos é imperativo para a segurança cirúrgica e a comunicação clínica eficaz.
Introdução
A anatomia topográfica é o alicerce da cirurgia. Contudo, existe uma dicotomia persistente entre a taxonomia descritiva moderna e a tradição oral dos centros cirúrgicos. Enquanto a academia privilegia termos locativos e funcionais, a prática diária — da passagem de plantão à descrição operatória — é dominada por epônimos. Estes nomes não são meros vestígios históricos; funcionam como “atalhos cognitivos” que evocam, simultaneamente, uma localização, uma relação anatômica complexa e, frequentemente, uma manobra cirúrgica específica. Este estudo revisa os principais epônimos do abdome, dissecando sua anatomia estrutural e sua relevância crítica na cirurgia geral e digestiva.
1. Parede Abdominal e Região Inguinal
A compreensão da estratigrafia da parede abdominal é o primeiro passo para o acesso seguro à cavidade peritoneal e para o reparo eficaz dos defeitos herniários.
Estratigrafia Subcutânea e Fascial
- Fáscia de Camper
- Anatomia: Camada superficial, de espessura variável e consistência adiposa, do tecido subcutâneo da parede abdominal anterior.
- Relevância Cirúrgica: A sua aproximação é frequentemente negligenciada, mas o manejo adequado do espaço morto nesta camada é vital na prevenção de seromas e infecções de sítio cirúrgico superficiais em laparotomias.
- Fáscia de Scarpa
- Anatomia: Camada membranosa profunda do tecido subcutâneo, contendo fibras elásticas amarelas, que se funde com a fáscia lata da coxa inferiormente e continua como Fáscia de Colles no períneo.
- Relevância Cirúrgica: Estrutura de fixação essencial no fechamento da parede abdominal. A sutura desta camada reduz a tensão sobre a pele e melhora o resultado estético da cicatriz. Devido à sua continuidade perineal, orienta a disseminação de extravasamentos de urina (fraturas de uretra) ou infecções necrotizantes (Síndrome de Fournier).
- Fáscia Inominada de Gallaudet
- Anatomia: A fáscia profunda aderida intimamente à aponeurose do músculo oblíquo externo e bainha do reto.
- Relevância Cirúrgica: Define o plano de dissecção “limpo” sobre a aponeurose durante o reparo de hérnias ou a confecção de retalhos miocutâneos.
Anatomia Inguinal e o Assoalho Pélvico
- Ligamento de Poupart (Ligamento Inguinal)
- Anatomia: A borda inferior espessa e recorvada da aponeurose do oblíquo externo, estendendo-se da Espinha Ilíaca Antero-Superior (EIAS) ao tubérculo púbico.
- Relevância Cirúrgica: O marco anatômico absoluto que diferencia hérnias inguinais (acima) de femorais (abaixo) e serve como âncora inferior para reparos teciduais clássicos (técnica de Bassini) e fixação de telas (técnica de Lichtenstein).
- Ligamento de Gimbernat (Ligamento Lacunar)
- Anatomia: Uma extensão triangular das fibras do ligamento inguinal que se reflete posteriormente e se insere na linha pectínea.
- Relevância Cirúrgica: Forma a borda medial do anel femoral. Em casos de hérnia femoral encarcerada, este ligamento é frequentemente a estrutura constritora que deve ser incisada (com cautela devido à presença eventual da Corona Mortis vascular) para redução do conteúdo.
- Ligamento de Cooper (Ligamento Pectíneo)
- Anatomia: Espessamento do periósteo e fáscia ao longo da crista pectínea (pecten do púbis), posterior ao ligamento inguinal.
- Relevância Cirúrgica: Estrutura de ancoragem robusta utilizada na técnica de McVay e ponto de fixação crítico para telas em reparos laparoscópicos (TAPP/TEP), prevenindo a recidiva direta.
- Triângulo de Hesselbach
- Anatomia: Delimitado inferiormente pelo ligamento inguinal, lateralmente pelos vasos epigástricos inferiores e medialmente pela borda lateral do músculo reto abdominal.
- Relevância Cirúrgica: Local anatômico das hérnias inguinais diretas, resultantes da fraqueza da fáscia transversal, sem passagem pelo anel inguinal profundo.
- Orifício Miopectíneo de Fruchaud
- Anatomia: Área de fraqueza da parede abdominal inferior que engloba tanto a região inguinal quanto a femoral, delimitada pelo músculo oblíquo interno (superior), reto abdominal (medial), iliopsoas (lateral) e osso púbico (inferior).
- Relevância Cirúrgica: Conceito fundamental para a cirurgia moderna de hérnia. Estabelece que o tratamento definitivo deve cobrir toda esta área com material protético (tela) para prevenir todos os tipos de hérnias da virilha, princípio base das abordagens pré-peritoneais (Stoppa, TEP, TAPP).
2. Peritônio e Topografia Gastrointestinal
A semiologia abdominal e o acesso cirúrgico baseiam-se em projeções de superfície e recessos cavitários.
- Linha de Monro-Richter e Ponto de McBurney
- Anatomia: Linha traçada da EIAS à cicatriz umbilical. O Ponto de McBurney localiza-se na união do terço lateral com os dois terços mediais desta linha.
- Relevância Cirúrgica: Projeção clássica da base do apêndice cecal. Guia a incisão oblíqua (McBurney) para apendicectomias abertas e é o ponto de máxima dor à descompressão na apendicite aguda.
- Ponto de Lanz
- Anatomia: Ponto na junção do terço direito com o terço médio da linha bi-ilíaca.
- Relevância Cirúrgica: Representa a projeção variável do ápice de um apêndice pélvico longo, importante no diagnóstico diferencial de patologias anexiais em mulheres.
- Ponto de Murphy
- Anatomia: Ponto situado abaixo do rebordo costal direito, na linha hemiclavicular (borda lateral do reto abdominal).
- Relevância Cirúrgica: Local de palpação do fundo da vesícula biliar. A interrupção súbita da inspiração à palpação profunda (Sinal de Murphy) é altamente sugestiva de colecistite aguda.
- Espaços Peritoneais (Morison, Douglas, Proust, Winslow)
- Bolsa de Morison (Recesso Hepatorrenal): Espaço virtual entre o fígado e o rim direito. É o local mais dependente do abdome superior em decúbito dorsal; local primário de acúmulo de líquido livre (sangue/pus) detectável pelo FAST em trauma.
- Fundo de Saco de Douglas (Retouterino): Ponto mais declive da cavidade peritoneal em mulheres. Local de acúmulo de líquido e de implantes metastáticos palpáveis ao toque retal/vaginal (Prateleira de Blumer).
- Espaço de Proust (Retovesical): Correspondente masculino ao de Douglas, entre o reto e a bexiga/próstata. Relevante na dissecção oncológica do reto baixo.
- Forame de Winslow (Forame Epiploico): Comunicação entre a grande cavidade e a retrocavidade dos epíplons (bolsa omental). Clinicamente, é a via de acesso para a Manobra de Pringle (clampeamento do pedículo hepático para controle de hemorragia) e local potencial para hérnias internas.
3. Trato Digestório e Glândulas Anexas
Nesta região, os epônimos descrevem marcos críticos para ressecções oncológicas e reconstruções.
- Ângulo de His
- Anatomia: Ângulo agudo formado entre o esôfago abdominal e o fundo gástrico.
- Relevância Cirúrgica: Componente anatômico crucial do mecanismo valvular anti-refluxo. A restauração ou acentuação deste ângulo é o objetivo central das fundoplicaturas (Nissen, Toupet) no tratamento da DRGE.
- Espaço de Traube e Triângulo de Labbé
- Anatomia: Áreas de projeção gástrica na parede toracoabdominal.
- Relevância Cirúrgica: O Espaço de Traube (timpânico à percussão) torna-se maciço em casos de esplenomegalia ou derrame pleural. O Triângulo de Labbé é a área de contato do estômago com a parede abdominal anterior, local seguro para gastrostomias percutâneas ou cirúrgicas.
- Ligamento de Treitz
- Anatomia: Músculo suspensor do duodeno, marcando a transição duodeno-jejunal (ângulo de Treitz).
- Relevância Cirúrgica: Divisor anatômico e clínico entre hemorragia digestiva alta e baixa. Marco fundamental para a mobilização do intestino delgado e identificação da primeira alça jejunal em reconstruções (ex: Y de Roux).
- Vias Biliares: Calot, Heister, Vater, Oddi
- Triângulo de Calot (Cisto-Hepático): Delimitado pelo ducto cístico, ducto hepático comum e borda inferior do fígado. Contém a artéria cística e, frequentemente, o linfonodo de Mascagni. Sua dissecção meticulosa para obter a “Visão Crítica de Segurança” (Critical View of Safety) é mandatória para prevenir lesões iatrogênicas da via biliar principal na colecistectomia.
- Válvulas de Heister: Pregas espirais na mucosa do ducto cístico que podem dificultar a cateterização durante a colangiografia intraoperatória.
- Ampola de Vater e Esfíncter de Oddi: A confluência biliopancreática e seu aparelho esfincteriano. Alvo terapêutico nas papilotomias endoscópicas (CPRE) para tratamento de coledocolitíase ou pancreatite biliar.
- Ductos Pancreáticos (Wirsung e Santorini)
- Anatomia: Wirsung é o ducto principal; Santorini é o acessório.
- Relevância Cirúrgica: A variante Pancreas Divisum (falha na fusão dos ductos) drena a maior parte do pâncreas pelo ducto menor (Santorini), sendo causa de pancreatites recorrentes inexplicadas.
- Marcos Hepáticos (Glisson e Cantlie)
- Cápsula de Glisson: Bainha de tecido conjuntivo que envolve o fígado e a tríade portal intra-hepática. Importante na manobra de Pringle e nas hepatectomias.
- Linha de Cantlie: Linha imaginária que divide o fígado funcionalmente em lobos direito e esquerdo, estendendo-se do leito da vesícula biliar à veia cava inferior. Base para hepatectomias anatômicas maiores.
4. Vascularização Abdominal e Colateralização
O conhecimento das arcadas vasculares é determinante para a viabilidade de anastomoses intestinais.
- Tripus Halleri (Tronco Celíaco)
- Anatomia: Trifurcação da aorta abdominal em artéria gástrica esquerda, esplênica e hepática comum.
- Relevância Cirúrgica: O controle vascular proximal em cirurgias de trauma, gastrectomias e pancreatectomias. Variações anatômicas são frequentes e devem ser antecipadas em planejamentos pré-operatórios.
- Arcada de Riolan e Artéria Marginal de Drummond
- Anatomia: Sistemas de anastomose entre a artéria cólica média (ramo da mesentérica superior) e a cólica esquerda (ramo da mesentérica inferior).
- Relevância Cirúrgica: Garantem a irrigação do cólon, especialmente na flexura esplênica (ponto crítico de Griffiths). A preservação ou a integridade funcional destas arcadas é vital para evitar isquemia do coto colônico em retossigmoidectomias e cirurgias de aneurisma de aorta.
- Veias de Sappey e Retzius
- Anatomia: Veias acessórias do sistema porta. As de Sappey correm no ligamento falciforme; as de Retzius são anastomoses retroperitoneais.
- Relevância Cirúrgica: Em pacientes com hipertensão portal, estas veias dilatam-se dramaticamente. As veias de Sappey podem causar hemorragia maciça na incisão supraumbilical. A dissecção do retroperitônio em cirróticos pode encontrar o “sangramento em lençol” das veias de Retzius, de difícil controle.
5. Retroperitônio e Rins
A anatomia dos planos fasciais renais é a chave para a cirurgia oncológica retroperitoneal radical.
- Fáscia de Gerota (e suas lâminas Toldt e Zuckerkandl)
- Anatomia: A fáscia renal envolve o rim e a adrenal em um compartimento fechado. A lâmina anterior é frequentemente associada à Fáscia de Toldt (no contexto da fusão com o mesocólon), e a posterior à Fáscia de Zuckerkandl.
- Relevância Cirúrgica: A “Fáscia de Toldt” (ou linha de Toldt) representa o plano de clivagem avascular embrionário entre o mesocólon e o retroperitônio. A dissecção neste plano permite a mobilização incruenta do cólon direito e esquerdo, essencial em colectomias oncológicas laparoscópicas e abertas. A violação da Fáscia de Gerota propriamente dita é indicativa de invasão tumoral renal ou necessária para nefrectomias radicais.
Conclusão
O domínio dos epônimos da anatomia abdominal transcende o exercício de erudição histórica; constitui uma ferramenta de precisão técnica. Falar em “Triângulo de Calot” ou “Fáscia de Toldt” evoca, instantaneamente, uma estratégia cirúrgica e um plano de segurança. Para o cirurgião do aparelho digestivo, a fluência nesta linguagem clássica é tão fundamental quanto a destreza manual, permitindo a integração segura entre o conhecimento anatômico estático e a dinâmica do ato operatório.
Referência-Base: ten Donkelaar HJ, Quartu M, Kachlík D. An Illustrated Guide to Anatomical Eponyms. Springer, 2025.
Índice Biográfico dos Epônimos Citados
Abaixo listam-se os dados biográficos dos anatomistas e cirurgiões cujos epônimos foram discutidos neste artigo.
- Blumer, George (Reino Unido/EUA, 1858–1940)
- Calot, Jean-François (França, 1861–1944)
- Camper, Pieter (Holanda, 1722–1789)
- Cantlie, James (Escócia, 1851–1926)
- Colles, Abraham (Irlanda, 1773–1843)
- Cooper, Astley Paston (Reino Unido, 1768–1841)
- Douglas, James (Escócia, 1675–1742)
- Drummond, Hamilton (Reino Unido, 1882–1925)
- Fruchaud, Henri (França, 1894–1960)
- Gallaudet, Bern Budd (EUA, 1860–1934)
- Gerota, Dimitrie (Romênia, 1867–1939)
- Gimbernat, Antoni de (Espanha, 1734–1816)
- Glisson, Francis (Reino Unido, 1597–1677)
- Griffiths, Joseph (País de Gales, 1863–1945)
- Haller, Albrecht von (Suíça, 1708–1777)
- Heister, Lorenz (Alemanha, 1683–1758)
- Hesselbach, Franz Kaspar (Alemanha, 1759–1816)
- His, Wilhelm Jr. (Suíça, 1863–1934)
- Labbé, Léon (França, 1832–1916)
- Lanz, Otto (Suíça, 1865–1935)
- Mascagni, Paolo (Itália, 1755–1815)
- McBurney, Charles (EUA, 1845–1913)
- Monro, Alexander III (Escócia, 1773–1859)
- Morison, James Rutherford (Reino Unido, 1853–1939)
- Murphy, John Benjamin (EUA, 1857–1916)
- Oddi, Ruggero (Itália, 1864–1913)
- Poupart, François (França, 1661–1709)
- Pringle, James Hogarth (Escócia, 1863–1941)
- Proust, Robert (França, 1873–1935)
- Retzius, Anders (Suécia, 1796–1860)
- Richter, August Gottlieb (Alemanha, 1742–1812)
- Riolan, Jean (o Jovem) (França, 1580–1657)
- Santorini, Giovanni Domenico (Itália, 1681–1737)
- Sappey, Marie Philibert Constant (França, 1810–1896)
- Scarpa, Antonio (Itália, 1752–1832)
- Toldt, Carl (Áustria, 1840–1920)
- Traube, Ludwig (Alemanha, 1818–1876)
- Treitz, Václav (República Tcheca/Áustria, 1819–1872)
- Vater, Abraham (Alemanha, 1684–1751)
- Winslow, Jacob Benignus (Dinamarca/França, 1669–1760)
- Wirsung, Johann Georg (Alemanha, 1589–1643)
- Zuckerkandl, Emil (Áustria, 1849–1910)
Lifelong Learning na Cirurgia: como a Aprendizagem Autorregulada Define o Cirurgião que Você Vai Ser
Introdução
A cirurgia é uma das áreas da medicina que mais muda ao longo do tempo. Técnicas, tecnologias, diretrizes e condutas são revisadas continuamente. Ninguém termina a residência “pronto para tudo”. O que diferencia o cirurgião que se mantém competente e atualizado ao longo da carreira não é apenas o que aprendeu na formação inicial, mas a capacidade de continuar aprendendo de forma ativa, intencional e estruturada.
Esse é o núcleo do conceito de Lifelong Learning: o compromisso de adquirir, revisar e integrar novos conhecimentos desde o primeiro dia de faculdade até o último dia de atividade profissional. E, na prática, o que sustenta isso é um conjunto de habilidades chamado aprendizagem autorregulada.
O problema: bons alunos, maus aprendizes
Grande parte dos estudantes que chegam à residência é formada por “altos desempenhos acadêmicos”. Mas muitos:
- atribuem sucesso e fracasso quase sempre ao professor, ao serviço ou ao tipo de prova;
- não conseguem descrever com clareza como estudam;
- acreditam que aprender é algo que “acontece com eles”, não algo que podem controlar.
Esse modelo funciona em um ambiente escolar tradicional, com provas previsíveis e conteúdo delimitado. Em cirurgia, não. No centro cirúrgico, na UTI ou no pronto-socorro, o cirurgião depende de outra coisa: da capacidade de identificar o que não sabe, de estudar com foco e de ajustar a própria prática a partir de resultados reais.
É aqui que entra a aprendizagem autorregulada.
O que é aprendizagem autorregulada?
Aprendizagem autorregulada é o conjunto de hábitos, estratégias e atitudes que fazem o aluno assumir o controle do próprio processo de aprendizagem.
Um aprendiz autorregulado:
- pensa sobre como aprende (metacognição);
- acredita que é capaz de melhorar com esforço e estratégia (autoeficácia realista);
- organiza o ambiente, o tempo e os recursos para aprender melhor (comportamento ativo).
Na prática, isso aparece em três dimensões:
1. Metacognitiva
- Define objetivos de aprendizado (“quero entender critérios de indicação de neoadjuvância no pâncreas”, “quero melhorar decisão em vesícula difícil”).
- Planeja como chegar lá (o que ler, que casos observar, que vídeos rever).
- Monitora se está, de fato, avançando.
- Se autoavalia com honestidade ao final.
2. Motivacional
- Liga esforço a desempenho.
- Não se vê como “bom” ou “ruim”, mas como alguém em processo de desenvolvimento.
- Usa erros como feedback, não como sentença.
3. Comportamental
- Seleciona ativamente casos, plantões e oportunidades que trazem aprendizado.
- Busca ajuda, feedback e coaching quando necessário.
- Usa estratégias de estudo estruturadas, não apenas leitura passiva.
O ciclo da aprendizagem autorregulada
Você pode enxergar esse processo como um ciclo contínuo:
- Planejamento (forethought)
- O que quero aprender?
- Por que isso é importante agora?
- Quanto tempo vou dedicar? Com que materiais?
- Execução (performance)
- Implementar o plano (leitura, vídeo, simulação, prática supervisionada).
- Monitorar em tempo real: estou entendendo? estou apenas decorando? estou aplicando?
- Reflexão (self-reflection)
- O que funcionou? O que não funcionou?
- O problema foi falta de esforço, estratégia inadequada, falta de recurso ou algo fora do meu controle?
- O que vou manter, o que vou mudar no próximo ciclo?
Quem atribui tudo a “azar”, “caso difícil”, “erro do serviço” sai mais fraco do caso.
Quem atribui a fatores ajustáveis (estratégia, preparação, decisão) sai mais forte, mesmo depois de um erro.
Ferramentas práticas para residentes e cirurgiões
1. Autoavaliação de como você aprende
Não é apenas “sou bom ou ruim”, mas:
- eu planejo o que estudar ou vou “apagando incêndio”?
- eu mudo de estratégia quando não entendo um tema?
- eu reviso os casos difíceis depois do plantão?
- eu procuro ativamente feedback objetivo sobre minha performance?
Transformar isso em rotina escrita (um caderno, um app, uma planilha) ajuda a tirar a aprendizagem do improviso e colocá-la em modo profissional.
2. Coaching cirúrgico
Coaching não é “mais uma aula”; é uma conversa estruturada para:
- definir objetivos de melhoria claros (ex.: decisão de conversão; planejamento de colecistectomia difícil; comunicação com a equipe);
- identificar pontos cegos (o que você não está vendo sobre a própria prática);
- desenhar um plano concreto de treinamento e estudo.
Ferramentas de vídeo-coaching (assistir a uma operação sua com um colega experiente e revisar decisões, tempos e manobras) têm efeito duplo: refinam a técnica e amplificam a sua metacognição.
3. Leitura inteligente: saindo do “sublinhar tudo”
Um exemplo prático é a estratégia SQ3R para capítulos e diretrizes:
- Survey (percurso) – passar rapidamente pelos subtítulos, tabelas, figuras.
- Question (perguntas) – transformar subtítulos em perguntas (“quando indicar intervalo apendicectomia?”, “como manejar abscesso apendicular?”).
- Read (leitura) – ler com foco em responder suas próprias perguntas.
- Recall (recordar) – fechar o texto e anotar o que lembra de cabeça.
- Review (revisar) – conferir no texto e corrigir lacunas.
É mais trabalhoso do que reler passivamente, mas a retenção é muito maior — e é isso que interessa na prática cirúrgica.
Aplicando isso na formação cirúrgica
Na rotina de um serviço de cirurgia, aprendizagem autorregulada se traduz em ações muito concretas:
- Antes do plantão: definir 1–2 objetivos de aprendizado (por exemplo, “revisar escore de Alvarado e conduta em apendicite complicada”).
- Durante o plantão: escolher conscientemente 1–2 casos para estudar em profundidade depois.
- Após a cirurgia: registrar rapidamente:
- o que foi bem,
- o que não foi,
- o que você precisa estudar para a próxima situação semelhante.
No nível do serviço, vale estimular:
- discussão de M&M com foco em análise de processo, não apenas em “culpa”;
- preceptores que verbalizam seu raciocínio e seus próprios erros;
- metas claras por ano de residência (o que se espera que o R1, R2, R3 saiba de fato).
Pontos-chave para o cirurgião que quer ser lifelong learner
- Assuma o comando do próprio aprendizado – ninguém fará isso por você.
- Planeje o estudo como planeja uma cirurgia – com objetivo, estratégia e checagem.
- Use erro e desconforto como combustível, não como fonte de paralisia.
- Busque feedback específico, não elogios genéricos.
- Padronize suas estratégias de leitura e revisão, fuja do improviso.
Conclusão
Formar um cirurgião tecnicamente competente é obrigatório.
Formar um cirurgião capaz de continuar aprendendo, se adaptando e se avaliando ao longo da vida é o verdadeiro diferencial.
Lifelong learning não é um slogan bonito de documento institucional.
É uma competência prática, treinável, que começa na residência, mas precisa acompanhar cada decisão, cada leitura, cada caso difícil.
Quanto mais cedo você organizar o próprio processo de aprender, mais preparado estará para os desafios que ainda nem existem hoje – mas que certamente farão parte da cirurgia de amanhã.
Hashtags (SEO)
#LifelongLearningEmCirurgia #AprendizagemAutorregulada #FormaçãoDoCirurgião #EducaçãoMédicaContinuada #ResidênciaEmCirurgia
Gostou❔ Deixe seu comentário ✍️, compartilhe com seus colegas e mande sua dúvida pelo 💬 Chat On-line em nossa DM no Instagram.
COMPARAÇÃO ENTRE AS TÉCNICAS DE CORREÇÃO DA LESÃO INADVERTIDA DA VIA BILIAR: RAFIA PRIMÁRIA, COLEDOCODUODENOSTOMIA E HEPATICOJEJUNOSTOMIA
1. Introdução
A lesão inadvertida da via biliar (LVB) é a complicação mais grave da colecistectomia. A escolha da técnica reconstrutiva influencia diretamente:
- morbidade imediata,
- risco de estenose tardia,
- necessidade de reoperações,
- qualidade de vida do paciente.
As três técnicas clássicas de reparo são: rafia primária, coledocoduodenostomia (CDD) e hepaticojejunostomia (HJ). Cada uma tem indicações específicas, benefícios, riscos e performance diferente no longo prazo.
2. RAFIA PRIMÁRIA
1. Conceito
Sutura primária do ducto biliar lesado, podendo ou não incluir T-tube (Kehr).
2. Indicações Reais (muito restritas)
- Lesões pequenas (<3 mm).
- Lesões laterais (tipo Strasberg D).
- Lesões reconhecidas imediatamente, antes de isquemia ductal.
- Sem perda de ducto.
- Paciente estável e equipe experiente em HPB.
3. Vantagens
- Técnica simples, rápida.
- Preserva a anatomia original.
- Não exige anastomose complexa.
4. Desvantagens / Problemas Clássicos
- Alto risco de estenose tardia (20–40%).
- Risco de falha se há trauma térmico ou perda segmentar.
- Depende de tecido viável, o que raramente ocorre após clipagem ou eletrocoagulação.
- Necessita drenagem adequada (Kehr), aumento de morbidade.
5. Resultados na Literatura
- Bons resultados em lesões muito precoces e laterais.
- Em perdas ductais ou transecções completas, a falha é a regra.
→ Por isso, seu uso caiu drasticamente e é considerado excepcional.
3. COLEDOCODUODENOSTOMIA (CDD)
1. Conceito
Anastomose término-lateral entre ducto biliar comum e duodeno.
2. Indicações Clássicas
- Estenoses baixas do colédoco (tipos B ou C).
- Lesões distais de fácil acesso.
- Situações especiais: idade avançada, comorbidades, impossibilidade técnica de HJ, cirurgias prévias complexas.
3. Vantagens
- Técnica relativamente simples.
- Tempo cirúrgico menor que HJ.
- Acesso endoscópico pós-operatório facilitado.
4. Desvantagens
- Maior risco de refluxo enterobiliar.
- Colangite recorrente (10–25%).
- Maior incidência de estenose em comparação à HJ a longo prazo.
- Evitada em lesões altas (porta hepatis).
5. Resultados
- Boa taxa de permeabilidade inicial.
- Menor durabilidade em pacientes jovens.
- Taxa de reoperação superior à HJ em seguimento >5 anos.
→ Técnica válida em casos selecionados, mas não é padrão-ouro.
4. HEPATICOJEJUNOSTOMIA (HJ) – PADRÃO-OURO
1. Conceito
Anastomose término-lateral do ducto hepático (ou confluência) com alça jejunal em Roux-en-Y.
2. Indicações
- Transecções completas (Strasberg E1–E5).
- Lesões proximais ou com perda de ducto.
- Lesões tardias diagnosticadas em estenose.
- Reconstrução definitiva após falha de outros métodos.
- Lesão térmica ou isquemia ductal.
3. Vantagens
- Melhor durabilidade entre as técnicas.
- Menor risco de estenose tardia (5–15%).
- Melhor vascularização do ducto.
- Evita refluxo duodenal.
4. Desvantagens
- Cirurgia complexa, exige centro especializado.
- Risco de fístula biliar inicial.
- Acesso endoscópico pós-operatório difícil.
5. Resultados
- Padrão-ouro mundial em lesões significativas.
- Sucesso >85–90% em centros de referência.
- Menor risco de reoperação tardia.
→ É a técnica mais segura e duradoura para a maioria das lesões importantes.
5. TABELA COMPARATIVA
| Técnica | Indicação | Vantagens | Desvantagens | Êxito a longo prazo |
|---|---|---|---|---|
| Rafia primária | Lesões laterais pequenas, precoces | Preserva anatomia | Alta estenose | 60–80% |
| CDD | Lesões distais, baixa lesão | Técnica simples, acesso endoscópico fácil | Refluxo, colangite, mais estenose | 65–85% |
| HJ (Roux-en-Y) | Transecções, perdas ductais, lesões proximais | Melhor durabilidade e segurança | Cirurgia longa e complexa | 85–95% |
6. Avaliação crítica da literatura
- Timing importa mais que técnica
– Reconstrução precoce (<72h) tem melhores resultados se feita em centro especializado.
– Reconstruções tardias (6–12 semanas) podem reduzir edema e inflamação, facilitando HJ. - Presença de isquemia do ducto
– Torna a rafia primária praticamente contraindicada.
– Favorece HJ como opção definitiva. - Perícia cirúrgica
– Estudos multicêntricos mostram falha recorrente quando cirurgiões não HPB tentam reparo primário. - ODM (outcomes dependem do centro)
– HJ feita por especialistas tem estenose <10%.
– HJ feita em hospital não especializado chega a 30–40% de falha.
7. Conclusões práticas
1. RAFIA PRIMÁRIA
Reservada apenas para lesões pequenas, laterais e reconhecidas imediatamente. Deve ser exceção.
2. COLEDOCODUODENOSTOMIA
Útil em lesões baixas e pacientes idosos ou debilitados. Boa opção quando HJ é desproporcional ao risco.
3. HEPATICOJEJUNOSTOMIA – Padrão-ouro
Melhor técnica na imensa maioria das lesões graves. Deve ser realizada em centros de referência HPB.
Regra de ouro:
Transecção ductal = HJ em centro especializado.
QUANDO INDICAR A RADIOLOGIA INTERVENCIONISTA NAS LESÕES DAS VIAS BILIARES
1. Introdução
Lesões da via biliar (LVB) – sobretudo pós-colecistectomia, cirurgias hepáticas e transplante – são causas relevantes de morbidade. O manejo moderno é obrigatoriamente multidisciplinar, envolvendo cirurgia, endoscopia e radiologia intervencionista (RI).
A pergunta prática do cirurgião é:
“Em que momento eu devo chamar a radiologia intervencionista?”
A seguir, um guia objetivo, organizado por cenários clínicos.
2. Papel da Radiologia Intervencionista nas LVB
De forma geral, a RI é indicada para:
- Drenar: coleções biliares ou abscessos (biloma, coleções perihepáticas).
- Desobstruir: drenagem biliar percutânea (PTBD) em obstrução ou estenose.
- Modelar/Tratar estenoses: dilatação com balão e/ou stent em estenoses benignas ou neoplásicas.
- Tratar complicações vasculares associadas: pseudoaneurisma de artéria hepática, hemobilia, sangramento.
- Criar acesso para procedimentos combinados (rendezvous com endoscopia).
RI raramente é “primeira linha isolada”, mas é frequentemente essencial como ponte ou complemento de endoscopia/cirurgia.
3. Situações agudas: quando indicar RI?
3.1. Biloma e coleções biliares pós-operatórias
Indicar drenagem percutânea guiada por US/TC quando:
- Coleção sintomática (dor, febre, instabilidade).
- Coleção ≥ 3–5 cm ou progressiva em exames seriados.
- Sinais de sepse ou disfunção orgânica.
- Fístula biliar externa com grande débito associada a coleção interna.
Nesses casos, a drenagem percutânea:
- Controla sepse rapidamente.
- Permite análise do líquido (bile x pus).
- Facilita avaliação posterior por CPRE/PTBD.
3.2. Fístula biliar com anatomia difícil ou falha da CPRE
A CPRE é primeira linha para a maioria das fístulas.
A RI entra quando:
- CPRE falhou (papila inacessível, canulação impossível).
- Anatomia alterada (gastrectomia, bypass gástrico em Y-de-Roux, anastomose biliodigestiva alta).
- Fístula alta (hepático comum/proximal) com difícil acesso endoscópico.
Nesses casos, indica-se drenagem biliar percutânea trans-hepática (PTBD):
- Dreno interno–externo ou externo, desviando o fluxo e reduzindo pressão intraductal.
- Frequentemente associada à drenagem de coleções.
3.3. Hemobilia e pseudoaneurisma de artéria hepática
Lesões combinadas artério-biliares (p. ex. pós-clipagem, lesão térmica) podem cursar com:
- Hemobilia (TRIÁDE: dor abdominal, icterícia, hematêmese/melena).
- Sangramento intra-abdominal.
Indicação imediata de RI:
- Angiografia diagnóstica + embolização seletiva da artéria lesada.
- Evita reoperação em pedículo hepático inflamado e de alto risco.
Cirurgião deve suspeitar e acionar RI em:
- Sangramento digestivo tardio em paciente com LVB.
- Queda de Hb sem foco óbvio + drenagem biliar sanguinolenta.
4. Situações tardias: estenoses e reconstruções
4.1. Estenoses benignas pós-cirúrgicas (hepáticojejunostomia / LVB reparada)
A RI é especialmente útil em pacientes com anatomia não acessível por CPRE:
- Hepáticojejunostomia em Y-de-Roux.
- Reconstruções biliares altas.
Indicações típicas:
- Icterícia colestática / colangite tardia.
- Estenose diagnosticada em colangiorressonância ou PTC.
Técnicas:
- Acesso percutâneo ao sistema biliar.
- Dilatação com balão da estenose.
- Posicionamento de cateteres de calibração ou stents.
- Sessões seriadas ao longo de meses.
Em estritos pós-hepáticojejunostomia, a combinação RI + cirurgia é muitas vezes a melhor estratégia (tratamento híbrido em casos recidivantes).
4.2. Obstrução biliar maligna (colangiocarcinoma, metástases hiliares, pâncreas)
Embora aqui não haja “lesão iatrogênica”, na prática HPB o raciocínio é o mesmo:
Indicar PTBD quando:
- ERCP falhou ou não é possível.
- Obstrução hilar complexa (Bismuth III/IV).
- Necessidade de drenagem seletiva de segmentos específicos antes de grande hepatectomia.
É uma área clássica da RI, mas o cirurgião deve lembrar que lesão biliar complexa iatrogênica com estenose alta se comporta de forma semelhante.
5. Lesões de via biliar complexas: como integrar cirurgia, endoscopia e RI?
5.1. Lesão parcial do ducto biliar comum com fístula (Strasberg D)
- Primeira linha: CPRE + stent.
- Se CPRE falha → PTBD + drenagem de coleções por RI.
- Reconstrução cirúrgica planejada se não houver fechamento após suporte combinado.
5.2. Secção completa do ducto com perda de segmento (Strasberg E)
- RI tem função adjuvante, não curativa.
- Indicações:
- Drenagem de biloma/coleções.
- PTBD para controle de icterícia e sepse.
- Objetivo: estabilizar o paciente e planejar hepáticojejunostomia definitiva em centro de referência.
5.3. Fístulas de pequeno ducto (Luschka, coto cístico) com biloma
- Drenagem percutânea se coleção significativa.
- CPRE para reduzir pressão e favorecer cicatrização.
- RI é especialmente útil se o paciente estiver séptico ou longe de centro endoscópico.
6. Critérios práticos para o cirurgião saber “quando chamar a RI”
Em linguagem direta:
Chame Radiologia Intervencionista quando houver:
- Coleção intra-abdominal (biloma/abscesso) que:
- seja volumosa, sintomática ou séptica;
- não possa ser drenada cirurgicamente com baixo risco.
- Falha ou impossibilidade de CPRE em:
- fístulas biliares;
- obstrução/estenose biliar;
- anatomia alterada (Y-de-Roux, gastrectomia).
- Obstrução ou estenose alta (hilar, hepático comum proximal) onde:
- a drenagem seletiva de segmentos é desejada;
- o acesso endoscópico é tecnicamente desfavorável.
- Suspeita de complicação vascular associada à LVB:
- hemobilia, pseudoaneurisma de artéria hepática, sangramento sem foco claro.
- Paciente de alto risco cirúrgico, em que:
- a drenagem percutânea (biloma ou biliar) pode estabilizar o quadro até uma reconstrução eletiva.
7. Conclusão
A radiologia intervencionista é hoje pilar central no manejo das lesões de via biliar, especialmente:
- como ponte segura entre o evento agudo e a reconstrução definitiva;
- como alternativa em anatomias complexas ou inacessíveis à endoscopia;
- como ferramenta essencial no tratamento de complicações vasculares associadas.
Para o cirurgião HPB e digestivo, o ponto-chave não é “se” deve envolver a RI, mas em que momento:
Quanto mais precoce a integração cirurgião–endoscopista–radiologista, menor a chance de reoperações desnecessárias, falhas terapêuticas e perda definitiva da via biliar.
Hashtags (SEO)
#LesãoDeViaBiliar #RadiologiaIntervencionista #CirurgiaHepatobiliar #Biloma #DrenagemPercutanea
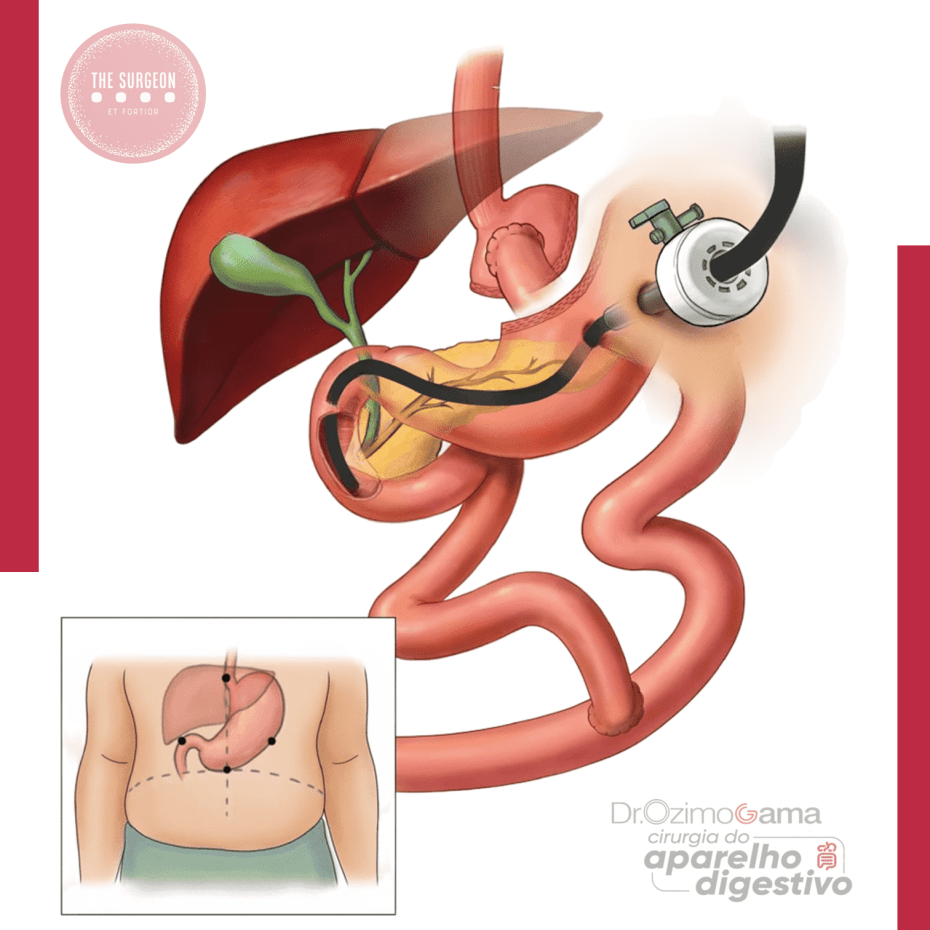
TRATAMENTO ENDOSCÓPICO DAS FÍSTULAS BILIARES PÓS-COLECISTECTOMIA: RESULTADOS, TÉCNICAS E AVALIAÇÃO CRÍTICA DA LITERATURA
Introdução
As fístulas biliares são uma complicação relativamente comum após colecistectomia, sobretudo nos casos difíceis, inflamatórios ou com lesão térmica inadvertida no ducto cístico. Embora a maioria das fístulas provenientes do coto cístico ou de pequenos ramos do ducto hepático direito sejam de baixo débito, seu manejo inadequado pode evoluir para sepse, coleções biliares ou necessidade de reoperação.
O tratamento endoscópico por CPRE (colangiopancreatografia retrógrada endoscópica) tornou-se o padrão-ouro para o manejo inicial dessas fístulas, com elevadas taxas de sucesso, baixa morbidade e impacto significativo na resolução precoce das complicações.
Este artigo revisa criticamente as técnicas endoscópicas disponíveis, seus resultados e as principais evidências da literatura.
Classificação das Fístulas Biliares Pós-Colecistectomia
As fístulas podem se originar de:
- Coto do ducto cístico – mais comum e com excelente resposta à CPRE.
- Ducto hepático direito acessório (ducto de Luschka).
- Lesão térmica do ducto hepático comum ou ducto biliar comum (DBC) – frequentemente associada a estenose.
- Lesões de alto débito (>300 mL/dia) – pior prognóstico, maior chance de lesão maior.
As fístulas tipo I (bile leak leve) respondem quase sempre ao tratamento endoscópico.
As fístulas tipo II, III e IV (maior lesão ductal) podem necessitar cirurgia reconstrutiva.
Racional do Tratamento Endoscópico
A CPRE atua em dois pilares fisiopatológicos:
1. Redução da pressão no ducto biliar
- Realizada por esfinterotomia e/ou stents biliares.
- Diminui o fluxo de bile para o local da fístula, favorecendo cicatrização.
2. Desvio do fluxo biliar
- O stent oferece um caminho de menor resistência.
- A fístula deixa de ser a via preferencial de drenagem.
Com isso, >90% das fístulas simples fecham em poucos dias.
Técnicas Endoscópicas
1. Esfinterotomia isolada
- Indicações: fístulas leves, baixo débito.
- Vantagem: técnica simples, baixo custo.
- Limitação: menor eficácia comparada ao uso de stent.
Taxas de sucesso relatadas: 70–85%.
2. Colocação de stent biliar plástico
- Técnica mais utilizada.
- Stents de 7 a 10 Fr, posicionados transpapilares.
- Associado ou não à esfinterotomia.
Taxas de sucesso: 85–95%
(Fechamento geralmente em 3–6 semanas.)
3. Stents totalmente cobertos (SEMS)
- Indicações:
- Fístulas persistentes após stent plástico.
- Estenose concomitante.
- Lesão térmica do hepático comum.
Taxas de sucesso: 90–100% em séries selecionadas.
Limitações: custo e risco de migração.
4. Retirada de cálculos associados
- Coledocolitíase residual pode perpetuar pressão intraductal.
- Papilotomia + extração de cálculos é essencial nesses casos.
5. Drenagem guiada por EUS
- Usada em casos complexos, falha da CPRE ou anatomia difícil.
- Promissora, mas ainda com dados limitados.
Resultados da Literatura
A maior parte dos estudos é formada por séries retrospectivas, mas os achados são consistentes:
• Sucesso global da CPRE no manejo de fístulas pós-colecistectomia: 85–95%
(meta-análises mostram média entre 88–92%)
• Tempo médio para fechamento da fístula: 2 a 6 semanas
• Principais fatores associados à falha:
- Estenose concomitante não tratada
- Lesão mais proximal (lesões tipo C/D de Strasberg)
- Fístulas de alto débito com lesão ductal maior
- Perfurações térmicas extensas
Complicações da CPRE
- Pancreatite pós-CPRE: 5–10%
- Sangramento: 1–3%
- Perfuração: <1%
Apesar disso, o risco é muito inferior ao de uma reoperação precoce.
Avaliação Crítica da Evidência
1. Estudos predominantemente retrospectivos
- Faltam ensaios clínicos randomizados comparando métodos endoscópicos.
- A heterogeneidade das classificações dificulta comparações.
2. Ausência de padronização universal
- Alguns centros utilizam stent + esfinterotomia como rotina.
- Outros reservam stent para casos complexos.
3. Papel crescente dos SEMS cobertos
- Cada vez mais estudados, principalmente para fístulas persistentes.
- Necessidade de mais estudos comparativos.
4. O dilema das lesões complexas
- Lesões de alto grau (como BDI tipo E de Strasberg) não devem ser tratadas exclusivamente por via endoscópica.
- A CPRE funciona como ponte para cirurgia de reconstrução (hepáticojejunostomia).
5. Importância da drenagem percutânea combinada
- Em casos com coleções biliares, a drenagem percutânea associada à CPRE acelera a resolução.
Quando o tratamento endoscópico falha?
A falha deve ser suspeitada quando:
- Vazamento persiste por >8 semanas.
- Débito permanece alto apesar do stent.
- Haja estenose proximal não tratada.
- Evidência de lesão de via biliar maior.
Nesses casos, a cirurgia reconstrutiva (hepáticojejunostomia) deve ser indicada.
Conclusão
A CPRE é o tratamento de primeira linha para as fístulas biliares pós-colecistectomia, oferecendo taxas de sucesso superiores a 85–95% nas fístulas simples e bom desempenho mesmo em cenários moderadamente complexos.
A literatura demonstra de forma consistente que:
- A endoscopia reduz morbidade
- Evita reoperações desnecessárias
- Promove fechamento rápido da fístula
- É custo-efetiva e amplamente disponível
No entanto, a interpretação crítica mostra que ainda faltam estudos prospectivos e padronização universal das técnicas. Para fístulas complexas ou quando associadas a estenose ductal significativa, a abordagem multidisciplinar é essencial.
A integração entre endoscopia, radiologia intervencionista e cirurgia HPB continua sendo a chave para reduzir complicações e preservar a via biliar.
Hashtags (SEO)
#FistulaBiliar #CPRE #CirurgiaDigestiva #Colecistectomia #LesãoDeViaBiliar
INTELIGÊNCIA ARTIFICIAL NA COLECISTECTOMIA LAPAROSCÓPICA: A NOVA FRONTEIRA DA SEGURANÇA CIRÚRGICA
A colecistectomia laparoscópica (LC) é um dos procedimentos mais realizados no mundo, mas ainda carrega um risco significativo de lesão de via biliar (BDI – bile duct injury), especialmente em situações inflamatórias ou anatômicas complexas. Nas últimas décadas, o Critical View of Safety (CVS) se consolidou como o padrão-ouro para evitar BDI, porém estudos mostram que sua identificação é altamente subjetiva e frequentemente superestimada pelos cirurgiões.
A inteligência artificial (IA), especialmente por meio de deep learning e visão computacional aplicada a vídeos cirúrgicos, surge como uma ferramenta promissora para padronizar, reconhecer e validar o CVS em tempo real, além de mapear estruturas anatômicas e zonas seguras e de risco durante a LC.
Este artigo resume de forma clara e aplicada o estado da arte da IA na colecistectomia.
IA e o Critical View of Safety (CVS)
Mesmo com o uso disseminado do CVS, a taxa de BDI permanece maior na LC do que na técnica aberta. Estudos revelam um dado alarmante: a maioria dos cirurgiões acredita ter alcançado o CVS, mas uma análise independente mostra o oposto.
- Em um estudo com 1108 LCs, cirurgiões relataram CVS em 80% dos casos.
- Revisores externos encontraram CVS verdadeiro em apenas 10,8%.
- Em todos os casos com BDI, o CVS não havia sido obtido.
A IA foi então proposta como forma de reduzir a subjetividade e melhorar a precisão do reconhecimento anatômico.
Modelos de IA para reconhecimento do CVS
Os primeiros modelos usaram milhares de imagens anotadas por cirurgiões experientes:
- Um deep neural network treinado com 201 vídeos e 2854 imagens alcançou:
- Acurácia: 71,9%
- Precisão: 71,4%
Outro estudo mais recente, com mais de 71 mil imagens, alcançou:
- Precisão: 0,97
- Acurácia: 0,83
Resultados muito superiores aos obtidos por humanos revisando apenas imagens estáticas.
IA para áreas seguras e zonas de risco (Go/No-Go)
O modelo GoNoGoNet, um dos mais influentes, usou semantic segmentation para marcar:
- Go zone — área segura de dissecção
- No-Go zone — região perigosa, associada a BDI
Resultados:
- Acurácia superior a 90% na maioria das estruturas.
- Cirurgiões que avaliaram vídeos com o suporte da IA corrigiram suas anotações em até 26,9%, sendo 70% correções que aumentavam a segurança.
Estudos subsequentes mostraram que:
- Vídeos com BDI tinham 33,6% mais interações em zonas No-Go.
- A IA demonstrou maior sensibilidade para regiões de risco do que o olho humano.
IA para reconhecimento de estruturas anatômicas
Modelos recentes conseguem identificar automaticamente:
- ducto cístico
- ducto biliar extra-hepático
- sulco de Rouvière
- margem inferior do segmento 4 (S4)
Esses pontos são cruciais na prevenção de BDI.
Em um estudo experimental:
- Cirurgiões mudaram sua interpretação anatômica em 25–30% dos casos após ver o vídeo anotado pela IA.
- A maioria considerou as correções mais seguras.
Em outro estudo, o modelo YOLOv3 foi integrado ao sistema laparoscópico da sala cirúrgica:
- Latência de apenas 0,09 s
- Reconhecimento de estruturas em 92% das etapas da LC
Esse é o primeiro passo real para um “copiloto cirúrgico” intraoperatório.
IA na identificação das fases cirúrgicas
Modelos baseados em redes temporais (MS-TCN, ResNet50, Cholec80) alcançaram:
- Acurácia de 78% a 91% na identificação das fases da LC
- Melhor desempenho nas etapas críticas:
- dissecção do triângulo de Calot
- clipagem
- liberação do leito da vesícula
Entretanto, eventos adversos diminuem o desempenho:
- Sem complicações: ~90%
- Com perfuração da vesícula: 87%
- Com grande vazamento biliar: 77%
Limitações atuais da IA na LC
As principais barreiras identificadas nos estudos:
1. Anotações humanas inconsistentes
• Pequeno número de cirurgiões anotadores
• Variabilidade inter-observador
• Falta de padronização global
2. Datasets pequenos e monocêntricos
• Reduz a generalização dos modelos
• Dificulta adaptação para diferentes câmeras, óticas e técnicas
3. Dificuldade com casos complexos
• Inflamação severa
• Cirurgias com fibrose, gordura densa ou sangramento
• Mudança de ângulo da ótica
4. Desafios práticos no intraoperatório
• Flicker de ROIs
• Distração visual
• Confiabilidade limitada em visão ampliada ou com fumaça
5. Falta de integração nativa
Apenas um estudo conectou a IA diretamente ao sistema laparoscópico (EndoALPHA).
Perspectivas Futuras
- Sistemas dual-AI integrados ao console laparoscópico
– Reconhecimento simultâneo de estruturas + fases cirúrgicas. - ROI dinâmico com “tiles” inteligentes, reduzindo flicker e adaptando-se a limites anatômicos.
- Combinação IA + ICG, ampliando reconhecimento além das capacidades isoladas de cada método.
- Incorporação ao treinamento cirúrgico e simulação, especialmente para residentes.
- Criação de guideline global para anotação, validação e uso clínico da IA.
- Suporte intraoperatório em tempo real, semelhante a um copiloto de aviação.
Conclusão
A inteligência artificial aplicada à colecistectomia laparoscópica está avançando rapidamente, com resultados promissores para:
- melhorar a identificação anatômica
- reforçar zonas de segurança
- padronizar o Critical View of Safety
- apoiar o treinamento e reduzir o risco de lesões de via biliar
Embora nenhum sistema esteja pronto para substituição completa do julgamento cirúrgico, o conjunto de evidências sugere que a IA será, em breve, parte essencial da estratégia global para aumentar a segurança da LC.
Como toda tecnologia emergente, o sucesso dependerá de:
- validação multicêntrica
- integração prática ao workflow operatório
- aceitação e liderança dos cirurgiões
A IA não substituirá o cirurgião.
Mas cirurgiões que usam IA substituirão os que não usam.
INTELIGÊNCIA ARTIFICIAL E CIRURGIA: O QUE O CIRURGIÃO PRECISA SABER HOJE
Introdução
A inteligência artificial (IA) deixou de ser um conceito futurista e entrou definitivamente na prática cirúrgica. Longe das versões “fortes” retratadas em filmes, as aplicações atuais são formas de IA estreita, projetadas para executar tarefas específicas com elevado desempenho. Na cirurgia, essas ferramentas já atuam na estratificação de risco, análise intraoperatória, previsão de desfechos e processamento de prontuários, com evidências crescentes de impacto na segurança, eficiência e precisão das decisões clínicas.
Este artigo apresenta, de forma didática e objetiva, as principais aplicações da IA ao ato cirúrgico moderno, suas limitações e o papel do cirurgião nessa transição tecnológica.
1. Fundamentos: o que de fato é Inteligência Artificial?
A IA é o campo que estuda algoritmos capazes de executar funções cognitivas. É composta por subáreas, sendo as mais relevantes para a cirurgia:
1.1 Machine Learning (ML)
Sistemas que aprendem padrões a partir de dados estruturados.
• Supervisionado: aprende com dados previamente rotulados.
• Não supervisionado: identifica padrões sem rótulos.
• Reforço: aprendizado por tentativa e erro.
1.2 Redes Neurais e Deep Learning
Capazes de extrair automaticamente características de dados complexos como imagens e vídeos.
• Especialmente úteis em laparoscopia, endoscopia e robótica.
1.3 Computer Vision (CV)
Permite que máquinas “vejam” e interpretem imagens cirúrgicas.
• Base da análise intraoperatória em tempo real.
1.4 Processamento de Linguagem Natural (NLP)
Interpreta texto livre, facilitando mineração de prontuários, relatórios operatórios e predição de risco.
2. Aplicações atuais na cirurgia
2.1 Predição pré-operatória de risco
Ferramentas como POTTER, MySurgeryRisk e modelos de aprendizado profundo superam calculadoras tradicionais (ASA, RCRI, NSQIP), oferecendo:
• Previsão personalizada de mortalidade e complicações.
• Integração em tempo real com prontuários eletrônicos.
• Aprendizado contínuo com novos dados.
Esses modelos já apresentam C-statistics superiores às ferramentas tradicionais, mostrando melhor acurácia na previsão de desfechos.
2.2 Análise intraoperatória por vídeo
Uma das fronteiras mais promissoras. A IA já consegue:
• Reconhecer fases cirúrgicas com >85% de acurácia em colecistectomias, gastrectomias e colectomias.
• Identificar estruturas anatômicas críticas.
• Estimar tempo cirúrgico restante.
• Detectar “near misses” e eventos de risco antes que causem dano.
Estudos mostram que até 70% das cirurgias apresentam um “near miss”. A IA pode atuar como um “copiloto” cirúrgico, alertando o cirurgião nesses momentos.
2.3 Suporte ao treinamento e avaliação de habilidades
A análise automatizada de vídeo já permite:
• Avaliar habilidades com base em padrões de movimento.
• Comparar a performance do cirurgião com especialistas (“global surgical consciousness”).
• Reduzir subjetividade na avaliação de residentes.
2.4 Integração com prontuário eletrônico (EMR)
Com NLP, a IA pode:
• Detectar infecções precocemente.
• Extrair automaticamente complicações de textos livres.
• Identificar pacientes de alto risco.
• Correlacionar decisões intraoperatórias a desfechos.
3. Limitações e riscos
Apesar do entusiasmo, a IA em cirurgia ainda é limitada:
• Modelos podem superajustar (overfitting) e não generalizar bem.
• Dados cirúrgicos são altamente variáveis e ruidosos.
• Lacunas éticas e jurídicas permanecem (privacidade de vídeo, responsabilidade por erros).
• Necessidade de padronização: cirurgiões diferem até na definição de fases cirúrgicas.
Portanto, o uso clínico deve ser crítico, criterioso e apoiado em validação robusta.
4. O papel do cirurgião na era da IA
A introdução da IA não substituirá o cirurgião — mas exigirá um profissional capaz de integrar tecnologia com julgamento clínico. O cirurgião deve:
• Entender princípios básicos de IA.
• Conhecer limitações dos modelos.
• Participar da criação de bases de dados cirúrgicas.
• Atuar na validação clínica de algoritmos.
• Liderar discussões éticas e regulatórias.
Como afirmam Hashimoto e col. (2020), a participação ativa da comunidade cirúrgica é essencial para garantir interoperabilidade, segurança e sustentabilidade dessas tecnologias.
Conclusão
A IA está transformando a cirurgia em múltiplas frentes — do risco pré-operatório ao suporte intraoperatório e análise pós-operatória. A integração com sistemas de visão computacional, prontuários eletrônicos e mecanismos de aprendizado contínuo aponta para um futuro onde o cirurgião terá ferramentas mais precisas para decisões mais seguras.
O desafio atual não é se a IA entrará no centro cirúrgico, mas como garantiremos que essa entrada ocorra de forma ética, segura e cientificamente validada.
CUSTO E VALOR NA HERNIOPLASTIA INGUINAL: O QUE REALMENTE IMPORTA PARA O PACIENTE E PARA O SISTEMA DE SAÚDE
Introdução
A discussão sobre “qual é a melhor técnica de hernioplastia inguinal” não pode ser feita apenas com base em recidiva ou complicações.
No cenário atual de restrição de recursos e busca por eficiência, o valor cirúrgico importa tanto quanto o desfecho clínico.
Segundo o modelo clássico de Michael Porter, Valor = Qualidade / Custo.
Quando os resultados clínicos entre técnicas são equivalentes, quem determina o valor final é o custo total envolvido — variável e fixo.
O estudo analisado comparou 100 procedimentos consecutivos em cada modalidade:
- Hernioplastia aberta
- Laparoscópica
- Robótica
A seguir, o que realmente importa quando comparamos custo, eficiência e valor em cada abordagem.
1. Entendendo Custos: Variáveis × Fixos
1.1. Custos variáveis (por caso)
Incluem:
- Materiais descartáveis
- Mão de obra direta
- Overhead variável por procedimento
Resultados:
- Laparoscópica: 1,02× o custo da aberta (diferença não significativa).
- Robótica: 2,11× o custo da aberta e 2,06× o custo da laparoscópica (P < 0,001).
Ou seja: a laparoscopia custa praticamente o mesmo que a aberta, mas a robótica mais que dobra o custo.
1.2. Custos fixos (capital + manutenção)
Incluem:
- Torre laparoscópica
- Robô cirúrgico
- Contratos de manutenção
- Amortização por caso
Quando esses custos entram na conta:
- Laparoscópica: 1,03× o custo da aberta
- Robótica: 3,18× o custo da aberta
A robótica triplica o custo total da operação.
2. O Impacto no Valor Cirúrgico
Se assumirmos que as taxas de recidiva são equivalentes (o que os dados atuais sustentam para as três técnicas), então:
- Laparoscopia → reduz o valor do procedimento em apenas 2–3%, o que é compensado por:
- menos dor
- retorno mais rápido às atividades
- melhor satisfação do paciente
Com isso, a laparoscopia entrega mais valor ao paciente do que a via aberta.
- Robótica → reduz o valor em:
- 53% quando analisado apenas custo variável
- 69% quando incluídos custo fixo + manutenção
O principal fator destruidor de valor é o custo incremental da tecnologia, sem ganho proporcional em desfecho.
3. O Que Isso Significa na Prática?
A. Hernioplastia aberta
- Custo menor
– Pior recuperação e mais dor em muitos casos
= Valor intermediário
B. Hernioplastia laparoscópica
- Mesma taxa de recidiva
- Melhores resultados relatados pelo paciente
- Custos semelhantes à aberta
= Maior valor
C. Hernioplastia robótica
- Conforto operatório (para o cirurgião)
- Ergonomia superior
– Sem benefícios clínicos comprovados ao paciente
– 3× mais cara
= Menor valor global
4. Conclusão: Qual Técnica Oferece Mais Valor?
Com base na melhor evidência disponível:
- A laparoscopia é a técnica com melhor relação custo-benefício, entregando mais valor ao paciente.
- A via aberta permanece adequada em cenários de baixo recurso ou limitação técnica.
- A robótica, apesar de tecnologicamente avançada, não aumenta o valor cirúrgico, pois aumenta custos sem melhorar resultados.
Em uma era de medicina baseada em valor, mais tecnologia não significa melhor cuidado — especialmente quando o resultado final esperado (baixa recidiva) é o mesmo.
Hashtags (SEO)
#HerniaInguinal #CirurgiaDigestiva #Laparoscopia #CirurgiaRobótica #MedicinaBaseadaEmValor
BAILOUT MANEUVERS NA COLECISTECTOMIA LAPAROSCÓPICA: O QUE FAZER QUANDO A DISSECÇÃO SE TORNA PERIGOSA
Introdução
Embora a colecistectomia laparoscópica seja um procedimento rotineiro e de baixo risco, a chamada “vesícula difícil” representa um dos cenários mais desafiadores da cirurgia digestiva. Inflamação intensa, distorção anatômica e aderências densas podem transformar um procedimento simples em uma operação com potencial de causar lesões biliares e vasculares graves, com impacto direto na morbimortalidade do paciente.
Diante desse cenário, é essencial que todo cirurgião domine as Bailout Maneuvers — manobras de resgate seguras e bem estabelecidas para interromper a progressão de uma dissecção perigosa e evitar complicações catastróficas.
Reconhecendo o Momento de Parar: “Red Flags” Intraoperatórios
Muitas lesões graves ocorrem não por falta de habilidade técnica, mas por falha na percepção do risco. Alguns sinais de alerta devem fazer o cirurgião pausar, reavaliar e reorientar a dissecção:
- Artéria “cística” incomumente calibrosa (possível artéria hepática).
- Pulsação intensa atrás do suposto ducto cístico (pode ser ducto hepático comum).
- Clip grande incapaz de ocluir o ducto (sugere lesão do DBC ou DHC).
- Presença de bile no campo sem perfuração da vesícula.
Perceber esses red flags pode evitar até 18% das lesões vasculares e biliares relatadas em séries grandes.
Quando o Critical View of Safety Não é Possível
Há situações em que a inflamação é tão intensa que não existe plano anatômico seguro para liberar o Triângulo de Calot. Nesses casos, insistir é o caminho mais rápido para uma lesão maior.
O cirurgião deve reconhecer o limite técnico e aplicar uma manobra de resgate apropriada.
Principais Bailout Maneuvers
1. Abortar o Procedimento
A decisão mais segura em casos extremos:
- Campo tomado por fibrose, necrose ou tecido friável.
- Vesícula não identificável.
- Distinção anatômica impossível mesmo com auxílio.
Condutas subsequentes:
- Manter antibióticos.
- Considerar colecistostomia percutânea.
- Planejar colecistectomia intervalar após 8–12 semanas.
Apesar de não resolver a doença naquele momento, salva o paciente de lesões irreversíveis.
2. Fundus-First Cholecystectomy (Técnica Fundo-Primeiro)
Indicada quando o Calot é impenetrável.
Técnica:
- Iniciar a dissecção no fundo da vesícula.
- Avançar cuidadosamente em direção ao infundíbulo.
- Manter o plano “na parede da vesícula” para evitar o pedículo portal.
Pontos de atenção:
- Em inflamação severa, o cystic plate pode estar espessado e encurtado.
- A proximidade com o pedículo hepático aumenta conforme a inflamação destrói planos naturais.
Estudos mostram que a técnica fundo-primeiro reduz a necessidade de subtotal e pode diminuir o tempo de internação.
3. Subtotal Cholecystectomy (Colecistectomia Subtotal)
Opção preferível quando a anatomia permanece obscura após fundus-first.
Técnica geral:
- Abrir a vesícula.
- Aspirar conteúdo.
- Ressecar o máximo da parede possível.
- Ablacionar a mucosa remanescente.
Existem duas variantes:
A. Reconstituting (Reconstituinte)
- O remanescente é fechado com sutura.
- Menor risco de fístula biliar.
- Maior risco de vesícula residual sintomática.
B. Fenestrating (Fenestrante)
- O remanescente permanece aberto.
- Maior chance de fístula, mas geralmente autolimitada.
- Menor risco de doença residual.
Fechamento do cístico, quando indicado, deve ser interno, pois o fechamento externo pode ocluir o DHC/DBC.
4. Conversão para Cirurgia Aberta
Historicamente considerada a principal manobra de resgate, hoje sabemos que:
- A conversão não garante segurança.
- O risco de lesão biliar aumenta até 100 vezes em conversões não planejadas (NSQIP).
- A anatomia pode permanecer obscura mesmo no campo aberto.
Ainda é válida quando:
- Há sangramento importante.
- Há perda completa dos planos anatômicos.
- Há necessidade de controle manual do pedículo hepático.
Entretanto, em muitos casos, uma subtotal laparoscópica é mais segura do que uma conversão difícil.
Como Escolher a Manobra Correta?
A lógica de decisão deve considerar:
- Visibilidade anatômica (CVS alcançável ou não).
- Grau de inflamação.
- Experiência do cirurgião.
- Risco iminente de lesão maior.
- Disponibilidade de outro cirurgião para segunda opinião.
Regra prática:
Se você não consegue obter o Critical View of Safety e não está absolutamente certo do que está dissecando, é hora de um bailout.
Conclusão
A vesícula difícil exige maturidade cirúrgica, capacidade de reconhecer limites e domínio das manobras de resgate. Insistir em uma dissecção perigosa é receita para desastre. Saber abandonar, retroceder ou mudar a estratégia é parte fundamental da segurança do paciente e da preservação da via biliar.
A Evolução dos Traumas de Guerra: De Termópilas aos Drones na Ucrânia
A progressão dos traumas de guerra espelha a interseção entre inovações bélicas, avanços médicos e sistemas logísticos. Desde a Batalha de Termópilas em 480 a.C., símbolo da guerra organizada antiga, até o conflito atual na Ucrânia, os ferimentos evoluíram de penetrações de baixa energia para explosões complexas de alta energia. Este artigo examina a biomecânica dos traumas – como forças mecânicas interagem com tecidos humanos –, junto com causas de morte, taxas de mortalidade e medidas de tratamento ao longo de períodos históricos chave. Baseado em análises históricas, registros médicos militares e relatórios recentes, destaca uma tendência de redução na mortalidade por meio de inovações, mas temperada por ameaças emergentes como a tecnologia de drones.
A ERA ANTIGA: TERMÓPILAS E FERIMENTOS PRIMITIVOS
Nas guerras antigas, como as Greco-Persas incluindo Termópilas, o combate dependia de armas corpo a corpo como lanças, espadas, flechas e pedras. Biomecanicamente, esses causavam ferimentos penetrantes de baixa velocidade, com transferência de energia cinética limitada a lacerações diretas e cavitação ao longo do trajeto do projétil. Por exemplo, golpes de lança criavam cavidades permanentes no peito ou abdômen, lesando órgãos vitais com expansão temporária mínima devido à baixa velocidade (geralmente abaixo de 300 m/s). Forças contundentes de pedras ou escudos provocavam fraturas compressivas ou tórax instável, onde segmentos de costelas se soltavam, prejudicando a respiração por movimento paradoxal.
As causas de morte eram predominantemente falha orgânica imediata ou exsanguinação por rupturas vasculares, com infecções secundárias agravando os sobreviventes. Proxies históricos, como descrições na Ilíada de Homero (ligada a contextos semelhantes à Guerra de Troia), indicam que 63% dos traumas torácicos resultavam de lanças, levando a letalidade rápida por hemotórax ou pneumotórax. Taxas de mortalidade para ferimentos torácicos graves aproximavam-se de 70%, com 89% classificados como médios a graves; fatalidades gerais no campo de batalha eram altas, pois muitos feridos ficavam sem tratamento em meio ao combate.
Os tratamentos eram primitivos, dependendo de aplicações herbais, banhos quentes e ataduras básicas por figuras como Machaon nos textos antigos. Princípios hipocráticos enfatizavam a drenagem de pus, mas sem antissepsia, a gangrena era comum. Não havia evacuação organizada, com cuidados limitados a guerreiros de elite, destacando a alta letalidade da era por biomecânica não gerenciada, como sangramento descontrolado.
PRIMEIRA GUERRA MUNDIAL: A DESTRUIÇÃO INDUSTRIALIZADA
A Primeira Guerra Mundial (1914-1918) introduziu a destruição mecanizada por metralhadoras, projéteis de artilharia e rifles de alta velocidade (acima de 600 m/s). Biomecanicamente, esses criavam danos teciduais extensos por projéteis instáveis e fragmentação, formando grandes cavidades temporárias (até 10-15 cm de diâmetro) que esticavam tecidos elásticos como músculos, causando necrose tardia, enquanto órgãos inelásticos como o fígado se estilhaçavam. Ondas de explosão de projéteis adicionavam barotrauma primário, comprimindo estruturas cheias de ar como pulmões e levando a contusões pulmonares.
Infecções e doenças superavam ferimentos de combate como causas de morte, com 63.000 fatalidades por doenças nos EUA versus 51.000 por batalhas. Hemorragia e choque de fraturas compostas eram principais matadores relacionados a ferimentos, exacerbados pela contaminação das trincheiras. A mortalidade por ferimentos de batalha era de aproximadamente 21% (53.402 mortes de 257.404 casos), com fraturas de fêmur inicialmente em 80% de fatalidade antes de intervenções. Doenças como tifo e gripe ceifaram milhões globalmente, alterando a proporção doença-para-batalha para 2:1 em algumas forças.
Os tratamentos avançaram com sistemas de triagem priorizando cuidados salvadores, desenvolvidos por médicos franceses. Estações de Limpeza de Feridos forneciam debridamento, irrigação com solução Carrel-Dakin (à base de hipoclorito) e transfusões de sangue iniciais (introduzidas em 1916, embora arriscadas). Taxas de amputação eram de 12%, com mortalidade caindo para 5% pós-procedimento via técnicas estéreis e talas como o método Thomas. Evacuação por ambulâncias motorizadas reduziu atrasos, marcando uma virada do cuidado ad hoc antigo.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Antiga (Termópilas) | Penetração de baixa velocidade; laceração direta | Falha orgânica, infecção | ~70% para torácicos graves | Remédios herbais, ataduras |
| Primeira Guerra Mundial | Cavitação de alta velocidade; barotrauma por explosão | Infecção, doença, hemorragia | 21% geral por ferimentos | Debridamento, irrigação, transfusões iniciais |
SEGUNDA GUERRA MUNDIAL: REFINAMENTOS EM EVACUAÇÃO E ANTIMICROBIANOS
A Segunda Guerra Mundial (1939-1945) viu uso contínuo de explosivos e armas de fogo, com biomecânica enfatizando ferimentos secundários por fragmentos causando politrauma (ex.: 70-75% envolvimento de extremidades). Cargas moldadas em armas antitanque produziam jatos de alta velocidade, levando a spallation (fragmentação interna) e inalação de fumos tóxicos. Traumas contundentes terciários por deslocamentos adicionavam fraturas e síndrome compartimental.
Hemorragia e infecção permaneciam causas primárias, mas mortalidade por doenças caiu para uma proporção de 0,1:1 com vacinas. Mortalidade geral por ferimentos era de 3,3%, com abdominais em 21%. Queimaduras de veículos blindados afetavam 35% das baixas em contextos navais, complicando com lesões inalatórias.
Tratamentos incluíam fechamento primário atrasado (sucesso de 93%), debridamento e penicilina (produzida em massa até 1945), cortando taxas de infecção. Transfusões de sangue e plasma tornaram-se rotineiras, com envios aéreos garantindo suprimento. Cadeias de evacuação – de postos de socorro a hospitais de campo – levavam em média 12-15 horas, usando amputações circulares e gaze com vaselina. Sobrevivência para feridos atingiu 50%, acima dos 4% da Primeira Guerra.
GUERRA DA COREIA: MOBILIDADE E INOVAÇÕES VASCULARES
A Guerra da Coreia (1950-1953) apresentava ferimentos semelhantes de alta energia, mas o frio amplificava riscos de congelamento. Biomecanicamente, fragmentos causavam fraturas cominutivas, com sobrepressão por explosão levando a barotrauma em terrenos confinados. Causas de morte mudaram para hemorragia gerenciada, com doenças em proporção de 0,2:1. Mortalidade por ferimentos caiu para 2,4%, abdominais para 12%. Gangrena gasosa foi quase eliminada por antibióticos.
Hospitais Cirúrgicos Móveis do Exército (MASH) permitiam cuidados em 3-12 horas via helicópteros. Tratamentos incluíam reparos vasculares (taxa de amputação de 49,6% na Segunda Guerra para 20,5%), profilaxia com penicilina-estreptomicina e gesso funcional. Injeções de procaína tratavam congelamento, destacando adaptações ambientais.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Segunda Guerra Mundial | Fragmentação, spallation; toxicidade inalatória | Hemorragia, infecção | 3,3% geral | Penicilina, transfusões de sangue, fechamento atrasado |
| Guerra da Coreia | Explosões cominutivas; compressão por congelamento | Hemorragia gerenciada, infecção reduzida | 2,4% geral | Evacuação por helicóptero, cirurgia vascular, antibióticos |
GUERRA DO VIETNÃ: RESPOSTA RÁPIDA EM GUERRA DE SELVA
O Vietnã (1955-1975) envolvia balas de alta velocidade e minas, criando politrauma contaminado com balas instáveis (ex.: fragmentação do M-16 após 12 cm de penetração). Biomecânica incluía cavitação profunda por armadilhas, levando a perda tecidual massiva e infecções secundárias. Choque hipovolêmico e infecções causavam a maioria das mortes, com doenças em 0,2:1. Mortalidade hospitalar era de 2,6%, abdominais de 4,5%; transportes rápidos inflavam números ao salvar quem antes era considerado morto em ação.
Evacuações por helicóptero (1-6 horas) e lavagem pulsátil para debridamento eram chave. Antibióticos lidavam com resistências (ex.: Pseudomonas), com reparos venosos preservando membros. Registros do Wound Data and Munitions Effectiveness Team (WDMET) informavam cuidados, reduzindo falha renal para 0,17%.
GUERRA DA UCRÂNIA: TRAUMAS IMPULSIONADOS POR DRONES
Desde 2022, drones revolucionaram os traumas, entregando explosivos com precisão. Biomecanicamente, drones FPV causam explosões quaternárias: barotrauma primário em pulmões/GI, shrapnel secundário (81% dos ferimentos), deslocamentos terciários e queimaduras por cargas termobáricas. Shrapnel cria múltiplas cavidades pequenas, com queimaduras e remoção tecidual semelhantes a IEDs, mas em escalas menores. Explosões repetidas de baixo nível elevam biomarcadores para danos crônicos, adicionando tensão psicológica por zumbidos constantes e vigilância.
Sangramento por danos arteriais é a causa primária de morte, com drones responsáveis por até 80% das baixas militares e 27-30% das mortes civis em 2025 (ex.: 38 fatalidades civis por drones de curto alcance em janeiro). Mortalidade civil geral por drones: 395 mortos, 2.635 feridos (2022-2025). Relatórios indicam que drones causam 70-80% das mortes e ferimentos no campo, com 75% das lesões vindas deles em análises recentes, e evacuações atrasadas elevando morbidade por uso prolongado de torniquetes.
Tratamentos incluem torniquetes para hemorragia, debridamento, shunts vasculares e fechamento assistido a vácuo. Amputações são comuns para lesões em membros, com termografia auxiliando reconstrução. Desafios incluem drones alvejando médicos, atrasando cuidados e elevando mortalidade. Equipes Cirúrgicas Avançadas adaptam modelos da Segunda Guerra-Coreia, mas a complexidade demanda inovação contínua, como treinamento em resiliência para fadiga de batalha e uso de ultrassom portátil. A onipresença de drones cria “zonas de exclusão tecnológica”, complicando operações médicas e sugerindo robótica para evacuações.
| Período de Guerra | Biomecânica Principal dos Ferimentos | Causas Principais de Morte | Taxa de Mortalidade (Ferimentos) | Tratamentos Chave |
|---|---|---|---|---|
| Vietnã | Fragmentação instável; explosões por minas | Choque, infecção | 2,6% hospitalar | Lavagem pulsátil, reparos venosos |
| Ucrânia (2022-) | Cavitação por shrapnel de drones; queimaduras termobáricas | Sangramento, politrauma | Até 80% das baixas relacionadas a drones | Torniquetes, retalhos, termografia |
Em conclusão, os traumas de guerra evoluíram de penetrações diretas não gerenciadas para explosões multifacetadas, com mortalidade declinando por avanços logísticos e farmacológicos. No entanto, drones na Ucrânia sinalizam um possível ressurgimento na complexidade, necessitando pesquisa em biomecânica adaptativa e tratamentos. Como visto em lições de conflitos passados, a inovação médica pode mitigar esses desafios, mas requer investimento global para proteger tanto militares quanto civis em guerras futuras.
Principais Pontos sobre a Evolução dos Traumas de Guerra
- A história dos ferimentos de guerra reflete avanços em armamentos e medicina: de ferimentos penetrantes de baixa energia em batalhas antigas, como Termópilas, para traumas complexos de alta energia causados por explosivos e drones modernos, com mortalidade caindo de mais de 70% em ferimentos graves antigos para menos de 3% em conflitos do século XX, graças a evacuações rápidas e antibióticos.
- Causas de morte evoluíram: hemorragia e infecções dominavam antigamente, mas explosões e drones na Ucrânia adicionam danos por ondas de choque, queimaduras e traumas psicológicos, com drones responsáveis por até 80% das baixas em alguns relatórios recentes.
- Taxas de mortalidade diminuíram historicamente, mas guerras modernas como a da Ucrânia podem inverter isso devido a atrasos em evacuações e ataques a instalações médicas; evidências sugerem que drones causam mais baixas civis do que qualquer outra arma em 2025.
- Tratamentos progrediram de remédios herbais para cirurgias de controle de danos e uso de torniquetes, com inovações como drones de evacuação sendo exploradas para mitigar riscos atuais.
Biomecânica dos Traumas Pesquisas indicam que ferimentos antigos envolviam lacerações diretas com cavidades permanentes mínimas, enquanto armas modernas criam cavidades temporárias extensas, danificando tecidos elásticos e causando necrose. Na Ucrânia, drones FPV geram fragmentos múltiplos e barotrauma, amplificando lesões em órgãos.
Causas de Morte e Taxas de Mortalidade Hemorragia permanece a principal causa, mas infecções caíram com antibióticos. Na Primeira Guerra Mundial, mortalidade por ferimentos era de cerca de 21%, caindo para 2,4% na Guerra da Coreia. Na Ucrânia, drones causam 70-80% das mortes e ferimentos no campo de batalha, com atrasos em cuidados elevando morbidade.
Medidas de Tratamento Avanços incluem debridamento precoce, transfusões e vacinas. Na era moderna, ênfase em treinamento de resiliência para estresse por drones e uso de ultrassom portátil para detecção precoce. Recomendações incluem integrar controle de estresse operacional em frentes avançadas.
Estratégias Cirúrgicas para diminuir as complicações das anastomoses biliodigestivas
1. Introdução
As anastomoses biliodigestivas — especialmente a hepáticojejunostomia em Y-de-Roux e a coledocojejunostomia — são procedimentos essenciais no tratamento de estenoses benignas, lesões biliares iatrogênicas, tumores peri-hilares e reconstruções complexas.
Apesar da ampla experiência acumulada, complicações como fuga biliar, estenose anastomótica, colangite, falha funcional e sepse ainda ocorrem.
A seguir, um conjunto de estratégias cirúrgicas baseadas em princípios anatômicos, vascularização, técnica minuciosa e manejo pós-operatório para reduzir complicações.
2. Estratégia 1 — Escolha correta do nível da anastomose
A. Anastomose em ducto saudável
- Nunca anastomosar na área inflamada, isquêmica ou descolada de forma agressiva.
- Em estenoses pós-colecistectomia, subir para hepático comum alto ou confluência, garantindo boa vascularização.
B. Respeitar o “Princípio de Hepp–Couinaud”
- Criar hepáticojejunostomia lateral (em janela) no ducto esquerdo quando há estenose alta.
- Reduz tensão e melhora o fluxo.
3. Estratégia 2 — Criar uma anastomose ampla (≥ 1,5–2 cm)
Regra de ouro: anastomose pequena = estenose futura.
- Utilizar abertura generosa do ducto, espatulando as margens.
- Em ductos pequenos (< 4 mm), dilatar suavemente a parede anterior.
- Enxergar toda a circunferência do ducto antes de suturar.
4. Estratégia 3 — Técnica de sutura precisa
A. Fios finos e material adequado
- Polipropileno 5-0 ou 6-0; monofilamento absorvível em casos selecionados.
- Evitar multifilamento que favoreça infecção.
B. Padrão de sutura
- Sutura contínua na parede posterior → rápida e homogênea.
- Sutura interrompida na anterior → segurança e ajuste fino.
C. Pontos mucosa–mucosa
- Garantir perfeita aposição da mucosa biliar à mucosa jejunal.
- Evitar inclusão de tecido fibrótico ou adventícia espessa.
5. Estratégia 4 — Garantir vascularização ideal
Nunca desvascularizar o ducto biliar.
Princípios:
- Evitar dissecação circunferencial agressiva.
- Não esqueletonizar o hepático comum.
- Manter adventícia e plexos pericoledocianos.
Vascularização pobre = risco de necrose e deiscência → fuga biliar precoce → estenose tardia.
6. Estratégia 5 — Tensão zero
A. Tensão na alça = mau prognóstico
- Fazer alça jejunal longa e móvel, Y-de-Roux verdadeira.
- Mobilização mesentérica ampla.
- Evitar que a alça seja “puxada” para cima.
B. Anastomose deve repousar no plano natural, sem tração.
7. Estratégia 6 — Preparo adequado da alça jejunal
- Alça isoperistáltica preferencial.
- Mesentério sem torções.
- Alça livre de hematomas e isquemia.
- Evitar alça muito fina (risco de estase e colangite).
8. Estratégia 7 — Magnificação e visualização ideal
- Usar lupas, microscópio ou câmeras HD.
- Iluminação frontal/auxiliar.
- Aspirador fino dedicado.
- Irrigação suave e contínua.
9. Estratégia 8 — Drenagem seletiva (não obrigatória)
O uso de drenos é controverso. Indicar quando:
- Difícil visualização intraoperatória
- Campo inflamado
- Dúvida sobre qualidade da anastomose
- Ducto muito fino
- Reconstrução após lesão térmica extensa
Dreno não previne complicações, mas alerta precocemente.
10. Estratégia 9 — Evitar estase biliar
- Alça jejunal não deve ser comprimida por mesentério angulado.
- Anastomose deve ser ampla e elevada para evitar “kinking”.
- Em reconstruções altas, preferir derivação ampla tipo Hepp–Couinaud.
11. Estratégia 10 — Controle intraoperatório
A. Colangiografia intraoperatória
Avalia permeabilidade, nível da anastomose e ductos acessórios.
B. Colangioscopia intraoperatória
Identifica:
- Estenoses ocultas,
- Ducto residual pouco visível,
- Extensão de lesões biliares,
- Cálculos residuais.
Aumenta segurança e reduz reoperação.
12. Estratégia 11 — Manejo pós-operatório rigoroso
- Controle diário de bilirrubina, FA, GGT.
- Ultrassom precoce se febre/taquicardia.
- TC se suspeita de coleção.
- Tratamento agressivo de colangite.
Colangite recorrente = alerta para estenose precoce.
13. Estratégia 12 — Realização em centros especializados
Evidência clara:
Reconstruções realizadas por cirurgiões hepatobiliares experientes têm taxas de sucesso > 90%, enquanto em centros não especializados, as falhas podem superar 30–40%.
Conclusão
A segurança e durabilidade das anastomoses biliodigestivas dependem de:
- seleção adequada do nível da anastomose;
- anastomose ampla, mucosa–mucosa;
- técnica delicada e meticulosa;
- vascularização preservada;
- ausência de tensão;
- uso racional de colangiografia/colangioscopia;
- execução por cirurgião experiente em HPB.
“Uma anastomose biliodigestiva segura é construída com técnica, paciência e respeito absoluto à biologia.”
Dilatações assintomáticas do colédoco
1. Introdução
A dilatação do colédoco é um achado frequente em ultrassom abdominal, tomografia ou colangioressonância, muitas vezes incidental e sem correlação clínica direta. O grande desafio é distinguir dilatações benignas fisiológicas de dilatações que representam doença biliar subjacente (coledocolitíase, estenose, tumor periampular).
Apesar de ser assintomática, nunca deve ser ignorada. A propedêutica adequada evita diagnósticos tardios de patologias graves, especialmente colangiocarcinoma hiliar, tumor de cabeça de pâncreas e ampuloma.
2. Quais valores definir como “dilatação”?
Critério clássico:
- Colédoco > 6 mm em pacientes com vesícula biliar íntegra.
Ajustes por idade:
- +1 mm por década acima de 60 anos.
Após colecistectomia:
- Até 10 mm pode ser considerado fisiológico.
Pós-bariátrica (bypass gástrico):
- 8–10 mm pode ser considerado normal pelo menor estímulo colecisto-pancreático.
3. Causas de Dilatação Assintomática
A. Causas benignas e fisiológicas
- Idade avançada
- Pós-colecistectomia
- Uso crônico de opioides
- Pós-esfincterotomia
- Pós-cirurgia anatômica complexa (ex.: derivação biliar)
B. Causas patológicas silenciosas
(perigosas e frequentemente subdiagnosticadas)
- Coledocolitíase “oculta” (microcálculos, barro biliar)
- Estenose biliar benigna
- Neoplasia periampular
- Tumor de cabeça de pâncreas
- Colangiocarcinoma hiliar ou distal
- Pancreatite autoimune
- Doença parasitária (em áreas endêmicas)
4. Propedêutica: Como investigar?
1. História clínica e exame físico
Avaliar:
- Colangite prévia
- Cólica biliar antiga
- Histórico de pancreatite
- Cirurgias prévias
- Perda ponderal
- Icterícia transitória
- Uso de opioides
2. Exames laboratoriais
- Bilirrubinas
- FA e GGT (mais sensíveis)
- TGO/TGP
- Amilase/lipase se suspeita de pancreatite
- IgG4 em casos selecionados
Mesmo pequenas elevações sugerem obstrução parcial.
3. Exames de imagem (ordem racional)
A. Ultrassom (triagem inicial)
- Dilatação intra e extra-hepática?
- Sinais indiretos de cálculo?
- Pâncreas visível?
B. Colangioressonância (CRM)
Exame padrão para investigação inicial.
Detecta:
- Estenoses
- Cálculos pequenos
- Massa periampular
- Dilatação ductal segmentar
C. Tomografia (TC) com contraste
Boa para:
- Tumor pancreático
- Linfonodos
- Anatomia geral
D. Endoscopia digestiva alta
Para excluir lesões ampulares.
4. Exames de alta precisão (casos selecionados)
A. EUS (Ecoendoscopia)
Melhor método para microcálculos e tumores pequenos (<1 cm).
Indicado quando:
- CRM inconclusiva
- Dilatação persistente sem diagnóstico
- Suspeita de tumor periampular
B. CPRE
Não é exame diagnóstico.
Usar apenas quando tratamento é provável (ex.: cálculo visto no EUS).
C. Colangioscopia
Para:
- Estenose indeterminada
- Avaliação direta da mucosa
- Suspeita de malignidade
- Biópsia dirigida
5. Conduta: Quando tratar e quando observar?
A. Dilatação fisiológica
(Nenhuma alteração laboratorial ou estrutural)
→ Apenas seguimento clínico
Repetir imagem em 6–12 meses.
B. Dilatação com alterações laboratoriais
→ Investigar com CRM → EUS
Se persistirem enzimas alteradas, mesmo sem imagem positiva:
→ EUS + CPRE terapêutica (se necessário)
C. Cálculo ou barro biliar detectado
→ CPRE com extração
→ Avaliar necessidade de colecistectomia (se vesícula presente)
D. Estenose benigna
→ Dilatação + stent plástico (técnica sequencial)
ou
→ Avaliação cirúrgica se estenose alta ou recorrente
E. Suspeita de malignidade
→ EUS + colangioscopia + biópsia
→ Encaminhar para equipe HPB
F. Anatomia alterada (bypass gástrico)
→ Preferir EUS transgástrico ou CPRE via gastrotomia laparoscópica
(técnica híbrida)
6. Quando a dilatação assintomática é grave?
Alta preocupação quando há:
- Dilatação > 12–15 mm
- Dilatação progressiva ao longo do tempo
- Dilatação segmentar intra-hepática
- Achado de “cut-off” abrupto no colédoco
- Elevação de FA/GGT persistente
- Perda de peso, anemia ou prurido
Esses pacientes merecem investigação completa e rápida.
Conclusão
A dilatação assintomática do colédoco nunca é um achado trivial. Embora parte delas seja fisiológica, uma fração significativa pode representar doença biliar silenciosa, incluindo neoplasias de alta letalidade.
A propedêutica deve ser estruturada, escalonada e guiada pelo risco, começando com CRM, laboratórios e evoluindo para EUS, CPRE ou colangioscopia conforme necessário.
Tratamento depende da causa:
- Fisiológica → observar.
- Litíase → tratar.
- Estenose → dilatar ou reconstruir.
- Tumor → investigação HPB.
“Via biliar dilatada sem explicação é diagnóstico que não pode esperar.”
Colangioscopia intraoperatória: Quando, como e quem deve fazer?
1. Quando indicar colangioscopia intraoperatória?
A colangioscopia intraoperatória é um recurso de alta precisão para avaliação direta do ducto biliar. As indicações reais são pontuais e estratégicas, especialmente quando a decisão cirúrgica depende de informação imediata e fidedigna.
Principais indicações:
A. Suspeita de coledocolitíase residual
- Coledoco dilatado (>8 mm)
- Icterícia pré-operatória
- Pancreatite biliar
- Colangite
- USG/CRM sugerindo cálculo, mas sem certeza
- Colangiografia inconclusiva ou contraditória
Objetivo: excluir ou remover cálculo residual imediatamente.
B. Avaliação de estenoses biliares indeterminadas
(Ao explorar a via biliar ou durante cirurgia hepato-biliar complexa).
- Diagnóstico e caracterização de estenoses benignas vs malignas
- Verificação de margem em cirurgia oncológica
- Lesão biliar pós-colecistectomia para avaliar extensão
C. Cirurgias biliares complexas
- Cisto de colédoco
- Colangiocarcinoma (para avaliar extensão intraductal)
- Doenças pediátricas biliares (em centros especializados)
D. Reoperação ou reexploração das vias biliares
- Falha prévia de abordagem endoscópica
- Anatomia alterada (Bypass gástrico – Y-de-Roux, gastrectomia)
- Dúvida intraoperatória sobre presença de cálculos
2. Como realizar a colangioscopia intraoperatória?
Existem dois sistemas utilizados:
A. Colangioscopia por canal operatório (Endoscópio flexível)
Técnica mais comum no intraoperatório.
Passo a passo:
- Acesso por coledocotomia ou pela coto do cístico dilatado.
- Lavado contínuo com soro morno.
- Introdução suave do colangioscópio sob visão direta.
- Avaliação sistemática:
- Hepático comum → hepáticos → lobares
- Permeabilidade
- Presença de cálculos, barro, estenoses, massa
- Remoção de cálculos com:
- Basket
- Balão
- Irrigação pressurizada
Vantagens:
- Alta definição
- Visualização direta da mucosa
- Detecção de cálculos pequenos e impactados
B. Sistema de colangioscopia digital descartável (SpyGlass®)
Mais caro, porém mais eficiente.
Vantagens:
- Resolução superior
- Canal de trabalho amplo
- Permite biópsia dirigida
- Indispensável para estenoses indeterminadas e tumores biliares
Pontos técnicos fundamentais
- Pressão baixa de irrigação para evitar bacteremia e extravasamento
- Evitar traumatismo da mucosa
- Manter campo biliar estéril (antibiótico profilático)
- Documentar achados com vídeo/foto
3. Quem deve realizar a colangioscopia intraoperatória?
A resposta é direta: cirurgião do aparelho digestivo com experiência em via biliar, endoscopista ou cirurgião hepatobiliar.
Pré-requisitos ideais:
- Treinamento formal em cirurgia hepatopancreatobiliar (HPB)
- Domínio de exploração laparoscópica e aberta da via biliar
- Capacidade de interpretar estenoses, cálculos e lesões mucosas
- Familiaridade com equipamentos ópticos flexíveis e rígidos
- Treinamento conjunto com endoscopia terapêutica
Em centros de referência, a colangioscopia intraoperatória é feita:
- Pelo cirurgião HPB, quando conduzindo colecistectomias complexas ou revisões de via biliar;
- Pelo cirurgião do aparelho digestivo, quando abordando coledocolitíase em anatomia alterada;
- Pelo endoscopista avançado, quando há integração com cirurgia laparoscópica (procedimentos híbridos).
4. A colangioscopia intraoperatória substitui a colangiografia?
Não.
Ela complementa.
- Colangiografia = método de rastreio
- Colangioscopia = método de confirmação e tratamento
A decisão correta é: colangioscopia quando a colangiografia NÃO responde tudo.
5. Situações em que deve ser evitada
- Coledoco muito fino (<5 mm)
- Infecção ativa não controlada
- Dúvida anatômica que contraindique manipulação
- Falta de equipamento ou profissional treinado
Conclusão
A colangioscopia intraoperatória é uma ferramenta precisa, segura e extremamente útil na via biliar, desde que usada no contexto certo.
Quando usar?
- Suspeita de cálculo residual
- Estenose indeterminada
- Cirurgias biliares complexas
- Revisões e reoperações
Como usar?
- Através de coledocotomia ou coto do cístico
- Com ótica flexível ou sistema digital
- Com técnica delicada, irrigação e limpeza sistemática
Quem deve fazer?
- Cirurgião HPB ou cirurgião do aparelho digestivo experiente em via biliar, ou endoscopista avançado em centros híbridos.
“O cirurgião domina o que vê. Ver o ducto biliar por dentro é decidir com precisão.”
Litíase Biliar Após Cirurgia Bariátrica: Quando e Como Abordar?
Introdução
A litíase biliar é uma complicação frequente após a cirurgia bariátrica, especialmente em procedimentos que resultam em perda ponderal rápida, como o bypass gástrico em Y-de-Roux (RYGB) e a gastrectomia vertical (sleeve). A incidência de formação de cálculos pode atingir 30–40% nos primeiros 12 a 18 meses, com até 15% dos pacientes evoluindo com sintomas ou complicações como colecistite aguda, pancreatite biliar ou coledocolitíase. O manejo adequado depende do tipo de procedimento bariátrico, da anatomia reconstruída e da apresentação clínica.
Mecanismos Fisiopatológicos
A rápida perda de peso induz:
- Supersaturação biliar por colesterol,
- Diminuição da motilidade da vesícula biliar,
- Aumento de mucinas e nucleação,
- Redução da ingesta lipídica estimuladora de contração biliar.
Tais fatores explicam por que a litíase se desenvolve principalmente nos primeiros 6–12 meses pós-operatórios.
Quando Suspeitar e Quando Intervir
1. Paciente assintomático com cálculos
Não há indicação de colecistectomia profilática na maioria dos centros. A conduta é expectante, exceto em casos especiais:
- Cálculos > 1 cm,
- Vesícula completamente preenchida,
- História prévia de pancreatite biliar,
- Paciente com acesso cirúrgico futuro dificultado.
Alguns grupos utilizam ursofalk (ácido ursodesoxicólico) 300–600 mg/dia por 6 meses, capaz de reduzir a incidência de litíase sintomática, sobretudo em RYGB.
2. Paciente sintomático (cólica biliar ou colecistite leve)
A conduta é colecistectomia laparoscópica, idealmente realizada por cirurgião com experiência em pacientes pós-bariátricos, que apresentam:
- Adhesões frequentes,
- Alterações anatômicas de troca de porta,
- Maior fragilidade tecidual.
3. Colecistite aguda moderada/grave
Seguir o protocolo de Tokyo Guidelines:
- Antibióticos,
- Colecistectomia de urgência se possível,
- Drenagem percutânea em casos selecionados.
4. Pancreatite biliar
Após estabilização clínica, realizar colecistectomia.
O timing depende do tipo de bariátrica:
- Sleeve: anatomia normal → colecistectomia na mesma internação.
- Bypass gástrico: alta chance de coledocolitíase → investigar colédoco antes de operar.
Coledocolitíase no Paciente Bariátrico: Como Abordar?
A abordagem depende profundamente da anatomia pós-cirúrgica:
A. Gastrectomia Vertical (Sleeve)
Anatomia do duodeno e papila mantida.
→ CPRE convencional possível.
Tratamento padrão:
- CPRE diagnóstica + terapêutica,
- Esfincterotomia,
- Balão/ Basket,
- Colecistectomia subsequente.
B. Bypass Gástrico em Y-de-Roux (RYGB)
É o cenário mais desafiador. O acesso à papila é bloqueado pela alça alimentar.
Opções:
- CPRE por enteroscopia de duplo balão
- Técnica exigente, limitada a centros avançados.
- Sucesso ~70%.
- CPRE transgástrica laparoscópica
- A via mais resolutiva em muitos centros.
- Procedimento combinado: laparoscopia cria gastrotomia no estômago excluído → endoscopista acessa a papila.
- Alta taxa de sucesso > 95%.
- CPRE guiada por EUS (EDGE)
- Criação de uma fístula temporária entre o pouch e o estômago excluído usando LAMS.
- Opção moderna e altamente eficaz.
- Exige endoscopista avançado.
- Exploração cirúrgica da via biliar
- Via transcística ou coledocotomia laparoscópica, quando experiência disponível.
Quando Operar Antes da Bariátrica?
Indicações de colecistectomia prévia:
- Cálculos sintomáticos,
- Cálculos > 1,5 cm,
- Vesícula em porcelana,
- Pólipos > 1 cm,
- Suspeita de malignidade,
- História prévia de pancreatite biliar.
Para pacientes assintomáticos, a tendência atual é não operar previamente, exceto quando o acesso futuro pode ser especialmente difícil (superobesidade com grande parede abdominal ou barreira logística).
Pontos-Chave
- A litíase biliar é comum após cirurgia bariátrica (30–40%).
- Colecistectomia profilática não é indicada rotineiramente.
- A abordagem depende do tipo de cirurgia:
- Sleeve → CPRE convencional possível.
- Bypass → técnicas avançadas (enteroscopia, EDGE, CPRE transgástrica).
- O tratamento deve ser individualizado e multidisciplinar.
- A cirurgia bariátrica exige planejamento prévio quanto ao risco de litíase.
Conclusão
A litíase biliar no pós-operatório de cirurgia bariátrica não é apenas frequente, mas também clinicamente relevante, podendo gerar quadros graves como pancreatite e colangite. O sucesso do manejo está em reconhecer o tipo de reconstrução do trato gastrointestinal, antecipar dificuldades técnicas e atuar com abordagem multidisciplinar envolvendo cirurgião bariátrico, cirurgião hepatobiliar e endoscopista avançado.
“A cirurgia moderna exige não apenas técnica, mas estratégia.” — Adaptado de Halsted
Ambroise Paré (1510–1590): o Cirurgião que Não Sabia Latim
Introdução
Ambroise Paré é reconhecido como um dos pais da cirurgia moderna. Nascido em 1510, na pequena vila de Bourg-Hersent, próximo a Laval, na França, viveu em uma época de intensas transformações políticas, religiosas e científicas. Sem formação médica formal e sem domínio do latim — idioma obrigatório da ciência de seu tempo —, Paré rompeu barreiras sociais e acadêmicas para revolucionar a cirurgia com base em observação empírica, técnica precisa e humanidade.

Formação e Primeiros Anos no Hôtel-Dieu
Paré iniciou sua trajetória como aprendiz de cirurgião-barbeiro, profissão considerada de segunda classe em relação aos médicos da época. Em Paris, conseguiu, aos dezenove anos, ingressar no Hôtel-Dieu, o mais antigo hospital da cidade, onde se destacou pela habilidade manual e pela curiosidade científica. Nessa instituição, conviveu com feridos, queimados e amputados de guerra, experiências que moldaram sua prática futura. Posteriormente, serviu como cirurgião militar do exército francês nas campanhas da Itália entre 1536 e 1545, período em que faria suas descobertas mais marcantes.
A Descoberta do Tratamento das Feridas por Arma de Fogo
Até o século XVI, acreditava-se que as feridas por projéteis de pólvora eram envenenadas e deviam ser cauterizadas com ferro em brasa ou óleo fervente, conforme ensinava o cirurgião italiano Giovanni da Vigo. Durante uma campanha militar em 1536, Paré ficou sem óleo fervente e improvisou um curativo com gema de ovo, óleo de rosas e terebintina. Na manhã seguinte, observou que os pacientes tratados com essa mistura estavam tranquilos e com feridas limpas, enquanto os cauterizados sofriam febre e dor intensa. Esse acaso deu origem à primeira grande inovação de Paré: o abandono do cautério e do óleo fervente, substituídos por curativos mais suaves e fisiológicos — um avanço que reduziu a mortalidade e o sofrimento dos soldados.
A Ligadura das Artérias e a Hemostasia Moderna
Outro marco histórico atribuído a Paré é a introdução da ligadura de vasos sanguíneos nas amputações.
Até então, a hemostasia era obtida cauterizando os cotos com ferro em brasa — prática cruel e ineficaz. Em 1552, Paré começou a usar pinças e fios de seda para ligar os vasos, técnica descrita em seu livro Traité de la Chirurgie (1564). Ele também projetou instrumentos como o “Bec de Corbin” (“bico de corvo”), precursor dos hemostatos modernos. Esse método, apesar de rudimentar, representou a fundação da cirurgia vascular moderna e foi adotado em toda a Europa.
Reconhecimento Real e Ascensão Profissional
Graças à sua habilidade e serenidade, Paré ganhou prestígio na corte francesa. Serviu como cirurgião de quatro reis:
Henrique II, Francisco II, Carlos IX e Henrique III.
Após a morte trágica de Henrique II em 1559 — em consequência de um ferimento craniano durante um torneio, atendido por Paré e Vesalius —, ele foi nomeado primeiro cirurgião do rei por Carlos IX e posteriormente cirurgião real de Henrique III, o posto mais alto que um cirurgião poderia ocupar na França renascentista. Mesmo sem formação universitária e alvo de críticas por “não saber latim”, Paré foi admitido na Confraria de São Cosme, onde obteve o título de mestre em cirurgia, superando o preconceito dos médicos letrados.
Obras, Invenções e Contribuições
Em 1564, publicou Dix Livres de la Chirurgie, e em 1575 reuniu todos os seus trabalhos em 27 volumes sob o título Les Œuvres de M. Ambroise Paré.
Entre suas contribuições destacam-se:
- Introdução da ligadura arterial;
- Uso de curativos terapêuticos não cáusticos;
- Desenvolvimento de membros artificiais e próteses oculares de metal e porcelana;
- Observação pioneira do fenômeno do “membro fantasma” em amputados;
- Correlação entre sífilis e aneurisma da aorta;
- Reintrodução de técnicas obstétricas antigas (como a versão podálica);
- Abordagem crítica ao uso de “pó de múmia” como medicamento, prática supersticiosa comum na época.
Fé, Humanismo e Legado
Ambroise Paré viveu em meio às Guerras Religiosas da França e sobreviveu ao Massacre da Noite de São Bartolomeu (1572), protegido pelo rei Carlos IX. Embora simpatizante dos huguenotes (protestantes calvinistas), manteve-se oficialmente católico para evitar perseguições. Seu lema, inscrito em sua cadeira na Confraria de São Cosme, resumia sua filosofia médica:
“Je le pansai, Dieu le guérit.”
(Eu o tratei, mas Deus o curou.)
Faleceu em 1590, aos 80 anos, em Paris. Foi sepultado com honras, e um monumento em sua memória foi erguido em Laval, sua cidade natal.
Conclusão
Ambroise Paré foi mais do que um cirurgião: foi um símbolo da medicina humanista, que uniu técnica e compaixão em uma era de brutalidade. Seu trabalho elevou a cirurgia de ofício manual a disciplina científica, lançando as bases da prática moderna e do ensino cirúrgico. De aprendiz barbeiro a cirurgião real, Paré encarnou o ideal atemporal do médico-cientista movido por propósito, disciplina e fé.
“Há cinco deveres da cirurgia: remover o que é supérfluo, restaurar o que foi deslocado, separar o que cresceu junto, reunir o que foi dividido e corrigir as imperfeições da natureza.” — Ambroise Paré
#HistóriaDaCirurgia #AmbroisePare #CirurgiaModerna #EducaçãoMédicaContinuada #HumanismoMédico
Evolução da Cirurgia Abdominal: das Incisões à Robótica
Introdução
A cirurgia abdominal representa um dos campos mais transformadores da medicina moderna. Desde os primeiros relatos de intervenções rudimentares no Egito Antigo até os procedimentos laparoscópicos e robóticos do século XXI, essa especialidade foi moldada por uma combinação de coragem, ciência e inovação. Entender a evolução da cirurgia abdominal é compreender a trajetória do próprio raciocínio cirúrgico — da exploração empírica à precisão tecnológica.
Origens Antigas e Pré-Anestésicas
Os primeiros registros datam de cerca de 1500 a.C., com relatos de drenagens de abscessos e tentativas de reparo de ferimentos abdominais em papiros egípcios. Na Grécia Clássica, Hipócrates e Galeno descreveram técnicas rudimentares de incisão e sutura, mas a ausência de anestesia e controle infeccioso limitava qualquer intervenção intra-abdominal. Durante séculos, abrir o abdome equivalia a uma sentença de morte. A verdadeira revolução só viria com três marcos fundamentais: anestesia, antissepsia e hemóstase.
O Século XIX: O Nascimento da Cirurgia Moderna
Em 1846, William T. G. Morton realizou a primeira anestesia com éter no Massachusetts General Hospital, permitindo intervenções prolongadas sem sofrimento. Pouco depois, Joseph Lister introduziu o conceito de antissepsia (1867), reduzindo drasticamente a mortalidade pós-operatória. Esses avanços abriram caminho para a “era heroica” da cirurgia abdominal. Em 1881, Theodor Billroth executou a primeira gastrectomia parcial com sucesso, e Carl Langenbuch realizou a primeira colecistectomia (1882), marco fundador da cirurgia hepatobiliar moderna.
A partir daí, o abdome deixou de ser território proibido e tornou-se o novo campo de batalha da ciência médica.
O Século XX: Consolidação e Aperfeiçoamento Técnico
O século XX testemunhou a consolidação das grandes operações digestivas: Whipple descreveu sua pancreatoduodenectomia em 1935; Halsted refinou a técnica de sutura e as bases da cirurgia oncológica; e os avanços na anestesia inalatória e transfusão sanguínea tornaram procedimentos cada vez mais complexos possíveis. Após as guerras mundiais, o desenvolvimento da cirurgia vascular, hepática e de trauma foi impulsionado pelo atendimento de feridos, transferindo o aprendizado militar para os hospitais civis.
A Revolução Laparoscópica
Em 1987, o francês Philippe Mouret realizou a primeira colecistectomia laparoscópica, inaugurando a era da cirurgia minimamente invasiva. A visão endoscópica e o controle de pequenos instrumentos trouxeram redução de dor, tempo de internação e morbidade. A laparoscopia transformou não apenas a técnica, mas também a mentalidade cirúrgica — substituindo força por precisão, e exposição por visualização.
O Século XXI: A Era Robótica e da Inteligência Artificial
A introdução da cirurgia robótica no início dos anos 2000 levou o conceito de precisão a um novo patamar. O sistema da Vinci®, com movimentos articulados e visão tridimensional, ampliou a capacidade de dissecção em espaços confinados e redefiniu o ensino cirúrgico. Hoje, a inteligência artificial generativa começa a ser usada no planejamento operatório, simulação de complicações e análise de prognóstico, inaugurando uma nova fase da medicina baseada em dados e aprendizado automatizado.
Conclusão
A história da cirurgia abdominal é uma narrativa de disciplina e superação: de um ato de desespero tornou-se um gesto de ciência e compaixão. Cada avanço — da antissepsia à robótica — ampliou a margem de segurança e reduziu o sofrimento humano. Mais do que uma linha do tempo, é uma linha de coragem. E o futuro, inevitavelmente, será moldado pela integração entre o bisturi humano e a inteligência das máquinas.
“A cirurgia é o encontro entre a precisão da técnica e a grandeza do propósito humano.” — Adaptado de Harvey Cushing
#CirurgiaAbdominal #HistóriaDaMedicina #CirurgiaMinimamenteInvasiva #RobóticaCirúrgica #EducaçãoMédicaContinuada
Tríade Cirúrgica
Introdução
A cirurgia é, antes de tudo, uma escola de caráter. O domínio técnico é apenas uma das faces da formação do cirurgião; o verdadeiro diferencial está na capacidade de manter disciplina, cultivar resiliência e agir com propósito. Esses três pilares sustentam a excelência médica em um ambiente de pressão constante, decisões imediatas e responsabilidade sobre vidas humanas.
“O cirurgião é um soldado em tempo de paz: luta sem armas, vence sem glória e serve sem descanso.”
A Disciplina: A Arte da Constância
A disciplina é a virtude silenciosa que transforma talento em competência. Ela se manifesta nas pequenas rotinas diárias: revisar casos antes do plantão, estudar o que foi operado, cuidar dos detalhes do pós-operatório e seguir protocolos com rigor. Para o cirurgião, disciplina não é rigidez, mas constância inteligente. É o hábito de fazer o certo mesmo quando ninguém observa, o compromisso de preparar-se continuamente para o imprevisto. A disciplina cria confiança — no bisturi, na equipe e no paciente.
Sem disciplina, não há precisão; sem precisão, não há vitória. A disciplina do cirurgião é o equivalente à prontidão de combate: preparação constante, execução impecável e obediência rigorosa aos princípios da técnica e da ética.
A Resiliência: A Força que se Refaz
O caminho cirúrgico é inevitavelmente marcado por falhas, complicações e perdas. O cirurgião que não aprende a lidar com o insucesso corre o risco de se quebrar por dentro. A resiliência é a capacidade de aprender com o erro sem se paralisar, de suportar a frustração sem perder a ética, e de recomeçar com humildade e vigor. Mais do que resistir, o cirurgião resiliente se transforma. Ele reconhece a dor como instrumento de amadurecimento profissional e humano, e compreende que nenhuma cicatriz — seja no paciente, seja nele próprio — é em vão.
Toda missão tem perdas. Toda operação tem falhas. O cirurgião que não suporta a frustração é como o soldado que abandona o posto na primeira ofensiva. A resiliência é a blindagem emocional que permite ao médico enfrentar a adversidade, aprender com o erro e voltar mais forte. O cirurgião resiliente não nega a dor — ele a transforma em disciplina moral. Sabe que cada complicação é uma batalha perdida, mas não a guerra. Sabe que a autocrítica é o campo de treinamento da sabedoria.
O Propósito: O Norte que Sustenta a Jornada
Sem propósito, a técnica torna-se mecânica. O propósito é o eixo moral que dá sentido às longas horas de centro cirúrgico, às privações e às renúncias pessoais. Ele nasce do reconhecimento do valor da vida humana e da consciência de que cada ato cirúrgico é um gesto de confiança depositado pelo paciente. Trabalhar com propósito é lembrar-se diariamente de por que se escolheu operar — e não apenas de como se opera. É compreender que cada decisão impacta uma biografia, não apenas um prontuário.
O propósito é a bússola que guia o soldado e o cirurgião no caos. Sem ele, a técnica vira rotina, o bisturi vira arma e o ofício perde o sentido. O propósito é servir: servir à vida, à verdade e ao dever. É ele que dá coragem para entrar no centro cirúrgico às 3h da manhã, que sustenta o comando em meio à fadiga e que transforma o ato cirúrgico em missão.
Conclusão
Disciplina mantém o foco; resiliência sustenta o espírito; propósito dá direção.
Juntos, formam o tripé que sustenta o cirurgião diante da complexidade da vida moderna e da impermanência dos resultados.
Em um mundo que valoriza a pressa, o cirurgião deve cultivar o tempo — o tempo de estudar, de refletir, de cuidar e de evoluir. A grandeza profissional não se mede pelo número de cirurgias realizadas, mas pela qualidade humana com que cada uma é conduzida.
“A mão que opera deve ser firme; o coração que conduz, compassivo; e a mente que decide, disciplinada.”
#CirurgiaComPropósito #ResiliênciaMédica #DisciplinaCirúrgica #FormaçãoMédica #LiderançaNaSaúde
Transplante Hepático na Insuficiência Hepática Aguda
Critérios, Sobrevida e Complicações
Introdução
A insuficiência hepática aguda (IHA) é uma condição rara, mas potencialmente fatal, caracterizada pela instalação rápida de disfunção hepática em pacientes previamente hígidos, acompanhada de coagulopatia e encefalopatia. Sem tratamento definitivo, a mortalidade ultrapassa 80% em muitos cenários. O transplante hepático é a única terapia curativa em casos graves, sendo fundamental identificar precocemente quais pacientes se beneficiam da cirurgia. Para isso, critérios clínicos e laboratoriais foram desenvolvidos e validados, destacando-se os de King’s College e Clichy, além de ferramentas modernas como o MELD score.
Critérios Prognósticos
- King’s College Criteria: o mais utilizado mundialmente, com alta especificidade. Diferencia critérios para intoxicação por paracetamol e demais etiologias.
- Critérios de Clichy: aplicados sobretudo em hepatite fulminante viral, baseados na dosagem do fator V associado à encefalopatia.
- MELD score: escore inicialmente criado para hepatopatia crônica, mas útil também em IHA; valores ≥ 30 associam-se a prognóstico ruim.
Sobrevida e Complicações Pós-Transplante
- Sobrevida:
- 1 ano: 65–80%
- 5 anos: 55–70%
- Melhor prognóstico em intoxicação por paracetamol, pior em hepatite fulminante viral e drogas idiossincráticas.
- Complicações:
- Infecções (até 40%)
- Disfunção primária do enxerto (10–15%)
- Trombose da artéria hepática (~5%)
- Complicações biliares (10–20%)
Apesar da maior mortalidade precoce, pacientes que superam o período inicial apresentam sobrevida comparável aos transplantados por doença crônica.
Tabela Comparativa dos Principais Critérios e Resultados
| Critério / Indicador | Definição / Parâmetro | Aplicação | Sobrevida Pós-TH (1 ano / 5 anos) | Principais Complicações |
|---|---|---|---|---|
| King’s College (Paracetamol) | pH < 7,30 OU INR > 6,5 + Cr > 3,4 + encefalopatia III–IV | Mais utilizado mundialmente | 65–80% / 55–70% | Infecção, disfunção precoce do enxerto |
| King’s College (Outras causas) | INR > 6,5 OU ≥ 3 critérios (idade, etiologia, intervalo, INR, bilirrubina) | Prognóstico em IHA não-paracetamol | 65–75% / 55–65% | Infecção, complicações biliares |
| Clichy | Encefalopatia + Fator V < 20% (<30 anos) ou < 30% (>30 anos) | Mais usado em hepatite fulminante viral | 60–70% / 50–60% | Disfunção primária do enxerto |
| MELD ≥ 30 | Escore baseado em INR, bilirrubina e creatinina | Complementar à decisão | Variável conforme etiologia | Complicações infecciosas e vasculares |
Conclusão
O transplante hepático é a única alternativa eficaz para pacientes com IHA sem perspectiva de regeneração espontânea. A aplicação correta dos critérios de King’s College e Clichy, associada ao uso de escores como o MELD, permite identificar precocemente os candidatos ideais. Apesar da maior taxa de complicações infecciosas e disfunção do enxerto no pós-operatório imediato, os resultados a longo prazo são satisfatórios, com sobrevida em 5 anos alcançando 70% em centros especializados. A decisão deve sempre ser multidisciplinar, precoce e baseada em critérios clínico-laboratoriais, maximizando as chances de sucesso.
“A oportunidade de indicar o transplante não se repete: reconhecer o momento certo é o maior desafio do hepatologista e do cirurgião.” — Adaptado de Roger Williams
Gostou ❔ Deixe seu comentário ✍️, compartilhe em suas redes sociais ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#TransplanteHepático #InsuficiênciaHepáticaAguda #Hepatologia #CirurgiaHepatobiliar #EducaçãoMédicaContinuada
Neoadjuvância no Adenocarcinoma de Pâncreas: Evidências e Aplicações Clínicas
Introdução
O adenocarcinoma de pâncreas é uma das neoplasias mais agressivas do trato gastrointestinal, caracterizada por diagnóstico tardio, baixa ressecabilidade e alta taxa de recorrência. Apesar dos avanços técnicos e terapêuticos, a sobrevida global em 5 anos permanece inferior a 10%. A terapia neoadjuvante (quimioterapia com ou sem radioterapia antes da cirurgia) vem ganhando papel central, especialmente em tumores localmente avançados, borderline ressecáveis e, em casos selecionados, em tumores ressecáveis. Seu objetivo é tratar micrometástases precoces, aumentar taxas de ressecção R0 e selecionar pacientes com melhor biologia tumoral para cirurgia.
Racional da Neoadjuvância
- Controle sistêmico precoce: trata micrometástases antes da cirurgia.
- Seleção de pacientes: evita cirurgias em pacientes com progressão rápida da doença.
- Maior chance de completar quimioterapia: até 50% dos pacientes não recebem adjuvância após pancreatectomia devido à morbidade.
- Aumento da taxa de R0: margens negativas são fator prognóstico chave.
Evidências Recentes
- Borderline ressecável: ensaios mostram benefício claro. Regimes como FOLFIRINOX ou gemcitabina/nab-paclitaxel aumentam ressecabilidade e sobrevida global.
- Ressecáveis: ainda em estudo, mas meta-análises sugerem equivalência ou até superioridade em pacientes com alto risco prognóstico (ex.: CA 19-9 elevado).
- Radioterapia: pode melhorar controle local, mas seu papel isolado ainda é controverso.
O estudo PREOPANC (fase III, multicêntrico) demonstrou que quimiorradioterapia neoadjuvante não aumentou complicações maiores, fístula pancreática ou mortalidade pós-operatória, reforçando a segurança da estratégia.
Critérios para Indicar Neoadjuvância em Tumores Ressecáveis
- Marcadores de alto risco:
- CA 19-9 >150–250 U/mL
- Tumor ≥35 mm
- Albumina ≤3,5 g/dL
- Relação neutrófilo-linfócito ≥3,5
- Dupan-2 ≥750 U/mL
- Achados clínicos e radiológicos:
- Linfonodomegalia suspeita
- Invasão vascular incipiente
- Sinais sistêmicos de agressividade
- Preocupação com adesão ao adjuvante:
- Pacientes frágeis ou comorbidades que dificultem completar quimioterapia após cirurgia.
Taxa de Abandono da Neoadjuvância
- Varia entre 15–30% nos principais estudos.
- Motivos: toxicidade da quimioterapia, progressão tumoral, deterioração clínica ou recusa do paciente.
- Estratégia exige acompanhamento multidisciplinar próximo e suporte intensivo para reduzir perdas.
Segurança Cirúrgica
Estudos randomizados e coortes demonstram que a neoadjuvância não aumenta o risco de complicações pós-operatórias, incluindo fístula pancreática, infecção intra-abdominal e mortalidade hospitalar. Em alguns trabalhos, observou-se até redução da incidência de fístula pancreática clinicamente relevante.
Pontos-Chave para a Prática
- Padrão atual: recomendado em borderline ressecáveis.
- Ressecáveis: indicado em casos com fatores de alto risco.
- Regimes preferenciais: FOLFIRINOX (pacientes com bom performance status); gemcitabina/nab-paclitaxel em casos selecionados.
- Multidisciplinaridade é essencial: decisão deve envolver oncologia, cirurgia, radiologia e gastroenterologia.
Conclusão
A terapia neoadjuvante no adenocarcinoma de pâncreas representa um avanço significativo, transformando a abordagem tradicional baseada em cirurgia seguida de adjuvância. Atualmente, é padrão para tumores borderline ressecáveis, considerada em tumores ressecáveis com alto risco prognóstico, e investigada em ensaios clínicos como alternativa ao tratamento clássico. O futuro caminha para uma estratificação mais refinada baseada em biomarcadores, imagem funcional e resposta precoce ao tratamento, permitindo individualizar a neoadjuvância e maximizar o benefício oncológico.
“O futuro da cirurgia oncológica não é apenas retirar o tumor, mas entender a biologia da doença e intervir no momento certo.” — Veronesi
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#PancreaticCancer #Neoadjuvância #OncologiaCirúrgica #CirurgiaDigestiva #EducaçãoMédicaContinuada
Colecistostomia: Indicações e Resultados na Prática Cirúrgica
Introdução
A colecistostomia é um procedimento fundamental no arsenal terapêutico para o manejo da colecistite aguda em pacientes de alto risco cirúrgico. Enquanto a colecistectomia laparoscópica permanece como tratamento definitivo, a colecistostomia surge como uma alternativa segura, minimamente invasiva e eficaz, especialmente em pacientes com comorbidades significativas ou contraindicações à anestesia geral.
No Brasil, onde o acesso à saúde ainda enfrenta desafios estruturais, a colecistostomia percutânea tem ganhado espaço como opção terapêutica em hospitais públicos e privados. Estudos locais destacam sua aplicação em pacientes idosos, portadores de doenças cardiovasculares avançadas ou em uso de anticoagulantes, perfis frequentes em nossa população.
Este artigo abordará as indicações, técnicas, complicações e resultados da colecistostomia, com foco na prática do cirurgião do aparelho digestivo.
Indicações da Colecistostomia
A colecistostomia é indicada principalmente em:
- Colecistite aguda refratária ao tratamento clínico em pacientes com alto risco cirúrgico (ASA ≥ III).
- Pacientes com distúrbios de coagulação (em uso de anticoagulantes ou com cirrose hepática).
- Colecistite acalculosa em pacientes críticos (ex.: internados em UTI).
- Gestantes no terceiro trimestre com colecistite aguda.
- Obstrução biliar em pacientes inaptos para CPRE.
Segundo as Diretrizes de Tóquio (TG13), a colecistostomia é recomendada para colecistite moderada (grau II) ou grave (grau III) não responsiva a antibioticoterapia. No Brasil, dados do Colégio Brasileiro de Cirurgiões apontam que até cerca de 5% dos casos de colecistite aguda podem ser tratados inicialmente com colecistostomia, especialmente em hospitais de referência.
Técnicas de Colecistostomia
- Percutânea (Guiada por US/TC):
- Via transhepática (preferencial para reduzir vazamento biliar).
- Via transperitoneal (menos utilizada devido a maior risco de complicações).
- Técnica de Seldinger ou trocarte direto.
- Laparoscópica:
- Opção quando a colecistectomia é inicialmente planejada, mas abortada devido a inflamação intensa.
- Cirúrgica Aberta:
- Reservada para falha das técnicas minimamente invasivas.
Aplicação na Cirurgia Digestiva
A colecistostomia tem papel crucial no manejo escalonado da colecistite aguda:
- Fase aguda: Alívio da sepse biliar.
- Fase definitiva: Decisão entre remoção do cateter ou colecistectomia tardia.
Estudos brasileiros demonstram que 30-40% dos pacientes submetidos à colecistostomia evoluem para colecistectomia eletiva, enquanto os demais mantêm o cateter como tratamento definitivo, principalmente idosos e pacientes com comorbidades graves.
Pontos-Chave para o Cirurgião Digestivo
- Seleção adequada do paciente é fundamental para evitar complicações.
- Via transhepática reduz vazamento biliar e deslocamento do cateter.
- Antibioticoterapia pré e pós-procedimento é essencial para evitar sepse.
- Remoção do cateter após 4-6 semanas (avaliar maturação do trajeto fistuloso).
- Monitorar complicações:
- Deslocamento do cateter (27%).
- Vazamento biliar (3-6%).
- Abscesso peri-hepático (9%).
Conclusões Aplicadas à Prática
A colecistostomia é uma ferramenta valiosa no tratamento da colecistite aguda em pacientes de alto risco. No cenário brasileiro, onde a população envelhecida e as comorbidades cardiovasculares são prevalentes, seu uso deve ser considerado de forma individualizada.
Embora a colecistectomia laparoscópica permaneça como padrão-ouro, a colecistostomia oferece uma alternativa segura e eficaz, reduzindo mortalidade e tempo de internação. Futuros estudos prospectivos, como o CHOCOLATE trial, trouxeram mais evidências sobre seu papel definitivo.
“A cirurgia é a arte de salvar vidas, mas também de saber quando não operar.” — William Stewart Halsted
Gostou? ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Colecistostomia #CirurgiaDigestiva #ColecistiteAguda #MedicinaCirúrgica #ResidênciaMédica
Câncer Incidental da Vesícula Biliar após Colecistectomia Laparoscópica: Diagnóstico e Conduta Cirúrgica
Introdução
O câncer de vesícula biliar é a neoplasia maligna mais comum do trato biliar e o quinto tumor mais frequente do trato gastrointestinal. Apesar disso, apresenta baixa incidência global — cerca de 3 casos por 100.000 habitantes/ano nos EUA — e geralmente é diagnosticado em estágios avançados, com taxa de ressecabilidade de apenas 25% e sobrevida média de 3 a 11 meses. O cenário muda quando a doença é detectada precocemente, especialmente nos casos de câncer incidental da vesícula biliar (IGBC), diagnosticado após colecistectomia laparoscópica (LC) realizada por outra indicação. O IGBC ocorre em 0,3–2% das LC e representa cerca de dois terços dos casos potencialmente curáveis.
Epidemiologia e Relevância Clínica
- Incidência: 0,3–2% das colecistectomias laparoscópicas.
- Impacto prognóstico: sobrevida de 5 anos pode atingir 92–100% nos estágios iniciais (T1a).
- Risco de disseminação: perfuração vesicular e derrame biliar intraoperatório elevam o risco de recidiva peritoneal e em portais (até 40%).
Diagnóstico Pós-Operatório
Quando o câncer é detectado no exame histopatológico após a colecistectomia, o primeiro passo é:
- Revisar o laudo histológico — confirmando profundidade de invasão (T) e status da margem do ducto cístico.
- Estadiamento por imagem — geralmente com tomografia computadorizada (TC) de tórax, abdome e pelve, podendo ser complementada por ressonância magnética (RM/MRCP).
- Avaliar presença de doença irressecável — até 20% dos pacientes candidatos à reoperação apresentam doença avançada na reexploração.
Conduta por Estadiamento T
- Tis / T1a
- LC isolada é curativa se não houve perfuração vesicular nem extravasamento biliar.
- Metástases linfonodais ocorrem em apenas 2% dos casos.
- T1b / T2
- Indicam re-resecção radical, incluindo:
- Reseção não anatômica de segmentos hepáticos 4b e 5 (margem ≥ 2–3 cm).
- Linfadenectomia do ligamento hepatoduodenal.
- Avaliação da margem do ducto cístico; se positiva, ampliar ressecção.
- Sobrevida de 5 anos: 60–100% (T1b) e 54–100% (T2).
- Indicam re-resecção radical, incluindo:
- T3 / T4
- Ressecção curativa apenas se R0 possível.
- Sobrevida de 5 anos em doença nodal negativa: até 77%.
- Em linfonodos positivos além do hepatoduodenal: prognóstico extremamente reservado (<1%).
Suspeita Intraoperatória
Fatores que devem acender o alerta:
- Vesícula endurecida e espessada.
- Infiltração hepática.
- Paciente com mais de 60 anos, especialmente com quadro de empiêma vesicular.
- Achados de imagem pré-operatória: espessamento focal ou difuso da parede, linfonodomegalias, vesícula pouco distendida.
Conduta recomendada:
- Se possível, interromper a cirurgia e encaminhar para estadiamento completo antes de definir tratamento definitivo.
- Evitar biópsia intraoperatória devido a risco de erro amostral e disseminação.
- Em casos de alta suspeita e condições clínicas favoráveis, realizar colecistectomia radical imediata.
Técnica da Re-Ressecção Radical
- Abordagem convencionalmente aberta (há relatos laparoscópicos e robóticos).
- Ressecção hepática (segmentos 4b e 5).
- Linfadenectomia hepatoduodenal.
- Ressecção do ducto cístico ± hepático comum se margem comprometida.
- Não há benefício comprovado na ressecção sistemática dos portais de entrada da laparoscopia.
Pontos-Chave para o Cirurgião Digestivo
- Sempre encaminhar espécimes de colecistectomia para exame histopatológico.
- Documentar integridade da vesícula e ausência de extravasamento biliar.
- Em IGBC T1b ou superior, a re-resecção precoce oferece ganho substancial de sobrevida.
- Envolver equipe de cirurgia hepatobiliar e oncologia desde o início.
- RO (ressecção com margem livre) é o fator mais importante para prognóstico.
Conclusão Aplicada
O IGBC representa uma oportunidade única de diagnóstico precoce de um tumor tipicamente letal. A conduta correta, baseada no estadiamento, pode transformar um achado acidental em um caso curável. A chave está em:
- Reconhecimento imediato do risco.
- Estadiamento preciso.
- Tratamento radical oportuno.
“O cirurgião deve ter a coragem de parar quando é cedo demais e a ousadia de agir quando ainda é tempo.” — Halsted
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#CâncerDeVesícula #Colecistectomia #CirurgiaHepatobiliar #OncologiaCirúrgica #EducaçãoMédicaContinuada
Colecistectomia Laparoscópica em Pacientes Cirróticos: Estratégias, Truques e Cuidados Especiais
Introdução
A prevalência de colelitíase em pacientes com cirrose é significativamente maior que na população geral, atingindo quase um terço desses indivíduos. Em cerca de 80% dos casos, os cálculos são assintomáticos; no restante, manifestam-se como cólica biliar, colecistite ou icterícia obstrutiva. Apesar de a colecistectomia laparoscópica ser considerada padrão-ouro no tratamento da colelitíase sintomática, a presença de cirrose impõe desafios técnicos e riscos adicionais, como maior propensão a sangramento, infecção e insuficiência hepática pós-operatória. Em pacientes com Child-Pugh A ou B, a laparoscopia é segura quando realizada por equipes experientes e em centros com suporte especializado. Já na classe C, o risco é proibitivo, sendo preferível manejo não cirúrgico ou encaminhamento para avaliação de transplante hepático.
Avaliação Pré-Operatória e Estratificação de Risco
A decisão cirúrgica deve considerar:
- Classificação de Child-Pugh: risco de mortalidade de 10% (A), 30% (B) e até 80% (C) em cirurgias não hepáticas.
- MELD score: preditor acurado de mortalidade; acima de 25, a mortalidade em 90 dias pode chegar a 90%.
- Presença de hipertensão portal: aumenta risco de sangramento e dificulta a dissecção.
- Comorbidades associadas: insuficiência renal, cardiopatia e doença pulmonar podem agravar prognóstico.
Exames complementares recomendados incluem doppler hepático para avaliação vascular, tomografia ou ressonância contrastada para análise anatômica e rastreio de neoplasias associadas.
Indicações e Momentos da Cirurgia
- Colelitíase sintomática em Child A ou B, sem colecistite aguda grave, é indicação para colecistectomia laparoscópica.
- Colecistite aguda: pode-se optar por tratamento inicial não operatório (antibioticoterapia, drenagem percutânea ou stent transpapilar) e cirurgia eletiva tardia.
- Achado incidental de cirrose durante colecistectomia: considerar abortar procedimento e encaminhar para avaliação multidisciplinar, exceto se quadro for emergencial.
Truques e Estratégias Intraoperatórias
1. Acesso e posicionamento
- Uso preferencial da técnica aberta (Hasson) para o primeiro trocárter, evitando lesão da veia umbilical recanalizada.
- Colocação do porto de trabalho no quadrante superior esquerdo, para reduzir risco de sangramento da parede abdominal.
- Documentar grau de cirrose e hipertensão portal com fotos intraoperatórias.
2. Dissecção e controle vascular
- Expectativa de aderências neovascularizadas; dissecção com Harmonic® ou Ligasure™ reduz sangramento.
- No Calot difícil devido a colaterais, considerar colecistectomia parcial: deixar parte da parede vesicular aderida ao fígado, com cauterização da mucosa para prevenir mucocele.
- Em situações críticas, usar clipes Hem-o-lok®, Endoloop® ou sutura intracorpórea no coto cístico, preferindo ligadura segura antes da secção.
3. Estratégias alternativas
- Abordagem fundus-first pode ser útil, mas requer atenção para não lesar ducto biliar.
- Conversão para cirurgia aberta deve ser encarada como decisão de segurança, e não falha técnica.
4. Controle de sangramento
- Ter à disposição argon beam coagulator, hemostáticos tópicos e instrumentos de cirurgia aberta.
- Monitorar constantemente o campo cirúrgico; portos devem ser removidos sob visão direta.
Cuidados Pós-Operatórios
- Evitar sobrecarga hídrica para prevenir ascite e complicações respiratórias.
- Monitorar função renal e eletrólitos; hiponatremia é frequente.
- Utilizar albumina e dieta hipossódica conforme orientação hepatológica.
- Vigilância para complicações como sangramento, vazamento biliar e encefalopatia hepática.
Conclusão
A colecistectomia laparoscópica em pacientes cirróticos não é uma contraindicação absoluta, mas exige seleção criteriosa, preparo pré-operatório rigoroso e técnica cirúrgica refinada. Pacientes Child-Pugh A ou B, operados por equipes experientes em centros terciários, apresentam resultados seguros e comparáveis à população geral em termos de mortalidade. O conhecimento de truques técnicos, manejo de complicações e decisão sobre momento ideal da cirurgia são determinantes para o sucesso.
“O bom cirurgião sabe o que pode fazer; o excelente sabe o que deve fazer.” — Halsted
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#ColecistectomiaLaparoscopica #CirroseHepatica #CirurgiaHepatobiliar #LaparoscopiaAvançada #EducaçãoMédicaContinuada
Apendicectomia Laparoscópica: Truques e Dicas para uma Execução Segura e Eficiente
Introdução
A apendicectomia laparoscópica consolidou-se como o padrão-ouro no tratamento da apendicite aguda, sobretudo em centros com acesso à tecnologia minimamente invasiva. Com benefícios amplamente documentados — como menor dor pós-operatória, redução do tempo de internação e menor incidência de infecção de ferida —, a técnica exige, contudo, treinamento e atenção a detalhes técnicos. Este artigo oferece truques e dicas práticas que podem otimizar a performance do cirurgião, reduzir complicações e melhorar a curva de aprendizado da equipe assistente.
1. Posicionamento do Paciente e da Equipe
- Decúbito dorsal com leve Trendelenburg e rotação à esquerda facilita a exposição do quadrante inferior direito.
- Fixe o braço direito do paciente ao corpo para permitir amplo espaço de movimentação do cirurgião.
- Cirurgião à esquerda do paciente, assistente ao lado da perna esquerda, monitor preferencialmente à direita ou à cabeceira, na linha dos ombros.
Dica: Ajuste fino da inclinação da mesa pode ser decisivo para deslocar alças e expor o ceco sem necessidade de manobras agressivas.
2. Posicionamento dos Trocárteres
- Um padrão eficiente inclui:
- Trocárter de 10 mm umbilical (ótica).
- Trocárter de 5 mm em hipogástrio (instrumentação dominante).
- Trocárter de 5 mm em flanco esquerdo (tração e dissecção).
Truque: Em pacientes obesos, insira o trocárter ótico com cuidado em ângulo oblíquo para evitar desinserção do pneumoperitônio e garantir estabilidade.
3. Estratégias de Exposição
- Identifique o teniae coli do ceco e siga até a base do apêndice.
- Use pinça atraumática para tração superior do apêndice, expondo sua base.
- Em casos de aderências, libere-as com energia monopolar delicada ou tesoura, evitando avulsões inadvertidas.
Dica de ouro: Evite “lutar” contra aderências retrocecais. Mude o plano, reposicione a câmera, varie o ângulo de dissecção. Tempo gasto com exposição segura evita complicações graves.
4. Controle do Pedículo e Secção Apendicular
- O método mais utilizado é o uso de duas ligaduras com endoloop ou clips poliméricos (Hem-o-lok®), seguido de secção entre eles.
- Alternativamente, grampeadores laparoscópicos podem ser usados, especialmente em apêndices friáveis ou bases espessadas.
Truque técnico: Em apêndices muito inflamados, realize a ligadura mais distal antes da proximal, para reduzir o risco de ruptura ou vazamento ao manipular a base.
5. Retirada e Proteção da Cavidade
- Retire o apêndice com saco cirúrgico sempre que possível, evitando contaminação do trajeto do trocárter.
- Irrigue abundantemente a loja apendicular se houver peritonite localizada ou pus livre.
- Se necessário, coloque dreno tubular por 24 a 48 horas.
Dica prática: Em caso de dúvida quanto à integridade da base, deixe um fragmento do ceco visível e documente o aspecto final com imagem.
6. Situações Especiais
- Apêndice retrocecal: requer liberação ampla da reflexão lateral direita do cólon.
- Apendicite perfurada com abscesso: considere drenagem inicial guiada por imagem e apendicectomia em intervalo.
- Apendicite gestacional: ideal até o segundo trimestre. Atenção ao deslocamento anatômico do apêndice.
Truque anatômico: Em gestantes ou crianças, a mobilidade intestinal pode mascarar a localização clássica. Reforce a busca sistemática do apêndice pela convergência das teníases do ceco.
Conclusão
A apendicectomia laparoscópica é uma cirurgia segura, eficaz e que continua evoluindo com a incorporação de técnicas assistidas por imagem, navegação e inteligência artificial. No entanto, sua execução requer atenção a detalhes aparentemente simples, que fazem toda a diferença nos desfechos clínicos. O domínio dos truques e dicas técnicas aqui apresentados contribui significativamente para uma prática cirúrgica mais segura, eficiente e baseada em excelência técnica.
“A simplicidade técnica não dispensa o rigor; é justamente na cirurgia simples que se exige a perfeição.” — René Leriche
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Inteligência Artificial & Apendicite Aguda
Uso da Inteligência Artificial Generativa no Diagnóstico, Tratamento Cirúrgico e Avaliação de Prognóstico da Apendicite Aguda
Introdução
A apendicite aguda continua sendo a causa mais comum de abdome agudo cirúrgico no mundo, com incidência estimada de 100 a 150 casos por 100.000 habitantes ao ano. No Brasil, representa uma das principais causas de internação cirúrgica no SUS. Apesar de amplamente estudada e com tratamento bem estabelecido, a apendicite aguda ainda apresenta desafios clínicos, como o diagnóstico precoce, a decisão operatória e a estratificação de risco de complicações. Nesse contexto, a Inteligência Artificial Generativa (IAG) emerge como uma tecnologia disruptiva com potencial transformador na abordagem cirúrgica dessa condição.
Diagnóstico com Apoio de IA Generativa
O diagnóstico precoce da apendicite aguda depende da correlação de dados clínicos, laboratoriais e de imagem. A IAG, treinada com grandes volumes de dados de prontuários eletrônicos, imagens de tomografia e ultrassonografias, pode sintetizar essas informações em tempo real para gerar hipóteses diagnósticas com acurácia comparável — e, em alguns casos, superior — à avaliação humana isolada. Modelos generativos baseados em deep learning, como os transformers, são capazes de identificar padrões sutis de apresentação clínica atípica, sobretudo em populações vulneráveis, como crianças pequenas, idosos e gestantes. Um exemplo prático é o uso de sistemas que combinam texto livre do prontuário com imagens de abdome para gerar relatórios diagnósticos automatizados e sugerir scores clínicos (como Alvarado ou AIR score) com ajuste probabilístico personalizado, auxiliando o cirurgião na tomada de decisão.
Tratamento Cirúrgico Guiado por IA
A cirurgia laparoscópica é o padrão ouro no tratamento da apendicite aguda, mas a escolha do momento cirúrgico e a abordagem ideal ainda são decisões dependentes da experiência do cirurgião e das condições clínicas do paciente. A IAG pode ser integrada a sistemas de suporte intraoperatório, auxiliando em navegação cirúrgica assistida por imagem, sugestão de condutas intraoperatórias com base em bancos de dados operatórios, e até mesmo em treinamento em simulações realistas de apendicectomia via realidade aumentada e modelos generativos de anatomia personalizada.
Em cenários de medicina robótica, já há estudos utilizando IA generativa para gerar “scripts” de procedimentos otimizados e oferecer feedback em tempo real com base em dados históricos de performance técnica — promovendo cirurgias mais seguras, rápidas e com menor taxa de complicações.
Avaliação Prognóstica com IA Generativa
A estratificação de risco no pós-operatório é outro campo onde a IAG pode oferecer avanços concretos. A partir de redes neurais treinadas em desfechos de milhares de pacientes com apendicite, esses modelos podem prever probabilidades de complicações como abscesso residual, íleo paralítico ou necessidade de reabordagem, considerando variáveis clínicas, laboratoriais, intraoperatórias e demográficas. Além disso, ferramentas baseadas em IA podem gerar relatórios individualizados de prognóstico funcional, tempo estimado de retorno às atividades e risco de reinternação, permitindo ao cirurgião personalizar o plano de alta, acompanhamento ambulatorial e orientações ao paciente com maior precisão.
Pontos-Chave
- A IA generativa tem capacidade de analisar simultaneamente dados clínicos, laboratoriais e de imagem para melhorar o diagnóstico precoce da apendicite aguda.
- Pode ser integrada à decisão cirúrgica, tanto no pré quanto no intraoperatório, por meio de simulação, navegação assistida e análise de performance técnica.
- Modelos preditivos baseados em IA oferecem ferramentas objetivas para prognóstico pós-operatório personalizado, otimizando o seguimento do paciente.
- O uso de IAG deve ser encarado como ferramenta complementar ao raciocínio clínico do cirurgião, jamais como substituto da experiência e do julgamento médico.
- O avanço dessas tecnologias requer formação crítica e ética dos futuros cirurgiões, capacitando-os para liderar a incorporação segura e eficaz dessas ferramentas na prática assistencial.
Conclusão
A incorporação da Inteligência Artificial Generativa na cirurgia do aparelho digestivo representa um novo paradigma na medicina personalizada, baseada em dados e centrada no paciente. No caso da apendicite aguda — uma das mais frequentes emergências cirúrgicas —, o uso responsável e crítico da IAG pode aprimorar significativamente os pilares do cuidado: diagnóstico, decisão operatória e prognóstico. O cirurgião do futuro será, acima de tudo, um líder clínico capaz de integrar inteligência humana e artificial com sabedoria e discernimento ético.
“A tecnologia não substitui o cirurgião. Ela amplia sua visão, mas é o julgamento humano que decide a incisão.” — Sir John Black, ex-presidente do Royal College of Surgeons
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#InteligenciaArtificialNaMedicina #CirurgiaDoAparelhoDigestivo #ApendiciteAguda #DiagnósticoComIA #CirurgiaGuiadaPorDados
Íleo Adinâmico e Íleo Pós-Operatório: Compreensão Atual e Aplicações na Cirurgia Digestiva
Introdução
A motilidade intestinal no pós-operatório abdominal é uma área frequentemente mal compreendida, apesar de sua importância clínica. Termos como íleo pós-operatório (POI, do inglês postoperative ileus) e íleo adinâmico são frequentemente utilizados de forma intercambiável, mas representam entidades fisiopatológicas distintas. Compreender suas diferenças é crucial para a conduta apropriada e para evitar intervenções desnecessárias. Neste artigo, abordamos as bases fisiológicas e clínicas dessas condições, diferenciando-as de forma clara, com foco na prática da cirurgia digestiva.
Íleo Pós-Operatório (POI)
O POI é uma resposta fisiológica previsível que acomete predominantemente o estômago e o cólon nas primeiras 24 a 96 horas após uma laparotomia. Ao contrário da crença comum, o íleo verdadeiro do intestino delgado é raro nesse contexto. Estudos intraoperatórios demonstram que o intestino delgado mantém atividade contrátil mesmo durante o procedimento, enquanto o estômago e o cólon mostram inatividade motora, mesmo com estímulos.
A ausência de ruídos hidroaéreos típicos do POI deve-se, sobretudo, à falta de progressão do gás deglutido, não a uma ausência de motilidade intestinal. O mito de que a manipulação extensa do intestino delgado prolonga o POI foi desmentido por estudos experimentais, sugerindo que o trauma da parede abdominal e o uso de opioides são os principais fatores desencadeantes.
Com o avanço das técnicas minimamente invasivas e dos protocolos de recuperação precoce (ERAS), o impacto clínico do POI tornou-se cada vez menos relevante, sobretudo após cirurgias laparoscópicas, como a colectomia laparoscópica ou a gastrectomia subtotal com reconstrução em Y-de-Roux.
Íleo Adinâmico
O íleo adinâmico verdadeiro é uma condição patológica e generalizada, com bloqueio da motilidade em todo o trato gastrointestinal: estômago, intestino delgado e cólon. Trata-se de uma disfunção neuromuscular reflexa secundária, frequentemente associada a quadros sistêmicos como sepse, trauma retroperitoneal, hematomas ou cirurgias retroperitoneais (ex: transplante renal). Essas condições interferem na inervação autonômica visceral, levando à inatividade motora intestinal completa.
Clinicamente, manifesta-se com distensão abdominal, náuseas, vômitos, obstipação completa (fezes e flatos) e achados radiológicos de distensão gástrica, de delgado e cólon. Diferente da obstrução mecânica, a dor cólica é incomum. O diagnóstico é clínico e radiológico, sendo essencial excluir causas obstrutivas distais, como neoplasias retais (por proctoscopia ou enema opaco).
O tratamento é conservador e de suporte, com correção do fator causal subjacente (geralmente sepse). A introdução de pró-cinéticos ou intervenções cirúrgicas raramente se justifica e pode ser prejudicial.
Aplicação na Cirurgia Digestiva
Na cirurgia digestiva, o conhecimento detalhado desses fenômenos tem implicações práticas relevantes:
- A retomada precoce da dieta por sonda jejunal ou via oral, mesmo após grandes ressecções, é segura e desejável, uma vez que o intestino delgado se mantém funcional no pós-operatório imediato.
- O uso criterioso de opioides deve ser avaliado, já que retarda a motilidade gástrica e colônica e prolonga o POI.
- Cirurgias minimamente invasivas devem ser priorizadas sempre que possível, devido à menor resposta inflamatória e menor impacto na motilidade intestinal.
- Reconhecer o íleo adinâmico é essencial para evitar reoperações desnecessárias, já que o problema é sistêmico e autolimitado com a resolução da causa de base.
Pontos-Chave
- POI é uma resposta fisiológica transitória que acomete estômago e cólon, e não o intestino delgado.
- O íleo adinâmico é uma condição patológica que afeta todo o tubo digestivo, geralmente por causas sistêmicas ou retroperitoneais.
- Manipulação do intestino delgado não prolonga o POI — o trauma parietal e o uso de opioides são mais determinantes.
- A abordagem laparoscópica e os protocolos ERAS diminuem drasticamente a incidência e a gravidade do POI.
- O tratamento do íleo adinâmico é conservador e depende da resolução da causa sistêmica subjacente.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
Para o cirurgião do aparelho digestivo, distinguir entre POI e íleo adinâmico é essencial para um manejo racional e eficaz. A compreensão das bases fisiológicas permite evitar exames desnecessários, intervenções precipitadas e retardos no cuidado. Além disso, reforça a importância das estratégias modernas de recuperação precoce, da analgesia multimodal sem opioides, e da valorização das vias de acesso minimamente invasivas. Saber esperar e não intervir é, por vezes, o ato mais sofisticado da prática cirúrgica.
“Conhecer o tempo certo de não operar é tão vital quanto saber quando operar.” — William Stewart Halsted
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#IleoPosOperatorio #CirurgiaDigestiva #IleoAdinamico #ERASprotocol #Laparoscopia
Prevenção das Aderências Pós-Operatórias
Introdução
As aderências pós-operatórias continuam sendo uma das principais complicações após cirurgias abdominais, impactando diretamente a qualidade de vida dos pacientes e os custos para o sistema de saúde. No contexto brasileiro, estima-se que até 35% das reoperações abdominais sejam causadas por aderências, contribuindo para obstruções intestinais, infertilidade, dor abdominal crônica e aumento do tempo cirúrgico em procedimentos subsequentes. A profilaxia eficaz destas formações fibrosas representa, portanto, um imperativo técnico e ético para o cirurgião do aparelho digestivo.
Desenvolvimento
A fisiopatologia das aderências envolve uma resposta inflamatória exacerbada da cavidade peritoneal à manipulação cirúrgica, à presença de corpos estranhos e à isquemia tecidual. Tais estímulos promovem a deposição de fibrina, que, na ausência de fibrinólise adequada, culmina na formação de pontes fibrosas entre órgãos e estruturas adjacentes.
As medidas preventivas devem ser iniciadas desde o planejamento operatório. O uso de técnicas cirúrgicas meticulosas é um dos pilares da prevenção. Manipular delicadamente os tecidos, evitar dissecções desnecessárias, garantir hemostasia rigorosa e eliminar corpos estranhos (como talco das luvas ou gaze fragmentada) são princípios fundamentais. Além disso, a preferência pela abordagem laparoscópica sempre que possível se justifica, já que estudos demonstram que ela reduz significativamente a formação de aderências em comparação com a cirurgia aberta.
Outra estratégia complementar é o uso de barreiras mecânicas antiaderentes bioabsorvíveis. Produtos como a membrana de ácido hialurônico/carboximetilcelulose (Seprafilm®) e a solução de icodextrina 4% (Adept®) têm evidências robustas de eficácia, sendo recomendados especialmente em procedimentos ginecológicos e digestivos de médio e grande porte. Estudos recentes também apontam novas perspectivas com o uso de polímeros zwitteriônicos, capazes de prevenir completamente aderências em modelos animais, embora ainda sem validação clínica em humanos.
Aplicação na Cirurgia Digestiva
Na prática da cirurgia do aparelho digestivo, especialmente em procedimentos como colectomias, gastrectomias, ressecções intestinais e cirurgias hepatopancreatobiliares, a prevenção de aderências é estratégica para evitar complicações precoces e tardias. A obstrução intestinal por bridas, por exemplo, representa até 20% das admissões de emergência cirúrgica abdominal nos hospitais terciários brasileiros.
A adoção da laparoscopia como via de acesso padrão, sempre que factível, deve ser incentivada como política institucional. Do mesmo modo, o emprego racional de barreiras antiaderentes é indicado especialmente em pacientes com histórico de múltiplas cirurgias abdominais, doenças inflamatórias intestinais ou em situações em que a reintervenção futura é previsível (como na cirurgia oncológica com perspectiva de reabordagem).
Pontos-Chave
- Aderências pós-operatórias ocorrem em até 93% das laparotomias, sendo a causa de 60% das obstruções intestinais por bridas.
- Técnicas cirúrgicas meticulosas são a medida preventiva mais eficaz e de menor custo.
- A laparoscopia reduz substancialmente a formação de aderências em comparação com a cirurgia aberta.
- Barreiras antiaderentes, como Seprafilm® e Adept®, têm indicação em cirurgias abdominais de maior complexidade.
- Inovações como polímeros zwitteriônicos prometem novas abordagens, mas ainda requerem validação clínica.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A prevenção das aderências pós-operatórias deve ser abordada como uma extensão da técnica cirúrgica refinada. Incorporar práticas baseadas em evidências, priorizar o uso de tecnologias minimamente invasivas e adotar dispositivos antiaderentes quando indicados são medidas que não apenas melhoram os desfechos clínicos, mas também reduzem o ônus econômico e o sofrimento dos pacientes.
A educação continuada do cirurgião digestivo é fundamental para manter-se atualizado quanto às inovações tecnológicas e diretrizes internacionais. Em tempos de medicina baseada em valor, prevenir complicações previsíveis como as aderências é um ato de excelência e responsabilidade profissional.
“Jamais devemos nos contentar em apenas operar. Devemos operar melhor, sempre.” — Alexis Carrel
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
O Bisturi
Ele é longo e pontudo.
Não precisa ser afiado —
em sua forma reside
a perfeição. Um instrumento
para penetrar de um modo
limpo e preciso.
Seja em um corte
no flanco, nas costas
ou atravessando
tecidos delicados,
seu efeito é vital.
Na mão do cirurgião,
não traz a morte,
mas a cura:
ferida que sara,
toque que liberta,
anatomia exata
entre a mão e a vida.
Fundamentos Filosóficos da Prática Cirúrgica Contemporânea
Reflexões para Cirurgiões do Aparelho Digestivo
Introdução
A conexão entre cirurgia e filosofia vai além da prática técnica, envolvendo uma reflexão profunda sobre os impactos éticos e humanos da profissão. Inspirado pela obra de Yves Chapuis e por grandes pensadores como Sócrates e Heidegger, este artigo examina como fundamentos filosóficos moldam a prática cirúrgica contemporânea, especialmente na cirurgia do aparelho digestivo, propondo um diálogo entre técnica, ética e humanização. A prática cirúrgica exige mais do que habilidade técnica. Ela demanda do cirurgião um compromisso ético, psicológico e social. Exploramos aqui 10 tópicos que integram esses fundamentos, demonstrando sua relevância na cirurgia digestiva:
Aplicação na Cirurgia Digestiva: 10 Tópicos Fundamentais
- Cirurgia e Filosofia
A filosofia aplicada à cirurgia busca compreender as causas primeiras e os valores humanos. A prática cirúrgica, enquanto combate às doenças, exige reflexão sobre o impacto dos atos no paciente e na sociedade, alinhando técnica e moralidade. - Indignação como Ponto de Partida
Chapuis, ecoando Sócrates e Heidegger, destaca a indignação diante do sofrimento humano como o motor da filosofia e, por extensão, da cirurgia. O cirurgião não apenas observa, mas age para transformar a realidade, aliviando a dor e restaurando a dignidade. - Aspecto Moral da Cirurgia
O avanço técnico amplia as possibilidades terapêuticas, mas exige maior responsabilidade ética. Como afirmou Ollier: “Quanto mais poderosa a cirurgia se torna, mais moral ela deve ser.” No Brasil, isso é crucial para lidar com as disparidades no acesso a procedimentos como transplantes hepáticos. - Perigos da Supremacia Técnica
A dependência excessiva da tecnologia pode desumanizar a prática. Como alertou Jean-François Mattei: “Se o médico dedica mais tempo ao seu computador do que ao seu paciente, algo está errado.” O foco deve permanecer no paciente, não apenas na doença. - Desafios Psicológicos do Cirurgião
O desgaste emocional, a monotonia e a pressão social afetam a saúde mental dos cirurgiões. Reflexões regulares sobre os desafios enfrentados podem ajudar a equilibrar a prática com o bem-estar pessoal. - Necessidade de Adaptação e Aprendizado Contínuo
A resistência às mudanças limita o progresso. Para enfrentar as inovações como a laparoscopia ou a robótica, é necessário um aprendizado constante e uma mente aberta, valorizando o impacto positivo dessas técnicas no tratamento de doenças como câncer gástrico. - Dimensão Coletiva e Social
A prática cirúrgica não é isolada, mas afeta diretamente o acesso aos cuidados de saúde. No Brasil, regiões periféricas sofrem com a falta de infraestrutura e profissionais qualificados, destacando a necessidade de políticas públicas mais inclusivas. - Responsabilidade com o Corpo e o Tempo
A íntima relação do cirurgião com o corpo humano é única. Como reflete Saint Augustin: “Se você não me pergunta o que é o tempo, eu sei o que ele é; mas, assim que você me pergunta, eu já não sei mais.” O tempo dedicado ao paciente, muitas vezes negligenciado, é um reflexo do compromisso ético e técnico do cirurgião. - Confronto com a Morte
A cirurgia é uma batalha constante contra a morte. Valéry define-a como “Combater o mal com as próprias mãos armadas.”, enquanto Bichat a vê como “Destruir as forças que se opõem à vida” Este enfrentamento exige equilíbrio entre ousadia terapêutica e limites éticos. - Desafios Futuros com a Genética
Avanços na genética trazem novos dilemas, como intervenções preventivas em indivíduos predispostos a doenças digestivas. A antecipação ética e a relação de confiança com o paciente tornam-se ainda mais importantes.
Pontos-Chave
- Humanização: A cirurgia é uma prática que conecta técnica e cuidado humano.
- Ética e Progresso: Avanços devem ser acompanhados de responsabilidade moral.
- Educação e Reflexão: Cirurgiões precisam de constante aprendizado e autocrítica.
- Desafios Brasileiros: A desigualdade regional exige ações concretas para ampliar o acesso.
- Sustentabilidade e Futuro: A integração de novas tecnologias deve considerar a realidade socioeconômica.
Conclusões
A prática cirúrgica contemporânea requer dos profissionais uma abordagem que mescle técnica, ética e reflexão filosófica. Inspirados por Chapuis, podemos construir um modelo que priorize o paciente, enfrente desigualdades e avance de forma responsável. A cirurgia do aparelho digestivo, com seus desafios únicos, é um campo que exemplifica essa necessidade de equilíbrio.
Como disse René Leriche:
“Todo cirurgião carrega consigo um cemitério, ao qual ele deve voltar de tempos em tempos para meditar.”
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#CirurgiaDigestiva #FilosofiaNaMedicina #HumanizaçãoNaSaúde #InovaçãoCirúrgica #EducaçãoMédica
Colecistectomia Robótica
A colecistectomia, procedimento cirúrgico para remoção da vesícula biliar, pode ser realizada por diferentes abordagens, sendo as mais comuns a via laparoscópica (1985), a assistida por robô (1987) e a cirurgia convencional ou aberta (1882). Cada uma dessas técnicas tem vantagens e desvantagens em termos de custo, eficácia clínica e complexidade, o que torna a análise de custo-efetividade crucial para a escolha da abordagem mais adequada em cada caso.
Colecistectomia Laparoscópica (CVL)
A colecistectomia laparoscópica (CVL) é amplamente reconhecida como a técnica de escolha devido à sua eficácia, recuperação rápida e menor custo comparado a outras abordagens. Diversos estudos confirmam que a laparoscopia é a opção mais custo-efetiva para a maioria dos pacientes com doenças benignas da vesícula biliar, como a colelitíase, devido ao menor custo direto das etapas cirúrgicas.
Estudos de Custo-efetividade indicam que o custo variável direto para o cirurgião na execução de uma colecistectomia laparoscópica foi significativamente inferior ao de técnicas mais complexas. A via laparoscópica custou, em média, $929, enquanto a colecistectomia assistida por robô (ROBOSILS) alcançou o custo de $2,608. Isso demonstra que a laparoscopia tem uma vantagem substancial em termos de custo sem comprometer a qualidade do procedimento.
Colecistectomia Robótica
Por outro lado, a colecistectomia assistida por robô tem ganhado popularidade devido à promessa de maior precisão, visualização e controle durante a cirurgia. No entanto, sua viabilidade econômica tem sido amplamente discutida na literatura, com estudos demonstrando que, embora seja uma técnica segura, os custos hospitalares associados a ela são consideravelmente mais altos. Em um estudo de caso-controle, os custos totais hospitalares para a cirurgia robótica foram de $7,985.4, em comparação com $6,255.3 para a laparoscopia. A maior parte desses custos adicionais vem dos gastos com a amortização e os consumíveis do sistema robótico, que são significativamente mais caros. Outra análise revelou uma razão de custo-efetividade da cirurgia robótica de $1,795,735.21 por ano de vida ajustado pela qualidade, um valor muito acima dos limiares aceitáveis para a maioria dos sistemas de saúde. Além disso, um banco de dados nacional demonstrou que, enquanto a colecistectomia robótica pode oferecer benefícios como redução do tempo de internação, os custos globais continuam elevados devido aos custos variáveis e consumíveis associados ao uso da tecnologia robótica, fazendo com que essa técnica não seja necessariamente mais custo-efetiva, mesmo em contextos que poderiam sugerir vantagens clínicas.
Colecistectomia Convencional ou Abertia (Via Laparotômica)
A colecistectomia convencional, ou aberta, apresenta indicações específicas, sendo geralmente reservada para situações onde a abordagem laparoscópica não oferece segurança ou viabilidade adequada. Esta modalidade cirúrgica encontra suas principais indicações em casos de inflamação severa, particularmente na colecistite aguda complicada, onde o risco de complicações durante a laparoscopia se mostra elevado, bem como em pacientes com histórico de múltiplas intervenções abdominais prévias que resultaram em aderências extensas. A suspeita de malignidade da vesícula biliar também constitui indicação relevante para a abordagem aberta, dada a necessidade de acesso mais amplo para ressecção e avaliação adequada. Ademais, pacientes com condições médicas que contraindicam anestesia geral prolongada ou que apresentam comorbidades significativas com elevado risco cirúrgico podem ser considerados candidatos à abordagem convencional, especialmente quando se antecipa a possibilidade de conversão do procedimento laparoscópico. Em determinados grupos, como idosos ou portadores de hepatopatias avançadas, a escolha entre as abordagens laparoscópica e aberta demanda análise criteriosa, ponderando-se riscos e benefícios específicos de cada caso. Desta forma, a colecistectomia convencional mantém-se como opção cirúrgica fundamental para casos complexos ou de alto risco, nos quais a segurança do paciente constitui o principal determinante da escolha terapêutica.
Indicando a Melhor Abordagem para o Paciente
A escolha da abordagem cirúrgica para a colecistectomia pode ser influenciada por uma série de fatores, incluindo a condição clínica do paciente, as características da doença e os custos. A colecistectomia laparoscópica continua sendo a técnica de escolha para a maioria dos pacientes com doenças benignas da vesícula biliar devido à sua eficácia comprovada e menor custo. Além disso, estudos sugerem que a colecistectomia robótica pode ser considerada em casos específicos, como:
- Pacientes com doença hepática avançada, onde a probabilidade de conversão para cirurgia aberta é menor e o tempo de internação é reduzido.
- Contextos de treinamento cirúrgico, onde a precisão e a visualização aprimorada podem ser cruciais.
No entanto, mesmo nesses casos, é importante ponderar os custos adicionais envolvidos e os benefícios clínicos específicos que a cirurgia robótica pode oferecer, considerando que, em geral, as vantagens não são suficientes para justificar o alto custo.
Considerações Finais sobre a Abordagem Convencional e Robótica
Embora a colecistectomia robótica tenha algumas vantagens técnicas, ela não é superior em termos de desfechos clínicos quando comparada à laparoscopia, o que faz com que a escolha por esta última, em termos de custo-benefício, seja preferível na grande maioria dos casos. A colecistectomia laparoscópica oferece uma recuperação mais rápida, menor dor pós-operatória, menos complicações e melhores resultados estéticos, com menor tempo de internação. Além disso, a técnica laparoscópica convencional tem uma mortalidade operatória baixa e um risco de lesão do ducto biliar de aproximadamente 0,5%, valor que é três vezes maior que o encontrado na cirurgia aberta. Por outro lado, a colecistectomia robótica tem se mostrado vantajosa apenas em contextos específicos, como na redução da taxa de conversão para cirurgia aberta em colecistite aguda. Contudo, o risco aumentado de lesões do ducto biliar com a abordagem robótica é uma preocupação adicional que deve ser considerada.
Conclusão
Em resumo, enquanto a colecistectomia laparoscópica continua sendo a técnica de escolha padrão devido ao seu custo-benefício superior e resultados clínicos satisfatórios, a colecistectomia assistida por robô pode ser útil em casos específicos, como doença hepática avançada ou treinamento cirúrgico. No entanto, os custos elevados da técnica robótica não são amplamente justificados por melhorias nos desfechos clínicos, o que reforça a técnica laparoscópica como a abordagem mais custo-efetiva para a maioria dos pacientes.
Pontos-Chave
- Colecistectomia Convencional : Esta técnica é indicada em casos onde a abordagem minimamente invasiva não é viável, como em colecistite aguda complicada com fistulizações ou em pacientes com histórico de múltiplas cirurgias abdominais por laparotomia. No Brasil, cerca de 10% das colecistectomias ainda são realizadas de maneira convencional, de acordo com dados do DATASUS. O procedimento por via convencional apresenta uma taxa de complicações como lesão do ducto biliar em torno de 0.15%, porém, vem com um período de recuperação mais prolongado e uma maior dor pós-operatória.
- Colecistectomia Laparoscópica: Tornou-se o padrão-ouro para a maioria dos casos devido à sua menor invasividade, resultando em menor tempo de hospitalização e recuperação mais rápida. Estudos indicam que no Brasil, aproximadamente 90% das colecistectomias são laparoscópicas. No entanto, a laparoscopia ainda carrega um risco de lesão do ducto biliar que, embora baixo (cerca de 0.5%), é três vezes maior do que na técnica aberta.
- Colecistectomia Robótica: Oferece precisão e visualização 3D, mas com um custo significativamente mais alto. Dados recentes no Brasil mostram que a prática da cirurgia robótica está crescendo, embora ainda seja limitada devido ao custo e disponibilidade de equipamentos. A técnica robótica pode reduzir a necessidade de conversão para cirurgia aberta em pacientes com condições hepáticas complicadas, mas a incidência de lesão do ducto biliar é potencialmente maior, chegando a ser até 5 vezes mais alta do que na laparoscopia, segundo alguns estudos.
Aplicação na Cirurgia Digestiva
A escolha da técnica deve ser baseada na condição do paciente, na expertise do cirurgião e na disponibilidade de recursos. No contexto brasileiro, onde o acesso a tecnologia de ponta pode ser desigual, a laparoscopia tem sido a escolha preferencial por equilibrar custo e eficácia. Entretanto, a cirurgia robótica pode ser considerada em cenários específicos, como em pacientes com doença hepática avançada ou em instituições com investimento em tecnologia robótica.
- Custo: Laparoscópica < Convencional < Robótica.
- Segurança: Convencional e Laparoscópica com taxas comparáveis de complicações, embora com diferentes perfis de risco.
- Eficácia: Todas as técnicas são eficazes, mas a laparoscópica é a mais custo-efetiva para a maioria dos casos.
- Recuperação: Laparoscópica e Robótica oferecem recuperação mais rápida.
“Nós cirurgiões devemos compreender que a tecnologia é meramente um instrumento, não uma solução milagrosa, e sua aplicação deve ser ponderada com cautela, levando em conta o equilíbrio entre segurança, desfechos clínicos e custos assistenciais.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#Colecistectomia #Laparoscopia #CirurgiaRobótica #CustoEfetividade #SaúdeDigestiva
A Medicina que Salva
Ética, Ciência e Humanismo no Tratamento Cirúrgico das Doenças do Aparelho Digestivo.
Introdução
A prática médica moderna, especialmente no campo da cirurgia do aparelho digestivo, está ancorada no progresso científico e na inovação tecnológica. Contudo, a essência da medicina não reside apenas nos avanços técnicos, mas na aplicação ética e humanística do conhecimento para aliviar o sofrimento humano. C.S. Lewis, conhecido por sua contribuição literária e teológica, trouxe à luz princípios que, mesmo fora do contexto médico, iluminam os fundamentos éticos e morais que sustentam a arte de curar. Este artigo analisa como esses princípios podem ser aplicados à prática cirúrgica, destacando a importância de integrar ciência e humanidade na busca pela cura.
Desenvolvimento
A Importância da Imaginação na Medicina
A imaginação é uma ponte essencial para alcançar verdades profundas. Na cirurgia digestiva, a imaginação é fundamental para visualizar soluções complexas, como planejar abordagens minimamente invasivas ou interpretar imagens radiológicas de maneira criativa para encontrar o melhor caminho terapêutico. A capacidade de “ver além” permite ao cirurgião inovar e adaptar-se às necessidades do paciente.
Moralidade Objetiva e Decisões Médicas
O tratamento cirúrgico é uma terapêutica universal, aplicável a todos quando indicada. Na medicina, isso se traduz através da ética profissional, que guia decisões difíceis, como quando ponderar entre tratamentos curativos e paliativos. O respeito pela dignidade humana é um pilar que sustenta essas escolhas, especialmente na oncologia digestiva.
Sacrifício e Redenção no Cuidado com o Paciente
O sacrifício de Aslan em O Leão, a Feiticeira e o Guarda-Roupa reflete o compromisso cirúrgico com a entrega pessoal. A medicina exige sacrifícios diários, desde longas horas de trabalho até decisões que podem impactar emocionalmente. A redenção ocorre quando o esforço resulta em salvar vidas ou proporcionar conforto a quem sofre.
O Conflito Entre Bem e Mal nas Escolhas Terapêuticas
A luta entre bem e mal se manifesta diariamente na medicina, onde o cirurgião enfrenta dilemas éticos, como a aplicação de tecnologias avançadas em contextos que podem beneficiar ou prejudicar pacientes. O nosso compromisso com o bem-estar do paciente deve ser o norteador.
A Alegria como Reflexo da Cura
A alegria é um vislumbre do divino. Na cirurgia digestiva, a alegria está presente em momentos simples, como ver um paciente se recuperar após uma colectomia ou uma gastrectomia bem-sucedida. Esses momentos dão sentido ao trabalho árduo e reforçam a missão do cirurgião.
Humildade e Aprendizado Contínuo
A humildade, um valor essencial para Lewis, é igualmente crucial para o cirurgião. Reconhecer limitações, buscar aprendizado contínuo e valorizar o trabalho em equipe são atitudes que fortalecem a prática médica.
A Eternidade e o Impacto das Escolhas Médicas
Cada escolha cirúrgica tem um impacto que transcende o momento imediato, pois todos os seres humanos têm um valor eterno. O respeito a essa visão confere à medicina um caráter sagrado, onde cada paciente é tratado como único.
Fé e Razão na Tomada de Decisão
A fé e a razão não são antagônicas. Na medicina, a razão científica é enriquecida pela fé no potencial humano e no desejo de servir ao próximo, formando uma abordagem holística no cuidado.
Pequenos Gestos, Grandes Impactos
A clínica cirúrgica é enriquecida por atos simples de bondade. Para o cirurgião digestivo, isso pode significar ouvir atentamente as preocupações de um paciente ou explicar detalhadamente um procedimento. Pequenos gestos constroem confiança e humanizam a prática médica.
Livre-Arbítrio e Responsabilidade
Assim como o livre-arbítrio é central para o Cristianismo, a responsabilidade é uma constante na medicina. Cada decisão do cirurgião, desde indicar uma laparotomia exploratória até manejar complicações, deve ser guiada pela prudência e pela ética.
Aplicação na Cirurgia Digestiva Contemporânea
No contexto atual, a prática cirúrgica do aparelho digestivo enfrenta desafios complexos, como o tratamento de doenças oncológicas, o avanço da cirurgia robótica e a integração de abordagens multidisciplinares. Os princípios de C.S. Lewis reforçam a necessidade de alinhar o progresso científico com a compaixão, criando uma medicina que salva não apenas corpos, mas também pode ajudar a restaurar as almas – dos profissionais e dos pacientes.
Exemplos práticos incluem:
- A Ética na Cirurgia Bariátrica: Equilibrar a necessidade de ajudar o paciente com obesidade mórbida e o respeito à autonomia.
- A Inovação no Tratamento de Tumores Digestivos: Usar avanços como a ablação por radiofrequência com responsabilidade, priorizando o benefício do paciente.
- Humanização no Pós-Operatório: Incorporar cuidados centrados no paciente, promovendo recuperação emocional além da física.
Pontos-Chave
- A imaginação e a criatividade são indispensáveis para solucionar problemas médicos.
- Decisões éticas devem ser baseadas em uma moralidade universal que respeite a dignidade humana.
- A prática médica exige sacrifício pessoal e uma visão humanística do cuidado.
- A integração de fé e razão fortalece a abordagem holística no tratamento.
- Pequenos gestos de bondade humanizam a relação médico-paciente.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A prática da cirurgia do aparelho digestivo deve transcender a técnica, fundamentando-se nos princípios éticos e humanísticos que fizeram da medicina uma vocação sagrada. Inspirados pelos ensinamentos de C.S. Lewis, os cirurgiões podem abraçar uma abordagem que une ciência e compaixão, transformando a cirurgia em uma “medicina que salva” na mais ampla acepção.

“Você nunca encontrará um ser humano comum. Nunca conversou com um mero mortal.” — C.S. Lewis
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#MedicinaHumanista #CirurgiaDigestiva #EducaçãoMédica #ÉticaNaMedicina #MedicinaQueSalva
História da Anatomia Humana
A palestra “Fundamentos Históricos da Anatomia Humana” traça a evolução do estudo da anatomia desde a pré-história até a era moderna. A apresentação destaca a importância de figuras como Cláudio Galeno e Andreas Vesalius, enfatizando a transição de métodos não científicos para a observação direta e a metodologia científica. A influência da anatomia em outras áreas, como arte e filosofia, também é abordada, assim como a evolução das práticas éticas e regulamentações na aquisição de corpos para estudo. A palestra conclui sublinhando a importância inabalável do conhecimento anatômico para profissionais de saúde.
Highlights
- 🏛️ Da Pré-história ao Renascimento: A apresentação percorre a longa história do estudo anatômico, desde as primeiras representações artísticas pré-históricas até os trabalhos detalhados de Leonardo da Vinci e Michelangelo, mostrando como a compreensão do corpo humano evoluiu ao longo dos séculos.
- 🔬 Galeno vs. Vesalius: A palestra destaca a transição paradigmática na anatomia, contrastando o trabalho de Cláudio Galeno, baseado em dissecção animal e que perdurou por séculos, com a abordagem revolucionária de Andreas Vesalius, que enfatizou a observação direta do corpo humano.
- 🎨 A Arte e a Anatomia: A profunda interligação entre a anatomia e a arte renascentista é explorada, mostrando como a compreensão precisa do corpo humano influenciou a representação artística em esculturas e pinturas de grandes mestres.
- 📜 Evolução Ética e Regulamentação: A apresentação aborda as mudanças éticas e regulatórias no acesso a corpos para estudo, destacando o Ato de Anatomia de 1832 na Inglaterra como marco importante na normatização dessas práticas.
- 🧑⚕️ Importância Indispensável: A palestra conclui enfatizando a importância inabalável do conhecimento anatômico para a prática médica, desde o diagnóstico até os procedimentos cirúrgicos, salientando sua relevância em todas as fases da formação e da carreira de um profissional de saúde.
Andreas Vesalius
O Cirurgião Anatomista e Seu Legado no Conhecimento Médico
Introdução
Andreas Vesalius (1514–1564), frequentemente chamado de “pai da anatomia moderna”, é uma figura central na história da medicina. Sua abordagem inovadora ao estudo do corpo humano, fundamentada na dissecação e observação direta, transformou o conhecimento médico, especialmente no campo da cirurgia. Neste artigo, exploraremos como Vesalius, ao articular a importância da anatomia para a prática cirúrgica, moldou a base científica do tratamento das doenças do aparelho digestivo.
A Revolução de Vesalius: Do Teatro ao Livro
No Renascimento, a cirurgia era amplamente relegada a uma posição inferior em relação à medicina teórica. Cirurgiões eram vistos como “trabalhadores manuais”, enquanto os médicos universitários, alinhados aos ensinamentos de Galeno, raramente praticavam a dissecação. Vesalius desafiou esse paradigma. Em sua obra-prima, De humani corporis fabrica (1543), ele não apenas corrigiu erros galênicos, mas também destacou a relação intrínseca entre o conhecimento anatômico e a prática cirúrgica. A anatomia para Vesalius não era apenas um exercício acadêmico; era uma ferramenta essencial para o manejo cirúrgico. Ele mostrou que o entendimento detalhado da anatomia era vital para intervenções seguras, como a sutura de feridas intestinais ou a drenagem de abscessos abdominais. Embora a Fabrica não fosse destinada como um manual de cirurgia, sua ênfase no conhecimento anatômico estabeleceu os alicerces para técnicas cirúrgicas mais precisas no trato digestivo e além.
O Papel da Anatomia na Cirurgia do Aparelho Digestivo
Vesalius defendia que um cirurgião habilidoso devia conhecer profundamente a organização dos órgãos internos. Para ele, compreender a estrutura e a função do trato digestivo era crucial para evitar complicações iatrogênicas. Um exemplo prático disso está na importância da anatomia na sutura intestinal, onde a direção das fibras musculares influencia diretamente a recuperação pós-operatória e a funcionalidade do tecido. Além disso, Vesalius abordava a necessidade de ferramentas apropriadas para a prática anatômica e cirúrgica. Em sua Fabrica, ele ilustrou instrumentos que poderiam ser adaptados para dissecação e procedimentos cirúrgicos, desde lâminas para cortes delicados até agulhas curvas para suturas. Ele enfatizava que, embora os instrumentos fossem importantes, era a habilidade do cirurgião – adquirida por meio do estudo e prática – que determinava o sucesso do procedimento.
Desafios e Contribuições à Educação Médica
Vesalius enfrentou críticas intensas de seus contemporâneos, particularmente dos seguidores de Galeno, que viam sua abordagem como uma afronta às tradições estabelecidas. No entanto, ele persistiu, integrando a dissecção ao ensino médico e instigando seus alunos a examinarem corpos humanos reais, ao invés de dependerem exclusivamente de textos antigos. Esse método transformador, baseado na dissecção – o exame direto do corpo – permanece até hoje como pilar fundamental da formação em cirurgia. No tratamento das doenças do aparelho digestivo, a aplicação prática desse aprendizado reflete-se na precisão em procedimentos como gastrectomias, colecistectomias e ressecções intestinais, que requerem um conhecimento detalhado das relações anatômicas entre órgãos como estômago, fígado e intestinos.
Legado para a Cirurgia Moderna
Embora Vesalius tenha se distanciado da prática cirúrgica em seus escritos, ele elevou o status da anatomia ao patamar de ciência central à medicina, garantindo que futuros cirurgiões tivessem uma base sólida para o desenvolvimento de técnicas mais avançadas. Seu trabalho abriu caminho para o desenvolvimento da cirurgia, permitindo avanços que hoje salvam incontáveis vidas.
Conclusão
O legado de Andreas Vesalius não se limita à anatomia; ele inspirou gerações de médicos e cirurgiões a desafiar paradigmas, questionar autoridades e buscar a verdade diretamente no corpo humano. Suas contribuições à medicina moderna são um lembrete da importância de integrar o conhecimento científico ao cuidado do paciente.
“Eu quem sempre fui tão devotado aos escritos de Galeno, admito minha própria estupidez por confiar tanto em seus textos sem examinar a realidade com meus próprios olhos.” – Andreas Vesalius
Gostou❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#AnatomiaModerna #AndreasVesalius #EducaçãoMédica #CirurgiaDigestiva #HistóriaDaMedicina
Cronologia
1514 – Nasce filho de Isabel Crabbe e Andries van Wesel.
1530 – Matricula-se na Universidade de Louvain.
1533 – Inicia seus estudos de medicina na Universidade de Paris.
1536–1538 – Guerra entre Francisco I e Carlos V.
1536–1537 – Continua seus estudos médicos na Universidade de Louvain.
1537
- Fevereiro: Publicação de A Paráfrase do Nono Livro de Rhazes . . . por Andreas Vesalius de Bruxelas, Candidato em Medicina, em Louvain, por Rutgerus Rescius.
- Março: Publicação de A Paráfrase do Nono Livro de Rhazes por Andreas Vesalius, o Autor, em Basileia, por Robert Winter.
- 5 de dezembro: Recebe o título de doutor em medicina no palácio do Bispo de Pádua.
- 6 de dezembro: Nomeado para as cátedras de cirurgia na Universidade de Pádua.
- 6–24 de dezembro: Realiza dissecação pública em Pádua.
1538
- Publicação de Seis Tabelas Anatômicas em Veneza por Bernardino Vitali.
- Princípios de Anatomia Segundo a Opinião de Galeno por Johann Guinter, Ampliado e Corrigido por Andreas Vesalius de Bruxelas publicado em Veneza por Melchior Sessa.
1539
- Publicação de Uma Carta Ensinando que em Casos de Dor no Lado, Deve-se Cortar a Veia Axilar no Cotovelo Direito em Basileia por Robert Winter.
1540
- 15–24 de janeiro: Realiza dissecação pública na Universidade de Bolonha.
1540–1542
- Contribuições para as traduções latinas das Obras Completas de Galeno (1541–1542), publicadas em Veneza pela firma de Lucantonio Giunta.
1542
- Agosto: Envia os blocos de madeira e o texto da Fabrica para Johannes Oporinus em Basileia.
1543
- Janeiro: Chega a Basileia.
- Maio: Disseca e monta um esqueleto articulado de Jakob Karrer.
- Julho: Publicação de Sete Livros sobre a Estrutura do Corpo Humano e Epitome da Estrutura do Corpo Humano (Fabrica) em Basileia por Johannes Oporinus.
- Agosto: Edição alemã da Fabrica, traduzida por Albanus Torinus, publicada em Basileia por Oporinus.
- Agosto: Apresentação de cópias da Fabrica e Epitome a Carlos V, sendo nomeado médico imperial.
1544
- Realiza demonstrações anatômicas em Pisa a convite de Cosme de Médici.
- Casa-se com Anne van Hamme.
1545 – Nasce sua filha, Anne.
1546 – Publicação da Epístola Explicando o Método e Técnica de Administração da Raiz da China Fervida que o Invencível Carlos Recentemente Empregou em Basileia por Oporinus.
1555 – Segunda edição de Sete Livros sobre a Estrutura do Corpo Humano publicada em Basileia por Oporinus.
1556
- Recebe o título de Conde Palatino por Carlos V.
- Abdicação de Carlos V, que cede seus territórios espanhóis ao filho, Filipe II.
1559
- Enviado por Filipe II para atender ao ferimento de Henrique II, Rei da França, após um torneio.
- Muda-se para a corte de Filipe II em Madri.
1561 – Recebe uma cópia de Observações Anatômicas de Gabriel Falloppio.
1562 – Trata um ferimento de Don Carlos, filho de Filipe II.
1564
- Publicação de Uma Consideração sobre as Observações Anatômicas de Falloppio em Veneza por Francesco Franceschi.
- Março: Parte em peregrinação à Terra Santa e entrega um presente de Filipe para apoiar católicos em Jerusalém.
- 15 de outubro: Morre em seu retorno à Europa, na Ilha de Zakynthos.
Fundamentos do Transplante Hepático
O transplante hepático é uma das intervenções mais complexas e, ao mesmo tempo, revolucionárias no tratamento de doenças hepáticas terminais. Este procedimento, que substitui o fígado doente por um fígado saudável, é crucial para muitos pacientes que sofrem de doenças hepáticas graves e progressivas, além de algumas condições metabólicas e neoplásicas. Para os estudantes de medicina, residentes em cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo, compreender os fundamentos do transplante hepático é essencial para atuar na linha de frente do tratamento avançado de doenças hepáticas. Neste artigo, abordaremos as principais indicações, critérios de seleção, técnicas cirúrgicas, imunologia, complicações, prognóstico e perspectivas futuras do transplante hepático.
Introdução
O transplante hepático tornou-se a única opção viável para pacientes com falência hepática irreversível, proporcionando uma sobrevida significativamente prolongada e melhor qualidade de vida. Desde a sua primeira realização em humanos, ele se consolidou como o tratamento padrão para diversas doenças hepáticas. No entanto, as limitações relacionadas à disponibilidade de doadores e as complexidades inerentes ao procedimento tornam-no um desafio tanto para médicos quanto para a sociedade.
Evolução Histórica
A história do transplante hepático iniciou-se nos anos 1960, quando o Dr. Thomas Starzl realizou o primeiro transplante bem-sucedido de fígado. Desde então, avanços significativos na imunossupressão e na técnica cirúrgica melhoraram dramaticamente os resultados. O desenvolvimento de drogas imunossupressoras, como a ciclosporina, foi crucial para reduzir a rejeição e aumentar a sobrevida dos pacientes. Hoje, o transplante hepático é realizado globalmente com taxas de sucesso que eram inimagináveis no início da prática.
Indicações de Transplante Hepático
O transplante de fígado é indicado principalmente para pacientes com falência hepática crônica ou aguda que apresentam risco de morte em curto prazo. As principais indicações incluem:
- Cirrose hepática avançada, incluindo cirrose hepática por hepatite viral crônica, cirrose biliar primária e doença hepática alcoólica;
- Hepatocarcinoma em fase inicial, que pode ter cura através do transplante em pacientes selecionados;
- Doenças metabólicas hereditárias, como a doença de Wilson e deficiência de alfa-1 antitripsina;
- Insuficiência hepática aguda, geralmente causada por hepatite fulminante.
Critérios de Seleção dos Receptores
A seleção de candidatos é orientada principalmente pelo MELD (Model for End-Stage Liver Disease) para adultos e o PELD (Pediatric End-Stage Liver Disease) para crianças, ambos sistemas de pontuação que avaliam a gravidade da doença hepática. Pacientes com MELD ≥ 15 geralmente são considerados elegíveis, enquanto aqueles com hepatocarcinoma devem atender aos critérios de Milão para serem listados. Além dos critérios médicos, os candidatos são avaliados em relação ao apoio familiar, aderência ao tratamento e estabilidade psicológica, pois esses fatores influenciam significativamente o sucesso pós-transplante.
Critérios de Seleção dos Doadores
Os doadores de fígado podem ser falecidos ou vivos. Os critérios para doadores falecidos incluem idade adequada, ausência de doença hepática e compatibilidade sanguínea com o receptor. Nos doadores vivos, além desses critérios, a saúde física e mental do doador é minuciosamente avaliada. A utilização de doadores vivos tornou-se comum em alguns países, sendo especialmente importante em regiões com baixa disponibilidade de doadores falecidos.
Contraindicações Relativas e Absolutas
Certas condições médicas são consideradas contraindicativas para o transplante hepático. Contraindicações absolutas incluem:
- Neoplasias extra-hepáticas não controladas;
- Insuficiência cardíaca ou pulmonar grave;
- Uso ativo de substâncias ilícitas. As contraindicações relativas, por outro lado, são avaliadas caso a caso, incluindo infecções controladas pelo HIV, cânceres tratados e comorbidades cardiovasculares que podem ser manejadas clinicamente.
Tipos de Transplante Hepático e Fundamentos Técnicos
Os transplantes hepáticos podem ser classificados como transplante de fígado total, reduzido, split-liver (dividido entre dois receptores), ou de doadores vivos. Nos transplantes de doadores vivos, segmentos específicos do fígado, geralmente o lobo esquerdo para crianças e o lobo direito para adultos, são removidos do doador e transplantados para o receptor. Cada tipo requer preparo técnico especializado e apresenta desafios próprios, especialmente em relação à preservação do fluxo sanguíneo e à minimização de complicações vasculares.
Principais Complicações
As complicações podem ocorrer tanto no pós-operatório imediato quanto no longo prazo. As principais incluem:
- Rejeição aguda e crônica do enxerto;
- Complicações vasculares, como trombose da artéria hepática e obstrução da veia porta;
- Complicações biliares, incluindo estenoses e fístulas;
- Infecções devido à imunossupressão prolongada.
Imunologia dos Transplantes: Tipos de Rejeição e Imunossupressão
A rejeição é um dos maiores desafios no transplante hepático e pode ser classificada em rejeição celular aguda, geralmente controlada por imunossupressão, e rejeição crônica, que leva à perda do enxerto. Os principais medicamentos imunossupressores incluem ciclosporina, tacrolimus e micofenolato de mofetila. A terapia é cuidadosamente ajustada para minimizar a rejeição e evitar efeitos adversos, como infecções oportunistas e cânceres secundários.
Prognóstico do Transplante: Sobrevida Global e do Enxerto
A sobrevida após o transplante hepático continua a melhorar, com taxas de 1 ano superiores a 90% e sobrevida média de 20 anos para pacientes transplantados na década de 1990. O prognóstico depende de múltiplos fatores, incluindo a gravidade da doença original, a idade do receptor e as complicações pós-operatórias. O acompanhamento vitalício é necessário para monitorar a função do enxerto e ajustar a imunossupressão.
Perspectivas Futuras
As inovações no transplante hepático incluem avanços na perfusão de órgãos, que permite avaliar e condicionar o fígado doado antes do transplante, e o potencial uso de xenotransplantes e engenharia de tecidos. Esses avanços visam aumentar a disponibilidade de órgãos e melhorar a longevidade dos enxertos. O desenvolvimento de técnicas para induzir tolerância imunológica também promete reduzir a necessidade de imunossupressão a longo prazo, melhorando a qualidade de vida dos transplantados.
Conclusão
O transplante hepático é um procedimento altamente complexo, mas fundamental para pacientes com doenças hepáticas terminais. Para estudantes e residentes, entender os critérios de seleção, o manejo das complicações e os fundamentos técnicos é essencial para a prática em gastroenterologia e cirurgia hepática. À medida que a medicina avança, a capacidade de tratar pacientes com insuficiência hepática melhora continuamente, levando a novos horizontes na área de transplantes.
“Liver transplantation provides an opportunity for these patients to reclaim a healthy life, free from the shadow of chronic liver disease.” – Thomas Starzl
Gostou? Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#TransplanteHepático #Medicina #CirurgiaDigestiva #SaúdeHepática #Imunossupressão
Manejo das Afecções Biliares nos Pacientes Bariátricos
A cirurgia bariátrica é uma das principais intervenções no manejo da obesidade mórbida, proporcionando perda ponderal significativa e melhora nas comorbidades associadas, como diabetes tipo 2, hipertensão arterial e dislipidemia. Contudo, essa intervenção acarreta alterações fisiológicas importantes no trato gastrointestinal, que podem favorecer o surgimento de afecções biliares, como a colelitíase. Este artigo tem como objetivo discutir as afecções biliares em pacientes bariátricos, abrangendo desde a relação entre a obesidade e a doença biliar até o manejo das complicações biliares antes e após a cirurgia bariátrica, além de abordar a propedêutica diagnóstica e terapêutica no acesso às vias biliares em pacientes submetidos ao bypass gástrico.
Introdução
A obesidade é uma condição de saúde pública crescente no Brasil. De acordo com o IBGE, mais de 20% da população brasileira é considerada obesa, o que coloca esses indivíduos em maior risco de desenvolver doenças biliares, entre outras comorbidades. A colelitíase (formação de cálculos biliares) é particularmente prevalente nesta população, sendo exacerbada pela rápida perda de peso que segue a cirurgia bariátrica. Dada a relevância do tema, é fundamental que cirurgiões e residentes compreendam as implicações das afecções biliares nos pacientes bariátricos e as abordagens terapêuticas adequadas.
Doença Biliar e Obesidade
A colelitíase afeta de 2% a 15% da população geral, mas em pacientes obesos esse número é substancialmente maior. A obesidade é um fator de risco importante para a formação de cálculos biliares devido à supersaturação da bile com colesterol, hipomotilidade da vesícula biliar e fatores dietéticos. Estudos indicam que pacientes com Índice de Massa Corporal (IMC) superior a 40 têm um risco 8 vezes maior de desenvolver colelitíase em comparação com indivíduos de peso normal.
Nos pacientes bariátricos, a rápida perda de peso após a cirurgia é um dos maiores contribuintes para o surgimento de cálculos biliares. A prevalência de colelitíase em pacientes pós-cirurgia bariátrica varia entre 6,7% a 52,8%, sendo os cálculos de colesterol os mais comuns. Além disso, a hipomotilidade da vesícula biliar após o bypass gástrico favorece o acúmulo de bile e a formação de “sludge” biliar, o que pode evoluir para cálculos e suas complicações.
Manejo da Colelitíase e Suas Complicações Antes e Após a Cirurgia Bariátrica
O manejo da vesícula biliar no contexto da cirurgia bariátrica é um tema controverso. Alguns cirurgiões optam por realizar colecistectomia profilática durante a cirurgia bariátrica em pacientes com cálculos assintomáticos documentados. Essa abordagem visa prevenir complicações futuras, como a colecistite aguda ou pancreatite biliar, que podem ser mais difíceis de tratar após o bypass gástrico, devido às alterações anatômicas.
Entretanto, muitos cirurgiões adotam uma abordagem seletiva, realizando a colecistectomia apenas em pacientes com sintomas ou doenças biliares comprovadas por exames de imagem. Essa prática é sustentada pelo fato de que não há consenso claro sobre a necessidade de remover a vesícula biliar em todos os pacientes durante a cirurgia bariátrica. Estima-se que cerca de 4% dos pacientes necessitarão de colecistectomia após o bypass gástrico, um número que pode ser considerado aceitável para evitar a remoção profilática da vesícula em todos os casos.
As complicações da colelitíase, como a colecistite, pancreatite biliar e a coledocolitíase, podem ocorrer tanto no pré quanto no pós-operatório bariátrico. Nessas situações, o manejo segue as diretrizes usuais, com colecistectomia laparoscópica sendo a primeira linha de tratamento. No entanto, em pacientes submetidos ao bypass gástrico, a abordagem endoscópica para remover cálculos biliares pode ser mais desafiadora.
Propedêutica Radiológica e Endoscópica nos Pacientes Bariátricos
O diagnóstico das afecções biliares em pacientes bariátricos pode ser dificultado pela anatomia alterada após a cirurgia. A ultrassonografia abdominal, o exame de escolha para diagnosticar colelitíase na população geral, pode ter sensibilidade reduzida em pacientes obesos devido à presença de gordura abdominal. Além disso, após o bypass gástrico, o acesso ao trato gastrointestinal superior é limitado, o que dificulta exames como a colangiopancreatografia retrógrada endoscópica (CPRE).
A ressonância magnética com colangiopancreatografia (RMCP) e a colangiografia transhepática percutânea são métodos alternativos valiosos no diagnóstico de coledocolitíase e outras afecções das vias biliares em pacientes bariátricos. Em casos mais complexos, o uso de ultrassom intraoperatório durante a colecistectomia pode auxiliar no diagnóstico e manejo.
Prevenção da Colelitíase Após Cirurgia Bariátrica
A formação de cálculos biliares é especialmente comum durante a fase de perda rápida de peso que se segue à cirurgia bariátrica. Para mitigar esse risco, muitos especialistas recomendam o uso de ursodiol (ácido ursodesoxicólico), que atua na dissolução de colesterol e reduz a formação de cálculos biliares. Estudos indicam que a administração de 600 mg de ursodiol por dia durante os primeiros 6 meses após a cirurgia reduz significativamente a incidência de colelitíase, especialmente na fase de perda ponderal acelerada.
Acesso Endoscópico das Vias Biliares Após Bypass Gástrico
A alteração anatômica após o bypass gástrico de Roux-en-Y torna o acesso endoscópico convencional ao trato biliar, como a CPRE, extremamente difícil. Nesse cenário, uma abordagem bem-sucedida envolve a CPRE transgástrica percutânea assistida por laparoscopia, que permite acessar a árvore biliar via o estômago remanescente. Este procedimento, realizado em conjunto com especialistas em endoscopia, mostrou-se altamente eficaz, com taxas de sucesso superiores a 95% na canulação do ducto biliar comum.
Conclusão
As afecções biliares são um desafio comum e relevante em pacientes bariátricos, principalmente devido às alterações fisiológicas e anatômicas decorrentes da cirurgia e da rápida perda de peso. O manejo adequado envolve uma abordagem multidisciplinar, utilizando técnicas radiológicas avançadas e estratégias preventivas como o uso de ursodiol. O cirurgião digestivo deve estar apto a manejar as complicações biliares tanto no pré quanto no pós-operatório, considerando as particularidades anatômicas e fisiológicas desses pacientes.
“O entendimento das alterações morfofuncionais pós-bariátrica e sua relação com o sistema biliar é crucial para a condução segura e eficaz do paciente obeso no pós-operatório” Prof. Dr. Ozimo Gama
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Cuidados Perioperatórios nos Pacientes Cirróticos
Desafios e Estratégias para Otimização dos Resultados
A cirurgia em pacientes com cirrose hepática apresenta desafios únicos e um risco significativamente elevado de complicações. O manejo perioperatório adequado é essencial para minimizar complicações como sangramento, falência hepática e ascite, garantindo melhores desfechos. Este post visa abordar os cuidados perioperatórios em pacientes cirróticos, com foco em pré-avaliação, estratégias intraoperatórias e o manejo de complicações pós-operatórias, oferecendo orientações práticas para estudantes de medicina, residentes e cirurgiões digestivos.
A Importância do Manejo Perioperatório em Pacientes Cirróticos
Os pacientes com cirrose são particularmente vulneráveis durante procedimentos cirúrgicos devido à disfunção hepática subjacente e suas complicações associadas, como hipertensão portal, ascite e coagulopatia. A taxa de mortalidade desses pacientes pode ser até três vezes maior que a dos pacientes sem cirrose. A avaliação cuidadosa e o planejamento pré-operatório são cruciais para evitar falência hepática pós-operatória e outras complicações graves.
Avaliação Pré-Operatória: Estratificação de Riscos
A adequada estratificação de risco no pré-operatório é um dos pilares para o sucesso em cirurgias de pacientes cirróticos. Os seguintes aspectos são críticos:
- Estado geral do paciente: Comorbidades como insuficiência cardíaca e renal, além de diabetes mellitus, aumentam os riscos.
- Função hepática: Avaliações quantitativas, como a classificação Child-Pugh e o MELD, são essenciais para prever o risco de falência hepática. Cirurgias maiores devem ser evitadas em pacientes Child B ou C, exceto em casos extremamente selecionados.
- Hipertensão portal: A presença de hipertensão portal, frequentemente acompanhada de trombocitopenia e varizes esofágicas, é um fator importante que aumenta o risco de complicações pós-operatórias.
Além disso, a avaliação do volume do fígado remanescente é fundamental. Em pacientes com cirrose, é necessário um remanescente de pelo menos 50% do volume hepático funcional para evitar falência hepática.
Estratégias Intraoperatórias para Minimizar Complicações
Durante a cirurgia, o manejo adequado é essencial para minimizar complicações em pacientes cirróticos. As seguintes estratégias são recomendadas:
- Abordagem Anterior: Para pacientes com tumores hepáticos grandes, essa técnica evita a manipulação excessiva do fígado, reduzindo o risco de sangramento e disseminação tumoral.
- Ultrassonografia Intraoperatória: A ultrassonografia com Doppler é essencial para mapear o padrão vascular do fígado e orientar as ressecções hepáticas, maximizando a preservação de parênquima funcional.
- Occlusão Vascular e Controle de Sangramento: A técnica de clampagem intermitente do fluxo hepático é uma estratégia eficaz para reduzir a perda sanguínea sem comprometer a função hepática. Manter uma pressão venosa central baixa (<5 mmHg) também reduz o sangramento durante a transecção hepática.
- Uso de Dispositivos para Transecção Hepática: Técnicas como o uso do Cavitron Ultrasonic Surgical Aspirator (CUSA) e o uso de bisturis harmônicos são recomendadas para a dissecção cuidadosa do parênquima cirrótico, minimizando o risco de sangramento.
Complicações Pós-Operatórias e Manejo
Após a cirurgia, os pacientes cirróticos estão sujeitos a complicações específicas que exigem manejo adequado:
- Ascite Pós-Operatória: É uma complicação comum, ocorrendo em cerca de um terço dos pacientes cirróticos. O tratamento envolve reposição volêmica cuidadosa e, em alguns casos, o uso de diuréticos. A ascite pode causar dor, limitação respiratória e infecções, exigindo vigilância contínua.
- Falência Hepática: A falência hepática pós-operatória ainda é uma das principais causas de mortalidade hospitalar após ressecções hepáticas em cirróticos, com uma taxa de mortalidade de até 50% nos casos mais graves. O monitoramento de indicadores como o tempo de protrombina (TP) e bilirrubina sérica no pós-operatório imediato é essencial para detectar precocemente essa complicação.
- Fístulas Biliares: As fístulas biliares, com taxa de ocorrência em torno de 6%, podem levar a coleções abdominais e infecções graves. O tratamento envolve drenagem percutânea e, em alguns casos, cirurgia de revisão.
- Trombose Portal: A trombose da veia porta é uma complicação perigosa, especialmente em pacientes com hipertensão portal. O diagnóstico precoce através de ultrassonografia e tratamento com anticoagulantes são essenciais para evitar desfechos fatais.
- Infecções: Cirurgias prolongadas e a presença de ascite aumentam o risco de complicações sépticas. A profilaxia com antibióticos de amplo espectro é recomendada, e a infecção deve ser prontamente diagnosticada e tratada.
Pontos-Chave para a Prática do Cirurgião Digestivo
- Seleção do Paciente: A correta seleção dos pacientes, baseada na avaliação da função hepática, estado geral e hipertensão portal, é o principal determinante de bons resultados cirúrgicos.
- Técnicas Cirúrgicas Avançadas: O uso de estratégias como a ultrassonografia intraoperatória e a abordagem anterior melhora os resultados ao minimizar o trauma tecidual e a perda sanguínea.
- Manejo de Complicações: O reconhecimento precoce e o tratamento das complicações pós-operatórias, como ascite, fístulas biliares e trombose portal, são essenciais para a recuperação dos pacientes.
- Importância da Equipe Multidisciplinar: O sucesso na abordagem cirúrgica de pacientes cirróticos depende da colaboração entre cirurgiões, hepatologistas, anestesistas e intensivistas.
Conclusão
O manejo perioperatório de pacientes cirróticos requer uma abordagem cuidadosa e individualizada para reduzir complicações e otimizar os resultados cirúrgicos. A avaliação pré-operatória detalhada, o uso de técnicas intraoperatórias avançadas e o manejo adequado das complicações são fundamentais para o sucesso. Como Jacques Belghiti, renomado cirurgião hepático, destacou: “A cirurgia no paciente cirrótico não é apenas uma técnica, mas uma tarefa criteriosa de selecionar e manejar com precisão cada etapa do tratamento”.
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#cirurgia #cirroses #cuidadosperioperatórios #cirurgiahepática #saúde
Protocolo ERAS na Cirurgia Pancreática
Otimizando a Recuperação Pós-Operatória
A cirurgia pancreática, em especial a pancreatoduodenectomia (PD), é um procedimento de alta complexidade e associado a complicações significativas. Para melhorar os desfechos pós-operatórios e reduzir o tempo de internação, foi implementado o protocolo ERAS (Enhanced Recovery After Surgery), que visa otimizar a recuperação dos pacientes cirúrgicos. Este artigo explora como o ERAS impacta a morbimortalidade, seu custo-efetividade e as principais medidas assistenciais envolvidas, oferecendo pontos-chave para a prática cirúrgica no aparelho digestivo.
O que é o Protocolo ERAS?
O protocolo ERAS é uma abordagem multimodal que visa reduzir o estresse cirúrgico, acelerar a recuperação e diminuir as complicações no período pós-operatório. Originalmente desenvolvido para cirurgias colorretais, o ERAS foi adaptado para outras especialidades, incluindo a cirurgia pancreática. O primeiro conjunto de diretrizes para a pancreatoduodenectomia foi publicado em 2012, sendo atualizado em 2019 com 27 recomendações baseadas em evidências científicas. Entre os principais objetivos do ERAS estão a redução do tempo de internação hospitalar, a menor incidência de complicações como a fístula pancreática e o retardo no esvaziamento gástrico (DGE, na sigla em inglês).
Na cirurgia pancreática, a mortalidade tem sido reduzida para menos de 5% em centros de grande volume no Brasil, entretanto, a morbidade ainda é alta, variando de 30% a 60%. O protocolo ERAS surge como uma alternativa eficiente para padronizar o manejo perioperatório, reduzir complicações e encurtar a permanência hospitalar.
Impacto do ERAS na Morbimortalidade
Um dos principais benefícios do protocolo ERAS é a redução das complicações pós-operatórias, incluindo infecções de sítio cirúrgico, DGE e fístula pancreática. Estudos internacionais e revisões sistemáticas mostraram que pacientes submetidos ao ERAS apresentaram menor tempo de internação e menor incidência de complicações sem aumento nas taxas de readmissão ou mortalidade.
No Brasil, onde a pancreatoduodenectomia também é uma cirurgia de alto risco, a implementação do ERAS traz ganhos substanciais. Em centros que adotaram o protocolo, houve uma redução expressiva do tempo médio de internação, que pode superar 14 dias nos cuidados convencionais. Além disso, as taxas de complicações severas são minimizadas, o que impacta diretamente a recuperação global do paciente e a sua qualidade de vida pós-operatória.
Custo-Efetividade do ERAS
A implementação do protocolo ERAS tem demonstrado ser financeiramente vantajosa. A redução das complicações e do tempo de hospitalização leva a uma economia significativa de recursos, especialmente em procedimentos de grande porte, como as cirurgias pancreáticas. Estima-se que, em hospitais que seguem o ERAS, os custos totais com o tratamento de pacientes submetidos à pancreatoduodenectomia sejam reduzidos em até 20%, principalmente devido à diminuição do tempo de internação e ao uso racional de exames laboratoriais e diagnósticos.
Um estudo norte-americano relatou que a economia total foi de aproximadamente USD 5.300 por paciente após a adoção do ERAS, enquanto no Brasil, uma análise preliminar mostrou uma economia que pode atingir R$ 15.000 por paciente em algumas instituições de referência. Esses números refletem a importância da padronização dos cuidados e a utilização eficiente de recursos hospitalares.
Principais Medidas Assistenciais do ERAS
O protocolo ERAS engloba diversas intervenções que visam otimizar o cuidado perioperatório. Entre as principais medidas adotadas no contexto da cirurgia pancreática, destacam-se:
- Pré-Operatório:
- Pré-habilitação: Início de um programa de reabilitação com exercícios físicos e suporte nutricional de 3 a 6 semanas antes da cirurgia, especialmente em pacientes com perda de peso superior a 15% ou IMC abaixo de 18,5.
- Jejum: Limitação do jejum pré-operatório para 6 horas para sólidos e 2 horas para líquidos, além de carga de carboidratos até 2 horas antes da cirurgia.
- Drenagem biliar: Indicada apenas em casos específicos, como níveis de bilirrubina acima de 250 μmol/L ou colangite.
- Pós-Operatório:
- Remoção precoce de drenos: Drenos são removidos após 72 horas em pacientes de baixo risco.
- Alimentação precoce: Início de alimentação oral assim que o paciente tolerar, com a utilização de chicletes e medicamentos como alvimopan para acelerar a recuperação intestinal.
- Mobilização precoce: Incentivo à deambulação a partir do primeiro dia pós-operatório.
Principais Recomendações do Protocolo ERAS sobre Técnica Operatória
- Minimização do Trauma Cirúrgico:
- O protocolo ERAS recomenda que as técnicas operatórias minimizem o trauma tecidual, uma prática que inclui o uso de abordagens menos invasivas quando viável. Embora o PD laparoscópico ou robótico tenha sido considerado, o ERAS alerta que esses métodos devem ser realizados apenas em centros de alto volume e com experiência, pois ainda há preocupações sobre sua segurança em larga escala.
- A PD robótica não é recomendada pelo ERAS devido à falta de evidências robustas que garantam segurança e eficácia comparável à cirurgia aberta.
- Drenagem Profilática:
- A drenagem abdominal profilática continua a ser um ponto controverso no manejo de cirurgias pancreáticas. O ERAS sugere que a decisão sobre o uso de drenos profiláticos deve ser individualizada. Para pacientes considerados de baixo risco (com níveis de amilase abaixo de 5000 U/L no primeiro dia pós-operatório), a drenagem pode ser removida precocemente, geralmente em 72 horas, o que pode reduzir o risco de infecções e acelerar a recuperação.
- Prevenção de Hipotermia:
- O ERAS recomenda a manutenção da normotermia intraoperatória através de técnicas como o uso de cobertores aquecidos e o controle da temperatura de fluidos intravenosos. Isso é crucial para evitar complicações relacionadas à hipotermia, como o aumento do risco de infecções e atraso na cicatrização.
- Controle de Fluidos:
- O controle rigoroso de fluidos durante a operação é uma diretriz fundamental no protocolo ERAS. A sobrecarga de fluidos pode levar a edemas nos tecidos e complicações, como o retardo no esvaziamento gástrico. Assim, o uso de um algoritmo de fluidoterapia dirigida por metas é recomendado para evitar o excesso de fluidos.
- Uso de Analgesia Regional:
- Para o controle da dor, o protocolo ERAS favorece o uso de analgesia epidural torácica em cirurgias abertas, como a pancreatoduodenectomia. Essa abordagem reduz a necessidade de opioides, que podem estar associados a efeitos colaterais como íleo paralítico. Caso a analgesia epidural seja contraindicada, são sugeridas alternativas como o uso de cateteres de ferida preperitoneais.
- Prevenção de Fístulas Pancreáticas:
- Embora não seja uma recomendação direta sobre a técnica operatória, o protocolo ERAS menciona que o uso de análogos de somatostatina (como octreotida) não é recomendado de forma sistemática para prevenir fístulas pancreáticas, devido à falta de evidências conclusivas. A gestão de fístulas pancreáticas, portanto, deve ser cuidadosa e individualizada.
Considerações sobre Cirurgia Minimamente Invasiva:
O ERAS reconhece o potencial da cirurgia minimamente invasiva (como laparoscopia e cirurgia robótica) para reduzir o trauma cirúrgico e acelerar a recuperação. No entanto, essas técnicas exigem habilidades especializadas e devem ser realizadas apenas em centros com alto volume e vasta experiência nesses procedimentos. A segurança e eficácia dessas abordagens ainda estão sendo avaliadas em estudos, e o ERAS não as recomenda como padrão para todos os pacientes.
Pontos-Chave e Conclusões para a Prática do Cirurgião Digestivo
A implementação do ERAS na cirurgia pancreática oferece uma abordagem sólida e baseada em evidências para reduzir a morbimortalidade e melhorar os resultados cirúrgicos. O sucesso do protocolo depende do comprometimento de toda a equipe multidisciplinar e do acompanhamento rigoroso das diretrizes estabelecidas. Alguns pontos-chave que devem ser ressaltados incluem:
- Aderência às diretrizes: O nível de adesão ao protocolo está diretamente relacionado à redução de complicações. Estudos mostraram que pacientes com uma adesão superior a 70% ao protocolo ERAS apresentam menos complicações e menor tempo de internação.
- Educação contínua da equipe: Reuniões regulares para discutir a implementação e as barreiras encontradas são essenciais para o sucesso a longo prazo.
- Foco no paciente: A educação do paciente quanto às expectativas do tratamento e a importância da adesão ao plano cirúrgico também desempenha um papel fundamental na implementação bem-sucedida do ERAS.
Para o cirurgião digestivo, o ERAS representa uma mudança de paradigma, não apenas na forma de realizar cirurgias de alta complexidade, mas também na maneira de lidar com o cuidado pós-operatório. A adesão ao protocolo requer coordenação e disciplina, mas os resultados são claros: pacientes com recuperação mais rápida, menos complicações e melhor qualidade de vida após a cirurgia.
Considerações Finais
O protocolo ERAS veio para transformar a forma como abordamos a recuperação cirúrgica, especialmente em procedimentos complexos como a pancreatoduodenectomia. Ele não só melhora os desfechos clínicos, mas também otimiza a utilização de recursos hospitalares, beneficiando tanto os pacientes quanto o sistema de saúde. Para a prática do cirurgião do aparelho digestivo, o ERAS é uma ferramenta indispensável para garantir um tratamento mais seguro, eficaz e econômico.
Como disse Allen Oldfather Whipple, renomado cirurgião norte-americano: “O conhecimento sobre a operação não é o suficiente, é necessário também uma compreensão profunda do que ocorre depois dela para alcançar a cura.”
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#cirurgiaplástica #ERAS #cirurgiapancreática #recuperaçãoacelerada #saúde
Anatomia Cirúrgica das Vias Biliares Aplicada para Colecistectomia Segura
Por Prof. Dr. Ozimo Gama
- Membro Titular do Colégio Brasileiro de Cirurgiões
- Membro Titular do Colégio Brasileiro de Cirurgia Digestiva
A colecistectomia é um dos procedimentos cirúrgicos mais comuns para o tratamento de doenças da vesícula biliar, como colelitíase sintomática e colecistite aguda. A correta compreensão da anatomia cirúrgica das vias biliares é fundamental para garantir uma colecistectomia laparoscópica segura, evitando complicações graves, como lesões ao ducto biliar. Este texto é voltado para estudantes de medicina, residentes de cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo, abordando aspectos essenciais da anatomia cirúrgica das vias biliares com foco na segurança cirúrgica.

Introdução
A colecistectomia, seja ela aberta ou laparoscópica, envolve a retirada da vesícula biliar, um órgão pequeno, mas de grande importância no armazenamento e liberação de bile. As complicações durante a colecistectomia são frequentemente relacionadas ao desconhecimento ou à falta de reconhecimento das variações anatômicas das vias biliares. Estudos indicam que entre 0,3% a 1,5% dos pacientes podem apresentar lesões das vias biliares durante esse procedimento, o que pode resultar em morbidade significativa.
Anatomia Descritiva
As vias biliares consistem no sistema de ductos que transportam a bile do fígado para o duodeno. Esse sistema é composto pelos ductos biliares intra-hepáticos, que se unem para formar o ducto hepático comum. As vias biliares extra-hepática é formada pelo ducto cístico, que conecta a vesícula biliar ao ducto hepático, formando assim o ducto colédoco, que desemboca na ampola duodenal maior (Vater) no duodeno. A vesícula biliar é localizada na fossa cística da superfície visceral do fígado, e sua drenagem ocorre através do ducto cístico.
Dimensões Normais
O ducto hepático comum mede, em média, 4 a 5 cm de comprimento e tem um diâmetro aproximado de 4 a 6 mm. O ducto cístico, por sua vez, mede cerca de 2 a 4 cm de comprimento, com um diâmetro de 3 mm. Já o ducto colédoco tem, em média, 6 a 8 cm de comprimento e um diâmetro que varia de 6 a 8 mm.
Anatomia Topográfica e Relações Anatômicas
A vesícula biliar está situada entre o lobo direito e o lobo quadrado do fígado, na fossa vesicular. Suas relações anatômicas incluem o duodeno, a cabeça do pâncreas e o fígado. Na cirurgia, o triângulo Hepatocístico é uma das principais referências anatômicas. Esse triângulo é formado pelo ducto cístico, ducto hepático comum e a borda inferior do fígado, contendo a artéria cística, que é uma importante estrutura a ser identificada e preservada durante a colecistectomia.

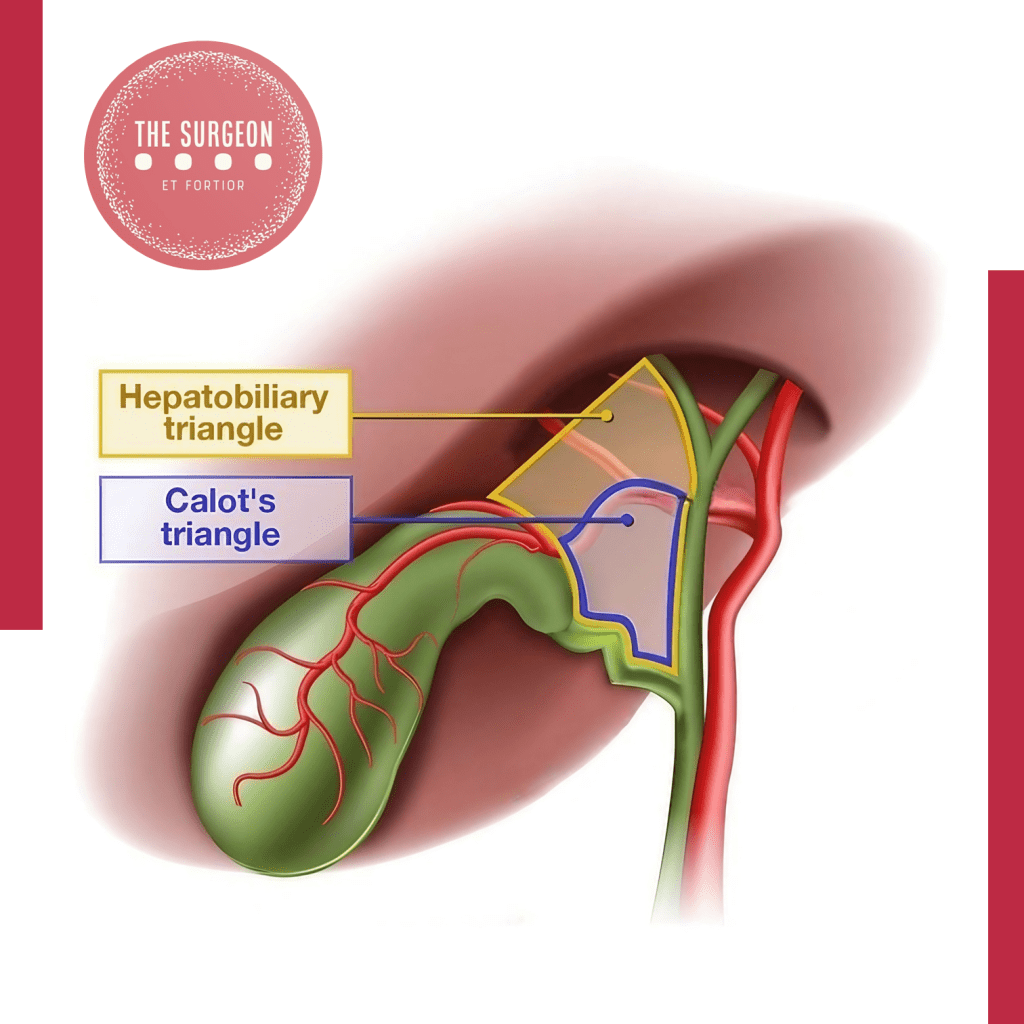
Variações Anatômicas Mais Relevantes
As variações anatômicas das vias biliares são comuns e podem estar presentes em até 25% da população. Entre as mais frequentes estão:
- Ducto cístico curto ou com inserção alta no ducto hepático.
- Ducto cístico espiralado, o que dificulta sua dissecção.
- Trifurcação dos ductos biliares, onde o ducto hepático direito se divide antes de formar o ducto hepático comum.
- Artéria cística de origem anômala, como proveniente da artéria hepática direita ou, em raros casos, da artéria hepática comum.
Essas variações anatômicas são importantes, pois lesões inadvertidas durante a dissecção podem causar complicações como fístulas biliares, estenoses e lesões vasculares.
Vascularização da Via Biliar
A vascularização das vias biliares é fornecida principalmente pela artéria cística, que se origina da artéria hepática direita. Adicionalmente, a artéria hepática direita e a artéria gastroduodenal fornecem ramos que contribuem para a irrigação do ducto hepático comum e colédoco. No entanto, a parte supraduodenal do colédoco é mais suscetível a isquemia, especialmente em casos de manipulação excessiva durante a cirurgia. A compreensão da rede arterial é vital para evitar lesões vasculares e garantir o sucesso da anastomose biliar, quando necessário.
Pontos de Reparo Anatômico para uma Colecistectomia Laparoscópica Segura
Para evitar lesões inadvertidas, os cirurgiões devem seguir alguns princípios básicos de segurança:
- Identificação do Triângulo de Calot: A correta visualização das estruturas no triângulo de Calot é essencial. A artéria cística deve ser cuidadosamente dissecada e clipada antes da secção.
- Dissecção crítica de segurança: O conceito de “visão crítica de segurança” implica a dissecção completa da vesícula do leito hepático, de forma que as únicas duas estruturas conectadas à vesícula sejam o ducto cístico e a artéria cística. Isso reduz significativamente o risco de lesões às vias biliares principais.
- Variações Anatômicas: O cirurgião deve estar ciente das possíveis variações anatômicas e, em caso de dúvida, deve recorrer a exames de imagem intraoperatórios, como a colangiografia.
Pontos-Chave e Conclusões Aplicadas à Prática do Cirurgião Digestivo
Para garantir uma colecistectomia segura, o conhecimento profundo da anatomia cirúrgica das vias biliares é indispensável. A incidência de lesões biliares pode ser minimizada seguindo protocolos rigorosos, como a dissecção anatômica cuidadosa, a utilização de exames complementares intraoperatórios e o respeito pelas variações anatômicas individuais. No Brasil, a colecistectomia laparoscópica é o padrão ouro, sendo realizada em aproximadamente 95% dos casos, com uma taxa de complicação de menos de 1%. No entanto, lesões das vias biliares ainda são uma causa significativa de morbidade pós-operatória, destacando a importância de uma educação anatômica continuada.
Em última análise, a chave para uma colecistectomia laparoscópica segura está na visualização precisa das estruturas anatômicas e na aplicação de técnicas cirúrgicas comprovadas. Seguindo esses princípios, é possível reduzir significativamente o risco de complicações graves e melhorar os resultados para os pacientes.
Como já afirmado por Lahey: “O reconhecimento e a compreensão das variações anatômicas nas vias biliares são fundamentais para evitar complicações nas cirurgias biliares.”
Gostou? Nos deixe um comentário ✍️, compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#AnatomiaBiliar #ColecistectomiaSegura #CirurgiaDigestiva #ViasBiliares #SegurançaCirúrgica
Incidentaloma Hepático
Estratégia Diagnóstica e Manejo Cirúrgico
A crescente utilização de exames de imagem na prática médica resultou no aumento da detecção de lesões hepáticas assintomáticas, conhecidas como incidentalomas hepáticos. Estes achados radiológicos, identificados incidentalmente em até 15% dos exames de imagem, levantam desafios diagnósticos e terapêuticos significativos para os cirurgiões do aparelho digestivo. Embora a maioria dessas lesões seja benigna, cerca de 10% podem ser malignas, exigindo uma avaliação detalhada para evitar procedimentos desnecessários e potencialmente arriscados. Este post aborda as estratégias diagnósticas e o manejo cirúrgico apropriado para o incidentaloma hepático, com foco na prática diária do cirurgião digestivo.
Introdução
O incidentaloma hepático é definido como uma lesão hepática assintomática detectada incidentalmente durante investigações radiológicas realizadas por outras razões. A maioria dessas lesões é benigna, como os hemangiomas e as hiperplasias nodulares focais (HNF), mas há casos em que o achado pode indicar uma lesão maligna, como o carcinoma hepatocelular ou metástases hepáticas. A identificação adequada e o manejo correto desses incidentalomas são essenciais para garantir que pacientes com lesões potencialmente graves sejam tratados precocemente, enquanto aqueles com lesões benignas possam evitar intervenções desnecessárias.
Diagnóstico Radiológico Diferencial
A principal ferramenta para a avaliação de incidentalomas hepáticos é a imagem, com a ultrassonografia (US), a tomografia computadorizada (TC) e a ressonância magnética (RM) sendo as modalidades mais comumente utilizadas. Cada exame possui características específicas que ajudam na classificação e distinção das lesões hepáticas.
- Ultrassonografia (US): Frequentemente o primeiro exame realizado, a ultrassonografia pode distinguir lesões císticas de sólidas. Lesões císticas simples geralmente não requerem tratamento, enquanto lesões císticas complexas podem demandar avaliação adicional com TC ou RM. Hemangiomas e hiperplasia nodular focal também podem ser identificados com precisão por ultrassom.
- Tomografia Computadorizada (TC): A TC com contraste é frequentemente utilizada para caracterizar lesões indeterminadas vistas em ultrassonografia. Lesões benignas, como hemangiomas, apresentam padrões típicos de realce nodular periférico com enchimento centrípeto nas fases tardias do contraste. Já as metástases hepáticas, comuns em pacientes com câncer gastrointestinal ou de mama, aparecem como lesões hipovasculares com margens mal definidas e heterogeneidade interna.
- Ressonância Magnética (RM): A RM, especialmente quando realizada com contraste específico para hepatócitos, oferece alta acurácia para a caracterização de lesões hepáticas. Ela é superior à TC na diferenciação entre lesões benignas, como hiperplasia nodular focal, e lesões malignas. Incidentalomas que mostram padrões típicos de hiperintensidade em T2, como os hemangiomas, podem ser facilmente identificados com a RM.
A relevância de cada modalidade de imagem varia de acordo com a característica da lesão e o risco do paciente. Lesões menores que 1 cm, também conhecidas como too small to characterize (TSTC), representam um desafio, pois não é possível determinar com segurança sua natureza com base em imagens iniciais. Nestes casos, o seguimento por imagem a curto prazo é recomendado.
Indicação de Manejo Cirúrgico
A decisão de realizar intervenção cirúrgica em um paciente com incidentaloma hepático deve ser cuidadosamente avaliada. O manejo cirúrgico é geralmente indicado em três situações principais:
- Lesões malignas ou com suspeita de malignidade: Incidentalomas em pacientes com histórico de câncer ou em casos onde as características radiológicas sugerem malignidade (margens irregulares, crescimento rápido ou captação anômala de contraste) devem ser tratados com ressecção cirúrgica. Carcinoma hepatocelular, metástases hepáticas e colangiocarcinoma estão entre as principais malignidades a serem excluídas.
- Lesões benignas com potencial de complicação: Hemangiomas de grande volume ou adenomas hepáticos, por exemplo, podem causar complicações, como sangramento ou transformação maligna, especialmente em lesões maiores que 5 cm. Nesses casos, a ressecção pode ser indicada para prevenir eventos adversos.
- Lesões sintomáticas: Embora a maioria dos incidentalomas seja assintomática, algumas lesões benignas, como angiomiolipomas grandes, podem causar desconforto abdominal ou hepatomegalia, justificando uma intervenção cirúrgica.
Além disso, é importante considerar o risco cirúrgico do paciente. Pacientes com cirrose avançada ou outras comorbidades significativas podem não ser candidatos adequados para ressecção hepática, sendo necessário um manejo mais conservador.
Pontos-Chave para a Prática Cirúrgica
- Risco de malignidade: Cerca de 10% dos incidentalomas hepáticos são malignos. A detecção precoce e a caracterização correta da lesão são fundamentais para otimizar o tratamento.
- Segurança do paciente: O excesso de investigação pode resultar em procedimentos desnecessários, aumentando o custo e o risco para o paciente. A avaliação criteriosa das imagens e a classificação do paciente em grupos de risco podem auxiliar na tomada de decisão clínica.
- Uso adequado de imagem: A ressonância magnética com contraste específico para hepatócitos é o exame de escolha em lesões indeterminadas e proporciona alta sensibilidade e especificidade para o diagnóstico de lesões benignas e malignas.
- Indicação cirúrgica: A cirurgia está indicada em lesões malignas, lesões benignas com risco de complicação e incidentalomas sintomáticos. Nos casos de lesões muito pequenas e indeterminadas, o seguimento clínico com exames seriados é uma alternativa válida.
Conclusão
O incidentaloma hepático representa um desafio comum na prática cirúrgica, especialmente com o aumento da utilização de exames de imagem. O manejo adequado dessas lesões requer uma abordagem balanceada entre evitar procedimentos desnecessários e tratar com eficácia os casos de malignidade ou complicação potencial. O cirurgião digestivo deve basear suas decisões em uma análise detalhada das características radiológicas, história clínica do paciente e potencial de complicações. Em última análise, o manejo desses incidentalomas deve ser individualizado para otimizar os resultados para o paciente.
Como sabiamente afirmou Henri Bismuth, um dos pioneiros da cirurgia hepática: “A arte de manejar o fígado vai além da técnica operatória; ela envolve a compreensão detalhada da fisiopatologia hepática e uma estratégia bem calculada de tratamento.”
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Surgical Treatment of Pyogenic Liver Abscess
Current Approach for Digestive Surgeons
Pyogenic liver abscess (PLA) is a severe infectious condition requiring rapid diagnosis and precise management. While antibiotics may suffice for small abscesses, larger or complicated cases often necessitate surgical intervention. This post aims to discuss surgical approaches in treating PLA, highlighting intervention criteria and best practices for digestive surgeons.
Introduction
Pyogenic liver abscess is an encapsulated collection of purulent material in the liver, frequently caused by bacterial infections originating from the biliary tract or intra-abdominal sources, such as diverticulitis. Management of this condition has evolved significantly, with surgery playing a key role when antibiotic treatment alone is insufficient. Global mortality from PLA ranges from 10% to 20%, and in Brazil, this rate may be higher depending on healthcare access.
Diagnosis and Classification
PLA diagnosis relies on imaging exams such as ultrasound (US) and computed tomography (CT), essential for identifying the extent of the lesion and determining therapeutic approaches. Small abscesses, typically less than 3-4 cm, can be treated with antibiotics, while larger lesions over 5 cm often require percutaneous or surgical drainage. In Brazil, Escherichia coli and Klebsiella pneumoniae are the most common causative agents. Immunocompromised patients, such as diabetics, have a higher risk of developing PLA, and aggressive management is crucial in this population.
Surgical Approach
Surgical intervention is indicated in cases of failed percutaneous drainage or when multiloculated abscesses are present, as well as in abscess rupture cases. Image-guided percutaneous drainage has a high success rate, around 90%, in unilocular abscesses. However, large, multiloculated, or highly viscous abscesses may require more aggressive surgical intervention.
Open surgery, though less frequent due to the advent of minimally invasive techniques, remains necessary for complex multiloculated abscesses or unstable patients. Studies show that surgical management may be more effective for large abscesses (over 10 cm), especially in reducing hospital stay and ensuring complete infection resolution.
Application in Digestive Surgery
Digestive surgeons play a critical role in managing hepatic complications, such as pyogenic abscesses. In addition to mastering percutaneous drainage techniques, surgeons must be prepared to intervene surgically when necessary. Laparotomy or laparoscopy may be indicated in cases of drainage failure, multiloculated abscesses, or when extensive necrotic tissue debridement is required. In Brazil, data from the Ministry of Health show that complicated intra-abdominal infections are one of the leading causes of emergency hospital admissions. The growing prevalence of chronic diseases, such as diabetes and cirrhosis, increases the number of PLA cases, demanding that surgeons stay updated on the most effective therapeutic approaches.
Key Points
- Early Diagnosis: Imaging, especially contrast-enhanced CT, is crucial for identifying the size and location of liver abscesses.
- Surgical Intervention: Multiloculated or abscesses larger than 5 cm often require surgical intervention, especially if percutaneous drainage fails.
- Minimally Invasive Approach: Laparoscopy offers a less invasive alternative to open surgery, leading to faster recovery in specific cases.
- Comprehensive Digestive Surgeon Role: Surgical expertise is vital in managing large, multiloculated, or complicated abscesses, ensuring an effective and personalized approach.
Conclusion
The treatment of pyogenic liver abscess requires a multidisciplinary approach, with the digestive surgeon playing a crucial role in managing complex cases. The decision between percutaneous drainage and surgery depends on multiple factors, including abscess size, clinical response, and the patient’s overall condition. Technical expertise and precise surgical judgment are key to ensuring successful treatment outcomes.
As Henri Bismuth wisely stated: “Le traitement chirurgical n’est pas seulement une question de technique, mais de jugement. Le moment de l’intervention est aussi important que l’intervention elle-même.”
Liked it? Leave us a comment ✍️, share on social media, or send your question through 💬 Chat Online in our Instagram DM.
#LiverAbscess #DigestiveSurgery #AbscessTreatment #PercutaneousDrainage #HepaticAbscess
Tratamento da Colecistite Gangrenosa
Abordagem Cirúrgica e Opções Técnicos
A colecistite gangrenosa (CG) é uma complicação grave e rara da colecistite aguda, resultando em necrose da parede da vesícula biliar. Pacientes com múltiplas comorbidades, como diabetes e doenças cardiovasculares, são particularmente suscetíveis, com uma prevalência maior em idosos e homens. Este artigo explora o tratamento da colecistite gangrenosa, com ênfase na abordagem cirúrgica e nos desafios intraoperatórios.
Introdução
A colecistite gangrenosa ocorre devido à hipoperfusão da vesícula biliar, seja por obstrução aguda do ducto cístico ou por doenças arteriais, como a ateromatose da artéria cística. Essa obstrução leva a isquemia e posterior necrose da parede vesicular, culminando em complicações sérias, como perfuração e sepse. A rápida evolução para choque séptico exige diagnóstico e tratamento precoce. No Brasil, os casos de colecistite aguda são frequentes, mas as formas gangrenosas representam uma minoria, embora com alta mortalidade, especialmente em pacientes idosos e com doenças associadas. Em muitos centros, a mortalidade associada à CG pode chegar a 15%, sendo crucial que os cirurgiões digestivos estejam preparados para manejar essa condição.
Propedêutica Cirúrgica
O diagnóstico da colecistite gangrenosa é desafiador, uma vez que os sintomas muitas vezes se sobrepõem aos da colecistite aguda. Os pacientes apresentam dor no quadrante superior direito, febre e leucocitose significativa. A ultrassonografia frequentemente revela espessamento da parede vesicular e fluido pericolecístico, mas não consegue distinguir de forma confiável a forma gangrenosa da colecistite comum. Sinais clínicos de piora rápida e a evolução para síndrome de resposta inflamatória sistêmica (SIRS) indicam a necessidade de intervenção urgente. A resposta ao tratamento inicial, incluindo reposição volêmica e antibioticoterapia de amplo espectro, deve ser monitorada de perto. Quando a condição do paciente não melhora rapidamente, a intervenção cirúrgica é indicada.
Aplicação na Cirurgia Digestiva
A cirurgia de escolha para colecistite gangrenosa é a colecistectomia, sendo a laparoscopia o método preferido em centros com experiência em cirurgia minimamente invasiva. No entanto, a taxa de conversão para cirurgia aberta pode chegar a 75% em casos de CG, devido à presença de aderências densas, necrose ou dificuldade em visualizar as estruturas anatômicas cruciais, como o ducto biliar comum e a artéria cística.
Técnicas Cirúrgicas
- Abordagem convencional: A colecistectomia começa com a dissecção cuidadosa do triângulo de Calot, usando tração e contra-tração para melhor visualização. Deve-se evitar a dissecção agressiva da vesícula, especialmente quando há risco de perfuração, para minimizar a contaminação da cavidade abdominal com bile purulenta.
- Abordagem top-down: Em casos de inflamação severa, uma abordagem “fundus-first” pode ser adotada, dissecando a vesícula do fundo até a placa cística, o que pode reduzir o risco de lesões aos ductos biliares principais.
- Colecistectomia subtotal: Quando a inflamação ou necrose impede a dissecção segura, uma colecistectomia subtotal pode ser realizada. Embora essa abordagem evite lesões vasculares e biliares, ela pode estar associada a complicações tardias, como retenção de cálculos no coto cístico e necessidade de reintervenção.
Pontos-chave
- Risco aumentado: Pacientes com colecistite gangrenosa têm risco significativamente maior de morbidade e mortalidade, com taxas de conversão para cirurgia aberta superiores a 50%.
- Preferência pela colecistectomia: Embora a drenagem percutânea da vesícula possa ser uma opção temporária para pacientes com alto risco cirúrgico, a colecistectomia continua sendo o tratamento definitivo para CG, pois remove a fonte de infecção.
- Técnica laparoscópica: A laparoscopia é preferida quando viável, mas a conversão para laparotomia deve ser considerada precocemente quando houver dificuldades técnicas ou risco de lesão aos ductos biliares.
- Importância da colangiografia: Em casos de anatomia duvidosa ou inflamação grave, a colangiografia intraoperatória pode ajudar a definir as estruturas anatômicas e prevenir lesões aos ductos biliares.
Conclusão Aplicada à Prática do Cirurgião Digestivo
A colecistite gangrenosa representa um desafio considerável para cirurgiões digestivos, especialmente em pacientes idosos e com múltiplas comorbidades. A identificação precoce e a intervenção cirúrgica apropriada são cruciais para melhorar os resultados clínicos. Embora a colecistectomia laparoscópica seja ideal, a alta taxa de conversão para a técnica aberta demonstra a complexidade do procedimento. O manejo adequado da CG exige uma equipe cirúrgica experiente e a capacidade de tomar decisões intraoperatórias rápidas para minimizar as complicações.
“Não existe cálculo biliar inocente.”
– Charles Mayo (1865-1939)
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#colecistitegangrenosa
#cirurgiadigestiva
#colecistectomiaminimamenteinvasiva
#urgenciascirurgicas
#saudedigestiva
Tratamento da Coledocolitíase no Paciente com Bypass Gástrico
Abordagens Cirúrgicas e Opções Técnicas
A coledocolitíase, caracterizada pela presença de cálculos no ducto biliar comum (CBD), é uma complicação potencialmente grave que pode causar icterícia obstrutiva, pancreatite e colangite. Em pacientes que realizaram o bypass gástrico em Y-de-Roux, o tratamento dessa condição se torna mais complexo devido às alterações anatômicas decorrentes da cirurgia bariátrica, tornando o acesso ao ducto biliar pelo método endoscópico tradicional (CPRE) um desafio significativo.
Introdução
O bypass gástrico em Y-de-Roux é um dos procedimentos mais realizados no tratamento da obesidade mórbida, resultando em perda de peso significativa e, consequentemente, em uma maior prevalência de cálculos biliares. No entanto, as alterações anatômicas criadas por esse procedimento, como a separação do estômago proximal do restante do trato gastrointestinal, impedem o acesso ao ducto biliar pelo estômago convencional. Nesse contexto, o manejo da coledocolitíase, que afeta até 18% dos pacientes submetidos à colecistectomia, exige uma abordagem multidisciplinar e técnicas cirúrgicas e endoscópicas inovadoras.
Propedêutica Cirúrgica
O diagnóstico e o manejo da coledocolitíase em pacientes com bypass gástrico podem ser desafiadores, pois o acesso ao ducto biliar comum por vias endoscópicas tradicionais é inviável. Nestes casos, a apresentação clínica pode variar de sintomas leves de icterícia a quadros graves de colangite ascendente.
Para a avaliação inicial, a ultrassonografia abdominal continua sendo o exame de escolha para identificar dilatação do ducto biliar e a presença de cálculos. No entanto, devido à complexidade anatômica, exames como a colangiopancreatografia por ressonância magnética (MRCP) são frequentemente necessários para fornecer uma visão mais detalhada da anatomia do trato biliar.
Uma vez confirmado o diagnóstico de coledocolitíase, o tratamento deve ser instituído rapidamente, a fim de evitar complicações como abscessos hepáticos ou pancreatite biliar. As opções terapêuticas variam dependendo da gravidade da apresentação clínica e da disponibilidade de recursos no centro de tratamento. Para pacientes com colangite ascendente, por exemplo, a descompressão urgente do ducto biliar é necessária. Nestes casos, o uso de técnicas minimamente invasivas, como a colangiografia trans-hepática percutânea (PTC), pode ser a melhor alternativa quando a CPRE não é viável.
Em pacientes estáveis, várias abordagens cirúrgicas são possíveis. Uma técnica amplamente utilizada é a colecistectomia laparoscópica assistida por CPRE transgástrica. Nesse procedimento, um acesso cirúrgico é feito no estômago excluído do paciente, permitindo que o endoscópio seja inserido diretamente na papila duodenal, facilitando a remoção dos cálculos e a descompressão do ducto biliar.
Aplicação na Cirurgia Digestiva
A cirurgia digestiva avançada em pacientes com bypass gástrico exige um planejamento cuidadoso e uma abordagem técnica que considere as alterações anatômicas decorrentes da cirurgia bariátrica. As seguintes estratégias têm sido eficazes no manejo da coledocolitíase nesses pacientes:
- ERCP assistida por laparoscopia: Esse procedimento permite o acesso ao trato biliar usando técnicas combinadas de laparoscopia e endoscopia. A transposição do endoscópio pelo estômago excluído oferece uma alternativa eficiente para a remoção dos cálculos, com baixa taxa de complicações.
- Colangiografia trans-hepática percutânea (PTC): Em pacientes instáveis ou com contraindicações para procedimentos invasivos, a PTC é uma alternativa valiosa para a descompressão biliar emergente. Essa técnica minimamente invasiva pode ser associada à dilatação do esfíncter de Oddi para expulsar os cálculos.
- Exploração cirúrgica do ducto biliar comum: Em casos complexos, onde as técnicas minimamente invasivas falham, a exploração laparoscópica ou aberta do ducto biliar pode ser necessária. A abordagem transcística ou a coledocotomia são opções viáveis, dependendo da localização e do tamanho dos cálculos. A utilização de cateteres tipo Fogarty ou cestas de Dormia pode ser útil para a remoção dos cálculos.
- Técnicas reconstrutivas: Para pacientes com múltiplos cálculos biliares ou complicações, a realização de anastomoses biliodigestivas, como a coledocoduodenostomia ou a hepaticojejunostomia em Y-de-Roux, pode ser considerada. Essas abordagens proporcionam um alívio duradouro, evitando futuros episódios de obstrução.
Pontos-chave
- Pacientes com bypass gástrico em Y-de-Roux apresentam desafios únicos no tratamento da coledocolitíase devido à anatomia alterada do trato digestivo.
- A ERCP tradicional é inviável nesses pacientes, sendo substituída por abordagens combinadas, como a ERCP transgástrica assistida por laparoscopia ou a PTC.
- A ultrassonografia e a MRCP são ferramentas essenciais para o diagnóstico, enquanto a intervenção cirúrgica deve ser cuidadosamente planejada, considerando as condições clínicas e anatômicas do paciente.
- Em centros especializados, as taxas de sucesso com essas abordagens minimamente invasivas são altas, com complicações graves ocorrendo em menos de 10% dos casos.
Conclusão Aplicada à Prática do Cirurgião Digestivo
O tratamento da coledocolitíase em pacientes submetidos ao bypass gástrico representa um desafio técnico significativo, exigindo conhecimento especializado em cirurgia digestiva avançada e endoscopia. A abordagem ideal deve ser individualizada, considerando a condição clínica do paciente e as habilidades da equipe cirúrgica. As técnicas minimamente invasivas, como a ERCP assistida por laparoscopia, são as preferidas quando viáveis, mas o cirurgião deve estar preparado para realizar intervenções mais complexas, como a coledocotomia ou anastomoses biliodigestivas, quando necessário.
Nas palavras do cirurgião Steven Strasberg, “O sucesso na cirurgia hepato-biliar depende não apenas da habilidade técnica, mas também da capacidade de adaptar-se às circunstâncias anatômicas e clínicas únicas de cada paciente.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#coledocolitiase
#bypassgastrico
#cirurgiabariatrica
#cirurgiadigestiva
#cprefalha
Tratamento da Colelitíase no Paciente Cirrótico
Desafios e Abordagens Cirúrgicas
A colelitíase, ou presença de cálculos na vesícula biliar, é uma condição comum em cirróticos, mas seu manejo cirúrgico apresenta desafios únicos devido às complicações inerentes à cirrose e à hipertensão portal. A cirurgia, neste contexto, exige uma abordagem criteriosa para minimizar riscos e maximizar a segurança do paciente. Este artigo destina-se a estudantes de medicina, residentes de cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo, com o objetivo de fornecer uma visão abrangente sobre o tratamento da colelitíase no paciente cirrótico, destacando as melhores práticas, evidências científicas e diretrizes atuais.
Introdução
A cirrose é uma condição crônica que afeta severamente a função hepática e está associada a um aumento expressivo da morbidade e mortalidade perioperatória. No Brasil, a cirrose hepática é responsável por 10 mil óbitos anuais, segundo o Ministério da Saúde, o que reforça a importância do seu manejo adequado, sobretudo em contextos cirúrgicos. A prevalência de colelitíase em pacientes cirróticos pode chegar a 30%, sendo que esses pacientes estão sujeitos a complicações graves, como a hipertensão portal e a coagulopatia, que complicam a realização de uma colecistectomia.
Portanto, ao tratar a colelitíase em pacientes com cirrose, é fundamental um planejamento cirúrgico cuidadoso e uma consideração detalhada dos riscos associados. O objetivo deste artigo é discutir as opções de tratamento para colelitíase em cirróticos, com ênfase nas abordagens cirúrgicas e estratégias para otimizar a segurança e os resultados.
Colecistectomia x Cirrose Hepática
Pacientes com cirrose hepática apresentam risco aumentado de complicações cirúrgicas, em parte devido à hipertensão portal, varizes perivesiculares, coagulopatia e distorção anatômica do fígado. A avaliação pré-operatória inclui a classificação do paciente pelo escore Child-Pugh ou MELD (Model for End-stage Liver Disease), que ajudam a prever a mortalidade pós-operatória. Um escore MELD acima de 14, por exemplo, indica um risco significativo de mortalidade após procedimentos cirúrgicos.
A colecistectomia é a principal intervenção cirúrgica para o tratamento da colelitíase, e a via laparoscópica é preferida, sempre que possível, devido aos seus benefícios em comparação com a abordagem aberta. Estudos mostram que a colecistectomia laparoscópica (LC) em pacientes cirróticos está associada a menor perda de sangue, menos infecções de ferida e internação hospitalar mais curta. No entanto, o risco de complicações, como hemorragia, é cerca de 20% maior em comparação com pacientes sem cirrose.
Em pacientes cirróticos com colecistite aguda, a situação torna-se ainda mais delicada. Nestes casos, o manejo não operatório inicial é preferível, incluindo o uso de antibióticos e drenagem percutânea da vesícula biliar (colecistostomia), especialmente para pacientes com cirrose avançada (Child-Pugh C ou MELD elevado). Caso a condição clínica do paciente melhore com o manejo não cirúrgico, uma colecistectomia eletiva pode ser planejada após seis semanas, garantindo uma melhor estabilidade hemodinâmica e recuperação hepática.
Aplicação na Cirurgia Digestiva
No contexto da cirurgia digestiva, a principal abordagem para pacientes cirróticos com colelitíase é a colecistectomia laparoscópica. A técnica laparoscópica oferece vantagens significativas em comparação com a abordagem aberta, como menor trauma cirúrgico, recuperação mais rápida e menor taxa de infecção. Contudo, devido à complexidade anatômica causada pela cirrose e presença de varizes, a dissecção deve ser realizada com extremo cuidado, evitando a manipulação excessiva da área do triângulo de Calot, onde as varizes são comuns.
Em casos de inflamação severa ou varizes pericolecísticas, a abordagem de colecistectomia subtotal pode ser preferida. Esta técnica permite remover a maior parte da vesícula, minimizando o risco de sangramento catastrófico que pode ocorrer durante a dissecção em áreas com hipertensão portal. O uso de dispositivos de energia para selar vasos sanguíneos e técnicas de hemostasia avançadas, como o uso de cola de fibrina e selantes de celulose, são fundamentais para evitar sangramentos.
Outro ponto relevante é a necessidade de otimização médica pré-operatória. Cirróticos devem ter a coagulopatia corrigida antes da cirurgia, com a administração de vitamina K e, em alguns casos, plasma fresco congelado. A função renal e cardíaca também deve ser monitorada rigorosamente, e a presença de ascite deve ser manejada com diuréticos. Em centros de referência, o uso de octreotida intravenosa para reduzir a pressão portal pode ser considerado.
Pontos-chave
- Risco aumentado: A cirrose aumenta significativamente a mortalidade cirúrgica, com uma taxa de até 60% em pacientes com Child-Pugh C.
- Abordagem laparoscópica: A colecistectomia laparoscópica é a abordagem preferida, com menor morbidade em comparação com a cirurgia aberta.
- Manejo não cirúrgico inicial: Em pacientes com colecistite aguda e alto risco cirúrgico, drenagem percutânea deve ser considerada antes de uma intervenção cirúrgica definitiva.
- Técnicas hemostáticas avançadas: O uso de dispositivos de energia e hemostáticos auxiliares é crucial para minimizar o risco de sangramento intraoperatório.
- Avaliação pré-operatória cuidadosa: A classificação Child-Pugh e o escore MELD são essenciais para prever a mortalidade e determinar a melhor abordagem terapêutica.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
O tratamento da colelitíase no paciente cirrótico exige uma abordagem individualizada e multidisciplinar. A escolha da intervenção, seja ela cirúrgica ou não, deve considerar a gravidade da doença hepática, a presença de complicações associadas e as habilidades técnicas do cirurgião. No Brasil, onde a cirrose é uma causa frequente de morbidade, é crucial que cirurgiões e equipes médicas sejam bem treinados para manejar essas situações com a maior segurança possível.
A colecistectomia laparoscópica permanece o padrão-ouro no manejo da colelitíase, mas cirurgiões devem estar preparados para adaptar suas técnicas e, quando necessário, optar por abordagens alternativas, como a colecistectomia subtotal. Além disso, a otimização pré-operatória e a correção das disfunções fisiológicas são fundamentais para reduzir complicações e melhorar os desfechos cirúrgicos.
Como afirmou Steven Strasberg, “A dissecção cuidadosa do triângulo de Calot e o uso de abordagens alternativas em cirurgias de risco são elementos essenciais para o sucesso cirúrgico em pacientes com complicações hepáticas.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
O Papel da CPRE no Tratamento da Pancreatite Aguda Biliar
A colangiopancreatografia retrógrada endoscópica (CPRE) tem sido estudada como uma potencial intervenção para reduzir a gravidade da pancreatite aguda biliar (AP). Contudo, diversas pesquisas sugerem que seu benefício é limitado a cenários clínicos específicos. Este artigo tem como objetivo explorar o papel da CPRE na pancreatite biliar, com base nas evidências mais recentes.
CPRE na Pancreatite Biliar Leve
Para pacientes com pancreatite biliar leve, estudos demonstram que a CPRE não traz benefícios significativos na redução da gravidade ou complicações da doença. Um estudo multicêntrico randomizado não encontrou vantagens na realização de CPRE urgente com esfinterotomia em comparação ao manejo conservador nesses pacientes.88,101 Portanto, a intervenção precoce com CPRE não é recomendada para casos de pancreatite biliar leve, com a prática de manejo conservador prevalecendo como a mais indicada.
CPRE na Pancreatite Biliar Grave
A CPRE também foi avaliada em pacientes com pancreatite biliar grave, mas os resultados não sustentam seu uso de rotina. Em um ensaio clínico multicêntrico, pacientes com pancreatite biliar grave, sem sinais de colangite, foram submetidos a CPRE urgente com esfinterotomia versus manejo conservador. Os resultados mostraram que a CPRE urgente não reduziu significativamente as complicações principais ou a mortalidade (38% no grupo CPRE vs 44% no grupo conservador).102 Esses achados indicam que, em pacientes com pancreatite biliar grave, sem colangite, o manejo conservador deve ser preferido.
Em outro estudo, os pesquisadores avaliaram a CPRE em pacientes com pancreatite grave e confirmação de cálculos ou lama biliar no ducto biliar comum por ultrassonografia endoscópica (EUS). Os resultados, novamente, mostraram que a CPRE com esfinterotomia não reduziu a mortalidade ou as principais complicações em comparação ao tratamento conservador em seis meses.103
Indicações Atuais da CPRE na Pancreatite Biliar
Com base nas evidências disponíveis, as diretrizes atuais recomendam que a CPRE não seja realizada de forma rotineira em casos de pancreatite biliar, independentemente da gravidade, na ausência de colangite.23
Contudo, a CPRE tem um papel crucial em dois cenários específicos:
- Colangite associada à pancreatite: A CPRE deve ser realizada urgentemente quando há suspeita ou confirmação de colangite concomitante.
- Coledocolitíase sintomática ou persistente: Nos casos de cálculos biliares no ducto biliar comum (coledocolitíase) que causam sintomas persistentes ou em pacientes sem resolução espontânea da obstrução, a CPRE é indicada eletivamente.
Conclusões
A CPRE tem um papel limitado no manejo da pancreatite biliar, sendo indicada apenas em casos de colangite ou coledocolitíase sintomática. Nos demais casos, o manejo conservador é preferível, conforme demonstrado por estudos recentes e as diretrizes atuais. Com esses achados, a CPRE deve ser utilizada de forma seletiva e com base em critérios clínicos rigorosos, evitando o uso desnecessário dessa intervenção em pacientes que podem ser manejados de forma conservadora.
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#CPRE #PancreatiteAguda #TratamentoPancreatite #Colangite #Coledocolitiase
Tratamento Cirúrgico da Pancreatite Aguda Biliar
Momento Ideal da Colecistectomia e Manejo da Necrose Pancreática Infectada
A pancreatite aguda biliar é uma condição inflamatória potencialmente grave, resultante da obstrução temporária ou permanente do ducto biliar por cálculos. A sua prevalência é significativa, sendo uma das causas mais comuns de pancreatite aguda em países como o Brasil, com uma incidência estimada de até 40% dos casos de pancreatite. O manejo cirúrgico desta doença, especialmente em casos complicados como a necrose pancreática infectada, é um desafio que requer decisões cuidadosas e multidisciplinares. Este artigo tem como objetivo educar estudantes de medicina, residentes em cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo sobre os princípios fundamentais do tratamento cirúrgico da pancreatite aguda biliar, com ênfase na colecistectomia e no manejo da necrose pancreática infectada.
Momento da Colecistectomia
A colecistectomia é o tratamento definitivo para a pancreatite biliar, pois remove a causa subjacente: os cálculos biliares. O momento ideal para realizar a colecistectomia em pacientes com pancreatite biliar aguda é uma decisão crucial, com implicações significativas para o prognóstico e o risco de complicações.
Nos casos de pancreatite biliar leve, a literatura recomenda fortemente a realização da colecistectomia durante a mesma internação hospitalar. Estudos demonstram que esta abordagem reduz significativamente o risco de novos episódios de pancreatite ou outras complicações relacionadas aos cálculos biliares. Um ensaio clínico multicêntrico relatou que 17% dos pacientes que tiveram a colecistectomia adiada experimentaram complicações relacionadas à presença de cálculos, em comparação com apenas 5% dos pacientes que realizaram a cirurgia durante a mesma internação. Além disso, não houve aumento nas taxas de complicações cirúrgicas naqueles submetidos à colecistectomia precoce.
Em pacientes com pancreatite moderadamente grave ou grave, o manejo é mais complexo. Diretrizes internacionais e especialistas recomendam que a colecistectomia seja adiada até que o paciente tenha se recuperado completamente e eventuais coleções líquidas pancreáticas ou necrose tenham se resolvido. No Brasil, os dados mostram que aproximadamente 10% a 20% dos pacientes com pancreatite aguda evoluem para formas mais graves com necrose, exigindo uma abordagem mais conservadora e cuidadosa. Um estudo retrospectivo indicou que a realização da colecistectomia antes de 8 a 10 semanas pode reduzir o risco de recorrência de eventos biliares sem aumentar o risco de complicações.
Indicações do Manejo Cirúrgico da Necrose Pancreática Infectada
A necrose pancreática, que ocorre em cerca de 10% a 20% dos casos de pancreatite aguda, é uma complicação grave que pode levar à infecção em aproximadamente um terço dos pacientes. A presença de necrose infectada está associada a altas taxas de morbidade e mortalidade, com estudos demonstrando uma mortalidade de 36% a 49,5% em pacientes com falência orgânica primária e necrose sobreposta infectada.
O manejo da necrose pancreática infectada tem evoluído significativamente nas últimas décadas. A abordagem minimamente invasiva em etapas é atualmente considerada a via preferida de tratamento, em oposição à cirurgia aberta tradicional, devido a menores taxas de complicações e mortalidade. Esta abordagem começa com a drenagem percutânea direcionada, seguida por procedimentos como drenagem transmural endoscópica e necrosectomia endoscópica direta, se necessário. Nos casos mais complexos, onde essas medidas não são eficazes, pode-se recorrer a desbridamento cirúrgico minimamente invasivo, que inclui técnicas como a necrosectomia retroperitoneal videoassistida.
A escolha da técnica e o momento da intervenção dependem da extensão da necrose, da presença de infecção e das condições clínicas do paciente. Em geral, a intervenção invasiva deve ser adiada por pelo menos 4 semanas após o início da pancreatite para permitir que a necrose se delimite (necrose encapsulada). Em pacientes gravemente doentes, com infecção progressiva ou falência de múltiplos órgãos, intervenções precoces podem ser consideradas, embora estudos recentes sugiram que o adiamento da drenagem pode reduzir a necessidade de múltiplas intervenções.
Pontos-Chave e Conclusões Aplicadas à Prática do Cirurgião Digestivo
- Colecistectomia Precoce em Pancreatite Biliar Leve: A colecistectomia na mesma internação reduz a recorrência de eventos biliares e não aumenta as complicações cirúrgicas. Este é o manejo recomendado para pacientes com pancreatite biliar leve.
- Adiar a Colecistectomia em Casos Graves: Em pacientes com pancreatite moderadamente grave ou grave, o adiamento da colecistectomia até a resolução de coleções ou necrose é indicado, geralmente em um intervalo de 8 a 10 semanas.
- Manejo Minimamente Invasivo da Necrose Pancreática Infectada: A abordagem em etapas minimamente invasiva deve ser priorizada, começando com drenagem percutânea. A intervenção cirúrgica deve ser reservada para casos refratários ou com deterioração clínica, com preferências por técnicas menos invasivas.
- Intervenção Invasiva Oportuna: O momento da intervenção deve ser cuidadosamente avaliado, com um atraso de pelo menos 4 semanas sendo o ideal para permitir a formação de necrose encapsulada. No entanto, intervenções precoces podem ser necessárias em casos críticos.
O manejo da pancreatite aguda biliar exige uma equipe multidisciplinar experiente e a consideração cuidadosa das condições clínicas do paciente. Cirurgiões do aparelho digestivo devem estar cientes das evidências mais recentes para otimizar os resultados cirúrgicos e minimizar complicações. Como disse William Osler, um dos grandes médicos da história: “A prática da medicina é uma arte, baseada na ciência.” A tomada de decisões cirúrgicas no manejo da pancreatite biliar reflete esta delicada interação entre arte e ciência, onde a experiência clínica e o julgamento são essenciais para alcançar os melhores resultados.
Gostou❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#PancreatiteAguda #TratamentoCirúrgico #Colecistectomia #NecrosePancreática #CirurgiaDigestiva
A Filosofia da Cirurgia
No contexto do tratamento cirúrgico das doenças do aparelho digestivo, a formação de um cirurgião vai além do domínio técnico. Inspirados nos ensinamentos de Galeno, renomado médico e filósofo da antiguidade, reconhecemos que o cirurgião ideal deve incorporar uma combinação de habilidades práticas e virtudes filosóficas que guiam sua conduta ética e profissional. Neste artigo, direcionado a estudantes de medicina, residentes de cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo, exploraremos as cinco virtudes fundamentais que definem o “cirurgião filósofo”. Essas virtudes não apenas aprimoram a prática clínica, mas também integram os princípios éticos da medicina na vida do cirurgião, como enfatizado por Galeno.
1. Proeficiência Técnica e Conhecimento Filosófico
O bom cirurgião não é apenas aquele que domina as técnicas cirúrgicas com excelência, mas também aquele que entende a importância de uma formação filosófica sólida. Para Galeno, a medicina e a filosofia caminham lado a lado, pois o cirurgião deve ser capaz de fazer julgamentos críticos baseados em raciocínios demonstrativos, além de possuir uma visão clara das causas e consequências de suas ações.
“O médico que pratica sua arte visando apenas ganhos pessoais é um charlatão; somente o conhecimento profundo do corpo e da alma o torna um verdadeiro mestre de sua prática.” – Galeno.
2. Ética e Humanidade
A prática cirúrgica, quando orientada pela ética e pelo respeito à vida humana, transforma-se em uma verdadeira arte de cura. Galeno reforça que o cirurgião não deve estar motivado por ambições financeiras ou status social, mas pela filantropia – o desejo genuíno de aliviar o sofrimento humano. Ao tratar doenças do aparelho digestivo, essa virtude é especialmente relevante, pois muitas intervenções são decisivas para a qualidade de vida do paciente.
“A medicina existe para o benefício da humanidade; aquele que visa o lucro ao invés de curar não é digno do título de médico.” – Galeno.
3. Disciplina e Dedicação Inabalável
A vida de um cirurgião é marcada por noites de estudo e jornadas exaustivas no centro cirúrgico. Para Galeno, a dedicação é a chave para o sucesso na prática médica. Ele descreve o verdadeiro cirurgião como alguém que sacrifica o próprio conforto pela excelência no cuidado dos pacientes, não permitindo que o cansaço ou a rotina afete sua atenção e precisão cirúrgica.
“Passei noites sem dormir por meus pacientes, pois, como um escravo do meu ofício, minha dedicação não conhecia limites.” – Galeno.
4. Humildade e Rejeição ao Luxo
A humildade, segundo Galeno, é uma virtude essencial para um cirurgião. Ele critica fortemente aqueles que se entregam ao luxo e à bajulação, comportamentos que desviam o foco do verdadeiro propósito da medicina. O cirurgião deve evitar a busca por reconhecimento superficial, concentrando-se na melhoria contínua de suas habilidades e na obtenção de resultados clínicos favoráveis para seus pacientes.
“O verdadeiro médico deve evitar o luxo e a bajulação; sua maior honra é a saúde de seus pacientes.” – Galeno.
5. Responsabilidade Social e Participação Comunitária
Para Galeno, a medicina vai além das portas dos hospitais. O cirurgião filósofo deve estar envolvido em sua comunidade, utilizando seu conhecimento e habilidades para o benefício da sociedade como um todo. Isso inclui atuar em programas de saúde pública, educação médica e prevenção de doenças. Um cirurgião deve ser um líder que promove a saúde e o bem-estar, não apenas em seus pacientes, mas na população em geral.
“A medicina, em sua essência, é uma arte pública, destinada a servir todos os membros da sociedade, do mais nobre ao mais humilde.” – Galeno.

Conclusão
As virtudes mencionadas por Galeno são mais do que um código moral; elas são uma diretriz prática para os cirurgiões que desejam integrar a filosofia em sua prática médica. Essas virtudes formam o alicerce de uma carreira dedicada não apenas à técnica cirúrgica, mas à verdadeira arte de cuidar da vida humana. Como futuros cirurgiões do aparelho digestivo, é fundamental adotar essas virtudes para uma prática cirúrgica que vá além da cura física, abraçando a responsabilidade ética e social que acompanha a medicina.
“Um médico que cura corpos, mas não cultiva o bem em sua alma, jamais será verdadeiramente completo.” – Galeno.
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#cirurgiageral #aparelhodigestivo #éticaemmedicina #galeno #cirurgiafilosófica
Cirurgia Bariátrica na Prevenção do Câncer de Mama
Impactos e Implicações para a Prática do Cirurgião Digestivo
A cirurgia bariátrica tem se destacado não apenas como uma opção eficaz no tratamento da obesidade mórbida, mas também na prevenção de diversas comorbidades associadas, incluindo o câncer de mama. Estudos recentes apontam que essa intervenção cirúrgica, amplamente realizada no Brasil, pode reduzir de forma significativa o risco de desenvolvimento do câncer de mama em mulheres obesas. Este texto tem como objetivo revisar as evidências científicas que relacionam a cirurgia bariátrica à prevenção do câncer de mama, destacando os mecanismos fisiológicos e os impactos práticos dessa abordagem para o cirurgião digestivo.
Introdução
A obesidade é um dos principais fatores de risco modificáveis para o câncer de mama, especialmente em mulheres pós-menopausa. No Brasil, o câncer de mama é o tipo mais comum entre as mulheres, representando cerca de 29,7% dos casos novos de câncer, segundo o Instituto Nacional de Câncer (INCA). A relação entre obesidade e câncer de mama ocorre por meio de múltiplos mecanismos, como o aumento da inflamação sistêmica, disfunção do tecido adiposo visceral e elevação dos níveis de insulina e estrógenos, que contribuem para o desenvolvimento e progressão tumoral. Nesse contexto, a cirurgia bariátrica emerge como uma intervenção preventiva, atuando na redução dos fatores de risco ao promover uma perda de peso sustentada e melhora das condições metabólicas.
Redução do Risco de Câncer de Mama
Estudos robustos indicam que a cirurgia bariátrica pode reduzir em até 49% o risco de câncer de mama em mulheres obesas. Uma meta-análise publicada na Surgery for Obesity and Related Diseases demonstrou que, em comparação com controles de peso semelhante, mulheres submetidas à cirurgia bariátrica apresentaram uma redução significativa na incidência de câncer de mama. A perda de peso substancial, aliada à melhora dos distúrbios metabólicos, reduz os níveis de inflamação sistêmica e limita a proliferação celular associada à carcinogênese.
Ademais, além de diminuir a incidência, há evidências que sugerem que a cirurgia bariátrica pode favorecer diagnósticos de câncer de mama em estágios mais precoces, o que tem implicações importantes para o prognóstico e a resposta ao tratamento oncológico. No Brasil, onde a obesidade afeta 25,9% da população adulta feminina, estratégias preventivas como a cirurgia bariátrica podem ter um impacto expressivo na saúde pública, reduzindo a carga de câncer de mama no país.
Mecanismos Fisiológicos
A cirurgia bariátrica promove a perda de peso significativa e melhora a síndrome metabólica, impactando diretamente nos mecanismos que ligam a obesidade ao câncer de mama. A diminuição da hiperinsulinemia, por exemplo, reduz a sinalização proliferativa mediada pela insulina, que favorece o crescimento de células tumorais. Outro fator importante é a redução da atividade da enzima aromatase, encontrada no tecido adiposo, que converte andrógenos em estrógenos e estimula o crescimento de tumores sensíveis a hormônios.
Além disso, a perda de tecido adiposo visceral após a cirurgia bariátrica diminui a liberação de citocinas pró-inflamatórias, como o fator de necrose tumoral-alfa (TNF-α) e a interleucina-6 (IL-6), que estão associadas à promoção do crescimento tumoral. A melhora no perfil inflamatório sistêmico pós-cirurgia contribui, portanto, para a redução do risco de desenvolvimento de câncer de mama.
Aplicação na Cirurgia Digestiva
Para o cirurgião do aparelho digestivo, entender a relação entre cirurgia bariátrica e prevenção do câncer de mama é fundamental na abordagem multidisciplinar de pacientes obesas. A decisão de indicar a cirurgia bariátrica deve levar em consideração não apenas os benefícios relacionados à perda de peso e controle de doenças metabólicas, mas também os potenciais ganhos em termos de prevenção oncológica. No Brasil, o número de cirurgias bariátricas cresceu significativamente, sendo realizadas mais de 100 mil cirurgias por ano, segundo a Sociedade Brasileira de Cirurgia Bariátrica e Metabólica (SBCBM). A utilização de procedimentos como o bypass gástrico e a gastrectomia vertical têm mostrado benefícios não apenas para a obesidade, mas também na redução do risco de câncer de mama, especialmente em pacientes com histórico familiar ou predisposição genética.
Pontos-chave para a Prática do Cirurgião Digestivo
- Seleção de Pacientes: A indicação de cirurgia bariátrica deve ser ponderada em mulheres obesas com fatores de risco adicionais para o câncer de mama, como histórico familiar e presença de síndrome metabólica.
- Acompanhamento Multidisciplinar: O cirurgião digestivo deve trabalhar em conjunto com oncologistas, endocrinologistas e ginecologistas para monitorar as pacientes pós-operatórias, otimizando a prevenção oncológica.
- Estudos Complementares: É essencial acompanhar as novas evidências que investigam o impacto da cirurgia bariátrica sobre o risco de diferentes tipos de câncer, incluindo o de mama, para uma prática baseada em evidências.
Conclusão
A cirurgia bariátrica apresenta benefícios amplamente documentados no tratamento da obesidade e suas complicações metabólicas. As evidências sugerem que essa intervenção também exerce um papel preventivo importante na redução do risco de câncer de mama, principalmente em mulheres com obesidade mórbida. Para o cirurgião digestivo, compreender essa relação pode ajudar na melhor indicação cirúrgica, oferecendo uma abordagem mais abrangente na promoção da saúde e na prevenção de doenças graves, como o câncer de mama.
Na prática diária, o cirurgião digestivo deve considerar a cirurgia bariátrica não apenas como uma ferramenta de controle de peso, mas também como uma intervenção com potenciais benefícios oncológicos, especialmente em pacientes obesas de alto risco. A vigilância contínua, o acompanhamento rigoroso e a atuação em equipes multidisciplinares são cruciais para garantir os melhores desfechos.
Como afirmou Halsted: “A cirurgia preventiva pode não apenas salvar vidas, mas também proporcionar uma qualidade de vida superior ao eliminar os riscos antes que eles se materializem.”
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags: #cirurgiabariátrica #câncerdemama #prevençãodecâncer #obesidadeemulheres #cirurgiãodigestivo