Cuidados Perioperatórios na Cirurgia Laparoscópica
Otimizando a Fisiologia e Acelerando a Recuperação no Aparelho Digestivo
Autor: Prof. Dr. Ozimo Gama Categoria: Cuidados Perioperatórios / Cirurgia Laparoscópica / Cirurgia do Aparelho Digestivo Tempo de Leitura: 10 minutos
Introdução
A cirurgia laparoscópica revolucionou a especialidade do aparelho digestivo ao atenuar o trauma cirúrgico, reduzir a resposta inflamatória sistêmica e proporcionar uma recuperação estética e funcional superior. No entanto, para o estudante de medicina, o residente de cirurgia geral e o pós-graduando, é imperativo compreender que incisões milimétricas não isentam o paciente da resposta endócrino-metabólica ao trauma. No Brasil, o Sistema Único de Saúde (SUS) realiza centenas de milhares de procedimentos videolaparoscópicos anualmente, desde colecistectomias até gastrectomias oncológicas complexas. Para maximizar os benefícios da via minimamente invasiva, a excelência no centro cirúrgico deve ser invariavelmente acompanhada por um manejo perioperatório de vanguarda. A transição dos cuidados dogmáticos do passado para protocolos modernos baseados em evidências — como o ERAS (Enhanced Recovery After Surgery) e o Projeto ACERTO (Aceleração da Recuperação Total Pós-Operatória), amplamente adotado e validado em território nacional — é o que define o cirurgião contemporâneo de alta performance.
A Fisiologia do Pneumoperitônio e a Quebra de Paradigmas
O manejo perioperatório na laparoscopia exige um profundo respeito pelas alterações fisiológicas induzidas pelo pneumoperitônio com dióxido de carbono (CO2). A insuflação abdominal eleva a pressão intra-abdominal (usualmente entre 12 e 15 mmHg), acarretando compressão da veia cava inferior, diminuição do retorno venoso, aumento da resistência vascular sistêmica e elevação das pressões das vias aéreas com risco de hipercapnia. A equipe cirúrgico-anestésica deve atuar em sintonia para mitigar estes efeitos.
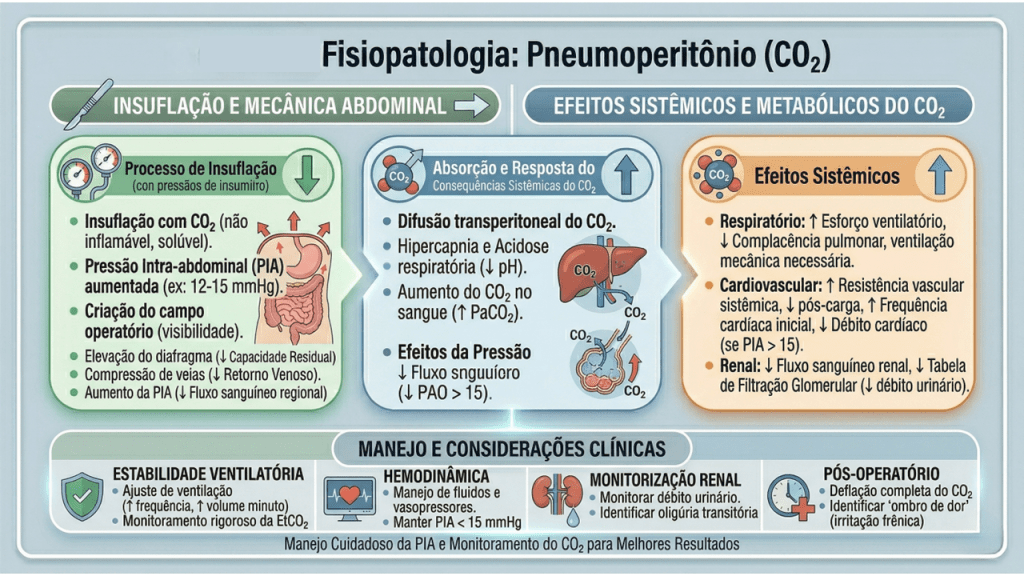
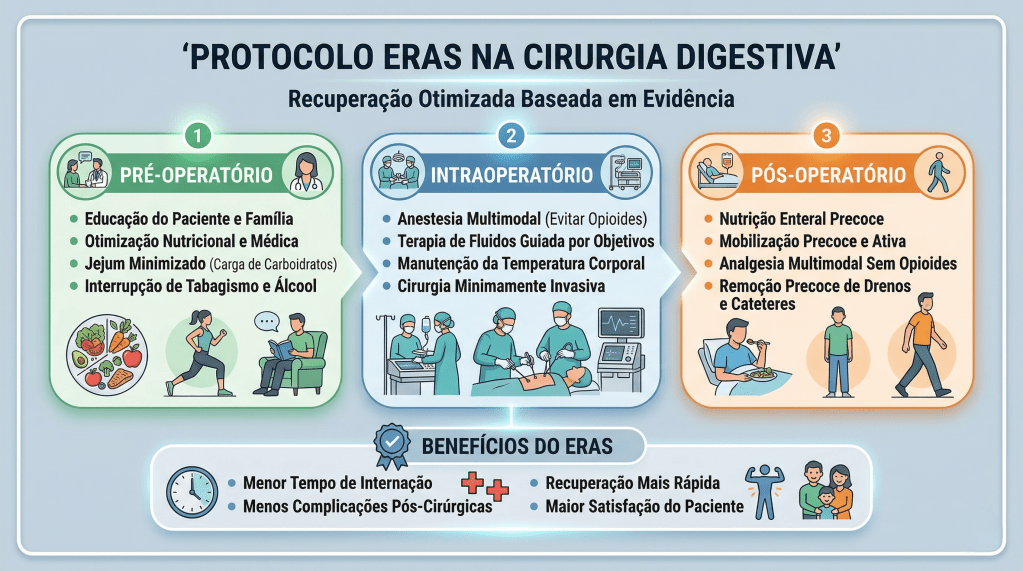
Sob a ótica dos protocolos de aceleração da recuperação, o manejo perioperatório é dividido em três fases cruciais, onde dogmas seculares foram cientificamente derrubados:
- Pré-Operatório (O Fim do Jejum Prolongado): O jejum absoluto de 8 a 12 horas está proscrito. Evidências robustas, corroboradas por dados do Projeto ACERTO no Brasil, demonstram que a oferta de líquidos claros enriquecidos com carboidratos até 2 horas antes da indução anestésica reduz a resistência insulínica pós-operatória, minimiza a perda de massa magra e diminui a ansiedade do paciente.
- Intraoperatório (A Terapia Hídrica Guiada por Metas): A hiper-hidratação venosa, outrora comum, resulta em edema esplâncnico e retardo do esvaziamento gástrico. A fluidoterapia deve ser restritiva ou guiada por metas (avaliando a variação do volume sistólico), garantindo perfusão tecidual sem encharcar o paciente. A analgesia deve ser preemptiva e multimodal, poupadora de opioides.
- Pós-Operatório (Mobilização e Realimentação): O repouso no leito prolongado favorece o tromboembolismo venoso (TEV) e a atelectasia pulmonar. O paciente deve ser encorajado a deambular no mesmo dia da operação.
Aplicação na Cirurgia Digestiva
Na cirurgia do trato gastrointestinal, a aplicação destes cuidados perioperatórios altera drasticamente os desfechos e o tempo de internação:
- Preparo Mecânico do Cólon (PMC): Em cirurgias colorretais laparoscópicas, o uso rotineiro do PMC foi abandonado por grande parte das diretrizes, pois causa desidratação, distúrbios hidroeletrolíticos e não reduz as taxas de deiscência anastomótica ou infecção de sítio cirúrgico (exceto quando associado a antibióticos orais específicos em cirurgias de reto baixo).
- Sondas e Drenos: A utilização profilática de sondas nasogástricas (SNG) e drenos cavitários não encontra mais respaldo na literatura para procedimentos eletivos sem complicações. A SNG prolonga o íleo adinâmico e predispõe a infecções respiratórias. O dreno deve ser uma exceção fundamentada, não a regra.
- Realimentação Precoce: O íleo pós-operatório é inerente à manipulação das alças, mas a dieta por via oral iniciada precocemente (muitas vezes nas primeiras 24 horas) estimula o reflexo gastrocólico, protege a barreira mucosa intestinal contra a translocação bacteriana e acelera a alta hospitalar. Estatísticas brasileiras apontam que a realimentação precoce em ressecções intestinais laparoscópicas reduz o tempo de internação em até 2 a 3 dias.
Pontos-Chave
- Avaliação Fisiológica: O pneumoperitônio exige monitorização hemodinâmica rigorosa devido às alterações cardiocirculatórias e ventilatórias.
- Jejum Abreviado: Utilização de maltodextrina a 12,5% até 2 horas antes da cirurgia é uma prática segura e metabolicamente protetora.
- Analgesia Opioid-Sparing: Uso de infiltração dos portais com anestésicos locais, anti-inflamatórios e bloqueios regionais (ex: TAP block) para evitar os efeitos eméticos e obstipantes dos opioides.
- Tromboprofilaxia: O uso de meias elásticas de compressão graduada, compressores pneumáticos intermitentes e heparina de baixo peso molecular é mandatório, dada a estase venosa promovida pela pressão intra-abdominal elevada.
- Racionalização de Dispositivos: Evitar o uso sistemático e prolongado de sondas vesicais de demora (SVD), SNG e drenos cavitários.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A destreza na dissecção laparoscópica e a precisão das anastomoses intracavitárias perdem parte de seu brilho se o paciente for submetido a um estresse fisiológico não gerenciado. O cirurgião do aparelho digestivo moderno deve compreender que o seu papel transcende a mecânica operatória; ele é o arquiteto da recuperação do paciente. Adoção de protocolos de cuidados perioperatórios como o ACERTO exige uma mudança de cultura institucional e o alinhamento de toda a equipe multidisciplinar (cirurgião, anestesiologista, enfermagem, nutrição e fisioterapia). Ao unirmos a mínima agressão tecidual da videolaparoscopia à máxima otimização metabólica do perioperatório, entregamos não apenas uma cirurgia segura, mas a restituição rápida e plena da qualidade de vida do nosso paciente.
“O principal objetivo da cirurgia moderna não é apenas curar a doença anatômica, mas suprimir a resposta ao trauma cirúrgico, proporcionando uma recuperação sem dor e sem risco.” — Henrik Kehlet, cirurgião dinamarquês e idealizador do protocolo ERAS.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
A Doutrina da Visão Crítica de Segurança (CVS) na Colecistectomia Laparoscópica
Fundamentos, Técnica e a Era da Inteligência Artificial
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 11 minutos)
Introdução
A colecistectomia laparoscópica é, indiscutivelmente, uma das intervenções cirúrgicas mais realizadas no planeta. Contudo, apesar de sua consolidação como um procedimento seguro, as lesões iatrogênicas da via biliar principal continuam a assombrar os blocos operatórios, configurando-se como uma das complicações mais temidas, onerosas e medicolegais da cirurgia moderna. Com o advento da laparoscopia no final dos anos 1980, observou-se um pico alarmante na incidência dessas lesões. Em resposta a esta crise global de segurança, o Dr. Steven Strasberg propôs, em 1995, a Visão Crítica de Segurança (CVS – Critical View of Safety). Mais do que uma simples manobra, o CVS transformou-se no dogma central da cirurgia biliar. Este artigo disseca o racional anatômico, a sistematização técnica rigorosa e as aplicações contemporâneas do CVS na prática do cirurgião do aparelho digestivo.
A Patogenia da Lesão e o Racional Anatômico
Para prevenir um erro, é imperativo compreender a sua gênese. A literatura clássica é taxativa: a esmagadora maioria das lesões biliares graves não ocorre por imperícia motora, mas por um erro de percepção visual conhecido como “Misidentification” (identificação errônea). A armadilha mais letal é a técnica da “Visão Infundibular”. Em cenários de inflamação aguda, fibrose ou tração inadequada, o ducto colédoco ou o ducto hepático comum alinham-se paralelamente à vesícula, mimetizando o ducto cístico. O cirurgião se convence de forma ilusória e perigosa de que “tem certeza de estar vendo o cístico”. Essa confiança excessiva, aliada à dissecção apressada, resulta na transecção trágica da via biliar principal. O CVS foi desenhado cirurgicamente para aniquilar esta ilusão de ótica.
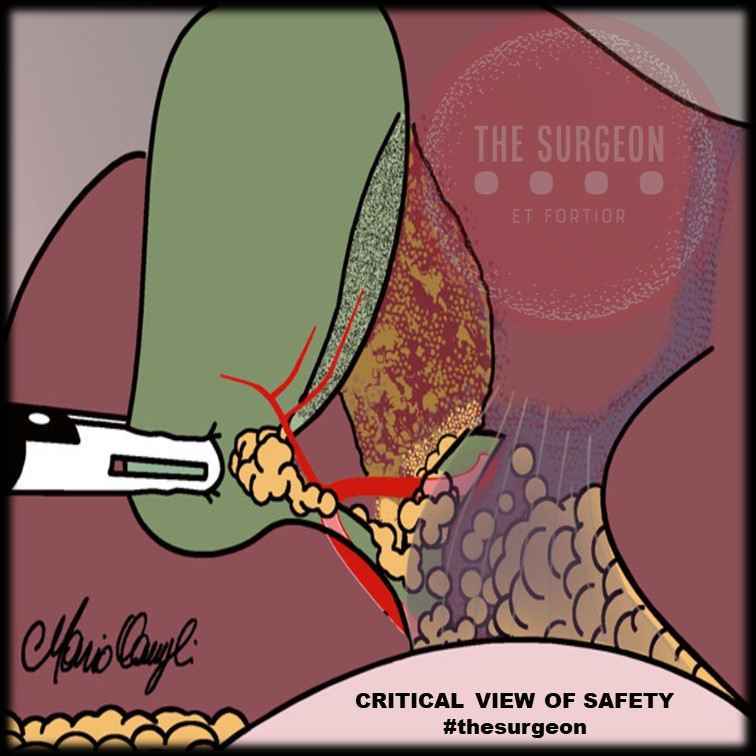
Os Três Critérios Obrigatórios do CVS
O CVS não é uma suposição; é um método de prova anatômica objetiva. Para declarar que o CVS foi atingido, todos os três critérios abaixo devem ser cumpridos simultaneamente antes do disparo de qualquer clipe ou grampeador:
- Dissecção Completa do Triângulo Hepatocístico: Toda a gordura e o tecido fibroso devem ser removidos do triângulo formado pelo ducto cístico, ducto hepático comum e borda hepática. A exposição deve ser visualizada nas faces anterior e posterior. Atenção: o ducto hepático comum não precisa (e não deve) ser dissecado ativamente, mas o triângulo deve estar limpo.
- Separação Parcial da Vesícula do Leito Hepático: O terço inferior da vesícula biliar (infundíbulo) deve ser completamente descolado da placa cística (leito hepático). Este passo, muitas vezes negligenciado, é o que garante que as estruturas dissecadas estão efetivamente a entrar na vesícula, e não a passar por ela.
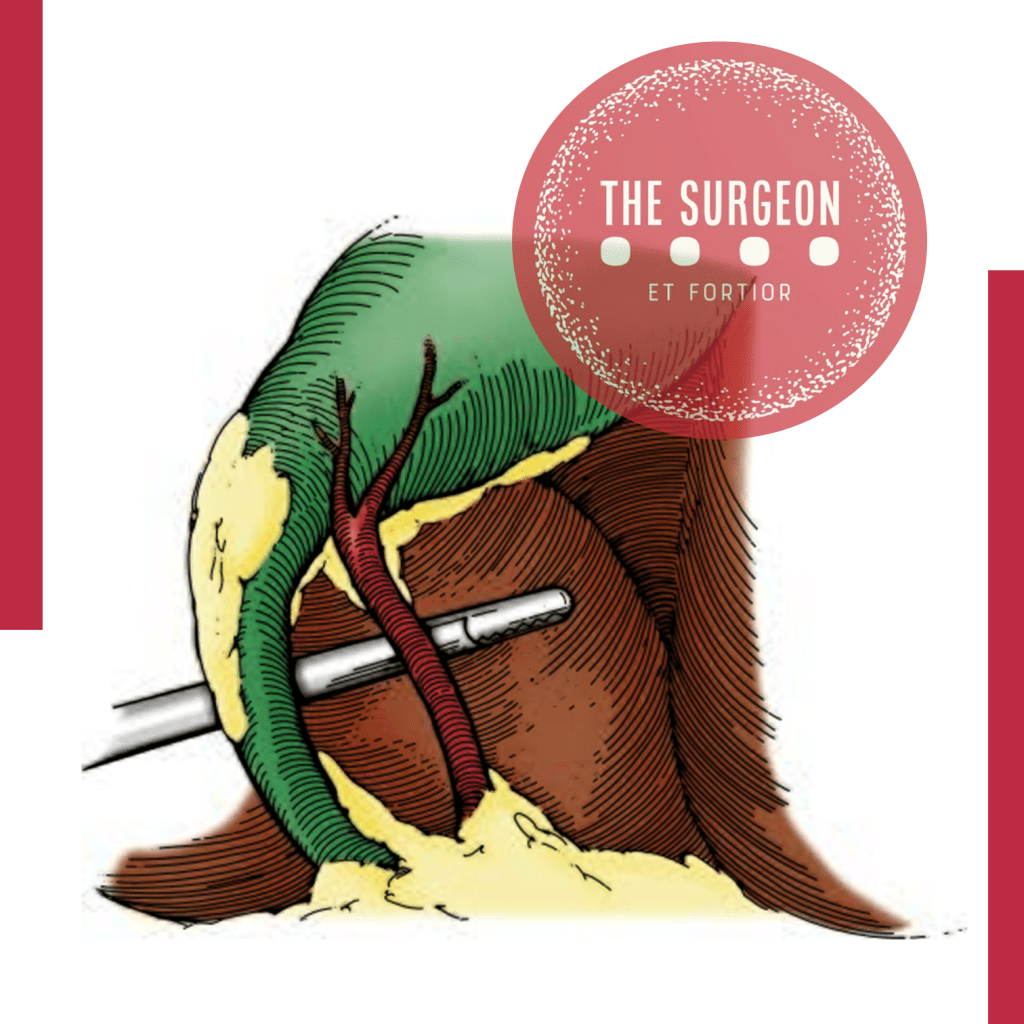
- Apenas Duas Estruturas Adentrando a Vesícula: Exclusivamente um ducto (ducto cístico) e uma artéria (artéria cística) devem ser vistos penetrando a vesícula. Ambos devem estar completamente isolados e identificáveis circunferencialmente (visão de 360 graus).
Robustez, Saídas e a Era Moderna
A eficácia do CVS é incontestável. Em grandes séries publicadas, como a de Yegiyants et al. (3.042 casos) e Avgerinos et al. (998 casos) em que o CVS foi estritamente aplicado, a taxa de lesão biliar grave foi de zero.
Cenários Adversos e Técnicas de Saída (Bail-Out Procedures)
O verdadeiro valor do CVS revela-se na dificuldade. Quando há inflamação severa, colecistite crônica esclerosante ou um Triângulo de Calot “obliterado” (frozen Calot), atingir o CVS torna-se perigoso ou impossível.
Para o cirurgião maduro, a incapacidade de completar o CVS não é um obstáculo a ser forçado, mas um alarme biológico de perigo iminente. Nestes casos, impõe-se a adoção imediata de técnicas de saída:
- Colecistectomia Subtotal (Fenestrada ou Reconstituída).
- Conversão precoce para cirurgia aberta.
- Realização de Colangiografia Intraoperatória.
Documentação Médico-Legal
A diretriz cirúrgica contemporânea exige que o CVS seja documentado. O cirurgião deve registrar uma fotografia ou um breve vídeo intraoperatório que demonstre inequivocamente a exposição anterior e posterior das estruturas isoladas. A inclusão desta prova no prontuário médico eletrônico protege o paciente contra complicações e o cirurgião em contextos litigiosos.
Cirurgia Robótica e Inteligência Artificial (IA)
Os princípios de Strasberg são universais e imutáveis, independentemente do instrumental. Na Cirurgia Robótica, a visão 3D ampliada de altíssima definição e a estabilidade das pinças facilitam o cumprimento escrupuloso dos três critérios. As perspetivas futuras são ainda mais fascinantes: algoritmos de Inteligência Artificial (IA) e visão computacional estão a ser treinados para reconhecer e validar os critérios do CVS em tempo real no monitor cirúrgico, funcionando como um checklist virtual que impede a clipagem até que o software confirme a segurança anatômica.
Pontos-Chave para a Prática Diária
- Prova, não Suposição: O CVS transforma um julgamento subjetivo (“parece o cístico”) numa evidência anatômica irrefutável.
- O Risco da Visão Infundibular: Confundir um infundíbulo dilatado com o ducto cístico é a principal via para a lesão do colédoco.
- Descolar para Comprovar: Não basta limpar o triângulo de Calot; a separação do terço inferior da vesícula do leito hepático (Critério 2) é essencial.
- Pausa Obrigatória: A identificação das duas únicas estruturas deve ser um “momento estático”, validado verbalmente por toda a equipe antes da transecção.
Conclusões Aplicadas
O Critical View of Safety transcendeu a classificação de “técnica cirúrgica” para se consolidar como o padrão-ouro e a obrigação ética universal na colecistectomia laparoscópica. A sua adoção obrigatória nos programas de residência médica aniquila a cultura do achismo visual, substituindo-a pelo rigor científico. Ao reconhecer que o principal inimigo do cirurgião biliar é a sua própria mente (na forma de ilusão de ótica), o CVS fornece a armadura necessária para proteger o doente de morbidades devastadoras. Na cirurgia do aparelho digestivo, a paciência não é apenas uma virtude; é uma técnica de salvamento.A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.”
“A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.” — Dr. Steven Strasberg, cirurgião e idealizador da Visão Crítica de Segurança.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
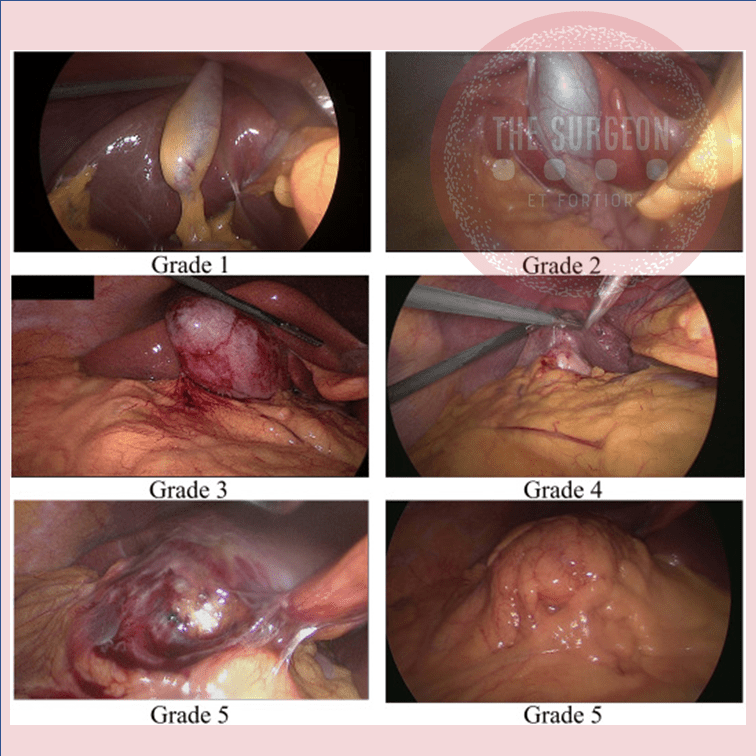
Apendicectomia Laparoscópica: Truques e Dicas para uma Execução Segura e Eficiente
Introdução
A apendicectomia laparoscópica consolidou-se como o padrão-ouro no tratamento da apendicite aguda, sobretudo em centros com acesso à tecnologia minimamente invasiva. Com benefícios amplamente documentados — como menor dor pós-operatória, redução do tempo de internação e menor incidência de infecção de ferida —, a técnica exige, contudo, treinamento e atenção a detalhes técnicos. Este artigo oferece truques e dicas práticas que podem otimizar a performance do cirurgião, reduzir complicações e melhorar a curva de aprendizado da equipe assistente.
1. Posicionamento do Paciente e da Equipe
- Decúbito dorsal com leve Trendelenburg e rotação à esquerda facilita a exposição do quadrante inferior direito.
- Fixe o braço direito do paciente ao corpo para permitir amplo espaço de movimentação do cirurgião.
- Cirurgião à esquerda do paciente, assistente ao lado da perna esquerda, monitor preferencialmente à direita ou à cabeceira, na linha dos ombros.
Dica: Ajuste fino da inclinação da mesa pode ser decisivo para deslocar alças e expor o ceco sem necessidade de manobras agressivas.
2. Posicionamento dos Trocárteres
- Um padrão eficiente inclui:
- Trocárter de 10 mm umbilical (ótica).
- Trocárter de 5 mm em hipogástrio (instrumentação dominante).
- Trocárter de 5 mm em flanco esquerdo (tração e dissecção).
Truque: Em pacientes obesos, insira o trocárter ótico com cuidado em ângulo oblíquo para evitar desinserção do pneumoperitônio e garantir estabilidade.
3. Estratégias de Exposição
- Identifique o teniae coli do ceco e siga até a base do apêndice.
- Use pinça atraumática para tração superior do apêndice, expondo sua base.
- Em casos de aderências, libere-as com energia monopolar delicada ou tesoura, evitando avulsões inadvertidas.
Dica de ouro: Evite “lutar” contra aderências retrocecais. Mude o plano, reposicione a câmera, varie o ângulo de dissecção. Tempo gasto com exposição segura evita complicações graves.
4. Controle do Pedículo e Secção Apendicular
- O método mais utilizado é o uso de duas ligaduras com endoloop ou clips poliméricos (Hem-o-lok®), seguido de secção entre eles.
- Alternativamente, grampeadores laparoscópicos podem ser usados, especialmente em apêndices friáveis ou bases espessadas.
Truque técnico: Em apêndices muito inflamados, realize a ligadura mais distal antes da proximal, para reduzir o risco de ruptura ou vazamento ao manipular a base.
5. Retirada e Proteção da Cavidade
- Retire o apêndice com saco cirúrgico sempre que possível, evitando contaminação do trajeto do trocárter.
- Irrigue abundantemente a loja apendicular se houver peritonite localizada ou pus livre.
- Se necessário, coloque dreno tubular por 24 a 48 horas.
Dica prática: Em caso de dúvida quanto à integridade da base, deixe um fragmento do ceco visível e documente o aspecto final com imagem.
6. Situações Especiais
- Apêndice retrocecal: requer liberação ampla da reflexão lateral direita do cólon.
- Apendicite perfurada com abscesso: considere drenagem inicial guiada por imagem e apendicectomia em intervalo.
- Apendicite gestacional: ideal até o segundo trimestre. Atenção ao deslocamento anatômico do apêndice.
Truque anatômico: Em gestantes ou crianças, a mobilidade intestinal pode mascarar a localização clássica. Reforce a busca sistemática do apêndice pela convergência das teníases do ceco.
Conclusão
A apendicectomia laparoscópica é uma cirurgia segura, eficaz e que continua evoluindo com a incorporação de técnicas assistidas por imagem, navegação e inteligência artificial. No entanto, sua execução requer atenção a detalhes aparentemente simples, que fazem toda a diferença nos desfechos clínicos. O domínio dos truques e dicas técnicas aqui apresentados contribui significativamente para uma prática cirúrgica mais segura, eficiente e baseada em excelência técnica.
“A simplicidade técnica não dispensa o rigor; é justamente na cirurgia simples que se exige a perfeição.” — René Leriche
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
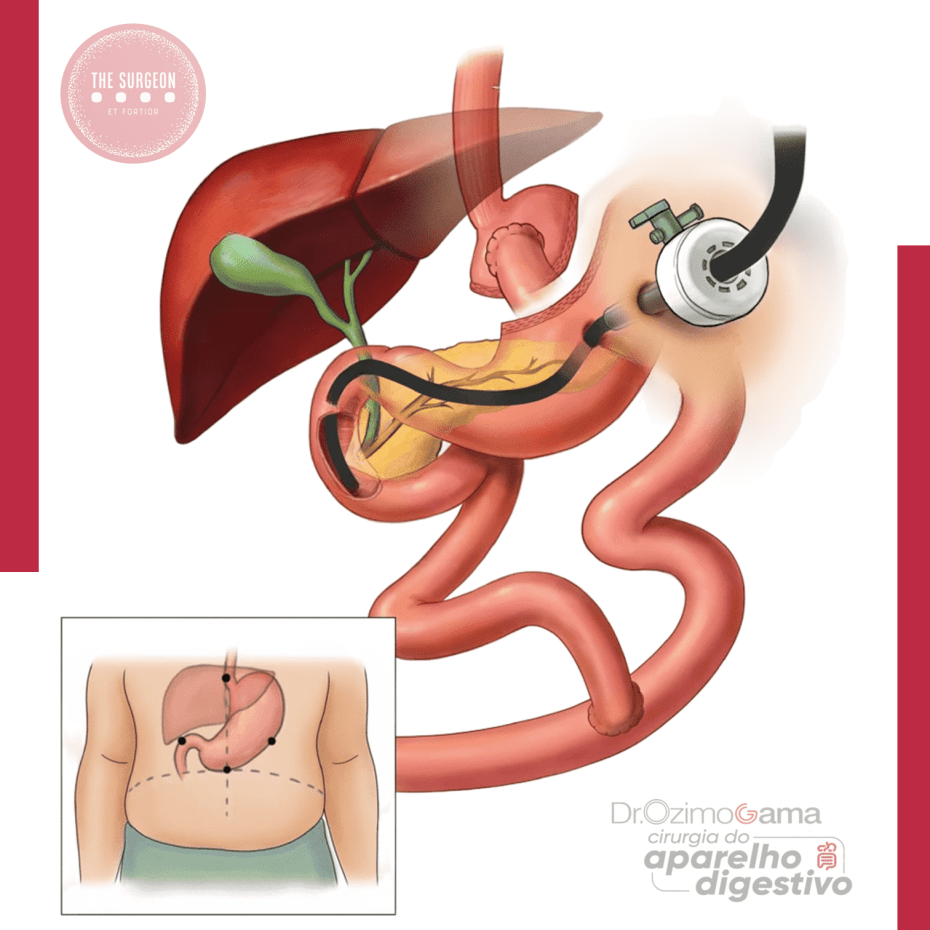
Tratamento da Coledocolitíase no Paciente com Bypass Gástrico
Abordagens Cirúrgicas e Opções Técnicas
A coledocolitíase, caracterizada pela presença de cálculos no ducto biliar comum (CBD), é uma complicação potencialmente grave que pode causar icterícia obstrutiva, pancreatite e colangite. Em pacientes que realizaram o bypass gástrico em Y-de-Roux, o tratamento dessa condição se torna mais complexo devido às alterações anatômicas decorrentes da cirurgia bariátrica, tornando o acesso ao ducto biliar pelo método endoscópico tradicional (CPRE) um desafio significativo.
Introdução
O bypass gástrico em Y-de-Roux é um dos procedimentos mais realizados no tratamento da obesidade mórbida, resultando em perda de peso significativa e, consequentemente, em uma maior prevalência de cálculos biliares. No entanto, as alterações anatômicas criadas por esse procedimento, como a separação do estômago proximal do restante do trato gastrointestinal, impedem o acesso ao ducto biliar pelo estômago convencional. Nesse contexto, o manejo da coledocolitíase, que afeta até 18% dos pacientes submetidos à colecistectomia, exige uma abordagem multidisciplinar e técnicas cirúrgicas e endoscópicas inovadoras.
Propedêutica Cirúrgica
O diagnóstico e o manejo da coledocolitíase em pacientes com bypass gástrico podem ser desafiadores, pois o acesso ao ducto biliar comum por vias endoscópicas tradicionais é inviável. Nestes casos, a apresentação clínica pode variar de sintomas leves de icterícia a quadros graves de colangite ascendente.
Para a avaliação inicial, a ultrassonografia abdominal continua sendo o exame de escolha para identificar dilatação do ducto biliar e a presença de cálculos. No entanto, devido à complexidade anatômica, exames como a colangiopancreatografia por ressonância magnética (MRCP) são frequentemente necessários para fornecer uma visão mais detalhada da anatomia do trato biliar.
Uma vez confirmado o diagnóstico de coledocolitíase, o tratamento deve ser instituído rapidamente, a fim de evitar complicações como abscessos hepáticos ou pancreatite biliar. As opções terapêuticas variam dependendo da gravidade da apresentação clínica e da disponibilidade de recursos no centro de tratamento. Para pacientes com colangite ascendente, por exemplo, a descompressão urgente do ducto biliar é necessária. Nestes casos, o uso de técnicas minimamente invasivas, como a colangiografia trans-hepática percutânea (PTC), pode ser a melhor alternativa quando a CPRE não é viável.
Em pacientes estáveis, várias abordagens cirúrgicas são possíveis. Uma técnica amplamente utilizada é a colecistectomia laparoscópica assistida por CPRE transgástrica. Nesse procedimento, um acesso cirúrgico é feito no estômago excluído do paciente, permitindo que o endoscópio seja inserido diretamente na papila duodenal, facilitando a remoção dos cálculos e a descompressão do ducto biliar.
Aplicação na Cirurgia Digestiva
A cirurgia digestiva avançada em pacientes com bypass gástrico exige um planejamento cuidadoso e uma abordagem técnica que considere as alterações anatômicas decorrentes da cirurgia bariátrica. As seguintes estratégias têm sido eficazes no manejo da coledocolitíase nesses pacientes:
- ERCP assistida por laparoscopia: Esse procedimento permite o acesso ao trato biliar usando técnicas combinadas de laparoscopia e endoscopia. A transposição do endoscópio pelo estômago excluído oferece uma alternativa eficiente para a remoção dos cálculos, com baixa taxa de complicações.
- Colangiografia trans-hepática percutânea (PTC): Em pacientes instáveis ou com contraindicações para procedimentos invasivos, a PTC é uma alternativa valiosa para a descompressão biliar emergente. Essa técnica minimamente invasiva pode ser associada à dilatação do esfíncter de Oddi para expulsar os cálculos.
- Exploração cirúrgica do ducto biliar comum: Em casos complexos, onde as técnicas minimamente invasivas falham, a exploração laparoscópica ou aberta do ducto biliar pode ser necessária. A abordagem transcística ou a coledocotomia são opções viáveis, dependendo da localização e do tamanho dos cálculos. A utilização de cateteres tipo Fogarty ou cestas de Dormia pode ser útil para a remoção dos cálculos.
- Técnicas reconstrutivas: Para pacientes com múltiplos cálculos biliares ou complicações, a realização de anastomoses biliodigestivas, como a coledocoduodenostomia ou a hepaticojejunostomia em Y-de-Roux, pode ser considerada. Essas abordagens proporcionam um alívio duradouro, evitando futuros episódios de obstrução.
Pontos-chave
- Pacientes com bypass gástrico em Y-de-Roux apresentam desafios únicos no tratamento da coledocolitíase devido à anatomia alterada do trato digestivo.
- A ERCP tradicional é inviável nesses pacientes, sendo substituída por abordagens combinadas, como a ERCP transgástrica assistida por laparoscopia ou a PTC.
- A ultrassonografia e a MRCP são ferramentas essenciais para o diagnóstico, enquanto a intervenção cirúrgica deve ser cuidadosamente planejada, considerando as condições clínicas e anatômicas do paciente.
- Em centros especializados, as taxas de sucesso com essas abordagens minimamente invasivas são altas, com complicações graves ocorrendo em menos de 10% dos casos.
Conclusão Aplicada à Prática do Cirurgião Digestivo
O tratamento da coledocolitíase em pacientes submetidos ao bypass gástrico representa um desafio técnico significativo, exigindo conhecimento especializado em cirurgia digestiva avançada e endoscopia. A abordagem ideal deve ser individualizada, considerando a condição clínica do paciente e as habilidades da equipe cirúrgica. As técnicas minimamente invasivas, como a ERCP assistida por laparoscopia, são as preferidas quando viáveis, mas o cirurgião deve estar preparado para realizar intervenções mais complexas, como a coledocotomia ou anastomoses biliodigestivas, quando necessário.
Nas palavras do cirurgião Steven Strasberg, “O sucesso na cirurgia hepato-biliar depende não apenas da habilidade técnica, mas também da capacidade de adaptar-se às circunstâncias anatômicas e clínicas únicas de cada paciente.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#coledocolitiase
#bypassgastrico
#cirurgiabariatrica
#cirurgiadigestiva
#cprefalha
Trauma Cirúrgico Controlado: Como Gerenciar a Resposta Metabólica em Intervenções Eletivas
INTRODUÇÃO
Sempre que ocorre uma agressão ao organismo, independentemente da causa, um conjunto complexo de respostas é desencadeado para manter a homeostase e a sobrevivência. Se a lesão for de pequena intensidade, o corpo rapidamente retoma o equilíbrio metabólico e imunológico. No entanto, em casos de traumas extensos, a resposta pode ser exacerbada, levando à disfunção orgânica e aumentando os riscos de mortalidade. Para o cirurgião, é crucial compreender os eventos endócrinos, metabólicos e imunológicos que ocorrem durante e após uma intervenção cirúrgica para manejar adequadamente essas respostas e minimizar complicações.
DESENVOLVIMENTO
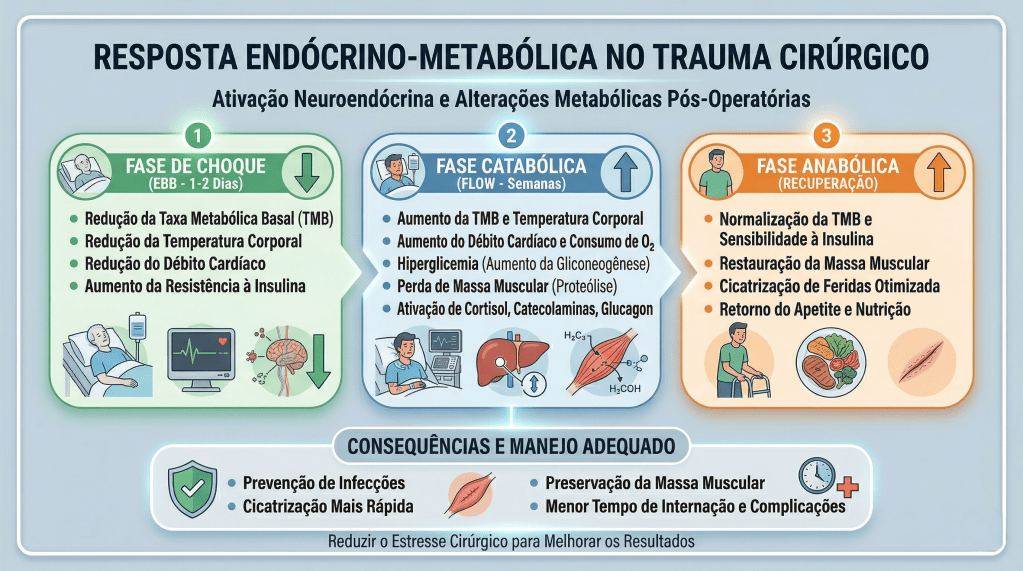
A resposta metabólica ao trauma pode ser dividida em três fases: Ebb, Flow e Anabólica. A fase Ebb ocorre imediatamente após o trauma e é caracterizada por instabilidade hemodinâmica, hipovolemia, e aumento de hormônios como catecolaminas e glicocorticoides. Nessa fase, apesar da elevação dos hormônios contrarreguladores, o metabolismo é diminuído. A fase Flow, que segue a fase Ebb, é marcada por hipermetabolismo, retenção hídrica e hiperglicemia, devido ao aumento dos hormônios catabólicos. A fase Anabólica, que pode durar meses, é quando o corpo começa a recuperar o peso e a massa muscular perdidos, com os níveis hormonais retornando ao normal.
APLICAÇÃO NA CIRURGIA DIGESTIVA
Em cirurgias eletivas, a resposta ao trauma pode ser melhor controlada. A avaliação pré-operatória, a escolha da técnica cirúrgica e o manejo adequado do jejum são essenciais para reduzir o impacto do trauma. A laparoscopia, por exemplo, deve ser priorizada sempre que possível, pois está associada a uma menor agressão tecidual e, consequentemente, a uma resposta metabólica mais branda. Além disso, controlar a ansiedade pré-operatória com benzodiazepínicos pode reduzir a liberação de catecolaminas, diminuindo os riscos de complicações como arritmias e hipertensão.
PONTOS-CHAVE
- A resposta ao trauma cirúrgico envolve fases distintas, cada uma com características metabólicas específicas.
- A laparoscopia é uma via cirúrgica que pode minimizar a resposta ao trauma.
- Controlar a ansiedade pré-operatória é fundamental para reduzir a liberação de catecolaminas e suas consequências.
CONCLUSÕES APLICADAS À PRÁTICA DO CIRURGIÃO DIGESTIVO
O manejo adequado da resposta ao trauma em cirurgias eletivas é vital para reduzir complicações e melhorar os resultados cirúrgicos. A compreensão das fases da resposta metabólica, a escolha de técnicas menos invasivas e a gestão da ansiedade pré-operatória são pilares essenciais para a prática do cirurgião digestivo. Com esses conhecimentos, os profissionais podem proporcionar uma recuperação mais rápida e segura aos seus pacientes. Como disse o renomado cirurgião Harvey Cushing: “A cirurgia é o mais elevado empreendimento humano, pois requer uma combinação de conhecimentos científicos, habilidade técnica e sensibilidade ética.”
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
“Aleijados Biliares”
O Flagelo Silencioso da Lesão do Ducto Biliar na Colecistectomia Laparoscópica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 8 minutos)

Introdução
No âmbito da cirurgia digestiva, poucas complicações causam tanto temor aos cirurgiões quanto a lesão do ducto biliar (LDB) durante a colecistectomia laparoscópica. Apesar de ser o procedimento padrão-ouro para o tratamento da colelitíase sintomática, esta intervenção carrega um perigo oculto que pode transformar uma operação de rotina num evento devastador, alterando irreversivelmente a vida tanto do doente quanto do cirurgião — criando o que a literatura clássica tragicamente apelida de “aleijados biliares”.
O Paradoxo Laparoscópico e o Impacto Socioeconómico
A colecistectomia laparoscópica, introduzida no final da década de 1980, revolucionou a cirurgia da vesícula biliar, proporcionando recuperações mais rápidas e menor dor pós-operatória. No entanto, trouxe consigo um risco aumentado de LDB em comparação com a abordagem aberta tradicional. Dados de um estudo do Colégio Brasileiro de Cirurgiões relataram uma taxa de LDB de 0,18% em colecistectomias laparoscópicas, um valor ligeiramente inferior à média global que oscila entre 0,3% e 0,7%. Contudo, as consequências da LDB são de uma gravidade extrema. Frequentemente exigem cirurgia reconstrutiva complexa (como a hepaticojejunostomia em Y de Roux) e resultam em significativa morbilidade, mortalidade e custos de saúde astronómicos. Um estudo brasileiro estimou que os casos de LDB conduzem a uma média de 22 dias adicionais de internamento hospitalar e a um aumento de 30 vezes nos custos hospitalares associados.
Pontos-Chave na Prevenção e Gestão da LDB
Para mitigar este flagelo, a abordagem cirúrgica deve assentar em quatro pilares fundamentais:
1. Fatores de Risco
Compreender e antecipar os fatores relacionados com o doente (ex: colecistite aguda, obesidade grave, variações anatómicas) e com o cirurgião (ex: inexperiência, fadiga, interpretação errónea da anatomia ou “ilusão de ótica” cirúrgica) é absolutamente crucial para o planeamento operatório.
2. Estratégias de Prevenção Inegociáveis
- Visão Crítica de Segurança (CVS): Esta técnica de identificação anatómica, defendida pelo Dr. Steven Strasberg, é a pedra basilar na prevenção da LDB. Exige a limpeza do triângulo hepatocístico, a separação do terço inferior da vesícula da placa cística e a visualização de apenas duas estruturas a adentrar a vesícula.
- Colangiografia Intraoperatória (CIO): Embora ainda alvo de algum debate quanto à sua utilização universal, a CIO é uma ferramenta valiosa que pode auxiliar de forma decisiva na identificação da anatomia biliar e no diagnóstico precoce de lesões.
- “Cultura de Segurança”: É imperativo adotar uma mentalidade que priorize a segurança do doente acima da conclusão do procedimento por via laparoscópica a todo o custo. A conversão para cirurgia aberta ou a realização de uma colecistectomia subtotal são sinais de maturidade cirúrgica, e nunca de fracasso.
3. Reconhecimento e Manejo Adequado
O reconhecimento precoce da LDB, preferencialmente no período intraoperatório, é vital. O reencaminhamento atempado e o reparo imediato ou diferido realizado por cirurgiões hepatobiliares experientes e dedicados proporcionam, inequivocamente, os melhores resultados a longo prazo.
4. Formação e Educação Médica Contínua
O treino baseado em simulação e a adoção de protocolos padronizados de segurança devem fazer parte da matriz formativa de qualquer serviço, ajudando a reduzir de forma drástica as taxas de LDB, especialmente entre os médicos internos (residentes) de cirurgia geral e digestiva.
Conclusões Aplicadas
Como cirurgiões digestivos, devemos permanecer em vigilância constante contra a ameaça da lesão do ducto biliar. Ao aderirmos de forma obstinada às técnicas cirúrgicas adequadas, mantendo um elevado índice de suspeição e fomentando uma cultura de segurança inabalável, podemos minimizar esta complicação potencialmente devastadora. A jornada para o “zero LDB” está em andamento, exigindo educação contínua, autorreflexão e um compromisso diário com a excelência na nossa prática.
“O desafio da cirurgia é a mão do cirurgião curar um doente pela precisão na estrutura e propósito.” – Joseph E. Murray, cirurgião plástico, pioneiro dos transplantes e Prémio Nobel da Medicina.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe nas suas redes sociais e|ou mande a sua dúvida pelo 💬 Chat On-line na nossa DM do Instagram.
Cirurgia Robótica
Introdução
A natureza humana do cirurgião sempre foi alcançar o máximo acesso à área de interesse cirúrgico enquanto causa o mínimo trauma ao paciente. Com a evolução das técnicas operatórias e tecnologias, essa capacidade avançou enormemente. Inicialmente, os cirurgiões desenvolveram a habilidade técnica para realizar incisões menores e mais estratégicas para o acesso cirúrgico aberto. Posteriormente, com a utilização de telescópios cirúrgicos e ferramentas de acesso mínimo, e agora com tecnologias assistidas por computador e robótica, os cirurgiões conseguem acessar áreas cirúrgicas de difícil abordagem com trauma secundário quase insignificante ao paciente.
A Revolução da Cirurgia Minimamente Invasiva
A revolução da cirurgia minimamente invasiva começou há três décadas com o advento da tecnologia de vídeo. Desde então, a abordagem laparoscópica a uma ampla variedade de procedimentos cirúrgicos tornou-se a escolha preferida, permitindo a realização de operações através de pequenas incisões, videotelescópios e instrumentos longos. Os benefícios dessas técnicas incluem menor tempo de internação, menos dor e cicatrizes, menor risco de infecção, menos perda de sangue e transfusões, retorno acelerado às atividades normais e recuperação mais rápida.
No entanto, a cirurgia laparoscópica exige um conjunto único de habilidades que pode ser difícil para alguns cirurgiões adquirirem. As limitações técnicas incluem falta de visualização tridimensional (3D), alcance limitado de movimento devido aos instrumentos rígidos, ergonomia pobre, movimentos contraintuitivos, amplificação do tremor fisiológico, sensibilidade tátil reduzida e o efeito fulcro.
Avanços com a Integração Robótica
A integração robótica permite que os cirurgiões realizem procedimentos minimamente invasivos mais complexos com visualização 3D aprimorada, maior destreza, aumento do alcance de movimento, melhor ergonomia e acesso superior a áreas de difícil alcance no corpo. Isso se traduz em procedimentos mais precisos e menos invasivos. A próxima geração de robôs está sendo desenvolvida para serem menores, mais inteligentes e com custos reduzidos, tornando-se cada vez mais comuns nas salas de cirurgia.
Para o paciente, um procedimento assistido por robô oferece todos os benefícios potenciais de uma operação minimamente invasiva. Estudos clínicos sugerem que a robótica pode ajudar os cirurgiões a proporcionar melhores resultados clínicos em certos contextos específicos.
Perspectivas Futuras e Desafios
A tecnologia robótica também possibilita a separação do cirurgião do paciente, seja em metros ou milhares de quilômetros. A teles cirurgia e o telementoring têm sido testados e demonstraram ser viáveis e benéficos. Exemplos incluem a remoção de uma vesícula biliar através do Oceano Atlântico e o mentoreamento de cirurgiões no Canadá. Outra tecnologia permite ao cirurgião realizar rondas remotamente, controlando movimentos robóticos via internet.
Os desafios incluem o desenvolvimento e aprimoramento das tecnologias e suas limitações, o treinamento e a expertise dos médicos para usar essas ferramentas de forma segura e eficaz, a incorporação das tecnologias mais recentes na educação cirúrgica e a gestão dos custos aumentados de maneira que permita maior acesso dos pacientes aos melhores cuidados disponíveis.
Contexto Brasileiro
No Brasil, a cirurgia robótica tem avançado significativamente, com instituições adotando plataformas robóticas para procedimentos complexos. Dados da Sociedade Brasileira de Cirurgia Minimamente Invasiva e Robótica (SOBRACIL) indicam um aumento no número de procedimentos realizados com auxílio de robôs, refletindo os avanços tecnológicos e os benefícios clínicos associados.
Conclusão
A cirurgia robótica representa uma evolução significativa na capacidade dos cirurgiões de realizar procedimentos complexos com precisão e segurança aprimoradas. À medida que a tecnologia avança, espera-se que a adoção de plataformas robóticas se torne cada vez mais comum, proporcionando aos pacientes os benefícios de técnicas minimamente invasivas e resultados clínicos superiores.
“A inovação é a capacidade de ver a mudança como uma oportunidade – não uma ameaça.” – Steve Jobs
Gostou? Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#CirurgiaRobótica #TecnologiaMédica #InovaçãoCirúrgica #Saúde #EducaçãoMédica
Diferenciação Morfológica do Ducto Cístico e do Ducto Colédoco no Contexto da Colecistectomia Laparoscópica
Introdução
Na cirurgia laparoscópica, a precisão na identificação e dissecação das estruturas anatômicas é fundamental para a prevenção de lesões iatrogênicas da via biliar. O conhecimento detalhado das variações anatômicas do ducto cístico e do ducto colédoco é essencial para evitar complicações graves. Conforme disse Lahey em 1954: “Cholecystectomy is a dangerous operation unless one realizes that variations are very common.” Assim, a terminologia anatômica deve ser correta, consistente e padronizada para evitar mal-entendidos.
Anatomia Normal
O ducto cístico conecta a vesícula biliar ao ducto hepático comum, formando o ducto colédoco. Este último é a principal via de passagem da bile do fígado para o duodeno. A correta identificação dessas estruturas é crucial durante a colecistectomia para evitar lesões.
Dimensões e Diâmetro
O ducto cístico geralmente tem um comprimento de 2-4 cm e um diâmetro de 1-5 mm. Já o ducto colédoco mede aproximadamente 6-8 cm de comprimento e tem um diâmetro que varia de 4-10 mm.
Trajeto e Topografia
O ducto cístico apresenta um trajeto sinuoso, que pode variar significativamente entre os pacientes. Ele se une ao ducto hepático comum para formar o ducto colédoco, que desce posteriormente ao duodeno até a ampola de Vater. A topografia dessas estruturas é fundamental para a correta dissecação durante a cirurgia.
Vascularização
O ducto cístico é irrigado principalmente pela artéria cística, um ramo da artéria hepática direita. O ducto colédoco, por sua vez, recebe irrigação de pequenos ramos axiais das artérias hepática direita, gástrica direita e pancreatoduodenal onde 60% é inferior e 40% é superior.
Epônimos e Terminologia Correta
Historicamente, a anatomia hepato-biliar tem sido confundida por diferentes nomenclaturas e epônimos imprecisos. O termo “triângulo de Calot”, descrito por Jean-Francois Calot em 1890, refere-se à área limitada pelo ducto cístico, o ducto hepático comum e a artéria cística. No entanto, o triângulo de Calot não é uma definição anatomicamente precisa, sendo preferível o termo “triângulo hepatocístico”, que é delimitado pelo ducto cístico e a vesícula biliar, o ducto hepático comum e a borda do fígado. Esta é uma área crítica onde ductos hepáticos direitos e a artéria hepática direita podem ser encontrados, sendo comumente obscurecida por inflamação aguda ou crônica.
Principais Variações Anatômicas e Suas Incidências
As variações anatômicas são comuns e devem ser antecipadas. Cerca de 20% dos ductos císticos seguem um trajeto paralelo ao ducto hepático comum, compartilhando uma bainha comum de tecido conjuntivo. Outros 5-10% têm um curso espiralado, cruzando o ducto hepático comum de forma ventral ou dorsal. As variações no trajeto e na união dos ductos são fatores de risco significativos para lesões durante a cirurgia.
Idiotopia em Relação ao Duodeno e Holotipia em Relação à Tríade Portal
O ducto colédoco passa diretamente atrás do duodeno, uma característica que o diferencia do ducto cístico durante a dissecção. Em relação à tríade portal, o ducto cístico e o ducto colédoco são componentes críticos, juntamente com a artéria hepática e a veia porta, sendo importantes marcos anatômicos para a orientação cirúrgica.
Sintopia Entre Eles
A relação espacial entre o ducto cístico e o ducto colédoco é complexa e varia com a inflamação e a dissecção cirúrgica. Durante a colecistectomia, a visualização clara do “triângulo hepatocístico” é fundamental para identificar corretamente o ducto cístico e evitar danos ao ducto colédoco.
Conclusão
O entendimento detalhado das variações anatômicas do ducto cístico e do ducto colédoco é crucial para a realização segura da colecistectomia laparoscópica. A aplicação do conceito da “vista crítica de segurança” ajuda a minimizar os riscos de lesões. Portanto, a terminologia precisa e o conhecimento das variações anatômicas são fundamentais para a prática cirúrgica segura.
Does routine performance of IOC prevent common bile duct injuries?
The main goals of IOC are to identify bile duct stones, clarify biliary anatomy, and prevent bile duct injuries. Indications for IOC during laparoscopic cholecystectomy may include jaundice or a history of jaundice, a history of pancreatitis particularly related to gallstone pancreatitis, elevated liver function tests, a common bile duct larger than 5-7mm in diameter, a cystic duct larger than 3mm in diameter, multiple small gallbladder stones, unclear anatomy, common bile duct stones visualized on preoperative ultrasound, possible bile duct injury or leak, and a short cystic duct.

The SAGES guidelines for the clinical application of laparoscopic biliary tract surgery recommended that IOC may decrease the risk of bile duct injury when used routinely and can allow access to the biliary tree for therapeutic intervention (Level II evidence, grade B recommendation). In a more recent guideline from the European Association for Endoscopic Surgery (EAES) regarding the prevention and treatment of bile duct injuries during laparoscopic cholecystectomy, the authors commented that the routine use of IOC remained controversial and that routine IOC could not be recommend based on the available literature. This guideline, however, indicated that IOC allows forearly identification of bile duct injuries as long as they are correctly interpreted.
Recommendation
Surgeons should use IOC liberally, be familiar with its indications, and become facile with the technique and interpretation of cholangiogram images. While IOC may decrease the risk of bile duct injury its routine use remains controversial; further high quality evidence is needed before routine IOC can be recommended. (Quality of evidence: +++, weak)
Safe Cholecystectomy | Bailout Procedures : When and How
Cholecystectomy is a common surgical procedure, with over 750,000 performed annually in the United States and 200,000 in Brazil. Popularized in the early 1990s, laparoscopic cholecystectomy (LC) is now considered the gold standard for routine cases of benign gallbladder and biliary pathology. LC has clear advantages over the traditional open approach, such as lower morbidity, less pain, and faster recovery. However, it is associated with a three to five times increase in bile duct injury (BDI). Major BDI can be a catastrophic complication, significantly increasing mortality. Additionally, patients who suffer a BDI often require further interventions, have a higher risk of additional complications, and experience a reduced quality of life. BDI is a common cause of legal litigation and remains one of the most frequent reasons for monetary compensation.
Correct Anatomical Identification The “classic injury” to the bile duct occurs when the common bile duct is mistaken for the cystic duct. This typically happens in the setting of severe acute or chronic inflammation, where the gallbladder may fuse to the lateral wall of the common hepatic duct, predisposing the surgeon to misidentify the biliary anatomy. This can result in a major BDI, where a segment of the common hepatic duct and bile duct is removed. Beyond this classic injury, other injuries to the biliary system can occur, such as sectional or segmental ducts disconnected from the liver with or without bile leakage, bile leakage from the cystic duct stump, long-term strictures due to thermal or iatrogenic damage, or combined vasculobiliary injuries.
Critical View of Safety (CVS) The Critical View of Safety (CVS), introduced by Strasberg et al. in 1995, is a method of safe anatomical identification that serves as a set of criteria to ensure the proper identification of the appropriate anatomy before ductal structures are ligated. These criteria include separating the lower end of the gallbladder from the liver to expose at least the lower third of the cystic plate, cleaning all fibrous and fatty tissue within the hepatocystic triangle, and seeing only two structures entering the gallbladder. The CVS mirrors the safe identification that occurs in traditional open cholecystectomy. While there are no level 1 data to support its use (due to the large sample size required to discriminate between an injury that occurs at a relatively low incidence), there is a body of literature of over 6000 cases where CVS was achieved without any major BDI.
Culture of Safety in Cholecystectomy (COSIC) Strict adherence to CVS is crucial to reducing BDI, but it is only part of the Culture of Safety in Cholecystectomy (COSIC), which requires that safety be at the forefront. Besides achieving CVS in total cholecystectomy cases, COSIC also requires proper patient selection and evaluation, adjustment of surgical technique in non-routine cases, use of bailout procedures, and avoiding complex cases when appropriate expertise is unavailable. The American Society of Gastrointestinal and Endoscopic Surgeons (SAGES) has developed a six-step program to enhance cholecystectomy safety:
- Understanding CVS and using it for identifying the cystic duct and artery.
- Considering an intraoperative pause before clipping or cutting any structure.
- Understanding aberrant anatomy.
- Liberal use of cholangiography or other intraoperative imaging means of the biliary system.
- Recognizing when dissection is approaching a significant danger zone and terminating the operation by a safe method, other than cholecystectomy, if the conditions around the gallbladder are too dangerous.
- Seeking assistance from another surgeon when conditions are difficult.
Bailout Procedures: When and How to Opt Deciding when to stop dissection of the hepatocystic triangle and opt for a bailout procedure rather than total cholecystectomy can be challenging. To make this decision before any biliary or vascular injury occurs, the surgeon must constantly ask: “Is it possible to safely achieve CVS?” When the answer is “No” or “I am not sure,” we recommend considering a bailout procedure. Early adoption of a bailout procedure is believed to reduce the difficulty of making this decision and avoid inadvertent injuries to the biliary system while trying to dissect in difficult and obstructed planes. It is essential always to remember that this operation is performed for benign pathology.
There are three clear bailout options in difficult cases:
- Stop the Operation (Stop, Drain, and Refer): Stopping the operation may conflict with the surgeon’s goal of “solving a problem,” but it should be considered and is a viable and safe option to avoid BDI. The patient should continue with a short course of antibiotics or even undergo postoperative drain placement and/or percutaneous cholecystostomy. A second attempt at cholecystectomy can be considered in 2–3 months.
- Surgical Cholecystostomy: The gallbladder’s fundus can be opened after placing a purse-string suture, the contents are aspirated, and a drainage catheter is placed in the gallbladder lumen. This method works as a temporary measure since definitive cholecystectomy will likely be necessary in 2–3 months.
- Subtotal “Fenestrating” Cholecystectomy: Subtotal cholecystectomy has been a surgical option for over 100 years. In 2016, an attempt was made to define subtotal cholecystectomy into two distinct subtypes to allow for improved study and dissemination of the technique. When a new gallbladder remnant is created, this is called “reconstituting” subtotal cholecystectomy. When the gallbladder is left open with a remaining portion, this is called “fenestrating” subtotal cholecystectomy. Recent systematic reviews have demonstrated the safety of these procedures. Fenestrating subtotal cholecystectomy is recommended as the most definitive bailout procedure.
Once the decision is made to proceed with fenestrating subtotal cholecystectomy, the surgeon should consider their experience and either convert to an open procedure or continue laparoscopically. This procedure can be safely performed laparoscopically with minimal “advanced” laparoscopic maneuvers, but it can also be easily performed using an open technique.
Fenestrating Subtotal Cholecystectomy Procedure The first step involves incising the anterior (peritonealized) wall of the gallbladder at the fundus. By initially leaving the gallbladder body intact, its contents can be evacuated more easily. It may be advisable to place a surgical sponge or “endobag” under the gallbladder to facilitate catching any stones that might spill upon opening. The incision should continue towards the infundibulum, removing most of the anterior wall of the gallbladder. A very important consideration of this technique involves leaving a portion of the anterior infundibulum wall intact to avoid inadvertent entry into the hepatoduodenal ligament. Once most of the anterior wall is removed and the gallbladder contents, including all stones, are evacuated, the internal aspect of the gallbladder can be examined. It is essential to identify whether continuous biliary drainage from the gallbladder is present. In most “difficult” gallbladders requiring fenestrating subtotal cholecystectomy, the cystic duct is obliterated and does not require formal ligation. However, in the rare instances where the duct is patent and bile continues to drain from it, the internal orifice of the cystic duct should be closed with non-permanent sutures from the internal aspect of the gallbladder. At no point should external ligation of the cystic duct be attempted, which could potentially injure the bile duct. A drain should be left in the hepatorenal recess. No drain is needed inside the gallbladder lumen. The drain should be monitored for biliary drainage. Although generally rare, if a postoperative biliary fistula occurs, standard management should proceed. Routine postoperative endoscopic sphincterotomy is not recommended unless the biliary fistula is persistent, as most of them are self-limiting.

The main goal of laparoscopic cholecystectomy is “safety first, total cholecystectomy second.” While most laparoscopic cholecystectomies are straightforward, the surgeon must always keep this safety culture at the forefront and remain vigilant to anticipate dangerous situations. COSIC will help minimize (or eliminate) BDI and assist the surgeon in managing difficult operating conditions or clinical scenarios. Safe management of the difficult gallbladder is possible with operational adjustments and liberal use of bailout procedures, specifically fenestrating subtotal cholecystectomy.
Prevention of Bile Duct Injury
Prevention of Bile Duct Injury During Laparoscopic Cholecystectomy
Introduction
Bile duct injury (BDI) during laparoscopic cholecystectomy is a significant surgical complication with profound clinical and medico-legal implications. The incidence of BDI ranges from 0.3% to 0.6%, despite advances in surgical techniques and imaging modalities. The prevalence of BDI remains concerning due to its association with high morbidity and mortality rates. Patients who suffer from BDI often face prolonged hospital stays, multiple surgeries, and long-term complications such as bile leakage, strictures, and secondary biliary cirrhosis. Medico-legally, BDI is one of the most common reasons for litigation against surgeons, often resulting in significant financial settlements and professional repercussions.
Questions and Answers
Question 1: What technique should be used to identify the anatomy during laparoscopic cholecystectomy?
Answer: The Critical View of Safety (CVS) is recommended for identifying the cystic duct and cystic artery.
Key Findings: The incidence of BDI was found to be 2 in one million cases using CVS, compared to 1.5 per 1000 cases with the infundibular technique.
Question 2: When should intraoperative cholangiography (IOC) be used?
Answer: IOC should be used in cases of anatomical uncertainty or suspicion of bile duct injury.
Key Findings: IOC aids in the prevention and immediate management of BDI by providing a precise assessment of biliary anatomy during surgery.
Question 3: What are the recommendations for managing patients with confirmed or suspected bile duct injury?
Answer: Patients with confirmed or suspected BDI should be referred to an experienced surgeon or a multidisciplinary hepatobiliary team.
Key Findings: Early referral to hepatobiliary specialists is associated with better long-term outcomes and lower complication rates.
Question 4: Should the “fundus-first” technique be used when CVS cannot be achieved?
Answer: Yes, the “fundus-first” technique is recommended when CVS cannot be achieved.
Key Findings: This technique is effective for safely dissecting the gallbladder in complex cases where anatomy is unclear.
Question 5: Should CVS be documented during laparoscopic cholecystectomy?
Answer: Yes, documenting CVS with double-static photographs is recommended.
Key Findings: Photographic documentation of CVS ensures correct anatomical identification and serves as a record for later review in case of complications.
Question 6: Should near-infrared biliary imaging be used intraoperatively?
Answer: The evidence for near-infrared biliary imaging is limited; thus, IOC is preferred.
Key Findings: IOC is more widely studied and proven effective in preventing BDI compared to near-infrared imaging.
Question 7: Should surgical risk stratification be used to mitigate the risk of BDI?
Answer: Yes, surgical risk stratification is recommended.
Key Findings: Risk stratification helps identify patients at higher risk of complications, aiding in surgical planning and decision-making.
Question 8: Should the presence of cholecystolithiasis be considered in risk stratification?
Answer: Yes, the presence of cholecystolithiasis should be considered in risk stratification.
Key Findings: Patients with cholecystolithiasis have a higher risk of complications during cholecystectomy, making it important to include this condition in risk assessments.
Question 9: Should immediate cholecystectomy be performed in cases of acute cholecystitis?
Answer: Yes, immediate cholecystectomy within 72 hours is recommended.
Key Findings: Surgery within 72 hours of the onset of acute cholecystitis symptoms is associated with lower complication rates and better patient recovery.
Question 10: Should subtotal cholecystectomy be performed in cases of severe inflammation?
Answer: Yes, subtotal cholecystectomy is recommended in cases of severe inflammation where CVS cannot be obtained.
Key Findings: In severe inflammation scenarios, subtotal cholecystectomy can facilitate the surgery and reduce the risk of BDI.
Question 11: Which approach is preferable, four-port laparoscopic cholecystectomy or reduced-port/single-incision?
Answer: Four-port laparoscopic cholecystectomy is recommended as the standard approach.
Key Findings: The four-port technique is the most studied, showing effectiveness and safety in performing cholecystectomies with lower complication risks.
Question 12: Should interval cholecystectomy be performed following percutaneous cholecystostomy?
Answer: Yes, interval cholecystectomy is recommended after initial stabilization with percutaneous cholecystostomy.
Key Findings: Interval cholecystectomy offers better long-term outcomes and lower risk of recurrent complications compared to no additional treatment.
Question 13: Should laparoscopic cholecystectomy be converted to open in difficult cases?
Answer: Yes, conversion to open surgery is recommended in cases of significant difficulty.
Key Findings: Conversion to open surgery can prevent BDI in situations where laparoscopic dissection is extremely difficult or risky.
Question 14: Should a waiting time be implemented to verify CVS?
Answer: Yes, a waiting time to verify CVS is recommended.
Key Findings: A waiting time allows better anatomical evaluation before proceeding with dissection, reducing the risk of BDI.
Question 15: Should two surgeons be used in complex cases?
Answer: The presence of two surgeons can be beneficial in complex cases, although strong recommendations are not made due to limited evidence.
Key Findings: Some studies suggest that collaboration between two surgeons can improve anatomical identification and reduce complications in difficult cases.
Question 16: Should surgeons receive coaching on CVS to limit the risk or severity of BDI?
Answer: Yes, surgeons should receive coaching on CVS.
Key Findings: Surgeons who receive targeted coaching on CVS show improved anatomical identification and reduced rates of BDI.
Question 17: Should simulation or video-based education be used to train surgeons?
Answer: Yes, simulation or video-based education should be used.
Key Findings: These training methods enhance technical skills, increase surgical precision, and reduce the incidence of BDI during laparoscopic cholecystectomy.
Conclusion
The consensus recommendations provide evidence-based approaches to minimize bile duct injury during laparoscopic cholecystectomy. Practices such as the critical view of safety (CVS), intraoperative cholangiography (IOC), and early referral to specialists can significantly improve surgical outcomes and reduce complications. As famously stated, “The history of surgery is the history of the control of bleeding,” a phrase that underscores the importance of meticulous surgical technique and the prevention of complications like bile duct injuries.
Revisional Surgery for Weight Regain
- Introduction
Revisional bariatric surgery is a weight loss surgery for people who have not lost enough weight or have regained weight after their first bariatric surgery. It is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain.
- Causes of Weight Regain
There are many factors that can contribute to weight regain after bariatric surgery, including:
* Lack of adherence to dietary recommendations
* Hormonal/metabolic imbalance
* Mental health
* Physical inactivity
* Anatomic/surgical factors
* Medications
- Preoperative Evaluation
Before revisional surgery, patients will undergo a thorough evaluation to determine if they are eligible for the procedure. This evaluation will include a medical history and physical exam, as well as blood tests, imaging studies, and a psychological evaluation.
- Selecting the Type of Revisional Surgery
The type of revisional surgery that is best for a patient will depend on a number of factors, including the type of primary surgery they had, the cause of their weight regain, and their overall health. Some of the most common types of revisional surgery include:
* Roux-en-Y gastric bypass (RYGB)
* Sleeve gastrectomy (SG)
* Biliopancreatic diversion with duodenal switch (BPD/DS)
* Single anastomosis gastric bypass (OAGB)
* Sleeve gastrectomy with duodenal switch (SADI-S)
- Weight Loss Following Revisional Surgery
Weight loss after revisional surgery can be significant. In one study, patients who underwent revisional surgery for weight regain lost an average of 50–65.3% of their excess weight after 3 months and 50.1–79.1% of their excess weight after 12 months.
- Complications of Revisional Surgery
Revisional surgery is more complex and technically demanding than primary bariatric surgery, and therefore carries a higher risk of complications. Some of the most common complications of revisional surgery include:
* Hernia
* Anastomotic leak
* Stricture
* Marginal ulcer
* Wound infection
* Hemorrhage
* Perforation
* Obstruction
- Conclusion
Revisional bariatric surgery can be a successful treatment option for patients with weight regain. However, it is important to carefully consider the risks and benefits of the procedure before making a decision. Patients should also work closely with their healthcare team to choose the best type of revisional surgery for them and to prepare for the procedure and its aftermath. Revisional bariatric surgery is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain. It is important to work closely with your healthcare team to determine if you are eligible for the procedure and to choose the best type of surgery for you.
Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
Tratamento Cirúrgico da ACALASIA ESOFÁGICA
A acalasia, definida como a falha ou relaxamento incompleto do esfíncter esofágico inferior (EEI), acompanhada de aperistalse do corpo esofágico na ausência de obstrução mecânica, é o tipo mais comum de distúrbio da motilidade esofágica. Tem uma incidência de 1 em 100.000 pessoas, com uma prevalência de 10 em 100.000. Não há diferença na prevalência de gênero entre as idades de 30 e 60 anos. A causa primária da acalasia ainda é indeterminada, mas acredita-se que surja da degeneração das células ganglionares inibitórias no plexo miontérico do EEI e corpo esofágico. Fatores associados a um risco aumentado de acalasia incluem distúrbios virais/neurodegenerativos, síndrome de Down, diabetes mellitus tipo 1, hipotireoidismo e condições autoimunes, como a síndrome de Sjögren, o lúpus eritematoso sistêmico e a uveíte. Os casos familiares são raros.
O diagnóstico de acalasia deve ser suspeitado em pacientes com disfagia para sólidos e líquidos que não melhora com o uso de inibidores da bomba de prótons. Se não for tratada, a acalasia é uma doença progressiva que pode evoluir para megaesôfago e está associada a um aumento do risco de carcinoma de células escamosas do esôfago. Embora não existam critérios padronizados para determinar a gravidade da doença, o diâmetro e a confirmação do esôfago dentro da cavidade torácica são geralmente considerados os dois principais fatores. A escala de Eckardt é uma escala frequentemente usada para avaliar a gravidade da doença e a eficácia da terapia. Qualitativamente, a acalasia grave é definida como um diâmetro esofágico maior que 6 cm; a acalasia em estágio avançado inclui a angulação distal, um esôfago sigmóide/tortuoso com diâmetro maior que 6 cm ou um megaesôfago com diâmetro maior que 10 cm. Aproximadamente 5% a 15% das pessoas com acalasia evoluem para acalasia em estágio avançado, são geralmente resistentes aos tratamentos endoscópicos e cirúrgicos iniciais e, em última instância, requerem uma esofagectomia.
MIOTOMIA Á HELLER
A miotomia laparoscópica de Heller (LHM) com fundoplicatura parcial, desenvolvida como uma alternativa minimamente invasiva à miotomia anterior aberta tradicional e posteriormente à miotomia toracoscópica, tem sido o padrão ouro para o tratamento da acalasia nas últimas três décadas. O objetivo da miotomia é abrir completamente o EEI e aliviar a disfagia. A LHM proporciona alívio sintomático inicial da disfagia em cerca de 90% dos pacientes com tipos I e II de acalasia e em 50% dos pacientes com tipo III de acalasia, enquanto diminui as taxas de refluxo pós-operatório. Em comparação com os procedimentos de miotomia aberta, a LHM está associada a menor dor pós-operatória, menor tempo de internação hospitalar e retorno mais precoce à função. A LHM é indicada como tratamento de primeira linha para todos os candidatos à cirurgia com acalasia que estejam dispostos a se submeter à cirurgia ou para aqueles que falharam na dilatação endoscópica. A operação consiste em dividir os músculos do EEI, seguida por uma fundoplicatura para diminuir o refluxo pós-operatório. A fundoplicatura parcial é favorecida em relação à fundoplicatura total porque reduz a falha do tratamento. As duas principais complicações da cirurgia são perfuração da mucosa e DRGE.
Intraoperative cholangiography: Selective or Routine?
Intraoperative cholangiography (IOC), described by Mirizzi in 1932, represented a significant advance in the diagnosis of choledocolithiasis during cholecystectomy. The natural history of asymptomatic choledocolithiasis has been investigated in different populations and its therapeutic management continues to be controversial. IOC is traditionally advocated as a procedure to be adopted in all laparoscopic cholecystectomies since it permits to define the anatomy of the biliary tract and to detect common bile duct stones. However, in the laparoscopic era, technological advances in radiologic-endoscopic workup have markedly increased the costs of investigation of patients with suspected choledocolithiasis. The routine use of IOC has raised the question of which cases require the exploration of bile tract anatomy during surgery and whether there are methods to predict preoperatively unsuspected choledocolithiasis. Therefore, the most appropriate management of preoperatively unsuspected choledocolithiasis, i.e., the routine or selective use of IOC, still remains undefined. In this respect, well-defined criteria for the inclusion of patients with possible choledocolithiasis who should be submitted to cholangiography exist in the literature, but there is no safe approach to exclude asymptomatic patients without an indication for contrast examination.
A systematic literature search was performed by KOVACS N, et al (2022) using the following search keys: cholangiogra* and cholecystectomy. The primary outcomes were BDI and retained stone rate. To investigate the differences between the groups (routine IOC vs selective IOC and IOC vs no IOC), they calculated weighted mean differences (WMD) for continuous outcomes and relative risks (RR) for dichotomous outcomes, with 95% confidence intervals (CI). Of the 19,863 articles, 38 were selected and 32 were included in the quantitative synthesis. Routine IOC showed no superiority compared to selective IOC in decreasing BDI (RR = 0.91, 95% CI 0.66; 1.24). Comparing IOC and no IOC, no statistically significant differences were found in the case of BDI, retained stone rate, readmission rate, and length of hospital stay. They found an increased risk of conversion rate to open surgery in the no IOC group (RR = 0.64, CI 0.51; 0.78). The operation time was significantly longer in the IOC group compared to the no IOC group (WMD = 11.25 min, 95% CI 6.57; 15.93). So this data findings suggest that IOC may not be indicated in every case, however, the evidence is very uncertain.
Subtotal cholecystectomy for difficult acute cholecystitis
Laparoscopic cholecystectomy is considered the gold standard for treatment of benign gallbladder diseases. Cholecystectomy using this method can be completed in 90% of elective cholecystectomies and 70% of emergency cholecystectomies. Acute cholecystitis, especially if difficult, can change the above paradigm, resulting in open conversion or change of technique. The conditions that define a difficult cholecystectomy are as follows: necessity of conversion from laparoscopic to open surgery; duration of procedure greater than 180 min; blood loss greater than 300 ml; and urgent need for involvement of a more experienced surgeon. One of the “rescue” procedures to complete the surgery safely (both for the surgeons and patients) is subtotal cholecystectomy (STC). Open and laparoscopic subtotal cholecystectomy have been reported. For many surgeons, this is considered a bail out technique, and the timing of decision making is crucial to avoid catastrophic complications. Te capability to perform STC in laparoscopy is increasingly requested during difficult laparoscopic cholecystectomy. Difficult LC has a risk of BDI from 3 to 5 times higher in laparoscopy than open surgery. In case of operative difficulties of young surgeons mostly trained in laparoscopy the help of senior surgeons is strongly recommended. The purpose of the present study is to clarify how laparoscopic subtotal cholecystectomy may be used to complete a difficult cholecystectomy for acute cholecystitis without serious complications.

Biliary leakage represents the most frequent complication of incomplete resection of the gallbladder wall in cases of difficult acute cholecystectomy treated with subtotal cholecystectomy. This complication is rarely fatal but requires correct treatment. If bile leakage does not stop spontaneously seven days postoperatively, the possible treatments are endoscopic biliary sphincterotomy, endoscopic plastic stent, and a fully covered self-expanding metal stent. When performing closure of the gallbladder stump, suturing the anterior residual of both anterior and posterior wall represents the best method to have fewer complications. Complications, if not lethal, decrease the patient’s quality of life. Intraoperatively, it is of utmost importance to carefully expose the gallbladder stump to avoid left-in-place stones, wash the entire cavity and drain the abdomen. Bile duct injuries can be a significant complication in this type of surgery. Prevention of the lesions with conversion from laparoscopic to open, or the opinion of older surgeon in case of difficulties is strongly recommended. Mortality is a very rare complication. The limitations of our study are given by the heterogeneity of the techniques used for LSC and the lack of a long-term follow-up analyzing the related complications.
Aderências Pós-Operatórias
Do Manejo Conservador à Decisão Cirúrgica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
Introdução
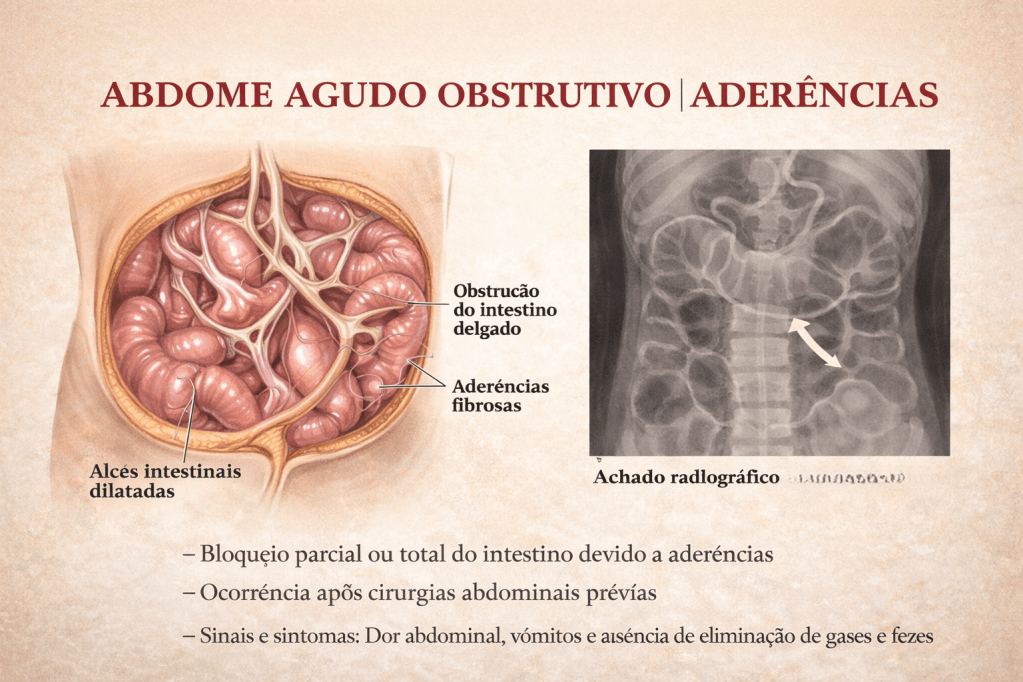
A obstrução intestinal é um dos cenários mais desafiadores e frequentes no pronto-socorro cirúrgico. Historicamente, o abdome agudo obstrutivo impunha o dilema imediato da exploração cirúrgica. Hoje, compreendemos que a etiologia dita o compasso da intervenção. Entre todas as causas de obstrução do intestino delgado, as aderências pós-operatórias (bridas) assumem o protagonismo absoluto. Para o estudante de medicina, o residente e o cirurgião do aparelho digestivo, dominar o manejo da obstrução por bridas é essencial. O desafio não reside apenas na técnica operatória da enterólise, mas sim no raciocínio clínico aguçado para responder à pergunta fundamental: quando persistir no tratamento conservador e quando a indicação cirúrgica se torna imperativa?
Cenário Brasileiro
A formação de aderências peritoniais é uma resposta fisiológica quase universal ao trauma cirúrgico (isquemia, manipulação, coágulos), ocorrendo em até 90% dos pacientes submetidos a laparotomias. Contudo, em uma parcela significativa, essa cicatrização aberrante leva à obstrução mecânica. Na prática da cirurgia geral no Brasil, a obstrução por bridas responde por cerca de 60% a 75% das admissões por obstrução do intestino delgado. Dados do Sistema Único de Saúde (DataSUS) indicam que milhares de laparotomias exploradoras são realizadas anualmente no país devido a quadros obstrutivos agudos, gerando elevado custo financeiro e considerável morbimortalidade, além de reinternações frequentes.
Fisiopatologia e Diagnóstico
O peritônio, quando lesado, inicia uma cascata inflamatória rica em fibrina. Em condições normais, a fibrinólise degrada essa matriz em poucos dias. O desequilíbrio nesse sistema — com falha na fibrinólise peritonia — resulta na organização da fibrina em tecido conjuntivo vascularizado: a aderência.
A Tríade Diagnóstica
O diagnóstico é eminentemente clínico e radiológico:
- Clínica: Dor abdominal em cólica, distensão, vômitos (fecaloides em fases tardias) e parada de eliminação de flatus e fezes.
- Laboratório: Hemograma, eletrólitos, lactato e gasometria são cruciais para rastrear sinais de isquemia ou necrose (leucocitose importante, acidose metabólica).
- Imagem: A Tomografia Computadorizada (TC) de abdome com contraste venoso substituiu o raio-X simples como padrão-ouro. A TC identifica o ponto de transição (zona de calibres distintos), o grau de obstrução, a presença de sofrimento de alça (espessamento parietal, pneumatose, gás no sistema porta) e descarta outras etiologias (hérnias internas, neoplasias).
Estratégias de Tratamento
O manejo da obstrução por bridas sofreu uma evolução paradigmática, distanciando-se de intervenções intempestivas em favor de protocolos baseados em evidências.
1. Tratamento Conservador (Não Operatório)
Na ausência de sinais de isquemia intestinal, perfuração ou peritonite localizada, a conduta inicial deve ser conservadora:
- Jejum (NPO) e descompressão com Sonda Nasogástrica (SNG).
- Ressuscitação volêmica vigorosa e correção de distúrbios hidroeletrolíticos.
- Protocolo com Contraste Hidrossolúvel: A administração de contraste hiperosmolar (ex: Gastrografin) via SNG não possui apenas valor diagnóstico (avaliar se o contraste atinge o cólon em 24h), mas também efeito terapêutico. A hiperosmolaridade atrai líquido para a luz intestinal, reduzindo o edema da parede e estimulando o peristaltismo, resolvendo o quadro obstrutivo em uma parcela significativa dos casos.
2. Tratamento Cirúrgico: O Timing Ideal
A falha do tratamento conservador (geralmente avaliada após 48 a 72 horas) ou o surgimento de sinais de alarme (taquicardia, febre, irritação peritoneal, acidose) indicam cirurgia de urgência.
- Laparotomia vs. Laparoscopia: A via aberta continua sendo o padrão em pacientes com múltiplas cirurgias prévias ou grande distensão abdominal. A abordagem laparoscópica é excelente em mãos experientes, mas reserva-se a casos selecionados (ex: suspeita de brida única, distensão moderada), exigindo extrema cautela na introdução do primeiro trocarte (técnica aberta de Hasson) para evitar enterotomias iatrogênicas.
- O Ato Cirúrgico: A lise de aderências deve ser restrita ao necessário para resolver a obstrução. Múltiplas enterólises desnecessárias aumentam o risco de lesões e a formação de novas bridas. Em caso de necrose, a ressecção do segmento isquêmico com anastomose primária ou estomia (a depender da estabilidade hemodinâmica) é imperativa.
Pontos-Chave para a Prática Diária
- Suspeição de Isquemia: Dor refratária a analgésicos, taquicardia inexplicada e acidose metabólica são gritos de socorro de uma alça intestinal isquêmica. O tratamento conservador é contraindicado.
- O Valor da TC: A tomografia não apenas confirma o diagnóstico, mas é o farol que guia a decisão entre a enfermaria e o centro cirúrgico.
- Prevenção Primária: O uso de barreiras antiaderentes ainda carece de evidências universais robustas, mas a técnica cirúrgica apurada, manipulação tecidual delicada e controle rigoroso da hemostasia continuam sendo a melhor profilaxia.
Conclusões Aplicadas
O tratamento da obstrução intestinal por bridas é a quintessência do bom senso cirúrgico. O cirurgião moderno deve possuir a paciência de um clínico para conduzir o tratamento conservador respaldado pelo contraste hidrossolúvel e, simultaneamente, a assertividade de um intervencionista para indicar a sala de cirurgia ao primeiro sinal de estrangulamento.Compreender que “operar a radiografia” é um erro crasso e que a reavaliação clínica seriada (de preferência pelo mesmo examinador) é a bússola do tratamento, molda o profissional de excelência que minimiza ressecções intestinais desnecessárias e reduz a morbidade pós-operatória.
“Na obstrução intestinal aguda, não deixe o sol se pôr ou nascer sobre o paciente sem uma decisão. Mas lembre-se: a cirurgia sem indicação precipita o desastre que buscava evitar.” — Aforismo clássico derivado dos ensinamentos de Sir Zachary Cope, pioneiro no estudo e diagnóstico do abdome agudo.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
The “BAD” Gallbladder
Once the decision for surgery has been made, an operative plan needs to be discussed and implemented. Should one initially start with laparoscopic surgery for the “bad gallbladder”? If a laparoscopic approach is taken, when should bail-out maneuvers be attempted? Is converting to open operation still the standard next step? A 2016 study published by Ashfaq and colleagues sheds some light on our first question. They studied 2212 patients who underwent laparoscopic cholecystectomy, of which 351 were considered “difficult gallbladders.” A difficult gallbladder was considered one that was necrotic or gangrenous, involved Mirizzi syndrome, had extensive adhesions, was converted to open, lasted more than 120 minutes, had a prior tube cholecystostomy, or had known gallbladder perforation. Seventy of these 351 operations were converted to open. The indications for conversion included severe inflammation and adhesions around the gallbladder rendering dissection of triangle of Calot difficult (n 5 37 [11.1%]), altered anatomy (n 5 14 [4.2%]), and intraoperative bleeding that was difficult to control laparoscopically (n 5 6 [1.8%]). The remaining 13 patients (18.5%) included a combination of cholecystoenteric fistula, concern for malignancy, common bile duct exploration for stones, and inadvertent enterotomy requiring small bowel repair. Comparing the total laparoscopic cholecystectomy group and the conversion groups, operative time and length of hospital stay were significantly different; 147 +- 47 minutes versus 185 +- 71 minutes (P<.005) and 3+-2 days versus 5+-3 days (P 5 .011), respectively. There was no significant difference in postoperative hemorrhage, subhepatic collection, cystic duct leak, wound infection, reoperation, and 30-day mortality.2 From these findings, we can glean that most cholecystectomies should be started laparoscopically, because it is safe to do so. It is the authors’ practice to start laparoscopically in all cases.
BAILOUT PROCEDURES
Despite the best efforts of experienced surgeons, it is sometimes impossible to safely obtain the critical view of safety in a bad gallbladder with dense inflammation and even scarring in the hepatocystic triangle. Continued attempts to dissect in this hazardous region can lead to devastating injury, including transection of 1 or both hepatic ducts, the common bile duct, and/or a major vascular injury (usually the right hepatic artery). Therefore, it is imperative that any surgeon faced with a bad gallbladder have a toolkit of procedures to safely terminate the operation while obtaining maximum symptom and source control, rather than continue to plunge blindly into treacherous terrain. If the critical view of safety cannot be achieved owing to inflammation, and when further dissection in the hepatocystic triangle is dangerous, these authors default to laparoscopic subtotal cholecystectomy as our bail-out procedure of choice. The rationale for this approach is that it resolves symptoms by removing the majority of the gallbladder, leading to low (although not zero) rates of recurrent symptoms. It is safe, and can be easily completed laparoscopically, thus avoiding the longer hospital stay and morbidity of an open operation. There is now significant data supporting this approach. In a series of 168 patients (of whom 153 were laparoscopic) who underwent subtotal cholecystectomy for bad gallbladders, the mean operative time was 150 minutes (range, 70–315 minutes) and the average blood loss was 170 mL (range, 50–1500 mL). The median length of stay for these patients was 4 days (range, 1–68 days), and there were no common bile duct injuries.23 There were 12 postoperative collections (7.1%), 4 wound infections (2.4%), 1 bile leak (0.6%), and 7 retained stones (4.2%), but the 30-day mortality was similar to those who underwent a total laparoscopic cholecystectomy. A systematic review and meta-analysis by Elshaer and colleagues showed that subtotal cholecystectomy achieves comparable morbidity rates compared with total cholecystectomy. These data support the idea that we should move away from the idea that the only acceptable outcome for a cholecystectomy is the complete removal of a gallbladder, especially when it is not safe to do so. This shift toward subtotal cholecystectomy has been appropriately referred to as the safety first, total cholecystectomy second approach.
Managing the “difficult” gallbladder
The gold standard for the surgical treatment of symptomatic cholelithiasis is conventional laparoscopic cholecystectomy (LC). The “difficult gallbladder” is a scenario in which a cholecystectomy turns into an increased surgical risk compared with standard cholecystectomy. The procedure may be difficult due to processes that either obscure normal biliary anatomy (such as acute or chronic inflammation) or operative exposure (obesity or adhesions caused by prior upper abdominal surgery). So, when confronted with a difficult cholecystectomy, the surgeon has a must: to turn the operation into a safe cholecystectomy, which can mean conversion (to an open procedure), cholecystostomy, or partial/ subtotal cholecystectomy. The surgeon should understand that needs to rely on damage control, to prevent more serious complications if choosing to advance and progress to a complete cholecystectomy.
When to Predict a Difficult Laparoscopic Cholecystectomy
A difficult cholecystectomy may be predicted preoperatively based on patient characteristics and ultrasound and laboratory findings. This is probably a very important step in mitigating the high risk associated with a difficult procedure and may serve either to reschedule the procedure or design intraoperative strategies of management to guarantee a safe performance of the surgical procedure.
The following situations are associated with a higher chance of a difficult cholecystectomy:
• Acute cholecystitis (more than 5 days of onset)
• Previous cholecystitis episode
• Male sex
• Obesity
• Cirrhosis
• Sclero-atrophic gallbladder
• Thick walls (>5 mm)
• Previous signs of canalicular dwelling (clinical and laboratory)
Through multivariate analysis, Bourgoin identified these elements of predictive help to identify difficult LC: male sex, previous cholecystitis attack, fibrinogen, neutrophil, and alkaline phosphatase levels. Another important point is the fact of conversion from a laparoscopic procedure to an open and traditional cholecystectomy, usually through a right subcostal incision. Conversion should not be considered as a personal failure, and the surgeon needs to understand the concept of “safety first,” considering that conversion is performed in order to complete the procedure without additional risks and preventing complications and not solving intraoperative complications. It is also useful to define a time threshold to aid in the decision to convert. It is not worth taking an hour and a half and still dissecting adhesions, preventing the correct visualization of the cystic pedicle. This time limit represents a method to prevent inefficiencies in the operating room (OR) schedule as well as additional expenditures.
A smart surgeon should rely to conversion in the following situations:
• Lack of progress in the procedure
• Unclear anatomy/any grade of uncertainty
• CVS not achieved
• Bleeding/vascular injury
• BD injury
• Lack of infrastructure, expertise, and support
Final Remarks
The primary goal of a laparoscopic cholecystectomy in the treatment of symptomatic cholelithiasis is the safe remotion of the gallbladder and the absence of common bile duct injury. Some tips to take into account:
– Never perform a laparoscopic cholecystectomy without a skilled surgeon close by.
– Beware of the easy gallbladder.
– Slow down, take your time.
– Knowledge is power, conversión can be the salvation!
– Do not repair a bile duct injury (unless you have performed at least 25 hepaticojejunostomies).
– Do not ignore postoperative complaints (pain, jaundice, major abdominal discomfort, fever)
Other options when confronted with a difficult laparoscopic cholecystectomy are:
– A percutaneous cholecystostomy, if the risk was identified preoperatively or the patient is a poor surgical candidate;
– An intraoperative cholangiography, which may aid in identifying an injury to the bile duct and solve it, if you are an experienced surgeon;
– A subtotal or partial cholecystectomy;
– Ask for help;
– Conversion to an open procedure;
POPF after Distal Pancreatectomy
Minimally Invasive Versus Open Techniques
Despite advances in laparoscopic and robotic approaches, the vast majority of distal pancreatectomies continue to be performed via an open approach. Recent retrospective data have demonstrated that minimally invasive distal pancreatectomy is associated with decreased blood loss and shorter hospital stays than open pancreatectomy. A large recent study utilizing the Nationwide Inpatient Sample database suggested, first, that the minimally invasive approach is becoming more widely utilized, increasing from 2.4 to 7.3 % over a study period from 1998 to 2009. Second, that study reported that the minimally invasive approach was associated with decreased length of stay as well as decreased incidence of infectious complications, bleeding complications, and blood transfusions. This population-based study echoes conclusions drawn by a large multi-institutional study performed several years previously. Drawing on a combined patient sample of 667 patients, with 24 % initially attempted laparoscopically, the authors were able to demonstrate lower overall complication rate, decreased blood loss, and shorter hospital stays among patients undergoing laparoscopic approach via a multivariate analysis.
Notably, there was no significant difference in the pancreatic leak rate between the open and laparoscopic approaches, although there was a nonsignificant trend favoring the laparoscopic approach. More recently, the robotic approach has generated significant interest as a technique for performing distal pancreatectomy. Retrospective analysis has suggested that the robotic approach is well suited for pancreatectomy. Fistula rates, however, remain a concern. A retrospective review of patients undergoing robotic pancreatic operations included 83 patients who underwent distal pancreatectomy. About 27 % were identified as having a ISPGF type A pancreatic leak; 12 and 4.8 % were identified as having a grade B or C leak, respectively.
Identifying Risk Factors
For pancreaticoduodenectomy (PTD) , a fistula risk score has been recently developed that has been shown to be highly predictive of POPF. This score assigns points based on gland texture, gland pathology, duct diameter, and intraoperative blood loss. In general, high blood loss, soft gland texture, and smaller duct diameter confer increased risk of POPF, whereas pancreatic adenocarcinoma and pancreatitis as the indication for PTD confer protection for the development of pancreatic fistula versus other diagnoses. Also of note, higher fistula risk scores correlated with greater incidence of clinically relevant (ISGPF grade B or C) fistula. The adaptation of this risk score to patients undergoing distal pancreatectomy is yet to be validated; however, at least one published study indicates that this scoring system may have limitations in the setting of distal pancreatectomy. In that study, risk factors for pancreatic fistula after stapled gland transection in patients undergoing distal pancreatectomy were examined, and in a multivariate analysis, only the presence of diabetes and the use of a 4.1-mm staple cartridge were associated with increased risk of pancreatic fistula formation.
Critical View Of Safety
“The concept of the critical view was described in 1992 but the term CVS was introduced in 1995 in an analytical review of the emerging problem of biliary injury in laparoscopic cholecystectomy. CVS was conceived not as a way to do laparoscopic cholecystectomy but as a way to avoid biliary injury. To achieve this, what was needed was a secure method of identifying the two tubular structures that are divided in a cholecystectomy, i.e., the cystic duct and the cystic artery. CVS is an adoption of a technique of secure identification in open cholecystectomy in which both cystic structures are putatively identified after which the gallbladder is taken off the cystic plate so that it is hanging free and just attached by the two cystic structures. In laparoscopic surgery complete separation of the body of the gallbladder from the cystic plate makes clipping of the cystic structures difficult so for laparoscopy the requirement was that only the lower part of the gallbladder (about one-third) had to be separated from the cystic plate. The other two requirements are that the hepatocystic triangle is cleared of fat and fibrous tissue and that there are two and only two structures attached to the gallbladder and the latter requirements were the same as in the open technique. Not until all three elements of CVS are attained may the cystic structures be clipped and divided. Intraoperatively CVS should be confirmed in a “time-out” in which the 3 elements of CVS are demonstrated. Note again that CVS is not a method of dissection but a method of target identification akin to concepts used in safe hunting procedures. Several years after the CVS was introduced there did not seem to be a lessening of biliary injuries.
Operative notes of biliary injuries were collected and studied in an attempt to determine if CVS was failing to prevent injury. We found that the method of target identification that was failing was not CVS but the infundibular technique in which the cystic duct is identified by exposing the funnel shape where the infundibulum of the gallbladder joins the cystic duct. This seemed to occur most frequently under conditions of severe acute or chronic inflammation. Inflammatory fusion and contraction may cause juxtaposition or adherence of the common hepatic duct to the side of the gallbladder. When the infundibular technique of identification is used under these conditions a compelling visual deception that the common bile duct is the cystic duct may occur. CVS is much less susceptible to this deception because more exposure is needed to achieve CVS, and either the CVS is attained, by which time the anatomic situation is clarified, or operative conditions prevent attainment of CVS and one of several important “bail-out” strategies is used thus avoiding bile duct injury.
CVS must be considered as part of an overall schema of a culture of safety in cholecystectomy. When CVS cannot be attained there are several bailout strategies such a cholecystostomy or in the case of very severe inflammation discontinuation of the procedure and referral to a tertiary center for care. The most satisfactory bailout procedure is subtotal cholecystectomy of which there are two kinds. Subtotal fenestrating cholecystectomy removes the free wall of the gallbladder and ablates the mucosa but does not close the gallbladder remnant. Subtotal reconstituting cholecystectomy closes the gallbladder making a new smaller gallbladder. Such a gallbladder remnant is undesirable since it may become the site of new gallstone formation and recurrent symptoms . Both types may be done laparoscopically.”
Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 1995;180:101-25.
Laparoscopic Liver Resection
INTRODUCTION
Laparoscopic hepatic resection is an emerging option in the field of hepatic surgery. With almost 3000 laparoscopic hepatic resections reported in the literature for benign and malignant tumors, with a combined mortality of 0.3% and morbidity of 10.5%, there will be an increasing demand for minimally invasive liver surgery. Multiple series have been published on laparoscopic liver resections; however, no randomized controlled trial has been reported that compares laparoscopic with open liver resection. Large series, meta-analyses, and reviews have thus far attested to the feasibility and safety of minimally invasive hepatic surgery for benign and malignant lesions.
THECNICAL ASPECTS
The conversion rate from a laparoscopic approach to an open procedure was 4.1%. The most common type of laparoscopic liver resection performed is a wedge resection or segmentectomy (45%), followed by left lateral sectionectomy (20%). Major anatomic hepatectomies are still less frequently performed: right hepatectomy (9%) and left hepatectomy (7%). Cumulative morbidity and mortality was 10.5% and 0.3%.
BENEFITS OF LAPAROSCOPIC APPROACH
More importantly, almost all the studies comparing laparoscopic with open liver resection consistently showed a significant earlier discharge to home after laparoscopic liver resection. Lengths of stay were variable based on the country of origin of the studies but were consistently shorter for laparoscopic liver resection. Three studies published in the United States presented a length of stay of 1.9 to 4.0 days after laparoscopic liver resection. Studies from Europe showed an average length of stay of 3.5 to 10 days whereas those from Asia reported an average of length of stay of 4 to 20 days after laparoscopic liver resection.
COST ANALYSIS
Vanounou and colleagues used deviation-based cost modeling to compare the costs of laparoscopic with open left lateral sectionectomy at the University of Pittsburgh Medical Center. They compared 29 laparoscopic with 40 open cases and showed that patients who underwent the laparoscopic approach faired more favorably with a shorter length of stay (3 vs 5 days, P<.0001), significantly less postoperative morbidity (P 5 .001), and a weighted-average median cost savings of $1527 to $2939 per patient compared with patients who underwent open left lateral sectionectomy.
SURGICAL MARGINS
Initial concerns about the adequacy of surgical margins and possible tumor seeding prevented the widespread adoption of laparoscopic resection approaches for liver cancers. In comparison studies, there were no differences in margin-free resections between laparoscopic and open liver resection. In addition, no incidence of port-site recurrence or tumor seeding has been reported. With more than 3000 cases of minimally invasive hepatic resection reported in the literature (and no documentation of any significant port-site or peritoneal seeding), the authors conclude that this concern should not prevent surgeons from accepting a laparoscopic approach.
SURVIVAL OUTCOMES
There were no significant differences in overall survival in the 13 studies that compared laparoscopic liver resection with open liver resection for cancer. For example, Cai and colleagues showed that the 1-, 3-, and 5-year survival rates after laparoscopic resection of HCC were 95.4%, 67.5%, and 56.2% versus 100%, 73.8%, and 53.8% for open resection. For resection of colorectal cancer liver metastasis, Ito and colleagues showed a 3-year survival of 72% after laparoscopic liver resection and 56% after open liver resection whereas Castaing and colleagues51 showed a 5-year survival of 64% after laparoscopic liver resection versus 56% after open liver resection.
CONCLUSION
Compared with open liver resections, laparoscopic liver resections are associated with less blood loss, less pain medication requirement, and shorter length of hospital stay. A randomized controlled clinical trial is the best method to compare laparoscopic with open liver resection; however, such a trial may be difficult to conduct because patients are unlikely to subject themselves to an open procedure when a minimally invasive approach has been shown feasible and safe in experienced hands. In addition, many patients would have to be accrued to detect a difference in complications that occur infrequently. Short of a large randomized clinical trial, meta-analysis and matched comparisons provide the next best option to compare laparoscopic with open liver resection. For laparoscopic resection of HCC or colorectal cancer metastases, there has been no difference in 5-year overall survival compared with open hepatic resection. In addition, from a financial standpoint, the minimally invasive approach to liver resection may be associated with higher operating room costs; however, the total hospital costs were offset or improved due to the associated shorter length of hospital stay with the minimally invasive approach.
Medically-Necessary, Time Sensitive: (MeNTS) Score
Operating During The COVID-19 Coronavirus Pandemic
“At the University of Chicago, members of the Department of Surgery decided to investigate this issue more precisely. As stay-at-home restrictions in some states are easing, and as non-emergency medical care is being reconsidered, how does one possibly triage the thousands upon thousands of patients whose surgeries were postponed? Instead of the term “elective,” the University of Chicago’s Department of Surgery chose the phrase “Medically-Necessary, Time Sensitive” (MeNTS). This concept can be utilized to better assess the acuity and safety when determining which patients can get to the operating room in as high benefit/low risk manner as possible. And unlike in any recent time in history, risks to healthcare staff as well as risks to the patient from healthcare staff, are now thrown into the equation. The work was published in the April issue of the Journal of the American College of Surgeons.
On March 17, 2020, the American College of Surgeons recommended that all “elective” surgeries be canceled indefinitely. These guidelines were published, stating that only patients with “high acuity” surgical issues, which would include aggressive cancers and severely symptomatic disease, should proceed. Based on the Elective Surgery Acuity Scale (ESAS), most hospitals were strongly encouraged to cancel any surgery that was not high acuity, including slow-growing cancers, orthopedic and spine surgeries, airway surgeries, and any other surgeries for non-cancerous tumors. Heart surgeries for stable cardiac issues were also put on hold. Patients and surgeons waited. Some patients did, indeed undergo non-Covid-19-related surgeries. But most did not. Redeployment is gradually turning to re-entry.
The re-entry process for non-urgent (yet necessary) surgeries is a complicated one. Decisions and timing, based on a given hospital’s number and severity of Covid-19 patients, combined with a given city or state’s current and projected number of Covid-19 cases, how sick those patients will be, and whether or not a second surge may come, involves a fair amount of guesswork. As we have all seen, data manipulation has become a daily sparring match in many arenas. The authors of the study created an objective surgical risk scoring system, in order to help hospitals across this country, as well as others across the world, better identify appropriate timing regarding which surgeries can go ahead sooner rather than later, and why. They factored several variables into their equation, to account for the multiple potential barriers to care, including health and safety of hospital personnel. They created scoring systems based on three factors: Procedure, Disease and Patient Issues.
CALCULATE MeNTS SCORE HERE
The authors of the study created an objective surgical risk scoring system, in order to help hospitals across this country, as well as others across the world, better identify appropriate timing regarding which surgeries can go ahead sooner rather than later, and why. They factored several variables into their equation, to account for the multiple potential barriers to care, including health and safety of hospital personnel. Each patient would receive an overall conglomerate score, based on all of these factors, with the lower risks giving them more favorable scores to proceed with surgery soon, and the higher risks giving patients a higher score, or higher risk regarding proceeding with surgery, meaning it may be safest, for now, to wait.
Dr. Jeffrey Matthews, senior author of the paper, and Department Chair at the University of Chicago, stated that this model is reproducible across hospital systems, in urban, rural, and academic settings. And in the event of potential unpredictable surges of Covid-19 cases, the scoring system “helps prioritize cases not only from the procedure/disease standpoint but also from the pandemic standpoint with respect to available hospital resources such as PPE, blood, ICU beds, and [regular hospital] beds.”
The scoring system is extremely new, and the coming weeks will reveal how patients, surgeons and hospitals are faring as patients without life-and-death emergencies and/or Covid-19 complications gradually begin filling the operating rooms and hospital beds. In addition, and perhaps just as important, the study authors note that creating systems whereby healthcare resources, safety, and impact on outcomes need to be considered more carefully for each patient intervention, the larger impact of each intervention on public health will be better understood: not only for today’s pandemic, but also in future, as yet unknown, global events.”
Source: Nina Shapiro, 2020
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
Covid-19 and Digestive Surgery
The current world Covid-19 pandemic has been the most discussed topic in the media and scientific journals. Fear, uncertainty, and lack of knowledge about the disease may be the significant factors that justify such reality. It has been known that the disease presents with a rapidly spreading, it is significantly more severe among the elderly, and it has a substantial global socioeconomic impact. Besides the challenges associated with the unknown, there are other factors, such as the deluge of information. In this regard, the high number of scientific publications, encompassing in vitro, case studies, observational and randomized clinical studies, and even systematic reviews add up to the uncertainty. Such a situation is even worse when considering that most healthcare professionals lack adequate knowledge to critically appraise the scientific method, something that has been previously addressed by some authors. Therefore, it is of utmost importance that expert societies supported by data provided by the World Health Organization and the National Health Department take the lead in spreading trustworthy and reliable information.
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
#Medicine #Surgery #GeneralSurgery #DigestiveSurgery
#TheSurgeon #OzimoGama
Laparoscopic JEJUNOSTOMY
Many oncological patients with upper gastrointestinal (GI) tract tumours, apart from other symptoms, are malnourished or cachectic at the time of presentation. In these patients feeding plays a crucial role, including as part of palliative treatment. Many studies have proved the benefits of enteral feeding over parenteral if feasible. Depending on the tumour’s location and clinical stage there are several options of enteral feeding aids available. Since the introduction of percutaneous endoscopic gastrostomy (PEG) and its relatively easy application in most patients, older techniques such as open gastrostomy or jejunostomy have rather few indications.
The majority of non-PEG techniques are used in patients with upper digestive tract, head and neck tumours or trauma that renders the PEG technique unfeasible or unsafe for the patient. In these patients, especially with advanced disease requiring neoadjuvant chemotherapy or palliative treatment, open gastrostomy and jejunostomy were the only options of enteral access. Since the first report of laparoscopic jejunostomy by O’Regan et al. in 1990 there have been several publications presenting techniques and outcomes of laparoscopic feeding jejunostomy. Laparoscopic jejunostomy can accompany staging or diagnostic laparoscopy for upper GI malignancy when the disease appears advanced, hence avoiding additional anaesthesia and an operation in the near future.
In this video the author describe the technique of laparoscopic feeding jejunostomy applied during the staging laparoscopy in patient with advanced upper gastrointestinal tract cancer with co-morbid cachexy, requiring enteral feeding and neoadjuvant chemotherapy.
Welcome and get to know our Social Networks through the following link
Ozimo Gama’s Social Medias
#AnatomiaHumana #CirurgiaGeral #CirurgiaDigestiva #TheSurgeon
Tratamento Cirúrgico do Abscesso Hepático Piogênico
Introdução
O abscesso hepático piogênico (AHP) é uma condição infecciosa grave caracterizada por uma coleção encapsulada de material purulento no fígado. Frequentemente, essa condição é resultante de infecções bacterianas, originárias do trato biliar ou de fontes intra-abdominais, como diverticulite. O manejo do AHP requer uma abordagem multidisciplinar, combinando diagnóstico rápido, antibioticoterapia e, em muitos casos, intervenção cirúrgica. No Brasil, a mortalidade associada a essa condição pode variar de 10% a 20%, sendo particularmente elevada em pacientes com comorbidades, como diabetes e cirrose. A presente revisão discute as abordagens cirúrgicas no tratamento do AHP, com ênfase nos critérios de intervenção, técnicas cirúrgicas e melhores práticas para o cirurgião do aparelho digestivo.
Diagnóstico e Classificação
O diagnóstico precoce do AHP é essencial para determinar a abordagem terapêutica mais adequada. Exames de imagem, como ultrassonografia (USG) e tomografia computadorizada (TC), são as ferramentas primárias para identificar a extensão da lesão e guiar a tomada de decisões. A classificação dos abscessos hepáticos baseia-se em seu tamanho e características morfológicas:
- Abscessos pequenos (menores que 3 cm) podem, muitas vezes, ser tratados com antibioticoterapia isolada.
- Abscessos maiores (geralmente >5 cm) e multiloculados exigem drenagem percutânea ou intervenção cirúrgica.
A etiologia do AHP no Brasil é predominantemente associada a bactérias como Escherichia coli e Klebsiella pneumoniae, e pacientes imunocomprometidos, como diabéticos, estão em maior risco de desenvolver complicações graves.
Abordagem Terapêutica
O tratamento do AHP é multimodal e deve ser adaptado à gravidade do caso, com o uso combinado de antibióticos, drenagem percutânea e intervenção cirúrgica, quando necessário. As diretrizes atuais propõem um algoritmo terapêutico baseado no tamanho e nas características dos abscessos.
1. Antibioticoterapia
A antibioticoterapia empírica deve ser iniciada imediatamente após o diagnóstico, visando cobertura para bactérias gram-negativas e anaeróbias. Ciprofloxacina ou cefixima combinadas com metronidazol são frequentemente utilizadas no manejo de abscessos hepáticos não complicados. A escolha do antibiótico deve ser ajustada conforme os resultados das culturas de sangue e de amostras do abscesso, garantindo uma abordagem personalizada.
2. Drenagem Percutânea
A drenagem percutânea, guiada por USG ou TC, é o tratamento de escolha para abscessos maiores que 3 cm e uniloculares (Tipo II). Esse método minimamente invasivo apresenta uma alta taxa de sucesso, próxima a 90%, sendo eficaz na maioria dos casos. No entanto, falhas podem ocorrer em abscessos multiloculados ou com conteúdo viscoso ou necrótico, situações em que a drenagem percutânea se torna inadequada, necessitando de intervenção cirúrgica.
3. Intervenção Cirúrgica
A cirurgia está indicada em abscessos multiloculados grandes (>3 cm, Tipo III), em abscessos que não respondem à drenagem percutânea ou na presença de complicações, como ruptura do abscesso. A cirurgia pode envolver drenagem minimamente invasiva ou ressecção hepática, dependendo da complexidade do abscesso e da experiência do cirurgião. Abscessos maiores que 10 cm apresentam maior risco de complicações, e nesses casos, a drenagem cirúrgica pode ser preferível. A laparotomia é recomendada em situações de peritonite ou quando o abscesso é de difícil acesso para drenagem percutânea.
4. Laparoscopia
A laparoscopia é uma alternativa minimamente invasiva à cirurgia aberta, indicada em abscessos uniloculares de tamanho moderado. Essa técnica oferece vantagens significativas, como menor tempo de internação e recuperação mais rápida, além de menor risco de complicações pós-operatórias.
Aplicação na Cirurgia Digestiva
O papel do cirurgião do aparelho digestivo é central no manejo dos abscessos hepáticos, especialmente em casos que requerem intervenção cirúrgica. A drenagem percutânea deve ser considerada a primeira linha de tratamento sempre que viável, mas o cirurgião deve estar preparado para realizar intervenções mais invasivas quando necessário. A laparoscopia tem demonstrado resultados promissores, reduzindo o tempo de internação e o risco de complicações. No Brasil, as infecções intra-abdominais complicadas são uma das principais causas de internação em emergências cirúrgicas, e o manejo adequado desses casos depende de uma sólida formação técnico-cirúrgica.
Algoritmo de Tratamento
Com base nas evidências disponíveis, um algoritmo de tratamento para o AHP pode ser delineado da seguinte forma:
- Abscessos pequenos (<3 cm, Tipo I): Tratamento com antibióticos isolados.
- Abscessos grandes uniloculares (>3 cm, Tipo II): Drenagem percutânea associada a antibioticoterapia.
- Abscessos grandes multiloculados (>3 cm, Tipo III): Intervenção cirúrgica.
Pontos-Chave
- Diagnóstico Precoce: O uso da TC com contraste é fundamental para o diagnóstico preciso do tamanho e da localização dos abscessos hepáticos, orientando a decisão terapêutica.
- Intervenção Cirúrgica: Abscessos multiloculados ou maiores que 5 cm frequentemente requerem intervenção cirúrgica, especialmente quando a drenagem percutânea falha.
- Abordagem Minimamente Invasiva: A laparoscopia oferece uma alternativa eficaz à cirurgia aberta, proporcionando uma recuperação mais rápida e com menor morbidade.
- Manejo Integral pelo Cirurgião Digestivo: O conhecimento técnico-cirúrgico é essencial para o manejo de abscessos hepáticos complexos, garantindo uma abordagem eficaz e personalizada.
Conclusão
O manejo do abscesso hepático piogênico exige uma abordagem multidisciplinar, sendo o cirurgião digestivo uma peça-chave no tratamento de casos complexos. A decisão entre drenagem percutânea e intervenção cirúrgica deve considerar múltiplos fatores, como o tamanho do abscesso, a resposta ao tratamento conservador e as condições clínicas do paciente. Como afirmou o Prof. Henri Bismuth: “Le traitement chirurgical n’est pas seulement une question de technique, mais de jugement. Le moment de l’intervention est aussi important que l’intervention elle-même.” Assim, o domínio técnico e a tomada de decisões precisas são fundamentais para o sucesso terapêutico no tratamento do AHP.
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#AbscessoHepatico #CirurgiaDigestiva #TratamentoAbscesso #DrenagemPercutanea #AparelhoDigestivo
Perioperative Fluid Management

In the setting of a normal ejection fraction, fluid is only administered when the expectation is that cardiac output will increase, and vasopressors are utilized if the aforementioned devices show fluid will not increase cardiac output. Excess fluid in certain general surgical cases can cause ileus and bowel edema, and in cardiac cases, it can cause hemodilution. Patients randomized to restricted and liberal fluid resuscitation strategies found a clear linear relationship between total fluids administered (and weight gain) and complications following colorectal surgery including pulmonary edema and tissue-healing complications. Further multiple studies exist demonstrating fewer complications with normovolemia than with liberal strategies of fluid resuscitation.
It must be understood that goal-directed therapy does, in no way, mean reduction in fluid administration. For some procedures, it may be necessary to administer more than anticipated fluid volumes (orthopedics), while for others, the opposite may be true (abdominal). Normovolemia is important to maintain perfusion without volume overload. Thus, the idea behind goaldirected therapy is to maintain zero fluid balance coupled with minimal weight gain or loss. Hypovolemia is associated with reduced circulating blood volume, decreased renal perfusion, altered coagulation, microcirculation compromise, and endothelial dysfunction, among other processes. Hypervolemia is associated with splanchnic edema, decreased pulmonary gas exchange secondary to pulmonary edema, impaired wound healing, anastomotic dehiscence, decreased mobility, altered coagulation, and endothelial dysfunction, amidst others processes.
Classroom: Perioperative Medicine
From a recent Cochrane review, there is no evidence that colloids are superior to crystalloid for resuscitation in patients. Therefore, crystalloid fluids should generally be the primary intravenous fluid during the perioperative course. In cardiac surgery, the utilization of 0.9% normal saline solution was associated with hyperchloremia and poor postoperative outcomes, including higher length of stay and increased mortality.118 Further, a more balanced crystalloid, such as Plasma-Lyte, was associated with improved outcomes in 22,851 surgical patients. In this study, there was a 2.05 odds ratio predictor of mortality with normal saline. Other complications such as acute kidney injury, gastrointestinal complications, major hemorrhage, and major infection were also increased in the group of
patients that were hyperchloremic after normal saline administration. Based on such evidence, it would seem prudent to proceed with a more balanced solution, such as PlasmaLyte, to reduce complications.
Principles of Surgical Resection of Hepatocellular Carcinoma
INTRODUCTION
There has been significant improvement in the perioperative results following liver resection, mainly due to techniques that help reduce blood loss during the operation. Extent of liver resection required in HCC for optimal oncologic results is still controversial. On this basis, the rationale for anatomically removing the entire segment or lobe bearing the tumor, would be to remove undetectable tumor metastases along with the primary tumor.
SIZE OF TUMOR VERSUS TUMOR FREE-MARGIN
Several retrospective studies and meta-analyses have shown that anatomical resections are safe in patients with HCC and liver dysfunction, and may offer a survival benefit. It should be noted, that most studies are biased, as non-anatomical resections are more commonly performed in patients with more advanced liver disease, which affects both recurrence and survival. It therefore remains unclear whether anatomical resections have a true long-term survival benefit in patients with HCC. Some authors have suggested that anatomical resections may provide a survival benefit in tumors between 2 and 5 cm. The rational is that smaller tumors rarely involve portal structures, and in larger tumors presence of macrovascular invasion and satellite nodules would offset the effect of aggressive surgical approach. Another important predictor of local recurrence is margin status. Generally, a tumor-free margin of 1 cm is considered necessary for optimal oncologic results. A prospective randomized trial on 169 patients with solitary HCC demonstrated that a resection margin aiming at 2 cm, safely decreased recurrence rate and improved long-term survival, when compared to a resection margin aiming at 1 cm. Therefore, wide resection margins of 2 cm is recommended, provided patient safety is not compromised.
THECNICAL ASPECTS
Intraoperative ultrasound (IOUS) is an extremely important tool when performing liver resections, specifically for patients with HCC and compromised liver function. IOUS allows for localization of the primary tumor, detection of additional tumors, satellite nodules, tumor thrombus, and define relationship with bilio-vascular structures within the liver. Finally, intraoperative US-guided injection of dye, such as methylene-blue, to portal branches can clearly define the margins of the segment supplied by the portal branch and facilitate safe anatomical resection.
The anterior approach to liver resection is a technique aimed at limiting tumor manipulation to avoid tumoral dissemination, decrease potential for blood loss caused by avulsion of hepatic veins, and decrease ischemia of the remnant liver caused by rotation of the hepatoduodenal ligament. This technique is described for large HCCs located in the right lobe, and was shown in a prospective, randomized trial to reduce frequency of massive bleeding, number of patients requiring blood transfusions, and improve overall survival in this setting. This approach can be challenging, and can be facilitated by the use of the hanging maneuver.

Multiple studies have demonstrated that blood loss and blood transfusion administration are significantly associated with both short-term perioperative, and long-term oncological results in patients undergoing resection for HCC. This has led surgeons to focus on limiting operative blood loss as a major objective in liver resection. Transfusion rates of <20 % are expected in most experienced liver surgery centers. Inflow occlusion, by the use of the Pringle Maneuver represents the most commonly performed method to limit blood loss. Cirrhotic patients can tolerate total clamping time of up to 90 min, and the benefit of reduced blood loss outweighs the risks of inflow occlusion, as long as ischemia periods of 15 min are separated by at least 5 min of reperfusion. Total ischemia time of above 120 min may be associated with postoperative liver dysfunction. Additional techniques aimed at reducing blood loss include total vascular isolation, by occluding the inferior vena cava (IVC) above and below the liver, however, the hemodynamic results of IVC occlusion may be significant, and this technique has a role mainly in tumors that are adjacent to the IVC or hepatic veins.

Anesthesiologists need to assure central venous pressure is low (below 5 mmHg) by limiting fluid administration, and use of diuretics, even at the expense 470 N. Lubezky et al. of low systemic pressure and use of inotropes. After completion of the resection, large amount of crystalloids can be administered to replenish losses during parenchymal dissection.
LAPAROSCOPIC RESECTIONS
Laparoscopic liver resections were shown to provide benefits of reduced surgical trauma, including a reduction in postoperative pain, incision-related morbidity, and shorten hospital stay. Some studies have demonstrated reduced operative bleeding with laparoscopy, attributed to the increased intra-abdominal pressure which reduces bleeding from the low-pressured hepatic veins. Additional potential benefits include a decrease in postoperative ascites and ascites-related wound complications, and fewer postoperative adhesions, which may be important in patients undergoing salvage liver transplantation. There has been a delay with the use of laparoscopy in the setting of liver cirrhosis, due to difficulties with hemostasis in the resection planes, and concerns for possible reduction of portal flow secondary to increased intraabdominal pressure. However, several recent studies have suggested that laparoscopic resection of HCC in patients with cirrhosis is safe and provides improved outcomes when compared to open resections.

Resections of small HCCs in anterior or left lateral segments are most amenable for laparoscopic resections. Larger resections, and resection of posterior-sector tumors are more challenging and should only be performed by very experienced surgeons. Long-term oncological outcomes of laparoscopic resections was shown to be equivalent to open resections on retrospective studies , but prospective studies are needed to confirm these findings. In recent years, robotic-assisted liver resections are being explored. Feasibility and safety of robotic-assisted surgery for HCC has been demonstrated in small non-randomized studies, but more experience is needed, and long-term oncologic results need to be studied, before widespread use of this technique will be recommended.
ALPPS: Associating Liver Partition with Portal vein ligation for Staged hepatectomy
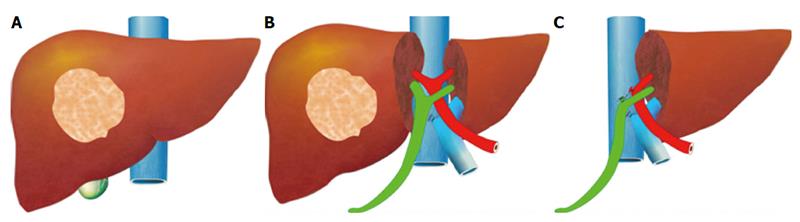
The pre-operative options for inducing atrophy of the resected part and hypertrophy of the FLR, mainly PVE, were described earlier. Associating Liver Partition with Portal vein ligation for Staged hepatectomy (ALPPS) is another surgical option aimed to induce rapid hypertrophy of the FLR in patients with HCC. This technique involves a 2-stage procedure. In the first stage splitting of the liver along the resection plane and ligation of the portal vein is performed, and in the second stage, performed at least 2 weeks following the first stage, completion of the resection is performed. Patient safety is a major concern, and some studies have reported increased morbidity and mortality with the procedure. Few reports exist of this procedure in the setting of liver cirrhosis. Currently, the role of ALPPS in the setting of HCC and liver dysfunction needs to be better delineated before more widespread use is recommended.
Management of gallbladder cancer

Gallbladder cancer is uncommon disease, although it is not rare. Indeed, gallbladder cancer is the fifth most common gastrointestinal cancer and the most common biliary tract cancer in the United States. The incidence is 1.2 per 100,000 persons per year. It has historically been considered as an incu-rable malignancy with a dismal prognosis due to its propensity for early in-vasion to liver and dissemination to lymph nodes and peritoneal surfaces. Patients with gallbladder cancer usually present in one of three ways: (1) advanced unresectable cancer; (2) detection of suspicious lesion preoperatively and resectable after staging work-up; (3) incidental finding of cancer during or after cholecystectomy for benign disease.
SURGICAL MANAGEMENT
Although, many studies have suggested improved survival in patients with early gallbladder cancer with radical surgery including en bloc resection of gallbladder fossa and regional lymphadenectomy, its role for those with advanced gallbladder cancer remains controversial. First, patients with more advanced disease often require more extensive resections than early stage tumors, and operative morbidity and mortality rates are higher. Second, the long-term outcomes after resection, in general, tend to be poorer; long-term survival after radical surgery has been reported only for patients with limited local and lymph node spread. Therefore, the indication of radical surgery should be limited to well-selected patients based on thorough preoperative and intra-operative staging and the extent of surgery should be determined based on the area of tumor involvement.
Surgical resection is warranted only for those who with locoregional disease without distant spread. Because of the limited sensitivity of current imaging modalities to detect metastatic lesions of gallbladder cancer, staging laparoscopy prior to proceeding to laparotomy is very useful to assess the
abdomen for evidence of discontinuous liver disease or peritoneal metastasis and to avoid unnecessary laparotomy. Weber et al. reported that 48% of patients with potentially resectable gallbladder cancer on preoperative imaging work-up were spared laparotomy by discovering unresectable disease by laparoscopy. Laparoscopic cholecystectomy should be avoided when a preoperative cancer is suspected because of the risk of violation of the plane between tumor and liver and the risk of port site seeding.
The goal of resection should always be complete extirpation with microscopic negative margins. Tumors beyond T2 are not cured by simple cholecystectomy and as with most of early gallbladder cancer, hepatic resection is always required. The extent of liver resection required depends upon whether involvement of major hepatic vessels, varies from segmental resection of segments IVb and V, at minimum to formal right hemihepatectomy or even right trisectionectomy. The right portal pedicle is at particular risk for advanced tumor located at the neck of gallbladder, and when such involvement is suspected, right hepatectomy is required. Bile duct resection and reconstruction is also required if tumor involved in bile duct. However, bile duct resection is associated with increased perioperative morbidity and it should be performed only if it is necessary to clear tumor; bile duct resection does not necessarily increase the lymph node yield.
Videos of Surgical Procedures

This page provides links to prerecorded webcasts of surgical procedures. These are actual operations performed at medical centers in the Brazil. Please note that you cannot send in questions by email, though the webcast may say that you can, because you are not seeing these videos live. The videos open in a second window. If you have a pop-up blocker, you will need to disable it to view the programs.
Videos of Surgical Procedures
Strangulation in GROIN HERNIAS
Importance
Declining Mortality Rates
In both the UK and the USA, the annual death rate due to inguinal and femoral hernias has significantly decreased over the past two to three decades. In the UK, deaths from these hernias declined by 22% to 55% between 1975 and 1990. Similarly, in the USA, the annual deaths per 100,000 population for patients with hernia and intestinal obstruction decreased from 5.1 in 1968 to 3.0 in 1988. For patients with obstructed inguinal hernias, 88% underwent surgery, with a remarkably low mortality rate of 0.05%. These improvements suggest that elective groin hernia surgery has played a crucial role in reducing overall mortality rates.
Elective Surgery and Strangulation Rates
Supporting this observation, the USA has lower rates of strangulation compared to the UK, possibly due to the threefold higher rate of elective hernia surgeries in the USA. Nevertheless, statistics indicate that the rate of elective hernia surgeries in the USA per 100,000 population decreased from 358 to 220 between 1975 and 1990, although this may be an artifact of data collection rather than a genuine decline.
Mortality Analysis from UK and Denmark Studies
During 1991–1992, the UK National Confidential Enquiry Into Perioperative Deaths investigated 210 deaths following inguinal hernia repair and 120 deaths following femoral hernia repair. This inquiry, which focuses on the quality of surgery, anesthesia, and perioperative care, found that many patients were elderly (45 were aged 80–89 years) and significantly infirm; 24 were ASA grade III and 21 ASA grade IV. The majority of postoperative mortality was attributed to preexisting cardiorespiratory issues.
A nationwide study in Denmark of 158 patients who died after acute groin hernia repair by Kjaergaard et al. also found that these patients were old (median age 83 years) and frail (>80% with significant comorbidity), with frequent delays in diagnosis and treatment. These findings highlight the need for high-quality care by experienced surgeons and anesthetists, especially for patients with high ASA grades.
Postoperative Care Recommendations
Postoperative care for these patients should occur in a high-dependency unit or intensive therapy unit. This might necessitate transferring selected patients to appropriate hospitals and facilities. Decisions about interventional surgery should be made in consultation with the relatives of extremely elderly, frail, or moribund patients, adopting a humane approach that may rule out surgery.
Emergency Admissions and Prioritization
Forty percent of patients with femoral hernias are admitted as emergency cases with strangulation or incarceration, while only 3% of patients with direct inguinal hernias present with strangulation. This disparity has implications for prioritizing patients on waiting lists when these hernias present electively in outpatient clinics.
Risk of Strangulation
A groin hernia is at its greatest risk of strangulation within three months of onset. For inguinal hernias, the cumulative probability of strangulation is 2.8% at three months after presentation, rising to 4.5% after two years. The risk is much higher for femoral hernias, with a 22% probability of strangulation at three months, rising to 45% at 21 months. Right-sided hernias have a higher strangulation rate than left-sided hernias, potentially due to anatomical differences in mesenteric attachment. The decline in hernia-related mortality in both the UK and USA underscores the importance of elective hernia surgery. Ensuring timely surgery, especially for high-risk femoral hernias, and providing high-quality perioperative care for elderly and frail patients are crucial steps in further reducing mortality and improving patient outcomes.
Evidence-Based Medicine
In a randomized trial, evaluating an expectative approach to minimally symptomatic inguinal hernias, Fitzgibbons et al. in the group of patients randomized to watchful waiting found a risk of an acute hernia episode of 1.8 in 1,000 patient years. In another trial, O’Dwyer and colleagues, randomizing patients with painless inguinal hernias to observation or operation, found two acute episodes in 80 patients randomized to observation. In both studies, a large percentage of patients randomized to nonoperative care were eventually operated due to symptoms. Neuhauser, who studied a population in Columbia where elective herniorrhaphy was virtually unobtainable, found an annual rate of strangulation of 0.29% for inguinal hernias.
Management of Strangulation
The diagnosis of hernias is primarily based on clinical symptoms and signs, supplemented by imaging studies when necessary. Pain at the hernia site is a constant symptom. In cases of obstruction with intestinal strangulation, patients may present with colicky abdominal pain, distension, vomiting, and constipation. Physical examination may reveal signs of dehydration, with or without central nervous system depression, especially in elderly patients with uremia, along with abdominal signs of intestinal obstruction.
Femoral hernias can be easily missed, particularly in obese women, making a thorough physical examination essential for an accurate diagnosis. However, physical examination alone is often insufficient to confirm the presence of a strangulated femoral hernia versus lymphadenopathy or a lymph node abscess. In such cases, urgent radiographic studies, such as ultrasound or CT scan, may be necessary.
The choice of incision depends on the type of hernia if the diagnosis is clear. When there is doubt, a half Pfannenstiel incision, 2 cm above the pubic ramus extending laterally, provides adequate access to all types of femoral or inguinal hernias. The fundus of the hernia sac is exposed, and an incision is made to assess the viability of its contents. If nonviability is detected, the transverse incision should be converted into a laparotomy incision, followed by the release of the constricting hernia ring, reduction of the sac’s contents, resection, and reanastomosis. Precautions must be taken to avoid contamination of the general peritoneal cavity by gangrenous bowel or intestinal contents.
In most cases, once the constriction of the hernia ring is released, circulation to the intestine is restored, and viability returns. The intestine that initially appears dusky or non-peristaltic may regain color with a short period of warming with damp packs. If viability is doubtful, resection should be performed. Resection rates are highest for femoral or recurrent inguinal hernias and lowest for simple inguinal hernias. Other organs, such as the bladder or omentum, should be resected as needed.
After peritoneal lavage and formal closure of the laparotomy incision, specific repair of the hernia should be performed. Prosthetic mesh should not be used in a contaminated operative field due to the high risk of wound infection. Hernia repair should follow the general principles of elective hernia repair. It is important to remember that in this predominantly frail and elderly patient group with a high postoperative mortality risk, the primary objective of the operation is to stop the vicious cycle of strangulation, with hernia repair being a secondary objective.
Key Point
The risk of an acute groin hernia episode is of particular relevance, when discussing indication for operation of painless or minimally symptomatic hernias. A sensible approach in groin hernias would be, in accordance with the guidelines from the European Hernia Society to advise a male patient, that the risk of an acute operation, with an easily reducible (“disappears when lying down”) inguinal hernia with little or no symptoms, is low and that the indication for operation in this instance is not absolute, but also inform, that usually the hernia after some time will cause symptoms, eventually leading to an operation. In contrast, female patients with a groin hernia, due to the high frequency of femoral hernias and a relatively high risk of acute hernia episodes, should usually be recommended an operation.
Minimally Invasive Approach to Choledocholithiasis
Introduction
The incidence of choledocholithiasis in patients undergoing cholecystectomy is estimated to be 10 %. The presence of common bile duct stones is associated with several known complications including cholangitis, gallstone pancreatitis, obstructive jaundice, and hepatic abscess. Making the diagnosis early and prompt management is crucial. Traditionally, when choledocholithiasis is identified with intraoperative cholangiography during the cholecystectomy, it has been managed surgically by open choledochotomy and place- ment of a T-tube. This open surgical approach has a morbidity rate of 10–15 %, mortality rate of <1 %, with a <6 % incidence of retained stones. Patients who fail endoscopic retrieval of CBD stones, as well as cases in which an endoscopic approach is not appropriate, should be explored surgically.
Clinical Manifestation
Acute obstruction of the bile duct by a stone causes a rapid distension of the biliary tree and activation of local pain fibers. Pain is the most common presenting symptom for choledocholithiasis and is localized to either the right upper quadrant or to the epigastrium. The obstruction will also cause bile stasis which is a risk factor for bacterial over- growth. The bacteria may originate from the duodenum or the stone itself. The combination of biliary obstruction and colo- nization of the biliary tree will lead to the development of fevers, the second most common presenting symptom of cho- ledocholithiasis. Biliary obstruction, if unrelieved, will lead to jaundice. When these three symptoms (pain, fever, and jaundice) are found simultaneously, it is known as Charcot’s triad. This triad suggests the diagnosis of acute ascending cholangitis, a potentially life-threatening condition. If not treated promptly, this can lead to hypotension and decreased metal status, both signs of severe sepsis. When combined with Charcot’s triad, this constellation of symptoms is commonly referred to as Reynolds pentad.
Laparoscopic common bile duct exploration
Laparoscopic common bile duct exploration (LCBDE) allows for single stage treatment of gallstone disease, reducing overall hospital stay, improving safety and cost-effectiveness when compared to the two-stage approach of ERCP and laparoscopic cholecystectomy. Bile duct clearance can be confirmed by direct visualization with a choledochoscope. But, before the advent of choledochoscope, bile duct clearance was uncertain, and blind instrumentation of the duct resulted in accentuated edema and inflammation. Due to advancement in instruments, optical magnification, and direct visualization, laparoscopic exploration of the CBD results in fewer traumas to the bile duct. This has led to an increasing tendency to close the duct primarily, reducing the need for placement of T-tubes. Still, laparoscopic bile duct exploration is being done in only a few centers. Apart from the need for special instruments, there is also a significant learning curve to acquire expertise to be able to perform a laparoscopic bile duct surgery.
Morbidity and mortality rates of laparoscopic exploration are comparable to ERCP (2–17 and 1–5 %), and there is no clear difference in primary success rates between the two approaches. However, the endoscopic approach may be preferable for elderly and frail patients, who are at higher risk with surgery. Patients older than 70–80 years of age have a 4–10 % mortality rate with open duct exploration. It may be as high as 20 % in elderly patients undergoing urgent procedures. In comparison, advanced age and comor- bidities do not have a significant impact on overall complication rates for ERCP. A success rate of over 90 % has been reported with laparoscopic CBD exploration. Availability of surgical expertise and appropriate equipment affect the success rate of laparoscopic exploration, as does the size, number of the CBD stones, as well as biliary anatomy. Over the years, laparoscopic exploration has become efficient, safe, and cost effective. Complications include CBD laceration, stricture formation, bile leak, abscess, pancreatitis, and retained stones.
In cases of failure of laparoscopic CBD exploration, a guidewire or stent can be passed through the cystic duct, common bile duct, and through the ampulla into the duodenum followed by cholecystectomy. This makes the identification and cannulation of the ampulla easier during the post- operative ERCP. Laparoscopic common bile duct exploration is traditionally performed through a transcystic or transductal approach. The transcystic approach is appropriate under certain circumstances. These include a small stone (<10 mm) located in the CBD, presence of small common bile duct (<6 mm), or if there is poor access to the common duct. The transductal approach is preferable in cases of large stones, stones in proximal ducts (hepatic ducts), large occluding stones in a large duct, presence of multiple stones, or if the cystic duct is small (<4 mm) or tortuous. Contraindications for laparoscopic approach include lack of training, and severe inflammation in the porta hepatis making the exploration difficult and risky.
Key Points
With advancement in imaging technology, laparoscopic and endoscopic techniques, management of common bile duct stone has changed drasti- cally in recent years. This has made the treatment of this condition safe and more efficient. Many options are now available to manage this condition, and any particular modality for treatment should be chosen carefully based on the patient related factors, institutional protocol, available expertise, resources, and cost-effectiveness.
Classroom: M.I.A. of Choledocholithiasis
Management of Complicated Appendicitis: Open or Laparoscopic Surgery?
Patients with acute appendicitis can present at different stages of the disease process, ranging from mild mucosal inflammation to frank perforation with abscess formation. The reported overall incidence of acute appendicitis varies with age, gender, and geographical differences. Interestingly, while the incidence of non-perforated appendicitis in the United States decreased between 1970 and 2004, no significant decline in the rate of perforated appendicitis was observed despite the increasng use of computed tomography (CT) and fewer negative appendectomies.
Of 32,683 appendectomies sampled from the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) hospitals between 2005 and 2008, 5,405 patients (16.5%) had a preoperative diagnosis of acute appendicitis with peritonitis/abscess.
The definition of complicated appendicitis varies slightly in the literature. Clinicopathological diagnoses (gangrenous, perforated, appendiceal abscess/phlegmon) of acute appendicitis are commonly used for its definition. Classically, patients at the extremes of age are more likely to present with complicated appendicitis. Similarly, pre-morbid conditions including diabetes and type of medical insurance are significantly associated with the risk of perforation.
The importance of early appendectomy has also been emphasized to prevent perforation of the appendix and the sub- sequent negative impact on patient outcomes. However, more recent meta-analysis data supports the safety of a relatively short (12–24 h) delay before appendectomy, which was not significantly associated with increased rate of complicated appendicitis. Teixeira et al. also showed that the time to appendectomy was not a significant risk factor for perforated appendicitis but did result in a significantly increased risk of surgical site infection.
The outcome of patients with complicated appendicitis is significantly worse than patients with uncomplicated appendicitis. A population-based study from Sweden showed that, in a risk-adjusted model, patients with perforated appendicitis were 2.34 times more likely to die after appendectomy than non- perforated appendicitis patients. Because of its higher mortality and morbidity in patients with complicated appendicitis, the management of complicated appendicitis has evolved significantly over the last few decades.
Open or Laparoscopic Surgery
Since the first laparoscopic appendectomy was described by Semm in 1983, multiple studies have compared operative time, complication rates, length of hospital stay, hospital cost, and other outcomes between open and laparoscopic appendectomy for acute appendicitis. The most recent Cochrane review included 67 studies showing that laparoscopic appendectomy was associated with a lower incidence of wound infection, reduced postoperative pain, shorter postoperative length of hospital stay, and faster recovery to daily activity. In contrast, reduced risk of intra-abdominal abscesses and shorter operative time were found as the advantages of open appendectomy.
Due to increased surgeon experience in uncomplicated appendicitis, laparoscopic appendectomy is more frequently attempted even in complicated appendicitis cases as an alternative approach to open appendectomy. Although the general surgical steps for complicated appendicitis are similar to those for uncomplicated appendicitis, the laparoscopic procedure can be more technically demanding. Therefore, conversion from laparoscopic appendectomy to open appendectomy can be expected.
Despite these concerns, the laparoscopic approach in patients with com- plicated appendicitis has been proven to be safe and comparable to open appendectomy. Retrospective studies using a large database in the United States uniformly showed more favorable clinical outcomes (mortality, morbidity, length of hospital stay, readmission rate) and hospital costs in patients who underwent laparoscopic appendectomy when compared to open appendectomy. The real risk of developing an intra- abdominal abscess after laparoscopic appendectomy remains unclear. A meta-analysis by Markides et al. found no significant difference in the intra-abdominal abscess rate between laparoscopic and open appendectomy for complicated appendicitis, whereas Ingraham et al. showed a higher likelihood of developing an organ-space surgical site infection in patients undergoing laparoscopic appendectomy.
Causas de conversão da VIDEOCOLECISTECTOMIA

Atualmente, a colecistectomia laparoscópica é a abordagem preferida para o tratamento da litíase biliar, representando cerca de 90% dos procedimentos realizados, uma marca alcançada nos Estados Unidos em 1992. A popularidade dessa técnica se deve a suas vantagens evidentes: menos dor no pós-operatório, recuperação mais rápida, redução dos dias de trabalho perdidos e menor tempo de hospitalização. Apesar de ser considerada o padrão-ouro na cirurgia biliar, a colecistectomia laparoscópica não está isenta de desafios. Entre 2% e 15% dos casos podem exigir a conversão para cirurgia convencional. Os motivos mais comuns para essa conversão incluem dificuldades na identificação da anatomia, suspeita de lesão da árvore biliar e controle de sangramentos. Identificar os fatores que contribuem para uma maior taxa de conversão é essencial para a equipe cirúrgica. Isso não apenas permite uma avaliação mais precisa da complexidade do procedimento, mas também ajuda na preparação do paciente para possíveis riscos e na mobilização de cirurgiões mais experientes quando necessário. Em um cenário onde a precisão e a segurança são cruciais, a compreensão dos desafios e a preparação adequada podem fazer toda a diferença no resultado da cirurgia.
Relacionados ao Paciente: 1. Obesidade (IMC > 35), 2. Sexo Masculino, 3. Idade > 65 anos, 4. Diabetes Mellitus e 5. ASA > 2.
Relacionadas a Doença: 1. Colecistite Aguda, 2. Líquido Pericolecístico, 3. Pós – CPRE, 4. Síndrome de Mirizzi e 5. Edema da parede da vesícula > 5 mm.
Relacionadas a Cirurgia: 1. Hemorragia, 2. Aderências firmes, 3. Anatomia obscura, 4. Fístulas internas e 5. Cirurgia abdominal prévia.
Laparoscopic Surgery for Morbid Obesity
The morbid obesity epidemic continues to spread throughout industrialized nations. It is a condition with a heterogeneous etiology, including genetic, psychosocial, and environmental factors. Prevention methods have currently been unable to halt the further spread of this disease. Obesity has been linked to increased healthcare costs, common physiologic derangements, reduced quality of life, and increased overall mortality. More than one third of adults and almost 17% of children in the United States are obese.
Medical therapy that can cause sustained significant weight loss may be years away. Bariatric surgery, when combined with a multidisciplinary team, continues to be the only proven method to achieve sustained weight loss in most patients. Bariatric procedures modify gastrointestinal anatomy and, in some cases, enteric hormone release to reduce caloric intake, reduce absorption, and alter metabolism to achieve weight loss. Currently, the three most common bariatric operations in the United States are Roux-en-Y gastric bypass, adjustable gastric band, and the vertical sleeve gastrectomy.
Como funciona o GRAMPEADOR INTESTINAL ?
A Busca pela Segurança e Eficiência na Cirurgia Moderna
Introdução O objetivo primordial de qualquer cirurgião, ao iniciar uma intervenção, é garantir que o procedimento seja seguro e eficiente. A “regra de ouro” da cirurgia moderna permanece clara: o ato operatório deve ser o mais breve possível, gerando o menor trauma tecidual e restaurando a função do órgão. O resultado esperado é sempre a minimização das intercorrências no pós-operatório. Embora a cirurgia atual atinja esses objetivos de forma bastante satisfatória, as complicações relacionadas às suturas e à cicatrização ainda são desafios presentes na rotina hospitalar.
A Evolução Histórica e os Desafios da Cicatrização A confiabilidade das suturas gastrointestinais, por exemplo, é uma conquista relativamente recente. Foi apenas no final do século XIX que a compreensão dos princípios básicos da cicatrização tecidual permitiu avanços significativos nesta área. Sabemos hoje que o sucesso do reparo tecidual não depende apenas da técnica empregada, mas é uma equação complexa que envolve o paciente e a área operada. Diversos fatores podem retardar ou prejudicar drasticamente a cicatrização, entre eles:
-
Isquemia (falta de suprimento sanguíneo);
-
Edema (inchaço excessivo);
-
Infecção local ou sistêmica;
-
Desnutrição do paciente.
O Fator Humano e o Surgimento da Tecnologia Existe uma variável crítica na cirurgia: a habilidade manual. Naturalmente, há uma variação na destreza e na técnica entre diferentes cirurgiões. Foi justamente essa disparidade que motivou o desenvolvimento de novos dispositivos cirúrgicos. O objetivo da inovação tecnológica na cirurgia é superar as diferenças individuais, funcionando como um equalizador. Ao utilizar dispositivos que padronizam etapas críticas (como as suturas mecânicas), permite-se que técnicas complexas sejam executadas adequadamente, independente de quem opera.
Conclusão Para que a medicina avance, uma técnica não pode depender apenas do talento de poucos. Ela deve ser reproduzível de forma confiável pelo maior número possível de cirurgiões. Somente através dessa padronização e do auxílio tecnológico é que os resultados cirúrgicos podem ser amplamente adotados, reconhecidos como eficazes e, acima de tudo, seguros para o paciente.
Laparoscopic Colorectal Surgery
With the introduction of laparoscopic colectomy nearly 20 years ago, a relatively slow adoption of laparoscopic colorectal surgery into surgical practice has taken place. It is estimated that 10% to 25% of all colorectal resections are performed utilizing laparoscopy. The persistent steep learning curve, the lack of high-volume colorectal surgery by general surgeons (who perform the bulk of colonic resection in the United States), and the modest advantages reported are but a few of the reasons that the percentage of laparoscopic colorectal procedures has not dramatically risen. With the publication of several large, prospective randomized trials for colon cancer, along with the interest in single-port surgery and natural orifice surgery, there appears to be a renewed interest in minimally invasive procedures for the colon and rectum. This chapter will provide an overview of these issues and offer a current assessment of the common diseases to which minimally invasive techniques have been applied.
Learning Curve
Numerous previous studies have evaluated the learning curve involved in laparoscopic colectomy. It is estimated by conventional laparoscopic techniques that the learning curve for laparoscopic colectomy is at least 20 cases but more likely 50 cases. The need to work in multiple quadrants of the abdomen, the need for a skilled laparoscopic assistant, and the lack of yearly volume has kept the learning curve relatively steep. The surgeon may also need to work in reverse angles to the camera. All of these combined add to the complexity of the procedure and result in the need to perform a number of cases before the surgeon and surgical team become proficient. More recent publications have suggested the learning curve is more than 20 cases. In a prospective randomized study of colorectal cancer in the United Kingdom, the CLASICC trial, surgeons had to perform at least 20 laparoscopic resections before they were allowed to enter the study. The study began in July 1996 and was completed in July 2002. Despite the surgeons’ prior experience, the rate of conversion dropped from 38% to 16% over the course of the study, suggesting that a minimum of 20 cases may not be enough to overcome the learning curve. In the COLOR trial from Europe, another prospective randomized study for colon cancer that required a prerequisite experience in laparoscopic colon resection before surgeons could enter patients in the study, surgeon and hospital volume were directly related to a number of operative and postoperative outcomes. The median operative time for high-volume hospitals (>10 cases/year) was 188 minutes, compared to 241 minutes for low-volume hospitals (<5 cases/year); likewise, conversion rates were 9% versus 24% for the two groups. High-volume groups also had more lymph nodes in the resected specimens, fewer complications, and shortened hospital stays. These two relatively recent multicenter studies suggest that the learning curve is clearly greater than 20 cases and that surgeons need to perform a minimum yearly number of procedures to maintain their skills.
Outcomes
There may not be another area in recent surgical history that has been more heavily scrutinized than laparoscopic colorectal surgery. The plethora of accumulated data allows a careful assessment of all outcome measures for nearly every colorectal disease and procedure. In comparison to conventional colorectal surgery, the benefits of laparoscopy for colorectal procedures compared to open techniques include a reduction in postoperative ileus, postoperative pain, and a concomitant reduction in the need for analgesics; an earlier tolerance of diet; a shortened hospital stay; a quicker resumption of normal activities; improved cosmesis; and possibly preservation of immune function. This is offset by a prolongation in operative time, the cost of laparoscopic equipment, and the learning curve for these technically challenging procedures. When reporting the outcomes of laparoscopic colectomy, a natural selection bias applies when comparing conventional and laparoscopic cases. The most complex cases are generally not suitable for a laparoscopic approach and therefore are performed via an open approach. Also, in many series the results of the successfully completed laparoscopic cases are compared to conventional cases, and the cases converted from a laparoscopic to a conventional procedure may be analyzed separately. Few studies, with the exception of the larger prospective randomized studies, leave the converted cases in the laparoscopic group as part of the “intention to treat” laparoscopic group. This clearly introduces selection bias.
Although the results of prospective randomized trials are available for almost every disease process requiring colorectal resection, the majority of studies of laparoscopic colectomy are retrospective case-control series or noncomparative reports. The conclusions regarding patient outcomes must therefore come from the repetitiveness of the results rather than the superiority of the study design. For any one study, the evidence may be weak; but collectively, because of the reproducibility of results by a large number of institutions, even with different operative techniques and postoperative management parameters, the preponderance of evidence favors a minimally invasive approach with respect to postoperative outcomes.
Operative Time
Nearly all the comparative studies provide information regarding operative times. The definition of the operative time may vary with each series, and there may be different groups of surgeons performing the laparoscopic and conventional procedures. With the exception of a few reports, nearly all studies demonstrated a prolonged operative time associated with laparoscopic procedures. In prospective randomized trials, the procedure was roughly 40 to 60 minutes longer in the laparoscopic groups. As the surgeon and team gain experience with laparoscopic colectomy, the operating times do reliably fall, but rarely do they return to the comparable time for a conventional approach.
Return of Bowel Activity and Resumption of Diet
Reduction in postoperative ileus is one of the proposed major advantages of minimally invasive surgery. Nearly all of the retrospective and prospective studies comparing open and laparoscopic colectomy have shown a statistically significant reduction in the time to passage of flatus and stool. Most series demonstrate a 1- to 2-day advantage for the laparoscopic group. Whether the reduction of ileus relates to less bowel manipulation or less intestinal exposure to air during minimally invasive surgery remains unknown. With the reduction in postoperative ileus, the tolerance by the patient of both liquids and solid foods is quicker following laparoscopic resection. The time to resumption of diet varies from 2 days to 7 days, but in the majority of comparative studies, this is still 1 to 2 days sooner than in patients undergoing conventional surgery. Again, the physician and patient were not blinded in nearly all studies, which may have altered patient expectations. However, the overwhelming reproducible data reported in both retrospective and prospective studies of laparoscopic procedures does likely favor a reduction of postoperative ileus and tolerance of liquid and solid diets.
Postoperative Pain
To measure postoperative pain, a variety of assessments have been performed to demonstrate a significant reduction in pain following minimally invasive surgery; some studies utilize an analog pain scale, and others measure narcotic requirements. Physician bias and psychologic conditioning of patients may interfere with the evaluation of postoperative pain. There are also cultural variations in the response to pain. Three of the early prospective randomized trials have evaluated pain postoperatively, and all three have found a reduction in narcotic requirements in patients undergoing laparoscopic colectomy. In the COST study, the need both for intravenous and oral analgesics was less in patients undergoing successfully completed laparoscopic resections. Numerous other nonrandomized studies have shown a reduction in postoperative pain and narcotic usage.
Length of Stay
The quicker resolution of ileus, earlier resumption of diet, and reduced postoperative pain has resulted in a shortened length of stay for patients after laparoscopic resection when compared to traditional procedures. Recovery after conventional surgery has also been shortened, but in the absence of minimally invasive techniques, it would seem unlikely that the length of stay could be further reduced. In nearly all comparative studies, the length of hospitalization was 1 to 6 days less for the laparoscopic group. Although psychological conditioning of the patient cannot be helped and likely has a desirable effect, the benefits of minimally invasive procedures on the overall length of stay cannot be discounted. The benefit, however, is more likely a 1 to 2 day advantage only. The more recent introduction of clinical pathways, both in conventional and laparoscopic surgery, has also narrowed the gap but appears to be more reliable in patients undergoing a minimally invasive approach.
Hospital Costs
One of the disadvantages of laparoscopy is the higher cost related to longer operative times and increased expenditures in disposable equipment. Whether the total cost of the hospitalization (operative and hospital costs) is higher following laparoscopic colectomy is debatable. A case-control study from the Mayo Clinic looked at total costs following laparoscopic and open ileocolic resection for Crohn’s disease (CD). In this study, 66 patients underwent laparoscopic or conventional ileocolic resection and were well matched. Patients in the laparoscopic group had less postoperative pain, tolerated a regular diet 1 to 2 days sooner, and had a shorter length of stay (4 vs. 7 days). In the cost analysis, despite higher operative costs, the overall mean cost was $3273 less in the laparoscopic group. The procedures were performed by different groups of surgeons at the institution, and although the surgeons may have introduced biases, this study was undertaken during the current era of cost containment, in which all physicians are encouraged to reduce hospital stays. The results are similar for elective sigmoid diverticular resection with a mean cost savings of $700 to $800. Clearly, if operative times and equipment expenditure are minimized, the overall cost of a laparoscopic resection should not exceed a conventional approach.
Tratamento Laparoscópica da DIVERTICULITE AGUDA
O aumento da prevalência de doença diverticular fez o seu adequado manuseio mais um assunto de debate constante. Especialmente para os casos de diverticulite, progresso considerável tem sido feito em termos de diagnóstico e tratamento. Diagnóstico apropriado em TC e técnicas intervencionistas são agora amplamente disponíveis, bem como agentes antimicrobianos eficazes. Finalmente, como a ressecção cirúrgica do cólon envolvido é a única maneira de erradicar definitivamente essa condição, a colectomia eletiva laparoscópica surgiu como uma opção segura e interessante entre as opções de tratamento. Embora tenha sido recentemente contestada sobre a sua progressão, a história natural da diverticulite é assumida como sendo a de recorrência ao longo do tempo, pelo menos, em um terço dos pacientes. O medo das complicações desta doença benigna e prevalente tem motivado sociedades médicas e cirúrgicas para produzir orientações e consensos sobre o assunto. A mortalidade geralmente vem de sepse recorrente e/ou operações de emergência para casos mais complicados. Como resultado, o procedimento cirúrgico mais realizado, a sigmoidectomia eletiva, é normalmente indicada para todos os casos complicados e muitos dos não-complicados. A abordagem laparoscópica para a colectomia esquerda tem evoluído e condições seguras são oferecidas aos pacientes, quando realizado por cirurgiões experientes em laparoscopia.
Lesão de Vias Biliares na Colecistectomia: Prevenção e Tratamento
A via laparoscópica tem sido reconhecida como padrão de excelência para a colecistectomias. Phillipe Mouret foi quem primeiro a realizou em 1987, mas outros procedimentos já haviam sido realizados por laparoscopia e foram descritos por ginecologistas. Desenvolvida no final da década de 80 e início dos anos 90, a videolaparoscopia mudou os conceitos de acesso cirúrgico e campo operatório, introduzindo a concepção de “cirurgia minimamente invasiva”.A colecistectomia é um dos procedimentos cirúrgicos mais realizados no mundo. Com o advento da videolaparoscopia, tornou-se uma cirurgia menos traumática, mais estética, com períodos mais curtos de internação. Em contrapartida, observou-se o aumento da incidência de lesões de via biliar extra-hepática quando comparado ao procedimento aberto, fato preocupante devido à morbidade elevada desse tipo de lesão, cuja mortalidade não é desprezível.