Cuidados Perioperatórios na Cirurgia Laparoscópica
Otimizando a Fisiologia e Acelerando a Recuperação no Aparelho Digestivo
Autor: Prof. Dr. Ozimo Gama Categoria: Cuidados Perioperatórios / Cirurgia Laparoscópica / Cirurgia do Aparelho Digestivo Tempo de Leitura: 10 minutos
Introdução
A cirurgia laparoscópica revolucionou a especialidade do aparelho digestivo ao atenuar o trauma cirúrgico, reduzir a resposta inflamatória sistêmica e proporcionar uma recuperação estética e funcional superior. No entanto, para o estudante de medicina, o residente de cirurgia geral e o pós-graduando, é imperativo compreender que incisões milimétricas não isentam o paciente da resposta endócrino-metabólica ao trauma. No Brasil, o Sistema Único de Saúde (SUS) realiza centenas de milhares de procedimentos videolaparoscópicos anualmente, desde colecistectomias até gastrectomias oncológicas complexas. Para maximizar os benefícios da via minimamente invasiva, a excelência no centro cirúrgico deve ser invariavelmente acompanhada por um manejo perioperatório de vanguarda. A transição dos cuidados dogmáticos do passado para protocolos modernos baseados em evidências — como o ERAS (Enhanced Recovery After Surgery) e o Projeto ACERTO (Aceleração da Recuperação Total Pós-Operatória), amplamente adotado e validado em território nacional — é o que define o cirurgião contemporâneo de alta performance.
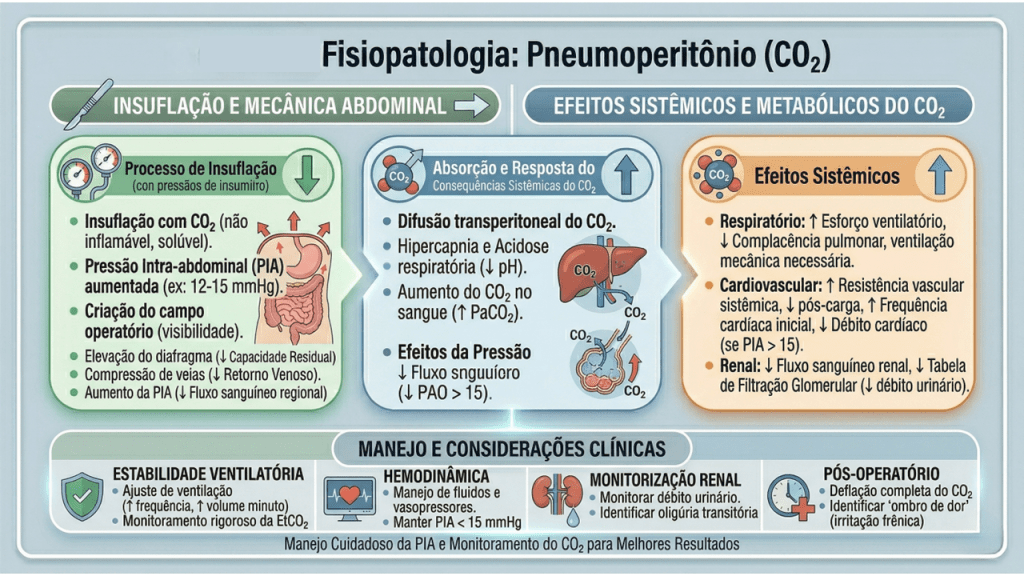
A Fisiologia do Pneumoperitônio e a Quebra de Paradigmas
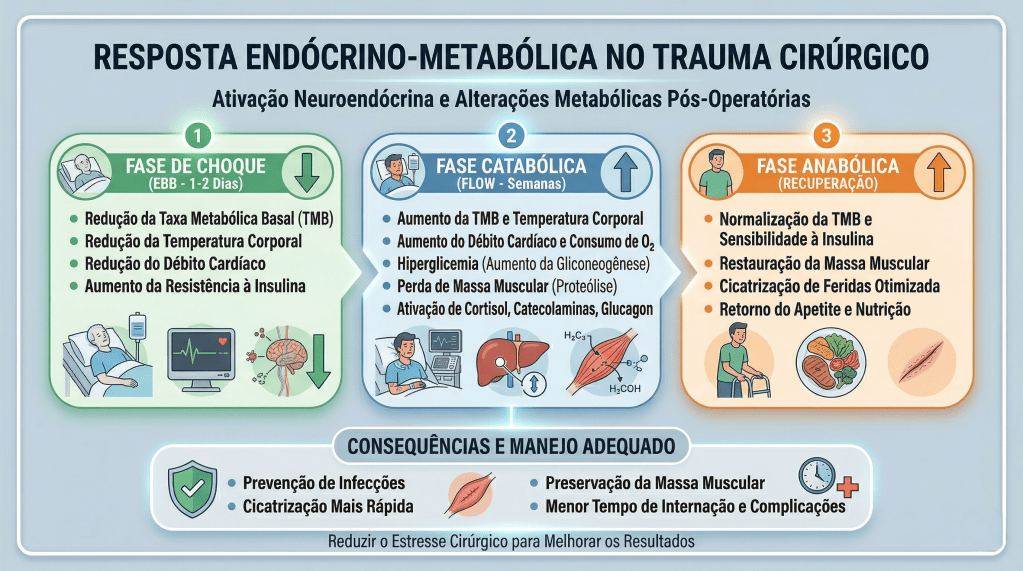
O manejo perioperatório na laparoscopia exige um profundo respeito pelas alterações fisiológicas induzidas pelo pneumoperitônio com dióxido de carbono (CO2). A insuflação abdominal eleva a pressão intra-abdominal (usualmente entre 12 e 15 mmHg), acarretando compressão da veia cava inferior, diminuição do retorno venoso, aumento da resistência vascular sistêmica e elevação das pressões das vias aéreas com risco de hipercapnia. A equipe cirúrgico-anestésica deve atuar em sintonia para mitigar estes efeitos.
Sob a ótica dos protocolos de aceleração da recuperação, o manejo perioperatório é dividido em três fases cruciais, onde dogmas seculares foram cientificamente derrubados:
- Pré-Operatório (O Fim do Jejum Prolongado): O jejum absoluto de 8 a 12 horas está proscrito. Evidências robustas, corroboradas por dados do Projeto ACERTO no Brasil, demonstram que a oferta de líquidos claros enriquecidos com carboidratos até 2 horas antes da indução anestésica reduz a resistência insulínica pós-operatória, minimiza a perda de massa magra e diminui a ansiedade do paciente.
- Intraoperatório (A Terapia Hídrica Guiada por Metas): A hiper-hidratação venosa, outrora comum, resulta em edema esplâncnico e retardo do esvaziamento gástrico. A fluidoterapia deve ser restritiva ou guiada por metas (avaliando a variação do volume sistólico), garantindo perfusão tecidual sem encharcar o paciente. A analgesia deve ser preemptiva e multimodal, poupadora de opioides.
- Pós-Operatório (Mobilização e Realimentação): O repouso no leito prolongado favorece o tromboembolismo venoso (TEV) e a atelectasia pulmonar. O paciente deve ser encorajado a deambular no mesmo dia da operação.
Aplicação na Cirurgia Digestiva
Na cirurgia do trato gastrointestinal, a aplicação destes cuidados perioperatórios altera drasticamente os desfechos e o tempo de internação:
- Preparo Mecânico do Cólon (PMC): Em cirurgias colorretais laparoscópicas, o uso rotineiro do PMC foi abandonado por grande parte das diretrizes, pois causa desidratação, distúrbios hidroeletrolíticos e não reduz as taxas de deiscência anastomótica ou infecção de sítio cirúrgico (exceto quando associado a antibióticos orais específicos em cirurgias de reto baixo).
- Sondas e Drenos: A utilização profilática de sondas nasogástricas (SNG) e drenos cavitários não encontra mais respaldo na literatura para procedimentos eletivos sem complicações. A SNG prolonga o íleo adinâmico e predispõe a infecções respiratórias. O dreno deve ser uma exceção fundamentada, não a regra.
- Realimentação Precoce: O íleo pós-operatório é inerente à manipulação das alças, mas a dieta por via oral iniciada precocemente (muitas vezes nas primeiras 24 horas) estimula o reflexo gastrocólico, protege a barreira mucosa intestinal contra a translocação bacteriana e acelera a alta hospitalar. Estatísticas brasileiras apontam que a realimentação precoce em ressecções intestinais laparoscópicas reduz o tempo de internação em até 2 a 3 dias.
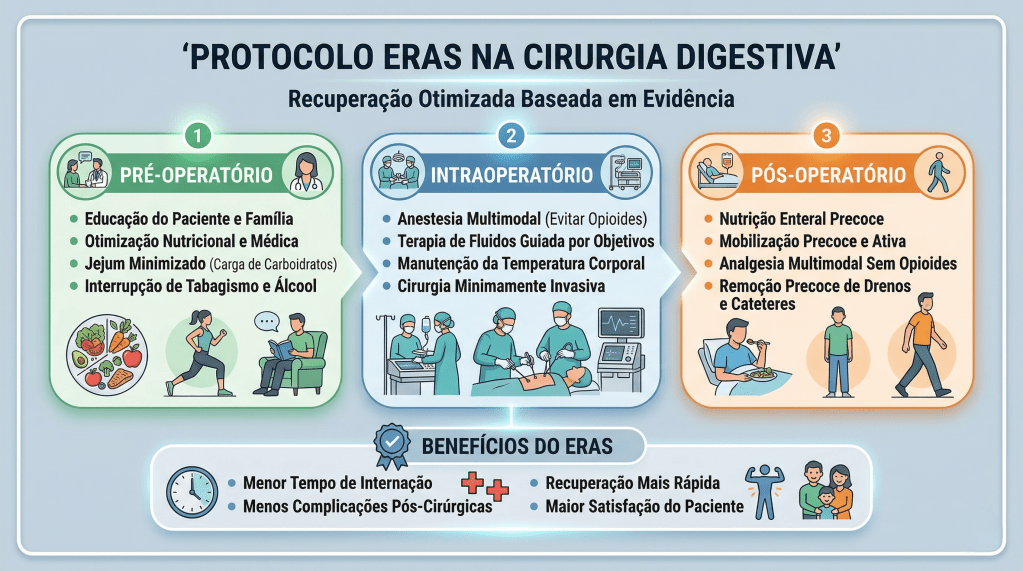
Pontos-Chave
- Avaliação Fisiológica: O pneumoperitônio exige monitorização hemodinâmica rigorosa devido às alterações cardiocirculatórias e ventilatórias.
- Jejum Abreviado: Utilização de maltodextrina a 12,5% até 2 horas antes da cirurgia é uma prática segura e metabolicamente protetora.
- Analgesia Opioid-Sparing: Uso de infiltração dos portais com anestésicos locais, anti-inflamatórios e bloqueios regionais (ex: TAP block) para evitar os efeitos eméticos e obstipantes dos opioides.
- Tromboprofilaxia: O uso de meias elásticas de compressão graduada, compressores pneumáticos intermitentes e heparina de baixo peso molecular é mandatório, dada a estase venosa promovida pela pressão intra-abdominal elevada.
- Racionalização de Dispositivos: Evitar o uso sistemático e prolongado de sondas vesicais de demora (SVD), SNG e drenos cavitários.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A destreza na dissecção laparoscópica e a precisão das anastomoses intracavitárias perdem parte de seu brilho se o paciente for submetido a um estresse fisiológico não gerenciado. O cirurgião do aparelho digestivo moderno deve compreender que o seu papel transcende a mecânica operatória; ele é o arquiteto da recuperação do paciente. Adoção de protocolos de cuidados perioperatórios como o ACERTO exige uma mudança de cultura institucional e o alinhamento de toda a equipe multidisciplinar (cirurgião, anestesiologista, enfermagem, nutrição e fisioterapia). Ao unirmos a mínima agressão tecidual da videolaparoscopia à máxima otimização metabólica do perioperatório, entregamos não apenas uma cirurgia segura, mas a restituição rápida e plena da qualidade de vida do nosso paciente.
“O principal objetivo da cirurgia moderna não é apenas curar a doença anatômica, mas suprimir a resposta ao trauma cirúrgico, proporcionando uma recuperação sem dor e sem risco.” — Henrik Kehlet, cirurgião dinamarquês e idealizador do protocolo ERAS.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Anatomia Cirúrgica da Região Inguinal
1.0 Introdução
A anatomia da região inguinal é notoriamente complexa, representando um desafio constante até mesmo para os cirurgiões mais experientes. Como afirmado de forma sucinta por Skandalakis et al. no início de seu trabalho seminal, a anatomia da região inguinal é “enigmática e confusa” (enigmatic and confusing). Este artigo tem como objetivo desmistificar essa complexidade, focando nas estruturas anatômicas cruciais e em suas relações, a fim de fornecer um guia claro e prático para estudantes de medicina, residentes e cirurgiões que atuam no reparo de hérnias inguinais. Uma compreensão precisa desta topografia não é apenas um exercício acadêmico, mas a fundação para a excelência cirúrgica.
2.0 Desvendando as Estruturas Anatômicas
2.1 As Paredes do Canal Inguinal
O canal inguinal é uma passagem oblíqua através das camadas inferiores da parede abdominal. Suas paredes são formadas por diversas estruturas musculoaponeuróticas, como visto na ilustração anatômica. Os limites são:
• Parede Anterior: Formada pela aponeurose do músculo oblíquo externo e, em sua porção lateral, reforçada pelo músculo oblíquo interno.
• Parede Posterior: Considerada a mais importante do ponto de vista anatômico e cirúrgico, é composta primariamente pela fusão da aponeurose do músculo transverso do abdome com a fáscia transversal. Em 23-25% dos indivíduos, é formada apenas pela fáscia transversal.
• Parede Superior (Teto): Composta pelas fibras arqueadas dos músculos oblíquo interno e transverso do abdome e suas aponeuroses.
• Parede Inferior (Assoalho): Constituída pelo ligamento inguinal e, mais medialmente, pelo ligamento lacunar.
A reconstrução desta parede posterior, especialmente quando deficiente, é o objetivo central de qualquer herniorrafia inguinal bem-sucedida.
2.2 As Camadas Musculoaponeuróticas Fundamentais
A parede abdominal é composta por três camadas de músculos planos. De superficial para profundo, temos o oblíquo externo, o oblíquo interno e o transverso do abdome. Embora todos sejam importantes, o músculo transverso do abdome assume um papel de destaque. De acordo com McVay, ele é a camada mais importante da parede abdominal. A integridade deste músculo e sua aponeurose é o que, em última análise, previne a formação de hérnias. Consequentemente, a mobilização e o uso da aponeurose deste músculo são manobras essenciais para um reparo anatômico e duradouro.
2.3 A Crucial Fáscia Transversal
A fáscia transversal é a lâmina de tecido conjuntivo que reveste a superfície interna da musculatura da cavidade abdominal. Ela é uma estrutura fundamental, contribuindo para a formação da fáscia espermática interna no funículo espermático e, como já mencionado, compondo a parede posterior do canal inguinal. Sua integridade é vital para a contenção visceral. Portanto, a dissecção e o reparo desta fáscia não são opcionais, mas sim o alicerce fundamental de uma herniorrafia sem tensão.
2.4 O “Mito” do Tendão Conjunto
O termo “tendão conjunto” é amplamente utilizado, mas sua existência anatômica como uma fusão distinta das fibras inferiores do oblíquo interno e do transverso do abdome é extremamente rara. A evidência anatômica mostra que essa configuração é mais uma exceção do que uma regra.
• Hollinshead considerou sua existência em apenas 5% dos corpos.
• Condon relatou uma incidência de apenas 3%.
• McVay acreditava que o “tendão conjunto” era um artefato de dissecação.
Por essa razão, o conceito de “área conjunta” (conjoined area) é um termo mais preciso e clinicamente relevante. Esta área refere-se à região medial da parede posterior, que inclui estruturas como o ligamento de Henle, a aponeurose do transverso do abdome e a borda lateral da bainha do reto, que são de fato utilizadas no reparo de hérnias.
2.5 Pontos de Referência Cirúrgicos Essenciais
A navegação segura na região inguinal exige o reconhecimento de marcos anatômicos chave:
• Triângulo de Hesselbach: É o local onde ocorre a maioria das hérnias inguinais diretas. Suas bordas, na definição moderna, são: os vasos epigástricos inferiores (borda superior), a bainha do músculo reto abdominal (borda medial) e o ligamento inguinal (borda lateroinferior).
• Ligamento de Cooper: Estrutura robusta formada pelo periósteo do ramo superior do púbis. É reforçado por múltiplas camadas, incluindo contribuições da fáscia transversal, do trato iliopúbico e da aponeurose do transverso do abdome. É um ponto de ancoragem fundamental em diversas técnicas de herniorrafia, como a de McVay.
• Trato Iliopúbico: Uma banda aponeurótica densa que se estende do arco iliopectíneo até o ramo superior do púbis, formando a margem inferior da camada musculoaponeurótica profunda. Condon o encontrou presente em 98% das dissecações. É uma estrutura profunda, paralela ao ligamento inguinal (que é mais superficial), e um marco essencial em reparos laparoscópicos e abertos.
3.0 Aplicação na Cirurgia Digestiva
3.1 Fisiologia e Mecanismos de Contenção da Hérnia
A integridade do canal inguinal normal é mantida por dois mecanismos fisiológicos dinâmicos:
1. Ação Esfincteriana: A fáscia transversal forma um “sling” (alça) ao redor do anel inguinal interno. A contração do músculo transverso do abdome tensiona essa alça, fechando o anel sob a borda muscular do oblíquo interno.
2. Ação de Obturador (“Shutter Action”): Durante a contração da musculatura abdominal (por exemplo, ao tossir), o arco aponeurótico do músculo transverso do abdome se move em direção ao trato iliopúbico e ao ligamento inguinal, reforçando e obliterando qualquer fraqueza na parede posterior.
3.2 Implicações Diretas na Herniorrafia
O conhecimento anatômico se traduz diretamente na estratégia cirúrgica. Como mencionado, a parede posterior é anatomicamente deficiente em 23-25% dos indivíduos, carecendo do suporte da aponeurose do músculo transverso. Nesses casos, a fáscia transversal torna-se a única barreira contra a herniação direta. O objetivo de uma herniorrafia eficaz não é apenas fechar um defeito, mas reconstruir a dinâmica da parede posterior. A compreensão precisa de quais estruturas estão presentes, quais são deficientes e quais podem ser utilizadas para um reparo sólido e sem tensão é o que diferencia um procedimento bem-sucedido de uma recidiva.
4.0 Pontos-Chave
1. A Complexidade é Real: A anatomia inguinal é intrincada e repleta de controvérsias terminológicas. Um estudo contínuo é necessário.
2. O Transverso do Abdome é Rei: A integridade do músculo transverso do abdome e sua aponeurose é o fator mais crítico na fisiopatologia e prevenção das hérnias inguinais.
3. O Tendão Conjunto é Raro: O “tendão conjunto” clássico é um achado raro (3-5%). O termo “área conjunta” é mais apropriado para descrever as estruturas utilizadas cirurgicamente.
4. A Parede Posterior é a Chave: O sucesso do reparo de uma hérnia inguinal depende fundamentalmente da reconstrução de uma parede posterior forte e sem tensão, utilizando as estruturas anatômicas corretas.
5. Conheça Seus Marcos: O Ligamento de Cooper, o Trato Iliopúbico e o Triângulo de Hesselbach são referências anatômicas indispensáveis para o cirurgião realizar um procedimento seguro e eficaz.
5.0 Conclusões Aplicadas à Prática Clínica
Apesar de séculos de estudo e de incontáveis procedimentos cirúrgicos, a região inguinal permanece um desafio anatômico. As variações individuais e as controvérsias na nomenclatura exigem do cirurgião mais do que a memorização de diagramas; exigem um conhecimento profundo e funcional. Esta compreensão detalhada da anatomia é a ferramenta mais poderosa que possuímos para avaliar defeitos herniários, escolher a técnica de reparo mais adequada e, por fim, garantir resultados cirúrgicos superiores e duradouros para nossos pacientes.
#AnatomiaCirurgica #HerniaInguinal #CirurgiaGeral #ResidenciaCirurgia #AparelhoDigestivo
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Conteúdo Extra
Lista Completa de Epônimos Anatômicos da Região Inguinal
Esta lista abrange as estruturas clássicas nomeadas em homenagem a anatomistas e cirurgiões, fundamentais para a compreensão cirúrgica da região inguiocrural.
1. Ligamentos e Estruturas Fibrosas
Os elementos estruturais que formam as paredes e o assoalho da região.
- Ligamento de Poupart
- Nome Moderno: Ligamento Inguinal.
- Quem foi: François Poupart (cirurgião francês, séc. XVII).
- Descrição: A borda inferior enrolada da aponeurose do músculo oblíquo externo. Estende-se da EIAS ao tubérculo púbico, formando o “assoalho” do canal inguinal.
- Ligamento de Gimbernat
- Nome Moderno: Ligamento Lacunar.
- Quem foi: Antonio de Gimbernat y Arbós (cirurgião espanhol, séc. XVIII).
- Descrição: Extensão posterior e triangular do ligamento de Poupart que se fixa no osso púbico. Forma a borda medial cortante do anel femoral.
- Ligamento de Cooper
- Nome Moderno: Ligamento Pectíneo.
- Quem foi: Astley Cooper (cirurgião inglês, séc. XIX).
- Descrição: Espessamento forte do periósteo (membrana do osso) ao longo do ramo superior do púbis. É o principal ponto de ancoragem óssea para telas em cirurgias complexas.
- Trato de Thomson
- Nome Moderno: Trato Iliopúbico.
- Quem foi: James Thomson (anatomista escocês, séc. XIX).
- Descrição: Espessamento da fáscia transversal profundo e paralelo ao ligamento inguinal. É o divisor profundo entre o canal inguinal (acima) e o canal femoral (abaixo).
- Ligamento de Henle
- Nome Moderno: Expansão lateral da bainha do músculo reto abdominal (frequentemente associado à foice inguinal/tendão conjunto).
- Quem foi: Friedrich Gustav Jakob Henle (anatomista e patologista alemão, séc. XIX).
- Descrição: Uma estrutura aponeurótica na parede posterior do canal inguinal, logo na borda do músculo reto abdominal. Ele forma o limite medial do Trígono de Hesselbach. Sua presença é variável; quando forte, protege contra hérnias diretas; quando ausente ou fraco, predispõe a elas.
- Ligamento de Colles (ou Ligamento Reflexo)
- Nome Moderno: Ligamento Inguinal Reflexo.
- Quem foi: Abraham Colles (cirurgião e anatomista irlandês, séc. XIX).
- Descrição: É uma camada de fibras derivadas da aponeurose do oblíquo externo do lado oposto, que cruzam a linha média e se inserem na crista púbica e na linha pectínea do lado afetado. Ele passa atrás do anel inguinal superficial e na frente do tendão conjunto, reforçando a porção medial do canal inguinal.
2. Fáscias e Camadas
As camadas de cobertura da parede abdominal anterior.
- Fáscia de Scarpa
- Nome Moderno: Lâmina membranácea da tela subcutânea.
- Quem foi: Antonio Scarpa (anatomista italiano, séc. XVIII/XIX).
- Descrição: A camada membranosa mais profunda da gordura abaixo da pele.
- Fáscia de Camper
- Nome Moderno: Lâmina adiposa da tela subcutânea.
- Quem foi: Petrus Camper (anatomista holandês, séc. XVIII).
- Descrição: A camada de gordura superficial e fofa logo abaixo da pele.
- Fáscia de Colles
- Nome Moderno: Fáscia membranácea do períneo.
- Quem foi: Abraham Colles (cirurgião irlandês, séc. XIX).
- Descrição: A continuação da Fáscia de Scarpa na região do períneo e escroto/lábios.
3. Espaços e Áreas Definidas
Marcos geográficos críticos para cirurgia e diagnóstico.
- Trígono de Hesselbach
- Nome Moderno: Trígono Inguinal.
- Quem foi: Franz Kaspar Hesselbach (anatomista alemão, séc. XIX).
- Descrição: A área triangular na parede posterior da virilha onde ocorrem as hérnias diretas. Delimitado pelos vasos epigástricos inferiores, a borda do músculo reto (Ligamento de Henle) e o ligamento inguinal.
- Orifício de Fruchaud
- Nome Moderno: Orifício Miopectíneo.
- Quem foi: Henri Fruchaud (cirurgião francês, séc. XX).
- Descrição: O “grande buraco” conceitual na parede abdominal que engloba todas as potenciais hérnias da virilha (direta, indireta e femoral), que deve ser coberto pela tela.
- Espaço de Bogros
- Nome Moderno: Espaço Retroinguinal (pré-peritoneal lateral).
- Quem foi: Jean-Annet de Bogros (anatomista francês, séc. XIX).
- Descrição: O espaço virtual atrás da fáscia transversal e à frente do peritônio, onde as telas são colocadas nas cirurgias laparoscópicas (TEP/TAPP).
- Espaço de Retzius
- Nome Moderno: Espaço Retropúbico (pré-vesical).
- Quem foi: Anders Retzius (anatomista sueco, séc. XIX).
- Descrição: O espaço atrás do osso púbico e à frente da bexiga, medial ao espaço de Bogros.
4. Linfonodos e Vasos
- Gânglio de Cloquet (ou Rosenmüller)
- Nome Moderno: Linfonodo inguinal profundo proximal.
- Quem foram: Jules Germain Cloquet (francês) e Johann Christian Rosenmüller (alemão).
- Descrição: O linfonodo localizado dentro do canal femoral, que pode simular uma hérnia se estiver inflamado.
- Corona Mortis (Termo clássico, não epônimo de pessoa)
- Tradução: Coroa da Morte.
- Descrição: Uma variação vascular perigosa (conexão entre vasos ilíacos externos e internos) que passa sobre o osso púbico, com risco de hemorragia grave se lesada durante a cirurgia.
A Doutrina da Visão Crítica de Segurança (CVS) na Colecistectomia Laparoscópica
Fundamentos, Técnica e a Era da Inteligência Artificial
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 11 minutos)
Introdução
A colecistectomia laparoscópica é, indiscutivelmente, uma das intervenções cirúrgicas mais realizadas no planeta. Contudo, apesar de sua consolidação como um procedimento seguro, as lesões iatrogênicas da via biliar principal continuam a assombrar os blocos operatórios, configurando-se como uma das complicações mais temidas, onerosas e medicolegais da cirurgia moderna. Com o advento da laparoscopia no final dos anos 1980, observou-se um pico alarmante na incidência dessas lesões. Em resposta a esta crise global de segurança, o Dr. Steven Strasberg propôs, em 1995, a Visão Crítica de Segurança (CVS – Critical View of Safety). Mais do que uma simples manobra, o CVS transformou-se no dogma central da cirurgia biliar. Este artigo disseca o racional anatômico, a sistematização técnica rigorosa e as aplicações contemporâneas do CVS na prática do cirurgião do aparelho digestivo.
A Patogenia da Lesão e o Racional Anatômico
Para prevenir um erro, é imperativo compreender a sua gênese. A literatura clássica é taxativa: a esmagadora maioria das lesões biliares graves não ocorre por imperícia motora, mas por um erro de percepção visual conhecido como “Misidentification” (identificação errônea). A armadilha mais letal é a técnica da “Visão Infundibular”. Em cenários de inflamação aguda, fibrose ou tração inadequada, o ducto colédoco ou o ducto hepático comum alinham-se paralelamente à vesícula, mimetizando o ducto cístico. O cirurgião se convence de forma ilusória e perigosa de que “tem certeza de estar vendo o cístico”. Essa confiança excessiva, aliada à dissecção apressada, resulta na transecção trágica da via biliar principal. O CVS foi desenhado cirurgicamente para aniquilar esta ilusão de ótica.
Os Três Critérios Obrigatórios do CVS
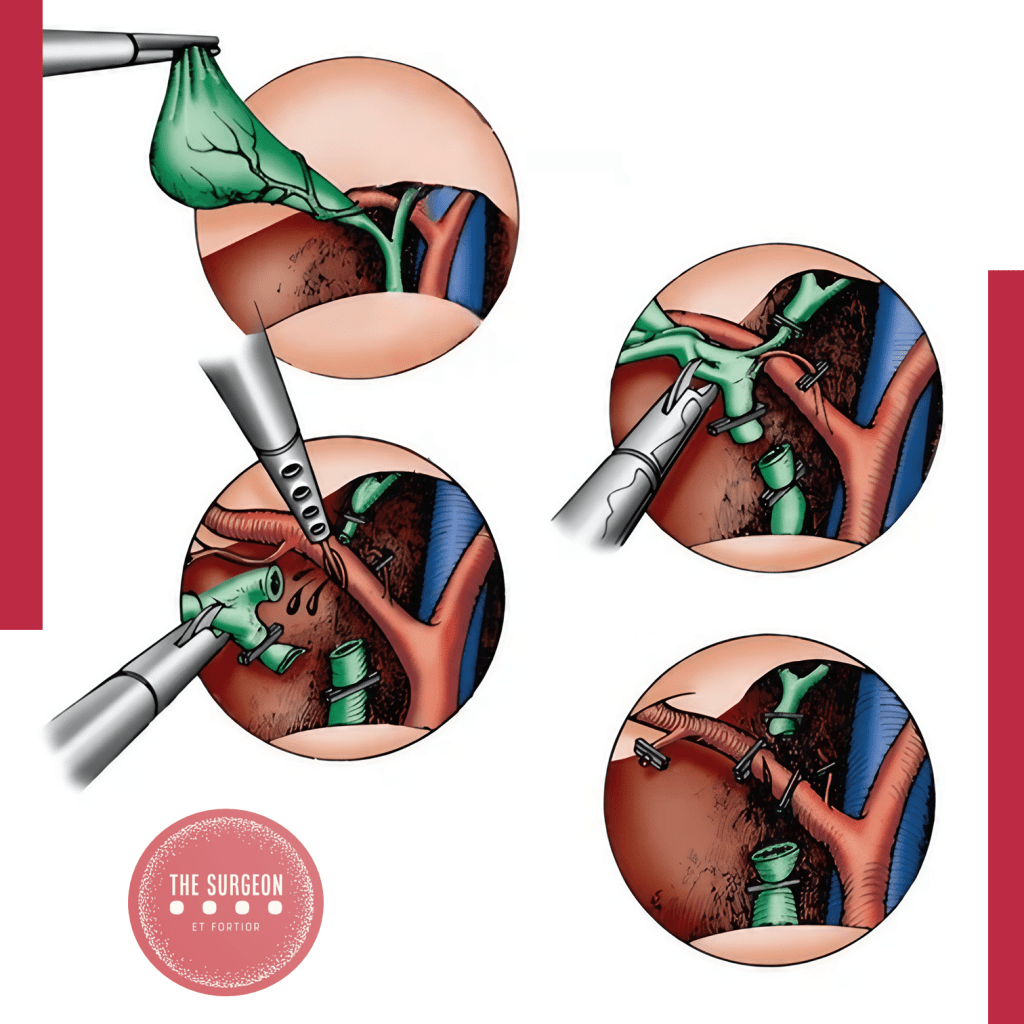
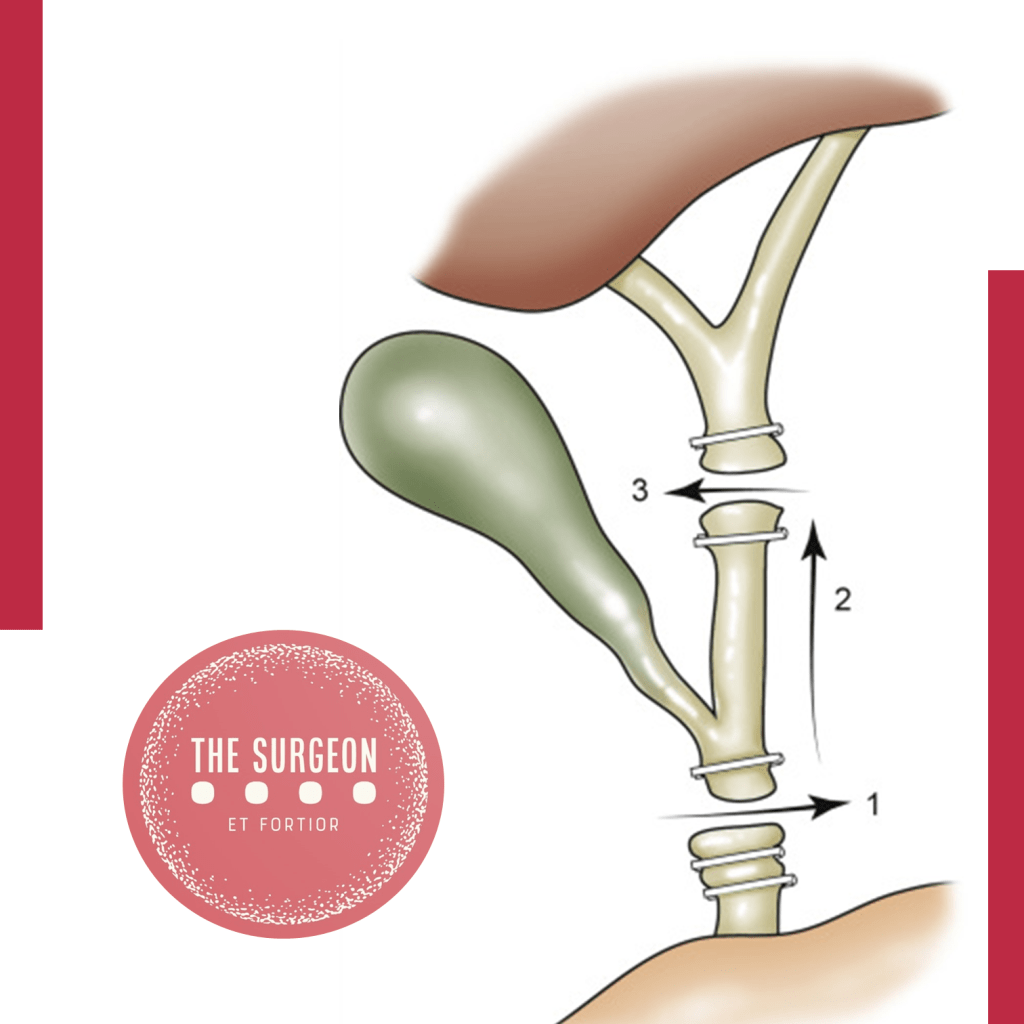
O CVS não é uma suposição; é um método de prova anatômica objetiva. Para declarar que o CVS foi atingido, todos os três critérios abaixo devem ser cumpridos simultaneamente antes do disparo de qualquer clipe ou grampeador:
- Dissecção Completa do Triângulo Hepatocístico: Toda a gordura e o tecido fibroso devem ser removidos do triângulo formado pelo ducto cístico, ducto hepático comum e borda hepática. A exposição deve ser visualizada nas faces anterior e posterior. Atenção: o ducto hepático comum não precisa (e não deve) ser dissecado ativamente, mas o triângulo deve estar limpo.
- Separação Parcial da Vesícula do Leito Hepático: O terço inferior da vesícula biliar (infundíbulo) deve ser completamente descolado da placa cística (leito hepático). Este passo, muitas vezes negligenciado, é o que garante que as estruturas dissecadas estão efetivamente a entrar na vesícula, e não a passar por ela.
- Apenas Duas Estruturas Adentrando a Vesícula: Exclusivamente um ducto (ducto cístico) e uma artéria (artéria cística) devem ser vistos penetrando a vesícula. Ambos devem estar completamente isolados e identificáveis circunferencialmente (visão de 360 graus).
Robustez, Saídas e a Era Moderna
A eficácia do CVS é incontestável. Em grandes séries publicadas, como a de Yegiyants et al. (3.042 casos) e Avgerinos et al. (998 casos) em que o CVS foi estritamente aplicado, a taxa de lesão biliar grave foi de zero.
Cenários Adversos e Técnicas de Saída (Bail-Out Procedures)
O verdadeiro valor do CVS revela-se na dificuldade. Quando há inflamação severa, colecistite crônica esclerosante ou um Triângulo de Calot “obliterado” (frozen Calot), atingir o CVS torna-se perigoso ou impossível.
Para o cirurgião maduro, a incapacidade de completar o CVS não é um obstáculo a ser forçado, mas um alarme biológico de perigo iminente. Nestes casos, impõe-se a adoção imediata de técnicas de saída:
- Colecistectomia Subtotal (Fenestrada ou Reconstituída).
- Conversão precoce para cirurgia aberta.
- Realização de Colangiografia Intraoperatória.
Documentação Médico-Legal
A diretriz cirúrgica contemporânea exige que o CVS seja documentado. O cirurgião deve registrar uma fotografia ou um breve vídeo intraoperatório que demonstre inequivocamente a exposição anterior e posterior das estruturas isoladas. A inclusão desta prova no prontuário médico eletrônico protege o paciente contra complicações e o cirurgião em contextos litigiosos.
Cirurgia Robótica e Inteligência Artificial (IA)
Os princípios de Strasberg são universais e imutáveis, independentemente do instrumental. Na Cirurgia Robótica, a visão 3D ampliada de altíssima definição e a estabilidade das pinças facilitam o cumprimento escrupuloso dos três critérios. As perspetivas futuras são ainda mais fascinantes: algoritmos de Inteligência Artificial (IA) e visão computacional estão a ser treinados para reconhecer e validar os critérios do CVS em tempo real no monitor cirúrgico, funcionando como um checklist virtual que impede a clipagem até que o software confirme a segurança anatômica.
Pontos-Chave para a Prática Diária
- Prova, não Suposição: O CVS transforma um julgamento subjetivo (“parece o cístico”) numa evidência anatômica irrefutável.
- O Risco da Visão Infundibular: Confundir um infundíbulo dilatado com o ducto cístico é a principal via para a lesão do colédoco.
- Descolar para Comprovar: Não basta limpar o triângulo de Calot; a separação do terço inferior da vesícula do leito hepático (Critério 2) é essencial.
- Pausa Obrigatória: A identificação das duas únicas estruturas deve ser um “momento estático”, validado verbalmente por toda a equipe antes da transecção.
Conclusões Aplicadas
O Critical View of Safety transcendeu a classificação de “técnica cirúrgica” para se consolidar como o padrão-ouro e a obrigação ética universal na colecistectomia laparoscópica. A sua adoção obrigatória nos programas de residência médica aniquila a cultura do achismo visual, substituindo-a pelo rigor científico. Ao reconhecer que o principal inimigo do cirurgião biliar é a sua própria mente (na forma de ilusão de ótica), o CVS fornece a armadura necessária para proteger o doente de morbidades devastadoras. Na cirurgia do aparelho digestivo, a paciência não é apenas uma virtude; é uma técnica de salvamento.A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.”
“A identificação deve ser um momento estático, após completa dissecação, e não um julgamento apressado.” — Dr. Steven Strasberg, cirurgião e idealizador da Visão Crítica de Segurança.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Apendicectomia Laparoscópica: Truques e Dicas para uma Execução Segura e Eficiente
Introdução
A apendicectomia laparoscópica consolidou-se como o padrão-ouro no tratamento da apendicite aguda, sobretudo em centros com acesso à tecnologia minimamente invasiva. Com benefícios amplamente documentados — como menor dor pós-operatória, redução do tempo de internação e menor incidência de infecção de ferida —, a técnica exige, contudo, treinamento e atenção a detalhes técnicos. Este artigo oferece truques e dicas práticas que podem otimizar a performance do cirurgião, reduzir complicações e melhorar a curva de aprendizado da equipe assistente.
1. Posicionamento do Paciente e da Equipe
- Decúbito dorsal com leve Trendelenburg e rotação à esquerda facilita a exposição do quadrante inferior direito.
- Fixe o braço direito do paciente ao corpo para permitir amplo espaço de movimentação do cirurgião.
- Cirurgião à esquerda do paciente, assistente ao lado da perna esquerda, monitor preferencialmente à direita ou à cabeceira, na linha dos ombros.
Dica: Ajuste fino da inclinação da mesa pode ser decisivo para deslocar alças e expor o ceco sem necessidade de manobras agressivas.
2. Posicionamento dos Trocárteres
- Um padrão eficiente inclui:
- Trocárter de 10 mm umbilical (ótica).
- Trocárter de 5 mm em hipogástrio (instrumentação dominante).
- Trocárter de 5 mm em flanco esquerdo (tração e dissecção).
Truque: Em pacientes obesos, insira o trocárter ótico com cuidado em ângulo oblíquo para evitar desinserção do pneumoperitônio e garantir estabilidade.
3. Estratégias de Exposição
- Identifique o teniae coli do ceco e siga até a base do apêndice.
- Use pinça atraumática para tração superior do apêndice, expondo sua base.
- Em casos de aderências, libere-as com energia monopolar delicada ou tesoura, evitando avulsões inadvertidas.
Dica de ouro: Evite “lutar” contra aderências retrocecais. Mude o plano, reposicione a câmera, varie o ângulo de dissecção. Tempo gasto com exposição segura evita complicações graves.
4. Controle do Pedículo e Secção Apendicular
- O método mais utilizado é o uso de duas ligaduras com endoloop ou clips poliméricos (Hem-o-lok®), seguido de secção entre eles.
- Alternativamente, grampeadores laparoscópicos podem ser usados, especialmente em apêndices friáveis ou bases espessadas.
Truque técnico: Em apêndices muito inflamados, realize a ligadura mais distal antes da proximal, para reduzir o risco de ruptura ou vazamento ao manipular a base.
5. Retirada e Proteção da Cavidade
- Retire o apêndice com saco cirúrgico sempre que possível, evitando contaminação do trajeto do trocárter.
- Irrigue abundantemente a loja apendicular se houver peritonite localizada ou pus livre.
- Se necessário, coloque dreno tubular por 24 a 48 horas.
Dica prática: Em caso de dúvida quanto à integridade da base, deixe um fragmento do ceco visível e documente o aspecto final com imagem.
6. Situações Especiais
- Apêndice retrocecal: requer liberação ampla da reflexão lateral direita do cólon.
- Apendicite perfurada com abscesso: considere drenagem inicial guiada por imagem e apendicectomia em intervalo.
- Apendicite gestacional: ideal até o segundo trimestre. Atenção ao deslocamento anatômico do apêndice.
Truque anatômico: Em gestantes ou crianças, a mobilidade intestinal pode mascarar a localização clássica. Reforce a busca sistemática do apêndice pela convergência das teníases do ceco.
Conclusão
A apendicectomia laparoscópica é uma cirurgia segura, eficaz e que continua evoluindo com a incorporação de técnicas assistidas por imagem, navegação e inteligência artificial. No entanto, sua execução requer atenção a detalhes aparentemente simples, que fazem toda a diferença nos desfechos clínicos. O domínio dos truques e dicas técnicas aqui apresentados contribui significativamente para uma prática cirúrgica mais segura, eficiente e baseada em excelência técnica.
“A simplicidade técnica não dispensa o rigor; é justamente na cirurgia simples que se exige a perfeição.” — René Leriche
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Inteligência Artificial & Apendicite Aguda
Uso da Inteligência Artificial Generativa no Diagnóstico, Tratamento Cirúrgico e Avaliação de Prognóstico da Apendicite Aguda
Introdução
A apendicite aguda continua sendo a causa mais comum de abdome agudo cirúrgico no mundo, com incidência estimada de 100 a 150 casos por 100.000 habitantes ao ano. No Brasil, representa uma das principais causas de internação cirúrgica no SUS. Apesar de amplamente estudada e com tratamento bem estabelecido, a apendicite aguda ainda apresenta desafios clínicos, como o diagnóstico precoce, a decisão operatória e a estratificação de risco de complicações. Nesse contexto, a Inteligência Artificial Generativa (IAG) emerge como uma tecnologia disruptiva com potencial transformador na abordagem cirúrgica dessa condição.
Diagnóstico com Apoio de IA Generativa
O diagnóstico precoce da apendicite aguda depende da correlação de dados clínicos, laboratoriais e de imagem. A IAG, treinada com grandes volumes de dados de prontuários eletrônicos, imagens de tomografia e ultrassonografias, pode sintetizar essas informações em tempo real para gerar hipóteses diagnósticas com acurácia comparável — e, em alguns casos, superior — à avaliação humana isolada. Modelos generativos baseados em deep learning, como os transformers, são capazes de identificar padrões sutis de apresentação clínica atípica, sobretudo em populações vulneráveis, como crianças pequenas, idosos e gestantes. Um exemplo prático é o uso de sistemas que combinam texto livre do prontuário com imagens de abdome para gerar relatórios diagnósticos automatizados e sugerir scores clínicos (como Alvarado ou AIR score) com ajuste probabilístico personalizado, auxiliando o cirurgião na tomada de decisão.
Tratamento Cirúrgico Guiado por IA
A cirurgia laparoscópica é o padrão ouro no tratamento da apendicite aguda, mas a escolha do momento cirúrgico e a abordagem ideal ainda são decisões dependentes da experiência do cirurgião e das condições clínicas do paciente. A IAG pode ser integrada a sistemas de suporte intraoperatório, auxiliando em navegação cirúrgica assistida por imagem, sugestão de condutas intraoperatórias com base em bancos de dados operatórios, e até mesmo em treinamento em simulações realistas de apendicectomia via realidade aumentada e modelos generativos de anatomia personalizada.
Em cenários de medicina robótica, já há estudos utilizando IA generativa para gerar “scripts” de procedimentos otimizados e oferecer feedback em tempo real com base em dados históricos de performance técnica — promovendo cirurgias mais seguras, rápidas e com menor taxa de complicações.
Avaliação Prognóstica com IA Generativa
A estratificação de risco no pós-operatório é outro campo onde a IAG pode oferecer avanços concretos. A partir de redes neurais treinadas em desfechos de milhares de pacientes com apendicite, esses modelos podem prever probabilidades de complicações como abscesso residual, íleo paralítico ou necessidade de reabordagem, considerando variáveis clínicas, laboratoriais, intraoperatórias e demográficas. Além disso, ferramentas baseadas em IA podem gerar relatórios individualizados de prognóstico funcional, tempo estimado de retorno às atividades e risco de reinternação, permitindo ao cirurgião personalizar o plano de alta, acompanhamento ambulatorial e orientações ao paciente com maior precisão.
Pontos-Chave
- A IA generativa tem capacidade de analisar simultaneamente dados clínicos, laboratoriais e de imagem para melhorar o diagnóstico precoce da apendicite aguda.
- Pode ser integrada à decisão cirúrgica, tanto no pré quanto no intraoperatório, por meio de simulação, navegação assistida e análise de performance técnica.
- Modelos preditivos baseados em IA oferecem ferramentas objetivas para prognóstico pós-operatório personalizado, otimizando o seguimento do paciente.
- O uso de IAG deve ser encarado como ferramenta complementar ao raciocínio clínico do cirurgião, jamais como substituto da experiência e do julgamento médico.
- O avanço dessas tecnologias requer formação crítica e ética dos futuros cirurgiões, capacitando-os para liderar a incorporação segura e eficaz dessas ferramentas na prática assistencial.
Conclusão
A incorporação da Inteligência Artificial Generativa na cirurgia do aparelho digestivo representa um novo paradigma na medicina personalizada, baseada em dados e centrada no paciente. No caso da apendicite aguda — uma das mais frequentes emergências cirúrgicas —, o uso responsável e crítico da IAG pode aprimorar significativamente os pilares do cuidado: diagnóstico, decisão operatória e prognóstico. O cirurgião do futuro será, acima de tudo, um líder clínico capaz de integrar inteligência humana e artificial com sabedoria e discernimento ético.
“A tecnologia não substitui o cirurgião. Ela amplia sua visão, mas é o julgamento humano que decide a incisão.” — Sir John Black, ex-presidente do Royal College of Surgeons
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#InteligenciaArtificialNaMedicina #CirurgiaDoAparelhoDigestivo #ApendiciteAguda #DiagnósticoComIA #CirurgiaGuiadaPorDados
Prevenção das Aderências Pós-Operatórias
Introdução
As aderências pós-operatórias continuam sendo uma das principais complicações após cirurgias abdominais, impactando diretamente a qualidade de vida dos pacientes e os custos para o sistema de saúde. No contexto brasileiro, estima-se que até 35% das reoperações abdominais sejam causadas por aderências, contribuindo para obstruções intestinais, infertilidade, dor abdominal crônica e aumento do tempo cirúrgico em procedimentos subsequentes. A profilaxia eficaz destas formações fibrosas representa, portanto, um imperativo técnico e ético para o cirurgião do aparelho digestivo.
Desenvolvimento
A fisiopatologia das aderências envolve uma resposta inflamatória exacerbada da cavidade peritoneal à manipulação cirúrgica, à presença de corpos estranhos e à isquemia tecidual. Tais estímulos promovem a deposição de fibrina, que, na ausência de fibrinólise adequada, culmina na formação de pontes fibrosas entre órgãos e estruturas adjacentes.
As medidas preventivas devem ser iniciadas desde o planejamento operatório. O uso de técnicas cirúrgicas meticulosas é um dos pilares da prevenção. Manipular delicadamente os tecidos, evitar dissecções desnecessárias, garantir hemostasia rigorosa e eliminar corpos estranhos (como talco das luvas ou gaze fragmentada) são princípios fundamentais. Além disso, a preferência pela abordagem laparoscópica sempre que possível se justifica, já que estudos demonstram que ela reduz significativamente a formação de aderências em comparação com a cirurgia aberta.
Outra estratégia complementar é o uso de barreiras mecânicas antiaderentes bioabsorvíveis. Produtos como a membrana de ácido hialurônico/carboximetilcelulose (Seprafilm®) e a solução de icodextrina 4% (Adept®) têm evidências robustas de eficácia, sendo recomendados especialmente em procedimentos ginecológicos e digestivos de médio e grande porte. Estudos recentes também apontam novas perspectivas com o uso de polímeros zwitteriônicos, capazes de prevenir completamente aderências em modelos animais, embora ainda sem validação clínica em humanos.
Aplicação na Cirurgia Digestiva
Na prática da cirurgia do aparelho digestivo, especialmente em procedimentos como colectomias, gastrectomias, ressecções intestinais e cirurgias hepatopancreatobiliares, a prevenção de aderências é estratégica para evitar complicações precoces e tardias. A obstrução intestinal por bridas, por exemplo, representa até 20% das admissões de emergência cirúrgica abdominal nos hospitais terciários brasileiros.
A adoção da laparoscopia como via de acesso padrão, sempre que factível, deve ser incentivada como política institucional. Do mesmo modo, o emprego racional de barreiras antiaderentes é indicado especialmente em pacientes com histórico de múltiplas cirurgias abdominais, doenças inflamatórias intestinais ou em situações em que a reintervenção futura é previsível (como na cirurgia oncológica com perspectiva de reabordagem).
Pontos-Chave
- Aderências pós-operatórias ocorrem em até 93% das laparotomias, sendo a causa de 60% das obstruções intestinais por bridas.
- Técnicas cirúrgicas meticulosas são a medida preventiva mais eficaz e de menor custo.
- A laparoscopia reduz substancialmente a formação de aderências em comparação com a cirurgia aberta.
- Barreiras antiaderentes, como Seprafilm® e Adept®, têm indicação em cirurgias abdominais de maior complexidade.
- Inovações como polímeros zwitteriônicos prometem novas abordagens, mas ainda requerem validação clínica.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A prevenção das aderências pós-operatórias deve ser abordada como uma extensão da técnica cirúrgica refinada. Incorporar práticas baseadas em evidências, priorizar o uso de tecnologias minimamente invasivas e adotar dispositivos antiaderentes quando indicados são medidas que não apenas melhoram os desfechos clínicos, mas também reduzem o ônus econômico e o sofrimento dos pacientes.
A educação continuada do cirurgião digestivo é fundamental para manter-se atualizado quanto às inovações tecnológicas e diretrizes internacionais. Em tempos de medicina baseada em valor, prevenir complicações previsíveis como as aderências é um ato de excelência e responsabilidade profissional.
“Jamais devemos nos contentar em apenas operar. Devemos operar melhor, sempre.” — Alexis Carrel
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
KYOTO IPMN GUIDELINES 2024
Introdução
A neoplasia mucinoso papilar intraductal do pâncreas (IPMN) representa um dos principais desafios no diagnóstico e manejo das lesões císticas pancreáticas. Com o avanço das técnicas diagnósticas e uma compreensão mais profunda da progressão dessas lesões, o Protocolo de Kyoto 2024 trouxe diretrizes atualizadas e baseadas em evidências para orientar a conduta cirúrgica. Este artigo tem como objetivo esclarecer os principais pontos das novas recomendações, abordando sua aplicação na prática do cirurgião do aparelho digestivo.
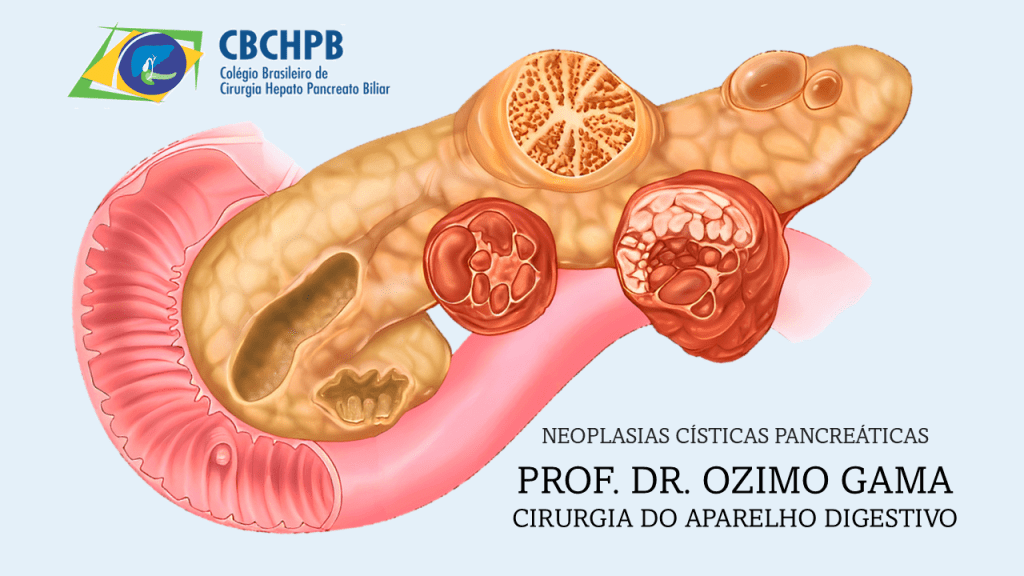
Classificação e Critérios de Risco
Os IPMNs são classificados em tipo ducto principal (MD-IPMN), tipo ducto secundário (BD-IPMN) e tipo misto (MT-IPMN). Os novos critérios de risco do Protocolo de Kyoto 2024 incluem:
- Estigmas de alto risco (HRS): icterícia obstrutiva, nódulo mural ≥5 mm, dilatação do ducto pancreático principal ≥10 mm e citologia suspeita ou positiva.
- Características preocupantes (WF): crescimento acelerado (>2,5 mm/ano), cistos >30 mm, nódulos murais <5 mm, espessamento da parede cística, entre outros.
O reconhecimento desses fatores é essencial para a decisão cirúrgica, diferenciando lesões de baixo risco daquelas que podem evoluir para displasia de alto grau ou carcinoma invasivo.
Indicação Cirúrgica
O Protocolo de Kyoto 2024 reforça a necessidade de uma abordagem personalizada, considerando o estado clínico do paciente, as condições anatômicas e a presença de comorbidades. As principais indicações cirúrgicas incluem:
- IPMNs do ducto principal devido ao alto risco de malignização.
- IPMNs do ducto secundário com estigmas de alto risco.
- IPMNs mistos com crescimento progressivo ou sintomatologia associada.
A decisão entre pancreatectomia parcial ou total depende da extensão da lesão e da histologia intraoperatória.
Aplicação na Cirurgia Digestiva
O manejo cirúrgico do IPMN deve seguir princípios oncológicos rigorosos, evitando pancreatectomias excessivas e preservando a função endócrina e exócrina do órgão. Algumas das principais estratégias incluem:
- Ressecção com margens negativas: intraoperatório, a análise histológica por congelação é essencial para guiar a extensão da ressecção.
- Pancreatectomia minimamente invasiva: avanços na laparoscopia e cirurgia robótica permitiram ressecções mais seguras e menos invasivas.
- Monitoramento pós-operatório: mesmo após a ressecção completa, a vigilância é recomendada devido ao risco de neoplasia residual ou carcinoma concomitante.
Estatísticas Relevantes
No Brasil, o câncer de pâncreas representa cerca de 2% de todos os tumores malignos, mas com alta letalidade. Estima-se que cerca de 10-15% dos casos de adenocarcinoma pancreático estejam associados a IPMNs. O aprimoramento das diretrizes pode impactar diretamente na detecção precoce e na sobrevida dos pacientes.
Pontos-chave
- O Protocolo de Kyoto 2024 reformulou a abordagem ao IPMN, priorizando critérios objetivos para indicação cirúrgica.
- O uso de nomogramas e avanços em EUS-FNA ampliaram a precisão diagnóstica.
- Critérios de descontinuidade de vigilância foram estabelecidos para pacientes com IPMNs pequenos e estáveis.
- O risco de PDAC concomitante exige monitoramento a longo prazo mesmo em lesões consideradas indolentes.
Conclusão
O manejo do IPMN requer uma abordagem multidisciplinar e individualizada. As novas diretrizes do Protocolo de Kyoto 2024 promovem um melhor refinamento na seleção de pacientes para cirurgia, minimizando riscos e otimizando resultados. Para o cirurgião digestivo, a compreensão detalhada dessas recomendações é essencial para um tratamento eficaz e baseado em evidências.
“A cirurgia pancreática não é apenas uma questão de técnica, mas de planejamento preciso, trabalho em equipe e respeito à biologia do tumor.” – Markus W. Büchler
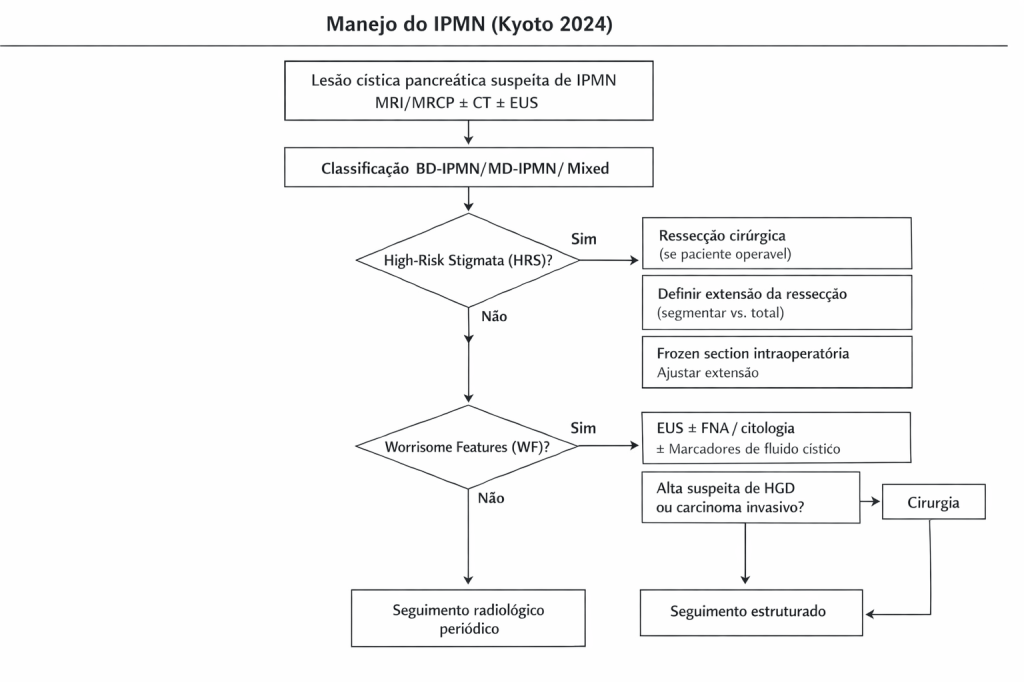
- Estigmas de alto risco (HRS): icterícia obstrutiva, nódulo mural ≥5 mm, dilatação do ducto pancreático principal ≥10 mm e citologia suspeita ou positiva.
- Características preocupantes (WF): crescimento acelerado (>2,5 mm/ano), cistos >30 mm, nódulos murais <5 mm, espessamento da parede cística, entre outros.
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Colecistectomia Robótica
A colecistectomia, procedimento cirúrgico para remoção da vesícula biliar, pode ser realizada por diferentes abordagens, sendo as mais comuns a via laparoscópica (1985), a assistida por robô (1987) e a cirurgia convencional ou aberta (1882). Cada uma dessas técnicas tem vantagens e desvantagens em termos de custo, eficácia clínica e complexidade, o que torna a análise de custo-efetividade crucial para a escolha da abordagem mais adequada em cada caso.
Colecistectomia Laparoscópica (CVL)
A colecistectomia laparoscópica (CVL) é amplamente reconhecida como a técnica de escolha devido à sua eficácia, recuperação rápida e menor custo comparado a outras abordagens. Diversos estudos confirmam que a laparoscopia é a opção mais custo-efetiva para a maioria dos pacientes com doenças benignas da vesícula biliar, como a colelitíase, devido ao menor custo direto das etapas cirúrgicas.
Estudos de Custo-efetividade indicam que o custo variável direto para o cirurgião na execução de uma colecistectomia laparoscópica foi significativamente inferior ao de técnicas mais complexas. A via laparoscópica custou, em média, $929, enquanto a colecistectomia assistida por robô (ROBOSILS) alcançou o custo de $2,608. Isso demonstra que a laparoscopia tem uma vantagem substancial em termos de custo sem comprometer a qualidade do procedimento.
Colecistectomia Robótica
Por outro lado, a colecistectomia assistida por robô tem ganhado popularidade devido à promessa de maior precisão, visualização e controle durante a cirurgia. No entanto, sua viabilidade econômica tem sido amplamente discutida na literatura, com estudos demonstrando que, embora seja uma técnica segura, os custos hospitalares associados a ela são consideravelmente mais altos. Em um estudo de caso-controle, os custos totais hospitalares para a cirurgia robótica foram de $7,985.4, em comparação com $6,255.3 para a laparoscopia. A maior parte desses custos adicionais vem dos gastos com a amortização e os consumíveis do sistema robótico, que são significativamente mais caros. Outra análise revelou uma razão de custo-efetividade da cirurgia robótica de $1,795,735.21 por ano de vida ajustado pela qualidade, um valor muito acima dos limiares aceitáveis para a maioria dos sistemas de saúde. Além disso, um banco de dados nacional demonstrou que, enquanto a colecistectomia robótica pode oferecer benefícios como redução do tempo de internação, os custos globais continuam elevados devido aos custos variáveis e consumíveis associados ao uso da tecnologia robótica, fazendo com que essa técnica não seja necessariamente mais custo-efetiva, mesmo em contextos que poderiam sugerir vantagens clínicas.
Colecistectomia Convencional ou Abertia (Via Laparotômica)
A colecistectomia convencional, ou aberta, apresenta indicações específicas, sendo geralmente reservada para situações onde a abordagem laparoscópica não oferece segurança ou viabilidade adequada. Esta modalidade cirúrgica encontra suas principais indicações em casos de inflamação severa, particularmente na colecistite aguda complicada, onde o risco de complicações durante a laparoscopia se mostra elevado, bem como em pacientes com histórico de múltiplas intervenções abdominais prévias que resultaram em aderências extensas. A suspeita de malignidade da vesícula biliar também constitui indicação relevante para a abordagem aberta, dada a necessidade de acesso mais amplo para ressecção e avaliação adequada. Ademais, pacientes com condições médicas que contraindicam anestesia geral prolongada ou que apresentam comorbidades significativas com elevado risco cirúrgico podem ser considerados candidatos à abordagem convencional, especialmente quando se antecipa a possibilidade de conversão do procedimento laparoscópico. Em determinados grupos, como idosos ou portadores de hepatopatias avançadas, a escolha entre as abordagens laparoscópica e aberta demanda análise criteriosa, ponderando-se riscos e benefícios específicos de cada caso. Desta forma, a colecistectomia convencional mantém-se como opção cirúrgica fundamental para casos complexos ou de alto risco, nos quais a segurança do paciente constitui o principal determinante da escolha terapêutica.
Indicando a Melhor Abordagem para o Paciente
A escolha da abordagem cirúrgica para a colecistectomia pode ser influenciada por uma série de fatores, incluindo a condição clínica do paciente, as características da doença e os custos. A colecistectomia laparoscópica continua sendo a técnica de escolha para a maioria dos pacientes com doenças benignas da vesícula biliar devido à sua eficácia comprovada e menor custo. Além disso, estudos sugerem que a colecistectomia robótica pode ser considerada em casos específicos, como:
- Pacientes com doença hepática avançada, onde a probabilidade de conversão para cirurgia aberta é menor e o tempo de internação é reduzido.
- Contextos de treinamento cirúrgico, onde a precisão e a visualização aprimorada podem ser cruciais.
No entanto, mesmo nesses casos, é importante ponderar os custos adicionais envolvidos e os benefícios clínicos específicos que a cirurgia robótica pode oferecer, considerando que, em geral, as vantagens não são suficientes para justificar o alto custo.
Considerações Finais sobre a Abordagem Convencional e Robótica
Embora a colecistectomia robótica tenha algumas vantagens técnicas, ela não é superior em termos de desfechos clínicos quando comparada à laparoscopia, o que faz com que a escolha por esta última, em termos de custo-benefício, seja preferível na grande maioria dos casos. A colecistectomia laparoscópica oferece uma recuperação mais rápida, menor dor pós-operatória, menos complicações e melhores resultados estéticos, com menor tempo de internação. Além disso, a técnica laparoscópica convencional tem uma mortalidade operatória baixa e um risco de lesão do ducto biliar de aproximadamente 0,5%, valor que é três vezes maior que o encontrado na cirurgia aberta. Por outro lado, a colecistectomia robótica tem se mostrado vantajosa apenas em contextos específicos, como na redução da taxa de conversão para cirurgia aberta em colecistite aguda. Contudo, o risco aumentado de lesões do ducto biliar com a abordagem robótica é uma preocupação adicional que deve ser considerada.
Conclusão
Em resumo, enquanto a colecistectomia laparoscópica continua sendo a técnica de escolha padrão devido ao seu custo-benefício superior e resultados clínicos satisfatórios, a colecistectomia assistida por robô pode ser útil em casos específicos, como doença hepática avançada ou treinamento cirúrgico. No entanto, os custos elevados da técnica robótica não são amplamente justificados por melhorias nos desfechos clínicos, o que reforça a técnica laparoscópica como a abordagem mais custo-efetiva para a maioria dos pacientes.
Pontos-Chave
- Colecistectomia Convencional : Esta técnica é indicada em casos onde a abordagem minimamente invasiva não é viável, como em colecistite aguda complicada com fistulizações ou em pacientes com histórico de múltiplas cirurgias abdominais por laparotomia. No Brasil, cerca de 10% das colecistectomias ainda são realizadas de maneira convencional, de acordo com dados do DATASUS. O procedimento por via convencional apresenta uma taxa de complicações como lesão do ducto biliar em torno de 0.15%, porém, vem com um período de recuperação mais prolongado e uma maior dor pós-operatória.
- Colecistectomia Laparoscópica: Tornou-se o padrão-ouro para a maioria dos casos devido à sua menor invasividade, resultando em menor tempo de hospitalização e recuperação mais rápida. Estudos indicam que no Brasil, aproximadamente 90% das colecistectomias são laparoscópicas. No entanto, a laparoscopia ainda carrega um risco de lesão do ducto biliar que, embora baixo (cerca de 0.5%), é três vezes maior do que na técnica aberta.
- Colecistectomia Robótica: Oferece precisão e visualização 3D, mas com um custo significativamente mais alto. Dados recentes no Brasil mostram que a prática da cirurgia robótica está crescendo, embora ainda seja limitada devido ao custo e disponibilidade de equipamentos. A técnica robótica pode reduzir a necessidade de conversão para cirurgia aberta em pacientes com condições hepáticas complicadas, mas a incidência de lesão do ducto biliar é potencialmente maior, chegando a ser até 5 vezes mais alta do que na laparoscopia, segundo alguns estudos.
Aplicação na Cirurgia Digestiva
A escolha da técnica deve ser baseada na condição do paciente, na expertise do cirurgião e na disponibilidade de recursos. No contexto brasileiro, onde o acesso a tecnologia de ponta pode ser desigual, a laparoscopia tem sido a escolha preferencial por equilibrar custo e eficácia. Entretanto, a cirurgia robótica pode ser considerada em cenários específicos, como em pacientes com doença hepática avançada ou em instituições com investimento em tecnologia robótica.
- Custo: Laparoscópica < Convencional < Robótica.
- Segurança: Convencional e Laparoscópica com taxas comparáveis de complicações, embora com diferentes perfis de risco.
- Eficácia: Todas as técnicas são eficazes, mas a laparoscópica é a mais custo-efetiva para a maioria dos casos.
- Recuperação: Laparoscópica e Robótica oferecem recuperação mais rápida.
“Nós cirurgiões devemos compreender que a tecnologia é meramente um instrumento, não uma solução milagrosa, e sua aplicação deve ser ponderada com cautela, levando em conta o equilíbrio entre segurança, desfechos clínicos e custos assistenciais.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#Colecistectomia #Laparoscopia #CirurgiaRobótica #CustoEfetividade #SaúdeDigestiva
História da Anatomia Humana
A palestra “Fundamentos Históricos da Anatomia Humana” traça a evolução do estudo da anatomia desde a pré-história até a era moderna. A apresentação destaca a importância de figuras como Cláudio Galeno e Andreas Vesalius, enfatizando a transição de métodos não científicos para a observação direta e a metodologia científica. A influência da anatomia em outras áreas, como arte e filosofia, também é abordada, assim como a evolução das práticas éticas e regulamentações na aquisição de corpos para estudo. A palestra conclui sublinhando a importância inabalável do conhecimento anatômico para profissionais de saúde.
Highlights
- 🏛️ Da Pré-história ao Renascimento: A apresentação percorre a longa história do estudo anatômico, desde as primeiras representações artísticas pré-históricas até os trabalhos detalhados de Leonardo da Vinci e Michelangelo, mostrando como a compreensão do corpo humano evoluiu ao longo dos séculos.
- 🔬 Galeno vs. Vesalius: A palestra destaca a transição paradigmática na anatomia, contrastando o trabalho de Cláudio Galeno, baseado em dissecção animal e que perdurou por séculos, com a abordagem revolucionária de Andreas Vesalius, que enfatizou a observação direta do corpo humano.
- 🎨 A Arte e a Anatomia: A profunda interligação entre a anatomia e a arte renascentista é explorada, mostrando como a compreensão precisa do corpo humano influenciou a representação artística em esculturas e pinturas de grandes mestres.
- 📜 Evolução Ética e Regulamentação: A apresentação aborda as mudanças éticas e regulatórias no acesso a corpos para estudo, destacando o Ato de Anatomia de 1832 na Inglaterra como marco importante na normatização dessas práticas.
- 🧑⚕️ Importância Indispensável: A palestra conclui enfatizando a importância inabalável do conhecimento anatômico para a prática médica, desde o diagnóstico até os procedimentos cirúrgicos, salientando sua relevância em todas as fases da formação e da carreira de um profissional de saúde.
Andreas Vesalius
O Cirurgião Anatomista e Seu Legado no Conhecimento Médico
Introdução
Andreas Vesalius (1514–1564), frequentemente chamado de “pai da anatomia moderna”, é uma figura central na história da medicina. Sua abordagem inovadora ao estudo do corpo humano, fundamentada na dissecação e observação direta, transformou o conhecimento médico, especialmente no campo da cirurgia. Neste artigo, exploraremos como Vesalius, ao articular a importância da anatomia para a prática cirúrgica, moldou a base científica do tratamento das doenças do aparelho digestivo.
A Revolução de Vesalius: Do Teatro ao Livro
No Renascimento, a cirurgia era amplamente relegada a uma posição inferior em relação à medicina teórica. Cirurgiões eram vistos como “trabalhadores manuais”, enquanto os médicos universitários, alinhados aos ensinamentos de Galeno, raramente praticavam a dissecação. Vesalius desafiou esse paradigma. Em sua obra-prima, De humani corporis fabrica (1543), ele não apenas corrigiu erros galênicos, mas também destacou a relação intrínseca entre o conhecimento anatômico e a prática cirúrgica. A anatomia para Vesalius não era apenas um exercício acadêmico; era uma ferramenta essencial para o manejo cirúrgico. Ele mostrou que o entendimento detalhado da anatomia era vital para intervenções seguras, como a sutura de feridas intestinais ou a drenagem de abscessos abdominais. Embora a Fabrica não fosse destinada como um manual de cirurgia, sua ênfase no conhecimento anatômico estabeleceu os alicerces para técnicas cirúrgicas mais precisas no trato digestivo e além.
O Papel da Anatomia na Cirurgia do Aparelho Digestivo
Vesalius defendia que um cirurgião habilidoso devia conhecer profundamente a organização dos órgãos internos. Para ele, compreender a estrutura e a função do trato digestivo era crucial para evitar complicações iatrogênicas. Um exemplo prático disso está na importância da anatomia na sutura intestinal, onde a direção das fibras musculares influencia diretamente a recuperação pós-operatória e a funcionalidade do tecido. Além disso, Vesalius abordava a necessidade de ferramentas apropriadas para a prática anatômica e cirúrgica. Em sua Fabrica, ele ilustrou instrumentos que poderiam ser adaptados para dissecação e procedimentos cirúrgicos, desde lâminas para cortes delicados até agulhas curvas para suturas. Ele enfatizava que, embora os instrumentos fossem importantes, era a habilidade do cirurgião – adquirida por meio do estudo e prática – que determinava o sucesso do procedimento.
Desafios e Contribuições à Educação Médica
Vesalius enfrentou críticas intensas de seus contemporâneos, particularmente dos seguidores de Galeno, que viam sua abordagem como uma afronta às tradições estabelecidas. No entanto, ele persistiu, integrando a dissecção ao ensino médico e instigando seus alunos a examinarem corpos humanos reais, ao invés de dependerem exclusivamente de textos antigos. Esse método transformador, baseado na dissecção – o exame direto do corpo – permanece até hoje como pilar fundamental da formação em cirurgia. No tratamento das doenças do aparelho digestivo, a aplicação prática desse aprendizado reflete-se na precisão em procedimentos como gastrectomias, colecistectomias e ressecções intestinais, que requerem um conhecimento detalhado das relações anatômicas entre órgãos como estômago, fígado e intestinos.
Legado para a Cirurgia Moderna
Embora Vesalius tenha se distanciado da prática cirúrgica em seus escritos, ele elevou o status da anatomia ao patamar de ciência central à medicina, garantindo que futuros cirurgiões tivessem uma base sólida para o desenvolvimento de técnicas mais avançadas. Seu trabalho abriu caminho para o desenvolvimento da cirurgia, permitindo avanços que hoje salvam incontáveis vidas.
Conclusão
O legado de Andreas Vesalius não se limita à anatomia; ele inspirou gerações de médicos e cirurgiões a desafiar paradigmas, questionar autoridades e buscar a verdade diretamente no corpo humano. Suas contribuições à medicina moderna são um lembrete da importância de integrar o conhecimento científico ao cuidado do paciente.
“Eu quem sempre fui tão devotado aos escritos de Galeno, admito minha própria estupidez por confiar tanto em seus textos sem examinar a realidade com meus próprios olhos.” – Andreas Vesalius
Gostou❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#AnatomiaModerna #AndreasVesalius #EducaçãoMédica #CirurgiaDigestiva #HistóriaDaMedicina
Cronologia
1514 – Nasce filho de Isabel Crabbe e Andries van Wesel.
1530 – Matricula-se na Universidade de Louvain.
1533 – Inicia seus estudos de medicina na Universidade de Paris.
1536–1538 – Guerra entre Francisco I e Carlos V.
1536–1537 – Continua seus estudos médicos na Universidade de Louvain.
1537
- Fevereiro: Publicação de A Paráfrase do Nono Livro de Rhazes . . . por Andreas Vesalius de Bruxelas, Candidato em Medicina, em Louvain, por Rutgerus Rescius.
- Março: Publicação de A Paráfrase do Nono Livro de Rhazes por Andreas Vesalius, o Autor, em Basileia, por Robert Winter.
- 5 de dezembro: Recebe o título de doutor em medicina no palácio do Bispo de Pádua.
- 6 de dezembro: Nomeado para as cátedras de cirurgia na Universidade de Pádua.
- 6–24 de dezembro: Realiza dissecação pública em Pádua.
1538
- Publicação de Seis Tabelas Anatômicas em Veneza por Bernardino Vitali.
- Princípios de Anatomia Segundo a Opinião de Galeno por Johann Guinter, Ampliado e Corrigido por Andreas Vesalius de Bruxelas publicado em Veneza por Melchior Sessa.
1539
- Publicação de Uma Carta Ensinando que em Casos de Dor no Lado, Deve-se Cortar a Veia Axilar no Cotovelo Direito em Basileia por Robert Winter.
1540
- 15–24 de janeiro: Realiza dissecação pública na Universidade de Bolonha.
1540–1542
- Contribuições para as traduções latinas das Obras Completas de Galeno (1541–1542), publicadas em Veneza pela firma de Lucantonio Giunta.
1542
- Agosto: Envia os blocos de madeira e o texto da Fabrica para Johannes Oporinus em Basileia.
1543
- Janeiro: Chega a Basileia.
- Maio: Disseca e monta um esqueleto articulado de Jakob Karrer.
- Julho: Publicação de Sete Livros sobre a Estrutura do Corpo Humano e Epitome da Estrutura do Corpo Humano (Fabrica) em Basileia por Johannes Oporinus.
- Agosto: Edição alemã da Fabrica, traduzida por Albanus Torinus, publicada em Basileia por Oporinus.
- Agosto: Apresentação de cópias da Fabrica e Epitome a Carlos V, sendo nomeado médico imperial.
1544
- Realiza demonstrações anatômicas em Pisa a convite de Cosme de Médici.
- Casa-se com Anne van Hamme.
1545 – Nasce sua filha, Anne.
1546 – Publicação da Epístola Explicando o Método e Técnica de Administração da Raiz da China Fervida que o Invencível Carlos Recentemente Empregou em Basileia por Oporinus.
1555 – Segunda edição de Sete Livros sobre a Estrutura do Corpo Humano publicada em Basileia por Oporinus.
1556
- Recebe o título de Conde Palatino por Carlos V.
- Abdicação de Carlos V, que cede seus territórios espanhóis ao filho, Filipe II.
1559
- Enviado por Filipe II para atender ao ferimento de Henrique II, Rei da França, após um torneio.
- Muda-se para a corte de Filipe II em Madri.
1561 – Recebe uma cópia de Observações Anatômicas de Gabriel Falloppio.
1562 – Trata um ferimento de Don Carlos, filho de Filipe II.
1564
- Publicação de Uma Consideração sobre as Observações Anatômicas de Falloppio em Veneza por Francesco Franceschi.
- Março: Parte em peregrinação à Terra Santa e entrega um presente de Filipe para apoiar católicos em Jerusalém.
- 15 de outubro: Morre em seu retorno à Europa, na Ilha de Zakynthos.
Tratamento da Coledocolitíase no Paciente com Bypass Gástrico
Abordagens Cirúrgicas e Opções Técnicas
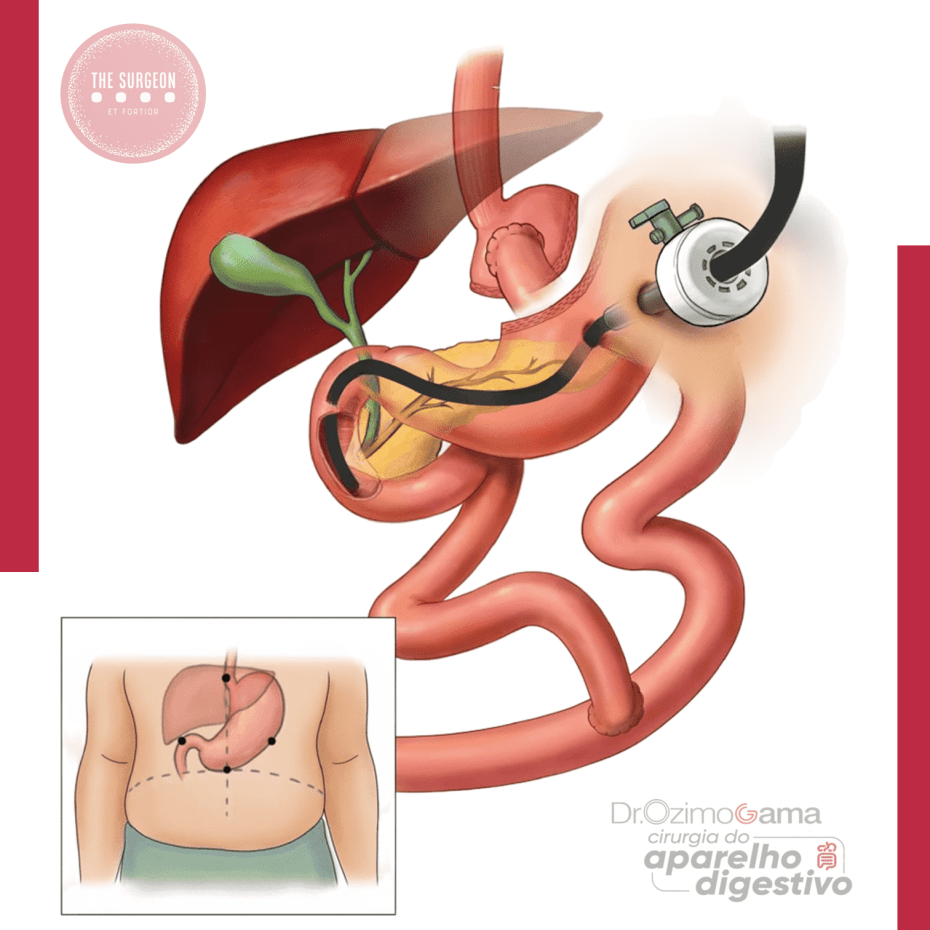
A coledocolitíase, caracterizada pela presença de cálculos no ducto biliar comum (CBD), é uma complicação potencialmente grave que pode causar icterícia obstrutiva, pancreatite e colangite. Em pacientes que realizaram o bypass gástrico em Y-de-Roux, o tratamento dessa condição se torna mais complexo devido às alterações anatômicas decorrentes da cirurgia bariátrica, tornando o acesso ao ducto biliar pelo método endoscópico tradicional (CPRE) um desafio significativo.
Introdução
O bypass gástrico em Y-de-Roux é um dos procedimentos mais realizados no tratamento da obesidade mórbida, resultando em perda de peso significativa e, consequentemente, em uma maior prevalência de cálculos biliares. No entanto, as alterações anatômicas criadas por esse procedimento, como a separação do estômago proximal do restante do trato gastrointestinal, impedem o acesso ao ducto biliar pelo estômago convencional. Nesse contexto, o manejo da coledocolitíase, que afeta até 18% dos pacientes submetidos à colecistectomia, exige uma abordagem multidisciplinar e técnicas cirúrgicas e endoscópicas inovadoras.
Propedêutica Cirúrgica
O diagnóstico e o manejo da coledocolitíase em pacientes com bypass gástrico podem ser desafiadores, pois o acesso ao ducto biliar comum por vias endoscópicas tradicionais é inviável. Nestes casos, a apresentação clínica pode variar de sintomas leves de icterícia a quadros graves de colangite ascendente.
Para a avaliação inicial, a ultrassonografia abdominal continua sendo o exame de escolha para identificar dilatação do ducto biliar e a presença de cálculos. No entanto, devido à complexidade anatômica, exames como a colangiopancreatografia por ressonância magnética (MRCP) são frequentemente necessários para fornecer uma visão mais detalhada da anatomia do trato biliar.
Uma vez confirmado o diagnóstico de coledocolitíase, o tratamento deve ser instituído rapidamente, a fim de evitar complicações como abscessos hepáticos ou pancreatite biliar. As opções terapêuticas variam dependendo da gravidade da apresentação clínica e da disponibilidade de recursos no centro de tratamento. Para pacientes com colangite ascendente, por exemplo, a descompressão urgente do ducto biliar é necessária. Nestes casos, o uso de técnicas minimamente invasivas, como a colangiografia trans-hepática percutânea (PTC), pode ser a melhor alternativa quando a CPRE não é viável.
Em pacientes estáveis, várias abordagens cirúrgicas são possíveis. Uma técnica amplamente utilizada é a colecistectomia laparoscópica assistida por CPRE transgástrica. Nesse procedimento, um acesso cirúrgico é feito no estômago excluído do paciente, permitindo que o endoscópio seja inserido diretamente na papila duodenal, facilitando a remoção dos cálculos e a descompressão do ducto biliar.
Aplicação na Cirurgia Digestiva
A cirurgia digestiva avançada em pacientes com bypass gástrico exige um planejamento cuidadoso e uma abordagem técnica que considere as alterações anatômicas decorrentes da cirurgia bariátrica. As seguintes estratégias têm sido eficazes no manejo da coledocolitíase nesses pacientes:
- ERCP assistida por laparoscopia: Esse procedimento permite o acesso ao trato biliar usando técnicas combinadas de laparoscopia e endoscopia. A transposição do endoscópio pelo estômago excluído oferece uma alternativa eficiente para a remoção dos cálculos, com baixa taxa de complicações.
- Colangiografia trans-hepática percutânea (PTC): Em pacientes instáveis ou com contraindicações para procedimentos invasivos, a PTC é uma alternativa valiosa para a descompressão biliar emergente. Essa técnica minimamente invasiva pode ser associada à dilatação do esfíncter de Oddi para expulsar os cálculos.
- Exploração cirúrgica do ducto biliar comum: Em casos complexos, onde as técnicas minimamente invasivas falham, a exploração laparoscópica ou aberta do ducto biliar pode ser necessária. A abordagem transcística ou a coledocotomia são opções viáveis, dependendo da localização e do tamanho dos cálculos. A utilização de cateteres tipo Fogarty ou cestas de Dormia pode ser útil para a remoção dos cálculos.
- Técnicas reconstrutivas: Para pacientes com múltiplos cálculos biliares ou complicações, a realização de anastomoses biliodigestivas, como a coledocoduodenostomia ou a hepaticojejunostomia em Y-de-Roux, pode ser considerada. Essas abordagens proporcionam um alívio duradouro, evitando futuros episódios de obstrução.
Pontos-chave
- Pacientes com bypass gástrico em Y-de-Roux apresentam desafios únicos no tratamento da coledocolitíase devido à anatomia alterada do trato digestivo.
- A ERCP tradicional é inviável nesses pacientes, sendo substituída por abordagens combinadas, como a ERCP transgástrica assistida por laparoscopia ou a PTC.
- A ultrassonografia e a MRCP são ferramentas essenciais para o diagnóstico, enquanto a intervenção cirúrgica deve ser cuidadosamente planejada, considerando as condições clínicas e anatômicas do paciente.
- Em centros especializados, as taxas de sucesso com essas abordagens minimamente invasivas são altas, com complicações graves ocorrendo em menos de 10% dos casos.
Conclusão Aplicada à Prática do Cirurgião Digestivo
O tratamento da coledocolitíase em pacientes submetidos ao bypass gástrico representa um desafio técnico significativo, exigindo conhecimento especializado em cirurgia digestiva avançada e endoscopia. A abordagem ideal deve ser individualizada, considerando a condição clínica do paciente e as habilidades da equipe cirúrgica. As técnicas minimamente invasivas, como a ERCP assistida por laparoscopia, são as preferidas quando viáveis, mas o cirurgião deve estar preparado para realizar intervenções mais complexas, como a coledocotomia ou anastomoses biliodigestivas, quando necessário.
Nas palavras do cirurgião Steven Strasberg, “O sucesso na cirurgia hepato-biliar depende não apenas da habilidade técnica, mas também da capacidade de adaptar-se às circunstâncias anatômicas e clínicas únicas de cada paciente.”
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#coledocolitiase
#bypassgastrico
#cirurgiabariatrica
#cirurgiadigestiva
#cprefalha
Prevenção da Hérnia Incisional
Um Guia Completo para Estudantes de Medicina e Residentes em Cirurgia
A hérnia incisional é uma complicação pós-operatória comum, especialmente após cirurgias abdominais, com taxas que variam de 1 a 20%. Além de causar dor e desconforto, essa condição pode gerar um grande impacto econômico e clínico para pacientes e o sistema de saúde. No Brasil, o aumento do índice de massa corporal (IMC) e a alta prevalência de cirurgias abdominais tornam a prevenção da hérnia incisional um tema de extrema relevância na prática cirúrgica. Este artigo abordará as principais técnicas de prevenção da hérnia incisional, com foco na escolha do tipo de incisão, técnica de fechamento e o uso de tela profilática, temas de suma importância para estudantes de medicina, residentes de cirurgia geral e pós-graduandos em cirurgia do aparelho digestivo.
Introdução
A hérnia incisional se desenvolve em locais de incisões cirúrgicas anteriores, onde a parede abdominal se enfraquece, permitindo que o conteúdo intra-abdominal protrua. Estima-se que 10% a 15% dos pacientes submetidos a laparotomias desenvolvam essa complicação, sendo que cerca de 400 mil cirurgias de reparo de hérnia incisional são realizadas anualmente na Europa e aproximadamente 348 mil nos Estados Unidos. No Brasil, os números são igualmente alarmantes, considerando a alta prevalência de cirurgias abdominais associadas a fatores de risco como obesidade e diabetes.
Fatores de Risco para a Hérnia Incisional
A formação da hérnia incisional está associada a uma série de fatores, tanto relacionados ao paciente quanto ao procedimento cirúrgico. Entre os fatores de risco mais comuns estão:
- Obesidade (IMC > 25 kg/m²): Pacientes obesos têm maior propensão ao desenvolvimento de hérnias, com taxas que variam de 25% a 50%. No Brasil, a obesidade é um fator crescente que precisa ser cuidadosamente avaliado no pré-operatório.
- Infecção no local cirúrgico: A infecção compromete a cicatrização adequada da incisão, aumentando o risco de herniação.
- Doenças crônicas como DPOC, diabetes e desnutrição: Esses pacientes apresentam maior risco de complicações pós-operatórias.
- Tabagismo e quimioterapia: Fatores que afetam a cicatrização e a integridade dos tecidos abdominais.
Técnicas de Fechamento da Parede Abdominal
A técnica utilizada para o fechamento da parede abdominal após a laparotomia é um dos fatores mais importantes na prevenção da hérnia incisional. Estudos recentes, revisados pela Sociedade Europeia de Hérnia, recomendam o uso de sutura contínua com monofilamento de absorção lenta e a técnica de pequenos pontos (<1 cm de distância), com uma relação comprimento de sutura/comprimento da ferida superior a 4:1. Esse protocolo baseia-se em evidências que demonstram uma redução de três vezes no risco de formação de hérnia quando a relação sutura/ferida é respeitada. Além disso, a técnica de pequenos pontos, com suturas próximas da borda da fáscia, diminui significativamente a formação de hérnias, conforme demonstrado em ensaios clínicos multicêntricos europeus.
Uso de Tela Profilática
Mesmo com técnicas de fechamento adequadas, alguns pacientes de alto risco podem se beneficiar do uso de tela profilática. Essa técnica é especialmente recomendada para pacientes submetidos a cirurgias com alto risco de hérnia incisional, como reparos de aneurismas de aorta abdominal (AAA) e cirurgias bariátricas laparotômicas. Em cirurgias de AAA, o uso de telas profiláticas pode reduzir a incidência de hérnias incisionais de 32% para menos de 10%, de acordo com estudos recentes. Na prática brasileira, ainda há um certo receio quanto ao uso de telas, especialmente em cirurgias contaminadas, como procedimentos colorretais. Contudo, estudos mostram que a aplicação de telas em pacientes de alto risco pode ser feita com segurança e eficácia, sem aumento significativo de complicações como infecções ou rejeição do material.
Aplicação na Cirurgia Digestiva
No contexto da cirurgia do aparelho digestivo, que inclui procedimentos complexos como ressecções colorretais e bariátricas, a prevenção da hérnia incisional deve ser uma preocupação constante. Pacientes submetidos a cirurgias digestivas frequentemente apresentam fatores de risco, como obesidade, diabetes e infecções pós-operatórias, aumentando significativamente as chances de complicações. Nessas cirurgias, a escolha correta do tipo de incisão e a técnica de fechamento são essenciais para minimizar o risco de hérnia. O uso de incisão não mediana, quando possível, e a técnica de pequenos pontos com a relação de sutura/ferida adequada, são práticas recomendadas. Além disso, em pacientes com maior risco, o uso de telas profiláticas deve ser seriamente considerado, especialmente em cirurgias abertas, como a bariátrica e o reparo de AAA.
Pontos-chave
- Conhecimento dos fatores de risco: Identificar pacientes de alto risco, como os com IMC elevado, histórico de infecção ou doenças crônicas, é fundamental para a escolha da melhor técnica cirúrgica.
- Escolha adequada da técnica de fechamento: Utilizar a técnica de pequenos pontos (<1 cm) com uma relação de sutura/comprimento da ferida >4:1 pode reduzir significativamente as taxas de hérnia incisional.
- Uso de telas profiláticas: Em pacientes de alto risco, como os submetidos a reparos de aneurismas ou cirurgias bariátricas, a tela profilática pode ser uma medida preventiva eficaz.
- Educação continuada: A formação de residentes e pós-graduandos em relação às técnicas atualizadas de fechamento abdominal é crucial para melhorar os resultados cirúrgicos e reduzir a incidência de hérnias incisionais.
Conclusão
A prevenção da hérnia incisional é um tema essencial para cirurgiões, especialmente aqueles que atuam no tratamento de doenças do aparelho digestivo. A adoção de técnicas de fechamento baseadas em evidências, como o uso de pequenos pontos e a proporção correta de sutura, pode reduzir significativamente as taxas de recidiva. Além disso, o uso de telas profiláticas em pacientes de alto risco oferece uma medida adicional de proteção, com resultados promissores. Embora as técnicas de fechamento de incisões sejam frequentemente subestimadas, sua importância na prevenção de complicações pós-operatórias, como as hérnias incisionais, não deve ser negligenciada. Investir em educação continuada e incentivar a prática baseada em evidências são os primeiros passos para garantir melhores resultados para os pacientes e reduzir a carga econômica e clínica associada às hérnias incisionais.
“O cirurgião deve não só curar, mas também prevenir.” — Ambroise Paré
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#CirurgiaDigestiva #HerniaIncisional #PrevençãoCirúrgica #EducaçãoMédica
Desvendando a Tela do Cirurgião
A Arte e a Ciência da Dissecção Abdominal
Introdução
A dissecção abdominal é a pedra angular da cirurgia digestiva, uma exploração meticulosa que revela a intrincada paisagem do abdômen humano. Para estudantes de medicina, residentes de cirurgia geral e especialistas de pós-graduação em cirurgia do sistema digestivo, dominar essa habilidade é fundamental. Este artigo mergulha nas nuances da dissecção abdominal, estabelecendo uma ponte entre o conhecimento anatômico e a perícia cirúrgica.
Importância do Tema
O abdômen, um compartimento complexo que abriga órgãos vitais, é delimitado superiormente pelo diafragma e inferiormente pela cavidade pélvica. O peritônio, uma membrana serosa, reveste essa cavidade e envolve seu conteúdo, criando um ambiente dinâmico para intervenção cirúrgica. No Brasil, as doenças do sistema digestivo representam uma parcela significativa dos procedimentos cirúrgicos. Segundo o Ministério da Saúde do Brasil, em 2022, aproximadamente 15% de todas as cirurgias realizadas no país estavam relacionadas ao sistema digestivo, ressaltando a importância da proficiência na dissecção abdominal.
O processo de dissecção abdominal começa com uma inspeção cuidadosa da cavidade. Os cirurgiões devem estar cientes de potenciais aderências, que ocorrem em quase 93% dos pacientes que passaram por cirurgias abdominais anteriores, conforme relatado pelo Colégio Brasileiro de Cirurgiões. Essas aderências podem complicar a dissecção e devem ser abordadas meticulosamente. À medida que progredimos pelos quadrantes abdominais, encontramos uma variedade de órgãos, cada um com sua relação única com o peritônio. O fígado, um órgão intraperitoneal que ocupa o quadrante superior direito e se estende para o esquerdo, serve como ponto de referência para orientação. Sua posição, dividida pelo ligamento falciforme, guia a abordagem do cirurgião às estruturas circundantes.
O estômago, outro órgão intraperitoneal, situa-se predominantemente no quadrante superior esquerdo. Sua posição e relação com o omento menor são cruciais em procedimentos como gastrectomias, que representam aproximadamente 5% de todas as cirurgias digestivas no Brasil, de acordo com o Instituto Nacional de Câncer (INCA). Os intestinos delgado e grosso apresentam um arranjo complexo que exige navegação cuidadosa. As porções secundariamente retroperitoneais, como o duodeno e partes do cólon, requerem uma abordagem diferente em comparação com suas contrapartes intraperitoneais. Compreender essas relações é essencial em procedimentos como cirurgias colorretais, que constituem cerca de 8% das operações do sistema digestivo no Brasil.
Pontos-Chave:
- Relações peritoneais: Distinguir entre órgãos intraperitoneais, retroperitoneais e secundariamente retroperitoneais é crucial para o planejamento e execução cirúrgica.
- Marcos anatômicos: Utilizar estruturas como o ligamento falciforme e inserções omentais auxilia na orientação e dissecção.
- Considerações vasculares: A consciência dos principais vasos e seus trajetos é vital para prevenir lesões iatrogênicas durante a dissecção.
- Contexto embriológico: Compreender a anatomia do desenvolvimento ajuda a entender reflexões peritoneais complexas e inserções ligamentares.
- Implicações patológicas: Reconhecer como as patologias alteram a anatomia normal é essencial para uma dissecção segura e eficaz.
Conclusões Aplicadas à Prática da Cirurgia Digestiva
O domínio da dissecção abdominal se traduz diretamente em melhores resultados cirúrgicos. Em procedimentos laparoscópicos, que agora representam mais de 60% das cirurgias abdominais nos hospitais terciários do Brasil, uma compreensão profunda das relações tridimensionais é indispensável. A capacidade de navegar pelas reflexões peritoneais e identificar estruturas-chave através de pontos de acesso mínimos depende fortemente do conhecimento fundamental adquirido através de técnicas de dissecção aberta. Além disso, em ressecções oncológicas complexas, onde a linfadenectomia radical é frequentemente necessária, o conhecimento íntimo do cirurgião sobre os espaços retroperitoneais e seus conteúdos pode significar a diferença entre resultados curativos e paliativos. Com o câncer gástrico sendo a terceira malignidade mais comum do sistema digestivo no Brasil, afetando aproximadamente 21.000 indivíduos anualmente, a importância da dissecção precisa não pode ser subestimada.
Em conclusão, a dissecção abdominal não é meramente uma habilidade técnica, mas uma forma de arte que requer refinamento constante. À medida que avançamos em técnicas minimamente invasivas e cirurgias robóticas, a base estabelecida pelos métodos tradicionais de dissecção permanece inestimável. O cirurgião que domina essa arte está bem equipado para enfrentar os desafios da cirurgia digestiva moderna, melhorando os resultados dos pacientes e avançando o campo.
Como disse o renomado cirurgião William Stewart Halsted: “A aquisição de habilidade técnica em cirurgia é diferente de qualquer outro campo profissional, pois trata constantemente de uma anatomia humana singular.”
DissecçãoAbdominal #CirurgiaDigestiva #AnatomiaCircúrgica #EducaçãoMédica #SaúdeBrasileira
Gostou? Deixe-nos um comentário ✍️, compartilhe em suas redes sociais e|ou envie sua pergunta via 💬 Chat Online em nossa DM do Instagram.
Ressecção Robótica do Câncer de Pâncreas
Introdução
O câncer de pâncreas continua sendo uma das malignidades mais letais, com um impacto significativo em todo o mundo. No Brasil, está entre as principais causas de mortalidade por câncer, com taxas de sobrevivência ainda desanimadoras, apesar dos avanços no tratamento. A ressecção cirúrgica oferece a única possibilidade curativa, mas a complexidade e a alta morbidade associadas às cirurgias abertas tradicionais têm limitado sua ampla adoção. Com o advento de técnicas minimamente invasivas, a cirurgia robótica pancreática surgiu como uma abordagem revolucionária, prometendo refinar e, possivelmente, redefinir o manejo cirúrgico das malignidades pancreáticas. Mas qual é o verdadeiro valor da ressecção robótica do câncer de pâncreas, e como isso se traduz na prática clínica?
Desenvolvimento do Tema
Desde sua criação, o procedimento de Whipple (pancreatoduodenectomia) tem sido o alicerce da cirurgia curativa para o câncer de pâncreas, embora com desafios significativos. As cirurgias abertas tradicionais, caracterizadas por extensos tempos de recuperação e altas taxas de complicações, têm exigido a busca por alternativas melhores. A evolução da cirurgia minimamente invasiva trouxe as técnicas laparoscópicas para o centro das atenções, mas sua aplicação generalizada na cirurgia pancreática foi limitada por dificuldades técnicas, especialmente em reconstruções complexas.
A introdução da cirurgia assistida por robô marcou um salto significativo. Em 2001, os primeiros casos de pancreatectomia distal robótica (PDR) e pancreatoduodenectomia robótica (PDR) foram realizados, inaugurando uma nova era na cirurgia pancreática. A plataforma robótica oferece várias vantagens distintas: visão estereotáxica, magnificação superior, estabilidade da plataforma e benefícios ergonômicos. Essas características permitem maior precisão e controle, especialmente no ambiente anatômico complexo do pâncreas.
Estudos recentes mostraram que as abordagens robóticas para a cirurgia pancreática não apenas são viáveis, mas também oferecem melhores resultados perioperatórios em comparação com as cirurgias abertas tradicionais. Esses resultados incluem menor perda sanguínea intraoperatória, menores períodos de internação hospitalar e redução das complicações respiratórias e infecções de feridas. Importante destacar que a cirurgia robótica demonstrou manter desfechos oncológicos comparáveis aos da cirurgia aberta, com alguns estudos sugerindo até melhores resultados em termos de ressecções com margens negativas (R0) e colheita de linfonodos.
No entanto, a adoção da cirurgia robótica pancreática não está isenta de desafios. A curva de aprendizado é íngreme, com melhorias significativas nos resultados observadas somente após o cirurgião ter realizado um número substancial de casos. Isso tem implicações para sua implementação, especialmente em centros de menor volume. No Brasil, onde os recursos de saúde podem variar significativamente entre as regiões, a concentração de expertise em centros de alto volume pode desempenhar um papel crucial para garantir a disseminação segura e eficaz da cirurgia robótica pancreática.
Pontos Principais
- Vantagens da Cirurgia Robótica Pancreática: A cirurgia robótica oferece melhor visualização, destreza e ergonomia, o que é particularmente benéfico no ambiente anatômico complexo do pâncreas. Isso se traduz em melhores resultados perioperatórios, incluindo menor perda de sangue e menores períodos de internação hospitalar, sem comprometer a eficácia oncológica.
- Curva de Aprendizado e Implementação: A adoção bem-sucedida da cirurgia robótica pancreática exige a superação de uma curva de aprendizado significativa. Centros de alto volume têm maior probabilidade de alcançar melhores resultados, destacando a importância da concentração de expertise, particularmente em regiões com recursos de saúde variáveis, como o Brasil.
- Desfechos Comparativos: Estudos que comparam cirurgias pancreáticas robóticas e abertas mostraram que as abordagens robóticas podem levar a recuperações mais rápidas e menos complicações, com desfechos oncológicos semelhantes ou até superiores. Isso posiciona a cirurgia robótica como uma ferramenta valiosa no arsenal cirúrgico para o câncer de pâncreas, especialmente em centros bem equipados.
- Contexto Brasileiro: No Brasil, o câncer de pâncreas é uma preocupação significativa de saúde pública, com necessidade de estratégias de tratamento mais eficazes. A adoção da cirurgia robótica pancreática pode desempenhar um papel fundamental na melhoria dos desfechos dos pacientes, especialmente se concentrada em centros de alto volume com a expertise e os recursos necessários.
- Direções Futuras: O futuro da cirurgia pancreática reside na contínua refinação e disseminação das técnicas robóticas. À medida que mais cirurgiões superam a curva de aprendizado e a tecnologia continua a avançar, o potencial para que a cirurgia robótica se torne o padrão de tratamento no câncer de pâncreas se torna cada vez mais provável.
A cirurgia robótica no tratamento do câncer pancreático, incluindo procedimentos complexos como a pancreatectomia distal e a pancreatoduodenectomia, está associada a uma série de riscos e complicações potenciais. Complicações comuns incluem fístula pancreática pós-operatória (POPF), que pode ocorrer em até 28% dos casos. A taxa de fístula pancreática clinicamente relevante (grau B/C) pode ser significativa, como observado em várias séries de casos. Outras complicações incluem sangramento intraoperatório, que pode levar à necessidade de conversão para cirurgia aberta. A taxa de conversão para cirurgia aberta varia, mas pode ser necessária em até 6% dos casos devido a falha de progressão ou sangramento. Complicações maiores, como infecções de ferida e necessidade de reoperação, também são preocupações importantes. A taxa de reoperação pode chegar a 7%. A mortalidade perioperatória é baixa, com taxas de mortalidade de 30 e 90 dias relatadas em 0,5% e 1%, respectivamente. No entanto, a morbidade geral pode ser alta, com complicações ocorrendo em até 63% dos pacientes, incluindo complicações graves (Clavien-Dindo ≥ IIIb) em 4% dos casos. O tempo operatório prolongado é outra consideração, com a pancreatoduodenectomia robótica frequentemente exigindo mais tempo do que a cirurgia aberta. Além disso, a cirurgia robótica pode estar associada a custos hospitalares mais elevados, apesar de uma menor taxa de complicações perioperatórias e menor tempo de internação. Em resumo, a cirurgia robótica para câncer pancreático é viável e segura em centros especializados, mas está associada a complicações significativas, como fístula pancreática, necessidade de conversão para cirurgia aberta e complicações maiores. A seleção cuidadosa dos pacientes e a experiência do cirurgião são cruciais para minimizar esses riscos.
References
Conclusão
A ressecção robótica do câncer de pâncreas representa um avanço significativo no tratamento cirúrgico de uma das malignidades mais letais. Seu valor reside não apenas nas melhorias técnicas que oferece, mas também no potencial de melhorar os desfechos dos pacientes através da redução da morbidade e da recuperação mais rápida. No entanto, os benefícios dessa tecnologia só podem ser plenamente realizados se for implementada em centros de alto volume com a expertise e os recursos necessários. Para o Brasil, isso significa um foco no desenvolvimento de centros de excelência onde os cirurgiões possam aprimorar suas habilidades e trazer todo o potencial da cirurgia robótica para combater essa doença desafiadora.
Como disse o grande cirurgião Dr. Harvey Cushing: “Um médico é obrigado a considerar mais do que um órgão doente, mais até do que o homem inteiro – ele deve ver o homem em seu mundo.” O advento da cirurgia robótica no tratamento do câncer de pâncreas exemplifica essa abordagem holística, combinando inovação tecnológica com uma profunda compreensão da condição humana para oferecer esperança onde antes havia pouca.
Gostou? Deixe-nos um comentário ✍️, compartilhe em suas redes sociais e|ou envie sua pergunta via 💬 Chat Online em nosso DM do Instagram.
#CâncerDePâncreas #CirurgiaRobótica #ProcedimentoWhipple #InovaçãoCirúrgica #CirurgiaDigestiva
Cirurgia Robótica no Tratamento do Câncer Pancreático: Avanço ou Risco?
Introdução
O câncer pancreático é uma das neoplasias mais desafiadoras na prática cirúrgica, com altas taxas de mortalidade e morbidade, mesmo em estágios iniciais. No Brasil, o Instituto Nacional de Câncer (INCA) estima que a incidência anual de câncer pancreático seja de aproximadamente 10 mil novos casos, com uma taxa de mortalidade de cerca de 95% nos primeiros cinco anos após o diagnóstico. A cirurgia é a única chance de cura, mas os desafios técnicos e a complexidade anatômica do pâncreas tornam a abordagem cirúrgica tradicional uma tarefa árdua, muitas vezes associada a complicações graves.
Com o avanço da tecnologia, a cirurgia robótica tem ganhado espaço como uma alternativa minimamente invasiva para o tratamento do câncer pancreático. Mas qual é o real valor dessa abordagem na prática clínica? Como ela se compara à cirurgia convencional em termos de eficácia, segurança e custo-benefício? Este post explora o estado atual da cirurgia robótica no tratamento do câncer pancreático, abordando desde a curva de aprendizado até os desfechos clínicos e as principais contra-indicações.
Desenvolvimento
A cirurgia robótica foi introduzida como uma evolução da cirurgia laparoscópica, oferecendo maior precisão e controle ao cirurgião, com benefícios como visão tridimensional, maior amplitude de movimentos e ergonomia aprimorada. No entanto, a implementação dessa tecnologia na cirurgia pancreática, especialmente na pancreatoduodenectomia (procedimento de Whipple) e na pancreatectomia distal, ainda é um tema de intenso debate.
Estudos recentes indicam que a cirurgia robótica pode reduzir o tempo de internação e a perda de sangue intraoperatória em comparação com a cirurgia aberta. No entanto, essas vantagens vêm acompanhadas de desafios significativos, incluindo uma curva de aprendizado íngreme e maior tempo operatório. De acordo com estudos realizados no Brasil, a curva de aprendizado para a pancreatoduodenectomia robótica pode variar entre 20 a 40 casos, com uma redução progressiva das complicações à medida que o cirurgião adquire mais experiência. Isso levanta a questão: será que todos os centros de cirurgia pancreática no Brasil estão preparados para adotar essa tecnologia de forma segura?
MEDICINA BASEADA EM EVIDÊNCIAS
A cirurgia robótica no tratamento do câncer pancreático, incluindo procedimentos complexos como a pancreatectomia distal e a pancreatoduodenectomia, está associada a uma série de riscos e complicações potenciais. Complicações comuns incluem fístula pancreática pós-operatória (POPF), que pode ocorrer em até 28% dos casos. A taxa de fístula pancreática clinicamente relevante (grau B/C) pode ser significativa, como observado em várias séries de casos. Outras complicações incluem sangramento intraoperatório, que pode levar à necessidade de conversão para cirurgia aberta. A taxa de conversão para cirurgia aberta varia, mas pode ser necessária em até 6% dos casos devido a falha de progressão ou sangramento. Complicações maiores, como infecções de ferida e necessidade de reoperação, também são preocupações importantes. A taxa de reoperação pode chegar a 7%. A mortalidade perioperatória é baixa, com taxas de mortalidade de 30 e 90 dias relatadas em 0,5% e 1%, respectivamente. No entanto, a morbidade geral pode ser alta, com complicações ocorrendo em até 63% dos pacientes, incluindo complicações graves (Clavien-Dindo ≥ IIIb) em 4% dos casos. O tempo operatório prolongado é outra consideração, com a pancreatoduodenectomia robótica frequentemente exigindo mais tempo do que a cirurgia aberta. Além disso, a cirurgia robótica pode estar associada a custos hospitalares mais elevados, apesar de uma menor taxa de complicações perioperatórias e menor tempo de internação. Em resumo, a cirurgia robótica para câncer pancreático é viável e segura em centros especializados, mas está associada a complicações significativas, como fístula pancreática, necessidade de conversão para cirurgia aberta e complicações maiores. A seleção cuidadosa dos pacientes e a experiência do cirurgião são cruciais para minimizar esses riscos.
Aplicação na Cirurgia Digestiva
A principal aplicação da cirurgia robótica no câncer pancreático tem sido na pancreatoduodenectomia e na pancreatectomia distal. Essas cirurgias, que envolvem a ressecção de partes significativas do pâncreas, são tradicionalmente associadas a altas taxas de complicações, como fístulas pancreáticas e infecções. A cirurgia robótica, com sua precisão aprimorada, tem o potencial de mitigar alguns desses riscos, mas a evidência atual ainda é limitada.
No Brasil, onde os custos são uma preocupação constante, a cirurgia robótica apresenta um desafio adicional. O custo médio de um procedimento robótico é significativamente maior que o da cirurgia aberta, com um aumento estimado de até 30% no valor total do procedimento, devido aos altos custos dos equipamentos e da manutenção do robô. Isso sem considerar a necessidade de treinamento específico e o tempo adicional de cirurgia, que pode impactar a disponibilidade de salas cirúrgicas.
Além disso, a literatura indica que as taxas de mortalidade em 30 dias não diferem significativamente entre a cirurgia robótica e a aberta, com ambos os métodos apresentando taxas em torno de 2% a 5% nos principais centros de referência. Entretanto, a incidência de complicações graves pode ser menor em centros de excelência que realizam um grande número de procedimentos robóticos.
Pontos-Chave
- Curva de Aprendizado e Expertise: A cirurgia robótica pancreática exige uma curva de aprendizado longa, com a aquisição de habilidades específicas ao longo de muitos casos, o que pode limitar sua aplicação em centros de menor volume.
- Custos Associados: O alto custo da cirurgia robótica, tanto em termos de equipamento quanto de manutenção, representa uma barreira significativa, especialmente em sistemas de saúde pública como o SUS no Brasil.
- Taxas de Complicações e Mortalidade: Embora a cirurgia robótica possa oferecer benefícios em termos de menor perda de sangue e recuperação mais rápida, as taxas de complicações graves e mortalidade não mostram uma vantagem clara sobre a cirurgia aberta, especialmente fora de centros de excelência.
- Contra-Indicações: Pacientes com tumores avançados ou invasão vascular significativa podem não ser candidatos ideais para a cirurgia robótica, onde a necessidade de ressecções complexas e múltiplas anastomoses pode superar os benefícios da abordagem minimamente invasiva.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A cirurgia robótica no tratamento do câncer pancreático representa uma fronteira promissora, mas ainda envolta em desafios. Para o cirurgião digestivo, a decisão de utilizar essa tecnologia deve ser baseada em uma avaliação criteriosa do perfil do paciente, da experiência da equipe cirúrgica e dos recursos disponíveis. Embora os benefícios potenciais sejam atraentes, eles precisam ser equilibrados com os custos e as limitações operacionais.
Em suma, a cirurgia robótica pode ser uma ferramenta valiosa, mas seu sucesso depende do contexto em que é aplicada. Centros com alta expertise e volume cirúrgico estão mais bem posicionados para maximizar os benefícios dessa abordagem, enquanto outros devem ponderar cuidadosamente antes de adotar essa tecnologia. Como o célebre cirurgião William Halsted disse uma vez: “O que o cirurgião precisa fazer pelo paciente é fazê-lo da melhor forma possível.” No caso da cirurgia robótica, isso significa não apenas adotar a tecnologia mais avançada, mas garantir que ela seja usada de forma segura e eficaz.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#CirurgiaRobótica #CâncerDePâncreas #Pancreatoduodenectomia #InovaçãoCirúrgica #CirurgiaDigestiva
Trauma Cirúrgico Controlado: Como Gerenciar a Resposta Metabólica em Intervenções Eletivas
INTRODUÇÃO
Sempre que ocorre uma agressão ao organismo, independentemente da causa, um conjunto complexo de respostas é desencadeado para manter a homeostase e a sobrevivência. Se a lesão for de pequena intensidade, o corpo rapidamente retoma o equilíbrio metabólico e imunológico. No entanto, em casos de traumas extensos, a resposta pode ser exacerbada, levando à disfunção orgânica e aumentando os riscos de mortalidade. Para o cirurgião, é crucial compreender os eventos endócrinos, metabólicos e imunológicos que ocorrem durante e após uma intervenção cirúrgica para manejar adequadamente essas respostas e minimizar complicações.
DESENVOLVIMENTO
A resposta metabólica ao trauma pode ser dividida em três fases: Ebb, Flow e Anabólica. A fase Ebb ocorre imediatamente após o trauma e é caracterizada por instabilidade hemodinâmica, hipovolemia, e aumento de hormônios como catecolaminas e glicocorticoides. Nessa fase, apesar da elevação dos hormônios contrarreguladores, o metabolismo é diminuído. A fase Flow, que segue a fase Ebb, é marcada por hipermetabolismo, retenção hídrica e hiperglicemia, devido ao aumento dos hormônios catabólicos. A fase Anabólica, que pode durar meses, é quando o corpo começa a recuperar o peso e a massa muscular perdidos, com os níveis hormonais retornando ao normal.
APLICAÇÃO NA CIRURGIA DIGESTIVA
Em cirurgias eletivas, a resposta ao trauma pode ser melhor controlada. A avaliação pré-operatória, a escolha da técnica cirúrgica e o manejo adequado do jejum são essenciais para reduzir o impacto do trauma. A laparoscopia, por exemplo, deve ser priorizada sempre que possível, pois está associada a uma menor agressão tecidual e, consequentemente, a uma resposta metabólica mais branda. Além disso, controlar a ansiedade pré-operatória com benzodiazepínicos pode reduzir a liberação de catecolaminas, diminuindo os riscos de complicações como arritmias e hipertensão.
PONTOS-CHAVE
- A resposta ao trauma cirúrgico envolve fases distintas, cada uma com características metabólicas específicas.
- A laparoscopia é uma via cirúrgica que pode minimizar a resposta ao trauma.
- Controlar a ansiedade pré-operatória é fundamental para reduzir a liberação de catecolaminas e suas consequências.
CONCLUSÕES APLICADAS À PRÁTICA DO CIRURGIÃO DIGESTIVO
O manejo adequado da resposta ao trauma em cirurgias eletivas é vital para reduzir complicações e melhorar os resultados cirúrgicos. A compreensão das fases da resposta metabólica, a escolha de técnicas menos invasivas e a gestão da ansiedade pré-operatória são pilares essenciais para a prática do cirurgião digestivo. Com esses conhecimentos, os profissionais podem proporcionar uma recuperação mais rápida e segura aos seus pacientes. Como disse o renomado cirurgião Harvey Cushing: “A cirurgia é o mais elevado empreendimento humano, pois requer uma combinação de conhecimentos científicos, habilidade técnica e sensibilidade ética.”
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
“Aleijados Biliares”
O Flagelo Silencioso da Lesão do Ducto Biliar na Colecistectomia Laparoscópica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 8 minutos)

Introdução
No âmbito da cirurgia digestiva, poucas complicações causam tanto temor aos cirurgiões quanto a lesão do ducto biliar (LDB) durante a colecistectomia laparoscópica. Apesar de ser o procedimento padrão-ouro para o tratamento da colelitíase sintomática, esta intervenção carrega um perigo oculto que pode transformar uma operação de rotina num evento devastador, alterando irreversivelmente a vida tanto do doente quanto do cirurgião — criando o que a literatura clássica tragicamente apelida de “aleijados biliares”.
O Paradoxo Laparoscópico e o Impacto Socioeconómico
A colecistectomia laparoscópica, introduzida no final da década de 1980, revolucionou a cirurgia da vesícula biliar, proporcionando recuperações mais rápidas e menor dor pós-operatória. No entanto, trouxe consigo um risco aumentado de LDB em comparação com a abordagem aberta tradicional. Dados de um estudo do Colégio Brasileiro de Cirurgiões relataram uma taxa de LDB de 0,18% em colecistectomias laparoscópicas, um valor ligeiramente inferior à média global que oscila entre 0,3% e 0,7%. Contudo, as consequências da LDB são de uma gravidade extrema. Frequentemente exigem cirurgia reconstrutiva complexa (como a hepaticojejunostomia em Y de Roux) e resultam em significativa morbilidade, mortalidade e custos de saúde astronómicos. Um estudo brasileiro estimou que os casos de LDB conduzem a uma média de 22 dias adicionais de internamento hospitalar e a um aumento de 30 vezes nos custos hospitalares associados.
Pontos-Chave na Prevenção e Gestão da LDB
Para mitigar este flagelo, a abordagem cirúrgica deve assentar em quatro pilares fundamentais:
1. Fatores de Risco
Compreender e antecipar os fatores relacionados com o doente (ex: colecistite aguda, obesidade grave, variações anatómicas) e com o cirurgião (ex: inexperiência, fadiga, interpretação errónea da anatomia ou “ilusão de ótica” cirúrgica) é absolutamente crucial para o planeamento operatório.
2. Estratégias de Prevenção Inegociáveis
- Visão Crítica de Segurança (CVS): Esta técnica de identificação anatómica, defendida pelo Dr. Steven Strasberg, é a pedra basilar na prevenção da LDB. Exige a limpeza do triângulo hepatocístico, a separação do terço inferior da vesícula da placa cística e a visualização de apenas duas estruturas a adentrar a vesícula.
- Colangiografia Intraoperatória (CIO): Embora ainda alvo de algum debate quanto à sua utilização universal, a CIO é uma ferramenta valiosa que pode auxiliar de forma decisiva na identificação da anatomia biliar e no diagnóstico precoce de lesões.
- “Cultura de Segurança”: É imperativo adotar uma mentalidade que priorize a segurança do doente acima da conclusão do procedimento por via laparoscópica a todo o custo. A conversão para cirurgia aberta ou a realização de uma colecistectomia subtotal são sinais de maturidade cirúrgica, e nunca de fracasso.
3. Reconhecimento e Manejo Adequado
O reconhecimento precoce da LDB, preferencialmente no período intraoperatório, é vital. O reencaminhamento atempado e o reparo imediato ou diferido realizado por cirurgiões hepatobiliares experientes e dedicados proporcionam, inequivocamente, os melhores resultados a longo prazo.
4. Formação e Educação Médica Contínua
O treino baseado em simulação e a adoção de protocolos padronizados de segurança devem fazer parte da matriz formativa de qualquer serviço, ajudando a reduzir de forma drástica as taxas de LDB, especialmente entre os médicos internos (residentes) de cirurgia geral e digestiva.
Conclusões Aplicadas
Como cirurgiões digestivos, devemos permanecer em vigilância constante contra a ameaça da lesão do ducto biliar. Ao aderirmos de forma obstinada às técnicas cirúrgicas adequadas, mantendo um elevado índice de suspeição e fomentando uma cultura de segurança inabalável, podemos minimizar esta complicação potencialmente devastadora. A jornada para o “zero LDB” está em andamento, exigindo educação contínua, autorreflexão e um compromisso diário com a excelência na nossa prática.
“O desafio da cirurgia é a mão do cirurgião curar um doente pela precisão na estrutura e propósito.” – Joseph E. Murray, cirurgião plástico, pioneiro dos transplantes e Prémio Nobel da Medicina.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe nas suas redes sociais e|ou mande a sua dúvida pelo 💬 Chat On-line na nossa DM do Instagram.
Endoscopia Gastrointestinal
Os endoscópios flexíveis modernos são verdadeiros prodígios da ciência médica. Esses dispositivos tubulares, finos e altamente flexíveis, iluminados por poderosas fontes de luz, possuem canais que permitem irrigação, sucção e limpeza de lentes, além de possibilitar a coleta de amostras de tecido, injeção de substâncias e a introdução de dispositivos auxiliares, como clipes e stents. Desde sua introdução ampla, essas ferramentas transformaram dramaticamente o cenário da prática cirúrgica. Procedimentos menos invasivos, realizados endoscopicamente em vez de cirurgicamente, revolucionaram a prática médica. A introdução da gastrostomia endoscópica percutânea (PEG) por Ponsky, a colangiopancreatografia retrógrada endoscópica (CPRE) descrita por McCune, a utilização do endoscópio flexível por Sugawa para identificar a origem de sangramentos gastrointestinais superiores durante a laparotomia, e o uso do endoscópio por Youmans Jr. para tratar sangramentos gastrointestinais superiores, marcaram o início de uma nova era. Cirurgiões sem habilidades endoscópicas começaram a perceber que estavam despreparados para o futuro da cirurgia digestiva.
A Explosão da Prática Endoscópica
Uma vez que essas ferramentas aumentaram o rendimento diagnóstico e a localização precisa de lesões, como cânceres do trato intestinal e locais de sangramento gastrointestinal, a prática endoscópica experimentou uma explosão contínua, especialmente após o advento da endoscopia terapêutica. Durante as décadas de 1970 e 1980, enquanto muitos cirurgiões digestivos focavam nos desenvolvimentos cirúrgicos tradicionais e em suas práticas operatórias intensas, outros continuaram a busca por métodos cada vez menos invasivos para diagnóstico e tratamento de problemas comuns, como demonstrado em vários estudos.
Sangramentos gastrointestinais, pólipos intestinais e cálculos no ducto biliar comum tornaram-se alvos dessas inovações. Logo, os métodos endoscópicos de tratamento mudaram o cenário da prática cirúrgica. O desenvolvimento contínuo da endoscopia flexível empurrou os limites do tratamento para além da sala de operações, e outros especialistas passaram a reivindicar essa ferramenta e o conjunto de procedimentos desenvolvidos em torno dela como seus, numa tentativa de excluir os cirurgiões da prática desse “estado da arte” em benefício de seus pacientes. No entanto, a endoscopia gastrointestinal flexível deve seu desenvolvimento à cirurgia e permanece bem estabelecida em nossa disciplina.
Conceitos Gerais
É impossível fornecer mais do que uma introdução a um campo tão amplo como a endoscopia cirúrgica em um texto único. Existem obras inteiras dedicadas a cada um dos procedimentos endoscópicos flexíveis comumente empregados na prática clínica. A aquisição de habilidades endoscópicas requer exposição clínica direta, uma experiência imersiva e o uso de uma ferramenta validada para avaliar a competência após o treinamento ministrado por um endoscopista experiente.
A Importância da Endoscopia na Cirurgia Gastrointestinal
A inclusão da endoscopia gastrointestinal flexível na prática cirúrgica melhora o desempenho e os resultados das cirurgias gastrointestinais. Em seu nível mais básico, o endoscópio é uma ferramenta que, quando usada para inspecionar ao final de uma operação envolvendo reconstrução ou anastomose do trato gastrointestinal, confirma para os pacientes e cirurgiões que os objetivos da cirurgia foram alcançados de forma segura. Todos os cirurgiões que operam no trato gastrointestinal devem ter grande facilidade no uso de endoscópios para testar anastomoses quanto a vazamentos, inspecionar superfícies mucosas para isquemia e avaliar a adequação de procedimentos reconstrutivos, como a fundoplicatura de Nissen, ou a completude de dissecações importantes para resultados ótimos, como durante a miotomia esofágica para acalasia. A endoscopia intraoperatória tornou-se uma parte crucial da cirurgia gastrointestinal.
Cirurgiões gerais, por exemplo, há muito tempo encontraram na endoscopia cirúrgica uma parte importante de suas práticas, oferecendo serviços de triagem, diagnóstico e terapêutica que, de outra forma, não estariam disponíveis em suas comunidades no interior do Brasil. Uma publicação de 2005 revelou que os cirurgiões gerais realizavam mais procedimentos endoscópicos do que procedimentos operatórios nos EUA, o que era essencial para suas comunidades e práticas. Esses resultados demonstraram também que, enquanto cirurgiões digestivos em centros urbanos realizavam menos procedimentos endoscópicos do que seus colegas cirurgiões gerais em centros menores, o número de procedimentos de endoscopia gastrointestinal flexível realizados por cirurgiões urbanos também era substancial, reafirmando que a endoscopia continua sendo uma ferramenta importante para o cuidado do paciente cirúrgico em todas as situações.
Impactos Econômicos e a Relevância Futura
Independentemente do ambiente de prática, os cirurgiões devem ser incentivados a manter suas habilidades endoscópicas e incorporá-las à prática para o benefício de seus pacientes e comunidades. Em meio à incerteza sobre políticas futuras que orientarão o sistema de saúde e a formação médica, é sensato, do ponto de vista econômico, colocar endoscópios flexíveis nas mãos dos cirurgiões digestivos. Estudos sobre a força de trabalho em saúde sugerem que a combinação de capacidades de especialistas e a superação das barreiras tradicionais de cuidado podem ser mais eficientes e melhorar a qualidade e a rapidez do atendimento.
Cirurgiões que combinam seu extenso conhecimento e experiência no tratamento de condições e sintomas, como doença do refluxo gastroesofágico, hemorragia gastrointestinal, malignidade gastrointestinal, disfagia e dor abdominal, provavelmente proporcionarão uma abordagem mais eficiente em termos de custos e resultados para esses pacientes, quando envolvidos precocemente, devido ao amplo espectro de capacidades diagnósticas e terapêuticas disponíveis. Ao eliminar atrasos e consultas desnecessárias com outros especialistas, o cirurgião é capaz de montar rapidamente uma avaliação que beneficia o paciente de maneira econômica.
Incorporar a endoscopia gastrointestinal flexível na prática cirúrgica atende às necessidades de nossos pacientes hoje e será um conjunto de habilidades cada vez mais importante para as futuras gerações de cirurgiões. O endoscópio flexível emergiu como uma plataforma empolgante sobre a qual se basearão futuros procedimentos cirúrgicos minimamente invasivos. Considerem o impacto positivo da triagem colonoscópica eficaz e da ablação endoscópica para pacientes com esôfago de Barrett, por exemplo, e o uso da ressecção endoscópica de mucosa (EMR) ou da dissecção endoscópica submucosa (ESD) para aqueles com câncer precoce de reto ou esôfago. Abordagens cirúrgicas tradicionais continuarão a ser menos necessárias, e terapias menos invasivas, baseadas na plataforma de endoscopia gastrointestinal flexível, emergirão como novos padrões em nossa abordagem a muitos pacientes. Cirurgiões digestivos que não desenvolvem e mantêm suas habilidades em endoscopia gastrointestinal perderão a oportunidade de participar da próxima época de nossa especialidade.
Considerações Práticas e Procedimentos Específicos
Aqueles que não aprenderam endoscopia durante o treinamento devem fazer esforços especiais para adquirir esse conhecimento e ganhar experiência clínica. A busca por oportunidades de fellowship está disponível para cirurgiões que podem se afastar de sua prática, e programas individualizados de instrução e avaliação são possíveis, variando de local para local. A mentoria é um marco da comunidade cirúrgica, e cirurgiões interessados em treinamento adicional e educação contínua visando à obtenção de maestria em novas habilidades devem buscar arranjos de mentoria que funcionem melhor em seus ambientes individuais.
Por fim, embora seja uma tarefa impossível discutir em detalhes todas as técnicas endoscópicas básicas e avançadas, os procedimentos fundamentais que serão empregados pela maioria dos cirurgiões são a esofagogastroduodenoscopia (EGD) e a colonoscopia. Estes devem ser aprendidos e dominados para que os cirurgiões possam oferecer aos pacientes uma escolha ótima de procedimentos e excelentes resultados clínicos.
Gostou ❔ Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Princípios Anatômicos da Laparotomia Exploradora
Introdução
A laparotomia, termo utilizado para descrever qualquer acesso aberto à cavidade peritoneal, abrange incisões na linha média, bem como abordagens paramedianas e oblíquas. É o método tradicional de acesso para a maioria das cirurgias viscerais, ainda sendo a abordagem preferida em casos de trauma, emergências e cirurgias extensas. A laparotomia, além de permitir um acesso amplo e direto, requer uma compreensão detalhada dos princípios anatômicos e técnicos que a sustentam, garantindo a eficácia e segurança do procedimento.
Preparação Pré-operatória
A preparação para uma laparotomia começa com uma série de medidas essenciais que visam otimizar as condições cirúrgicas e a segurança do paciente:
- Anestesia Geral (GA): A maioria das incisões laparotômicas, exceto as menores na porção inferior do abdômen, requer anestesia geral para proporcionar relaxamento muscular adequado e analgesia.
- Tipagem sanguínea e reserva: Dependendo do procedimento planejado, é necessário realizar tipagem e reserva ou compatibilização cruzada de sangue.
- Consentimento informado: Garantir que o paciente tenha consentido para outros procedimentos que possam ser necessários durante a cirurgia.
Posicionamento e Configuração da Sala Cirúrgica
- Cateterização uretral: Recomendada para a maioria das laparotomias, especialmente se houver patologia abdominal inferior, para garantir a descompressão da bexiga.
- Sonda nasogástrica (SNG): Indicada em casos de obstrução intestinal superior, para reduzir o risco de aspiração durante a indução anestésica.
- Posicionamento da mesa cirúrgica: A posição supina é a mais comum para cirurgias viscerais abertas, como aquelas envolvendo intestino delgado, intestino grosso e estômago. Outras posições, como Lloyd-Davis (supina com leve flexão e abdução dos quadris), podem ser utilizadas para acesso ao períneo e reto.
Passos da Cirurgia
- Incisão cutânea: Realizada com bisturi ou diatermia de corte com eletrodo de ponta fina, conforme a abordagem cirúrgica escolhida.
- Incisão no tecido adiposo: Utiliza-se diatermia combinada para reduzir o risco de sangramento.
Acesso pela Linha Média
- Incisão na fáscia da linha média: Realizada na linha alba, onde as fibras fasciais cruzadas obliquamente são identificadas e expostas. A fáscia é elevada com pinças para gerar pressão intra-abdominal negativa e incisa de forma aguda.
- Extensão do acesso: Realizada com diatermia combinada na linha média.
Acesso Paramediano
- Incisão na fáscia da bainha do reto: Realizada verticalmente com diatermia combinada.
- Musculatura do reto: As fibras musculares são separadas minimamente, sem divisão muscular significativa.
- Incisão peritoneal: Realizada após elevação com pinças e incisão aguda.
- Extensão do acesso: Com diatermia combinada verticalmente.
Acesso Oblíquo (e.g., Gridiron, Subcostal)
- Incisão na fáscia da bainha do reto: Realizada de forma oblíqua com diatermia combinada.
- Musculatura: Para incisões menores, a separação das fibras pode proporcionar acesso adequado. Incisões maiores podem requerer divisão muscular com diatermia de coagulação.
Procedimentos Básicos
- Avaliação das vísceras não-alvo: Embora a importância dessa prática tenha diminuído com o advento da imagem pré-operatória (como a tomografia computadorizada), ainda é realizada em uma progressão lógica, avaliando-se o intestino delgado, omento, cólon transverso, baço, estômago, fígado, vesícula biliar, rins, bexiga, útero e ovários.
- Avaliação do órgão-alvo: Deve-se considerar a ressecabilidade, extensão da ressecção e mobilidade das estruturas a serem anastomosadas.
Princípios Chave da Laparotomia de Emergência
- Controle de sangramento: Deve-se inicialmente controlar o sangramento por meio de pressão com compressas, removendo-as gradualmente para identificar e tratar os locais de sangramento.
- Lesões viscerais múltiplas: Prioriza-se o controle e fechamento das lesões ao invés da restauração anatômica imediata, que pode ser postergada para procedimentos subsequentes.
- Contaminação: Todas as áreas de contaminação devem ser identificadas e tratadas, com irrigação copiosa e uso de drenos de grande calibre para áreas altamente contaminadas.
Fechamento
- Peritônio: Deve ser aproximado, se possível, para reduzir o risco de aderências.
- Fáscia: Sempre fechada, geralmente com suturas absorvíveis pesadas.
- Pele: Pode ser fechada com suturas subcuticulares ou interrompidas, ou com clipes.
Complicações Específicas
As complicações associadas à laparotomia incluem infecção da ferida, com incidência de 2 a 30%, dependendo da patologia, e hérnia incisional, que pode ocorrer em até 30% dos casos, especialmente em pacientes com sepse, desnutrição ou idade avançada.
Conclusão
A laparotomia exploradora é um procedimento fundamental na cirurgia abdominal, exigindo um conhecimento profundo da anatomia e uma abordagem técnica precisa para garantir o sucesso cirúrgico e a segurança do paciente. Desde a preparação pré-operatória até o fechamento da incisão, cada passo deve ser realizado com atenção aos princípios anatômicos e técnicos, prevenindo complicações e otimizando os resultados.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
#LaparotomiaExploradora #CirurgiaAbdominal #EducaçãoMédicaContinuada #AnatomiaCirúrgica #ProcedimentosCirúrgicos
Cirurgia Robótica
Introdução
A natureza humana do cirurgião sempre foi alcançar o máximo acesso à área de interesse cirúrgico enquanto causa o mínimo trauma ao paciente. Com a evolução das técnicas operatórias e tecnologias, essa capacidade avançou enormemente. Inicialmente, os cirurgiões desenvolveram a habilidade técnica para realizar incisões menores e mais estratégicas para o acesso cirúrgico aberto. Posteriormente, com a utilização de telescópios cirúrgicos e ferramentas de acesso mínimo, e agora com tecnologias assistidas por computador e robótica, os cirurgiões conseguem acessar áreas cirúrgicas de difícil abordagem com trauma secundário quase insignificante ao paciente.
A Revolução da Cirurgia Minimamente Invasiva
A revolução da cirurgia minimamente invasiva começou há três décadas com o advento da tecnologia de vídeo. Desde então, a abordagem laparoscópica a uma ampla variedade de procedimentos cirúrgicos tornou-se a escolha preferida, permitindo a realização de operações através de pequenas incisões, videotelescópios e instrumentos longos. Os benefícios dessas técnicas incluem menor tempo de internação, menos dor e cicatrizes, menor risco de infecção, menos perda de sangue e transfusões, retorno acelerado às atividades normais e recuperação mais rápida.
No entanto, a cirurgia laparoscópica exige um conjunto único de habilidades que pode ser difícil para alguns cirurgiões adquirirem. As limitações técnicas incluem falta de visualização tridimensional (3D), alcance limitado de movimento devido aos instrumentos rígidos, ergonomia pobre, movimentos contraintuitivos, amplificação do tremor fisiológico, sensibilidade tátil reduzida e o efeito fulcro.
Avanços com a Integração Robótica
A integração robótica permite que os cirurgiões realizem procedimentos minimamente invasivos mais complexos com visualização 3D aprimorada, maior destreza, aumento do alcance de movimento, melhor ergonomia e acesso superior a áreas de difícil alcance no corpo. Isso se traduz em procedimentos mais precisos e menos invasivos. A próxima geração de robôs está sendo desenvolvida para serem menores, mais inteligentes e com custos reduzidos, tornando-se cada vez mais comuns nas salas de cirurgia.
Para o paciente, um procedimento assistido por robô oferece todos os benefícios potenciais de uma operação minimamente invasiva. Estudos clínicos sugerem que a robótica pode ajudar os cirurgiões a proporcionar melhores resultados clínicos em certos contextos específicos.
Perspectivas Futuras e Desafios
A tecnologia robótica também possibilita a separação do cirurgião do paciente, seja em metros ou milhares de quilômetros. A teles cirurgia e o telementoring têm sido testados e demonstraram ser viáveis e benéficos. Exemplos incluem a remoção de uma vesícula biliar através do Oceano Atlântico e o mentoreamento de cirurgiões no Canadá. Outra tecnologia permite ao cirurgião realizar rondas remotamente, controlando movimentos robóticos via internet.
Os desafios incluem o desenvolvimento e aprimoramento das tecnologias e suas limitações, o treinamento e a expertise dos médicos para usar essas ferramentas de forma segura e eficaz, a incorporação das tecnologias mais recentes na educação cirúrgica e a gestão dos custos aumentados de maneira que permita maior acesso dos pacientes aos melhores cuidados disponíveis.
Contexto Brasileiro
No Brasil, a cirurgia robótica tem avançado significativamente, com instituições adotando plataformas robóticas para procedimentos complexos. Dados da Sociedade Brasileira de Cirurgia Minimamente Invasiva e Robótica (SOBRACIL) indicam um aumento no número de procedimentos realizados com auxílio de robôs, refletindo os avanços tecnológicos e os benefícios clínicos associados.
Conclusão
A cirurgia robótica representa uma evolução significativa na capacidade dos cirurgiões de realizar procedimentos complexos com precisão e segurança aprimoradas. À medida que a tecnologia avança, espera-se que a adoção de plataformas robóticas se torne cada vez mais comum, proporcionando aos pacientes os benefícios de técnicas minimamente invasivas e resultados clínicos superiores.
“A inovação é a capacidade de ver a mudança como uma oportunidade – não uma ameaça.” – Steve Jobs
Gostou? Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags
#CirurgiaRobótica #TecnologiaMédica #InovaçãoCirúrgica #Saúde #EducaçãoMédica
A Morfologia Humana e suas Implicações na Estratégia Cirúrgica do Aparelho Digestório

Como cirurgiões do aparelho digestivo, é fundamental compreendermos a morfologia humana e suas implicações na estratégia cirúrgica. Assim como Sun Tzu enfatizou a importância do conhecimento do terreno e das condições de batalha para obter vitórias em operações militares, nós, enquanto cirurgiões, devemos dominar a anatomia e a fisiologia do aparelho digestivo para planejar e executar cirurgias com sucesso. A classificação de Sun Tzu dos diferentes tipos de terreno, como acessível, traiçoeiro, duvidoso, estreito, acidentado e distante, pode ser correlacionada com as variações anatômicas encontradas no aparelho digestivo.
Por exemplo, o estômago e o intestino delgado, com suas curvaturas e alças, podem ser considerados terrenos “acidentados”, exigindo maior destreza cirúrgica para navegação. Por outro lado, o reto e o cólon descendente, com seu trajeto mais retilíneo, podem ser vistos como terrenos “acessíveis e estreitos”. Já o fígado é “acessível”, mas o pâncreas é “distante e traiçoeiro” e as vias biliares – devido ao número expressivo de variações anatômicas – como terreno “duvidoso.”
Além disso, a compreensão das seis situações que apontam a derrota de um exército, como soldados desorganizados, disciplina negligente e oficiais fracos, pode ser aplicada à equipe cirúrgica. Um cirurgião habilidoso deve liderar sua equipe com eficácia, garantindo que todos os membros estejam bem treinados, disciplinados e trabalhando em harmonia para alcançar o objetivo comum de um resultado cirúrgico bem-sucedido.
Assim como Sun Tzu enfatizou a importância de conhecer o inimigo e a si mesmo para obter a vitória, nós, cirurgiões, devemos conhecer profundamente a anatomia e a fisiologia do aparelho digestivo, bem como nossas próprias habilidades e limitações. Essa compreensão nos permite planejar estratégias cirúrgicas adequadas, antecipando possíveis desafios e adaptando nossa abordagem conforme necessário.
Ao operar em “território inimigo”, ou seja, dentro da região abdominal distorcida por uma afecção, é crucial seguir os princípios gerais de Sun Tzu, como manter a coesão da equipe, garantir provisões adequadas (instrumentos e suprimentos cirúrgicos), manter o moral alto e conservar as energias da equipe. Isso nos permite executar cirurgias com eficiência e segurança, mesmo em situações desafiadoras de emergência.
Em suma, a morfologia humana e suas implicações na estratégia cirúrgica do aparelho digestivo são fundamentais para o sucesso de nossas intervenções. Ao aplicarmos os princípios de Sun Tzu, como conhecer o terreno (anatomia), adaptar nossas táticas às condições (variações anatômicas) e liderar nossa equipe com eficácia, podemos alcançar melhores resultados para nossos pacientes.
Como disse o lendário cirurgião William Halsted: “A cirurgia é uma ciência aprendida pela prática e aperfeiçoada pela anatomia”.Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram
#CirurgiaDigestiva #AnatomiaQuirúrgica #EstrategiaCirúrgica #AparelhoDigestivo #MorfologiaHumana
Prevenção da Lesão da Via Biliar
INTRODUÇÃO
Carl Langenbuch é creditado como o primeiro cirurgião a realizar uma colecistectomia aberta (CA) em 1882. Ele havia feito suas pesquisas em animais e cadáveres antes de realizar o primeiro procedimento em um ser humano. Langenbuch postulou que a remoção da vesícula biliar resultaria na extração dos cálculos biliares e do órgão que os produzia. Em 1985, E. Muhe, de Boblingen, Alemanha, realizou a primeira colecistectomia laparoscópica (CL), mas enfrentou grande oposição de seus colegas.
Três anos depois, um ginecologista francês, P. Mouret, realizou uma CL, o que influenciou F. Dubois e J. Perissat a desenvolverem suas técnicas para essa abordagem. A popularização dessa técnica nos Estados Unidos deve ser creditada a E.J. Reddick e D.O. Olsen, de Nashville, Tennessee, que realizaram seu primeiro caso em 1988 e estabeleceram os princípios da operação como ela é conhecida atualmente. Durante a década de 1990, foram descritas tentativas de reduzir ainda mais a abordagem laparoscópica minimamente invasiva para uma única incisão. O uso de trocartes e instrumentos de pequeno diâmetro, de 2 a 3 mm, conhecido como técnica de needlescopic, também foi experimentado. Nenhuma dessas técnicas ganhou aceitação geral devido à falta de benefícios comprovados.
A CL tornou-se o primeiro procedimento de uma revolução que mudou a maneira como a cirurgia abdominal estava sendo realizada. Nos 15 anos seguintes, uma abordagem laparoscópica foi relatada como viável para quase todos os procedimentos abdominais. Esse avanço resultou em um benefício significativo para o paciente na maioria dos procedimentos, devido às vantagens inerentes da técnica laparoscópica. Para a CL, no entanto, ainda há um risco aumentado de lesão do ducto biliar (LDB) em comparação com a agora histórica CA. O denominador comum na ocorrência de LDB é a falha em identificar claramente a anatomia do triângulo de Calot. Embora essa desvantagem persistente da CL não justifique a realização de uma CA, ela precisa ser continuamente abordada. Passos para prevenir LDB foram descritos nos primeiros anos da CL, e em 1995 Strassberg descreveu o termo “visão crítica de segurança” como o passo mais importante na prevenção de LDB durante o procedimento.
Passos Técnicos Importantes para Evitar Lesões do Ducto Biliar
A partir da técnica cirúrgica descrita, os principais passos para evitar lesões do ducto biliar (LDB) são:
- Máxima tração cefálica do fundo da vesícula biliar.
- Retração lateral e inferior (em direção ao pé direito) da bolsa de Hartmann, puxando-a para longe do fígado e evitando o alinhamento do ducto cístico com o ducto biliar comum.
- Inicie a dissecção na parte superior do colo da vesícula biliar e conduza-a de lateral para medial.
- Gire a bolsa de Hartmann medialmente para uma dissecção póstero-lateral da serosa da vesícula biliar.
- Liberte o colo da vesícula biliar do leito hepático, criando uma janela que seja tão grande quanto necessário para expor claramente a anatomia do triângulo de Calot e obter uma visão crítica de segurança.
- Coloque os clipes sob visualização direta de ambos os ramos em uma direção medial para lateral, a partir do porto subxifoide.
- Realize a colangiografia intraoperatória se houver quaisquer dúvidas sobre a anatomia biliar.
- Ao iniciar o descolamento subsequente da vesícula biliar do leito hepático, mantenha a dissecção próxima à parede da vesícula biliar e longe das estruturas do hilo do fígado. O uso criterioso de eletrocautério é recomendado.
Dicas e Truques para Evitar Complicações
- Assuma que cada caso tem um ducto cístico curto e/ou outras anomalias anatômicas. O objetivo é a dissecção do ducto cístico começando na junção com a vesícula biliar.
- Use a técnica da visão crítica de segurança para limpar o triângulo de Calot e individualizar, identificar e isolar completamente o ducto cístico e a artéria antes de dividi-los.
- Durante toda a operação, sempre tenha em mente que sua interpretação da anatomia pode estar errada ou equivocada; isso serve como uma salvaguarda para prevenir lesões dos ductos biliares (LDBs).
- Uma reação de pânico ao sangramento, resultando na aplicação de vários clipes ou cauterização em massa, leva ao desastre. A compressão, irrigação e sucção, e o uso de uma pinça de bico de pato para pegar o ponto de sangramento são recomendados. Esteja ciente do aumento de 16 vezes da visão laparoscópica.
- Cuidado com a retração excessiva da bolsa de Hartmann, que pode levar à interpretação errada do ducto biliar comum como sendo o ducto cístico. Ao se deparar com um ducto cístico largo, a colangiografia é obrigatória para garantir que é o ducto cístico e não o ducto biliar comum. Para o fechamento, podem ser colocadas 2 suturas endoloop em vez de clipes de titânio regulares.
Diretrizes de Tóquio 2018: Passos Seguros Durante Colecistectomia para Colecistite Aguda
As Diretrizes de Tóquio 2018 (TG18) foram uma conferência de consenso com mais de 60 especialistas de todo o mundo. Um dos objetivos do TG18 foi estratificar a colecistite aguda por gravidade e recomendar o tratamento com base na severidade. Os passos seguros do TG18 durante a colecistectomia para colecistite incluem os seguintes:
- Se a vesícula biliar estiver distendida e interferir na visão, ela deve ser descomprimida por aspiração com agulha.
- Retração eficaz da vesícula biliar para desenvolver um plano na área do triângulo de Calot e identificar seus limites (contra-tração).
- Iniciar a dissecção a partir da folha posterior do peritônio que cobre o colo da vesícula biliar e expor a superfície da vesícula biliar acima do sulco de Rouvière.
- Manter o plano de dissecção na superfície da vesícula biliar durante toda a colecistectomia laparoscópica (CL).
- Dissecar a parte inferior do leito da vesícula biliar (pelo menos um terço) para obter a visão crítica de segurança (CVS).
- Criar a visão crítica de segurança (CVS).
- Para hemorragia persistente, obter hemostasia principalmente por compressão e evitar o uso excessivo de eletrocautério ou clipes.
Lesão do Ducto Biliar (LDB) na Colecistectomia: Desafios e Prevenção
A lesão do ducto biliar (LDB) é uma complicação muito séria da colecistectomia. Após a curva de aprendizado inicial da laparoscopia (50 casos), estudos mais recentes indicam que as taxas de LDB durante a colecistectomia laparoscópica variam entre 0,08% e 0,5%, geralmente mais altas do que as taxas relatadas para a colecistectomia aberta (0,1%–0,2%). Contudo, a LDB pode ser subestimada em cirurgias abertas. A maioria das lesões de LDB (97%) resulta de erros na percepção visual, com falhas nas habilidades técnicas representando apenas 3%. Aproximadamente 25% das lesões são reconhecidas durante a operação inicial. Fatores que contribuem para lesões iatrogênicas incluem tração excessiva que distorce a anatomia, anomalias anatômicas, presença de inflamação, uso inadequado de cautério ou problemas relacionados ao controle de hemorragia intraoperatória, entre outros. Além disso, graus mais altos de gravidade da colecistite aguda estão associados a um maior risco de LDB. O manejo depende do momento em que a lesão é reconhecida, da localização da lesão e se há lesão vascular concomitante.
PREVENÇÃO DA LESÃO DO DUCTO BILIAR
Uma técnica cirúrgica adequada, conhecimento de possíveis anomalias anatômicas, obtenção da visão crítica de segurança e uso de colangiografia radiográfica e/ou fluorescente são medidas que ajudam a evitar a LDB. A visão crítica de segurança foi descrita por Strasberg e colegas como um método de identificação consistente do ducto cístico e da artéria. No entanto, pode ser difícil de obter em casos com inflamação severa. Consiste em cumprir 3 critérios: (1) o triângulo hepatocístico deve estar livre de gordura e tecido fibroso, (2) o terço inferior da vesícula biliar deve estar separado do fígado para expor a placa cística, e (3) dois e apenas dois estruturas devem ser vistas entrando na vesícula biliar. Essas duas estruturas devem ser visualizadas anteriormente e posteriormente com um laparoscópio angular ou “balançando” o fundo da vesícula. A visão crítica de segurança é amplamente apoiada pela opinião de especialistas como o método mais confiável de identificação anatômica, e, portanto, é recomendada como uma diretriz importante para colecistectomias laparoscópicas.
Se houver incerteza sobre a anatomia biliar ou suspeita de anomalias anatômicas, a colangiografia via vesícula biliar ou ducto cístico deve ser usada, se possível, para esclarecer as estruturas. Relatórios mostram que o uso de colangiografia pode reduzir o risco de lesões do ducto biliar, embora alguns estudos não tenham encontrado associação. No entanto, se uma lesão biliar for suspeitada intraoperatoriamente, a colangiografia intraoperatória é eficaz para aumentar as chances de reconhecimento precoce, melhorando os resultados no tratamento da LDB. A Sociedade Americana de Cirurgiões Gastrointestinais e Endoscópicos recomenda o “uso liberal” de colangiografia ou outras modalidades para delinear com precisão a anatomia cirúrgica. Alguns defensores sugerem o uso rotineiro de colangiografia, citando não apenas uma menor taxa de LDB, mas também a prevenção de lesões excisionais mais graves. A anatomia biliar também pode ser visualizada usando ultrassonografia intraoperatória, se a experiência estiver disponível.
Conclusão
A colecistectomia é um dos procedimentos da cirurgia do aparelho digestivo que mais comumente são realizados em todo o mundo, mas pode estar repleta de potenciais problemas. O Safe Cholecystectomy Task Force da Sociedade Americana de Cirurgiões Gastrointestinais Endoscópicos identificou vários fatores importantes para garantir a segurança na colecistectomia laparoscópica:
- Estabelecimento da visão crítica de segurança
- Compreensão da anatomia
- Obtenção de exposição adequada
- Solicitação de ajuda a um colega mais experiente
- Reconhecimento de quando converter ou abandonar o procedimento
As complicações após a colecistectomia incluem lesões ou vazamentos do ducto biliar, sangramento, lesões vasculobiliares combinadas, cálculos biliares retidos, cálculos biliares derramados, abscessos, estenoses, hérnias e síndrome pós-colecistectomia (PCS). Embora as taxas dessas complicações sejam geralmente muito baixas, a conscientização e uma alta suspeita clínica quando um paciente apresenta sintomas semanas, meses ou anos após a colecistectomia podem ajudar no diagnóstico e tratamento adequado.
Colecistectomia Segura: Preditores de Dificuldade Técnica
Introdução
A colecistectomia laparoscópica é amplamente reconhecida por suas vantagens sobre a colecistectomia aberta, incluindo redução da dor pós-operatória, estadias hospitalares mais curtas e tempos de recuperação mais rápidos. No entanto, a transição para técnicas laparoscópicas introduziu um maior risco de lesões do ducto biliar, com incidências relatadas sendo duas a três vezes maiores do que em procedimentos abertos. Dadas as graves consequências associadas à LDB, identificar preditores de colecistectomia difícil e implementar estratégias de manejo apropriadas são cruciais para melhorar os resultados dos pacientes.
Preditores Pré-operatórios de Colecistectomia Difícil
Gênero: O gênero masculino é um preditor significativo de colecistectomia difícil. Análises retrospectivas indicam taxas mais altas de aderências, desafios anatômicos, taxas de conversão e inflamação aguda ou crônica em pacientes do sexo masculino. Fatores contribuintes possíveis incluem a consulta médica tardia e a colecistite recorrente, levando a um aumento da fibrose. Além disso, diferenças fisiológicas, como maior massa muscular e menor circunferência torácica, podem complicar o procedimento.
Idade: A idade avançada (acima de 65-70 anos) está associada a taxas de conversão mais altas devido a históricos mais longos de doença calculosa biliar e frequentes ataques de colecistite. Apesar desses desafios, a colecistectomia laparoscópica em pacientes idosos geralmente resulta em menos complicações em comparação com procedimentos abertos.
Obesidade: O IMC mais alto introduz várias dificuldades técnicas, como obter acesso para a criação do pneumoperitônio, retração do fundo da vesícula biliar e dissecação do triângulo de Calot em um ambiente com excesso de gordura. Pacientes obesos também necessitam de cuidados anestésicos e de enfermagem especializados. Embora se notem tempos operatórios aumentados e taxas de conversão mais altas, a colecistectomia laparoscópica ainda oferece menor morbidade pós-operatória em comparação com a colecistectomia aberta.
Histórico de Colecistite Aguda, Colangite, Pancreatite e Icterícia: Essas condições levam a cicatrizes e fibrose, complicando a delineação anatômica e a dissecação. A colecistite recorrente pode encurtar o ducto cístico, fazendo com que a vesícula biliar pareça drenar diretamente no ducto biliar comum (DBC). A pancreatite aguda, embora não afete diretamente a vesícula biliar, pode causar aderências que dificultam a exposição operatória. A colangiopancreatografia retrógrada endoscópica (CPRE) prévia com stent também pode contribuir para dificuldades intraoperatórias.
Cirrose: A cirrose hepática apresenta um risco significativo de hemorragia devido a colaterais dilatados no triângulo de Calot, coagulopatia e trombocitopenia. A menor complacência do fígado fibroso complica ainda mais a retração da vesícula biliar e a exposição do triângulo de Calot. O uso de tesouras ultrassônicas e a manutenção de uma hemostasia meticulosa são essenciais nesses casos.
Laparotomia Anterior: Cirurgias abdominais anteriores, especialmente múltiplos procedimentos abdominais superiores, frequentemente resultam em aderências, complicando o acesso ao local operatório. O planejamento pré-operatório para o acesso peritoneal e o aconselhamento do paciente sobre a possibilidade de conversão são imperativos.
Fatores Intraoperatórios que Predizem Dificuldade
Aderências densas, vesícula biliar tensa (mucocele/empiema), colecistite aguda, anatomia pouco clara, triângulo de Calot congelado, síndrome de Mirizzi, fístula colecistoentérica, vesícula biliar intra-hepática e cálculos biliares grandes complicam significativamente a colecistectomia laparoscópica. O nível de dificuldade varia com a experiência do cirurgião. Aderências no triângulo de Calot, em particular, apresentam o maior risco de conversão para cirurgia aberta.
Estratégias de Manejo
Acesso Difícil: Locais alternativos para a criação do pneumoperitônio, como o ponto de Palmer, podem ser utilizados em pacientes com cirurgias abdominais superiores anteriores ou obesidade. As aderências devem ser abordadas com dissecação afiada ou métodos rombos, dependendo de sua densidade.
Estratégias de Resgate: Em casos em que a dissecção segura não é viável, estratégias como colecistectomia subtotal, colecistectomia do fundo para a tríade, colecistostomia tubular ou conversão para cirurgia aberta são recomendadas. A colangiografia intraoperatória ou ultrassonografia laparoscópica podem auxiliar na compreensão da anatomia biliar.
Síndrome de Mirizzi e Fístula Colecisto-colédoco: O manejo depende do tipo de síndrome de Mirizzi. A colecistectomia total ou subtotal é tipicamente suficiente para o Tipo I, enquanto casos mais complexos (Tipos II-IV) podem necessitar de coledocoplastia ou anastomose bilioentérica.
Conclusão
A transição para a colecistectomia laparoscópica, embora benéfica, introduz maiores riscos de complicações, como a lesão do ducto biliar. Identificar preditores pré-operatórios e intraoperatórios de colecistectomia difícil é essencial para planejar e executar intervenções cirúrgicas seguras. Os cirurgiões devem estar preparados para implementar várias estratégias de manejo para minimizar os riscos e garantir a segurança do paciente. Como Lahey afirmou apropriadamente, “O cirurgião que vê diante de si apenas a anatomia normal enfrentará mais dificuldades.”

Colecistectomia Segura: Plano R4U
O Imperativo do Sulco de Rouvière e a Visão Crítica de Segurança
Autor: Prof. Dr. Ozimo Gama
Afiliação: Membro Titular do Colégio Brasileiro de Cirurgiões
Membro Titular do Colégio Brasileiro de Cirurgia Digestiva
Introdução
A colecistectomia laparoscópica (CL) permanece como um dos procedimentos cirúrgicos mais executados globalmente. No entanto, sua ubiquidade não mitiga riscos inerentes; a variação anatômica dos ductos biliares, combinada à identificação visual incorreta, continua a precipitar complicações graves, notadamente as lesões iatrogênicas das vias biliares[1]. Neste cenário, a segurança do paciente transcende a destreza manual, residindo na identificação rigorosa de marcos anatômicos imutáveis. Entre estes, o sulco de Rouvière (SR) emerge não apenas como detalhe morfológico, mas como farol essencial para a orientação cirúrgica segura[2].
A Nomenclatura e a Realidade Anatômica
A precisão cirúrgica exige precisão anatômica. Na literatura, o SR é referenciado sob diversas nomenclaturas, incluindo “incisura dextra de Gans”[7] e, originalmente por Henri Rouvière, como “le sillon du processus caudé”[5]. Embora textos clássicos apresentem dados inconsistentes, estudos contemporâneos demonstram a presença do SR em até 80% dos fígados[3,6]. Tal prevalência torna o reconhecimento deste marco uma competência obrigatória. A relevância topográfica do SR é crítica, estabelecendo um divisor de águas anatômico: o ducto e a artéria cística localizam-se anterossuperiormente ao sulco, enquanto o ducto biliar comum (CBD) reside abaixo do seu nível. Evidências sugerem que dissecções iniciadas ventralmente ao SR reduzem significativamente a incidência de lesões ao CBD[2]. Ademais, o marco é instrumental para a execução segura de ressecções hepáticas segmentares[4].
A Integração com a Visão Crítica de Segurança (CVS)
A técnica da Critical View of Safety (CVS), preconizada por Strasberg, é o padrão ouro para minimizar riscos de lesão biliar[1]. Contudo, a aplicação da CVS deve ser ancorada em marcos confiáveis. Variações da árvore biliar extra-hepática afetam até um terço dos pacientes[3], mas o SR tende a manter-se no mesmo plano do ducto hepático comum e do CBD, facilitando a orientação tridimensional. A identificação do SR deve ser o passo preliminar que habilita a construção segura da CVS, mormente em casos de anatomia distorcida por inflamação.
Estratégias de Contingência: A Linha R4U
Em casos onde o SR está obscurecido por fibrose ou aderências, a segurança não pode ser comprometida. A linha R4U — estendendo-se da fissura umbilical para a direita do hilo — apresenta-se como alternativa viável para delimitar um plano seguro de dissecção[2]. Todavia, a utilização eficaz da linha R4U exige profundo conhecimento da anatomia hepática e treinamento contínuo em técnicas de segurança.
Conclusão
A prevenção de lesões na CL exige uma sistematização que transcende a técnica básica. A identificação rotineira do Sulco de Rouvière e a adesão estrita à CVS são imperativos éticos e técnicos. Ao integrar o conhecimento anatômico clássico com estratégias modernas, elevamos o padrão de cuidado. Como nos lembra a sabedoria cirúrgica secular:
“O cirurgião que ignora a anatomia esculpe o corpo humano como um cego esculpe a madeira.” — Guy de Chauliac (1300–1368), Chirurgia Magna.
Referências Bibliográficas
Referências
- Strasberg, S. M., et al. “The Critical View of Safety in Laparoscopic Cholecystectomy.”
- Hugh, T. B. “Anatomical Landmarks for Safe Laparoscopic Cholecystectomy.”
- Zubair, M., et al. “Anatomical Variations in the Extrahepatic Biliary Tree.”
- Couinaud, C. “Segmental Anatomy of the Liver.”
- Rouvière, H. “Le sillon du processus caudé.”
- Reynaud, C., Coucoravas, G., Giuly, J., et al. “Incisura Dextra de Gans.”
- Hugh, T. B. “Minimal Common Bile Duct Injury During Laparoscopic Cholecystectomy.”
- Gans, H. “Incisura Dextra in Classic Anatomical Literature.”
Variações Anatômicas da Árvore Biliar Extra-Hepática no Contexto da Colecistectomia Laparoscópica
Introdução A compreensão das variações anatômicas dos ductos biliares extra-hepáticos é essencial para a realização segura da colecistectomia laparoscópica. A terminologia frequentemente utilizada para descrever essas variações inclui termos como “acessório”, “anômalo” e “aberrante”. Entretanto, essas variações são naturais e suficientemente frequentes para serem antecipadas pelo cirurgião atento. A preferência é pelo termo “duto variante”.
Marcos Anatômicos Gerais para a Orientação
Durante a colecistectomia laparoscópica, é crucial reconhecer marcos anatômicos adjacentes para evitar lesões. O ligamento falciforme situa-se entre os segmentos hepáticos 3 e 4, e o ducto hepático comum (DHC) está próximo ao plano mediano do fígado entre os segmentos 4 e 5. O sulco de Rouvière é uma fissura que contém porções do pedículo portal direito e demarca o plano ventral-dorsal do ducto biliar comum (DBC), auxiliando na orientação durante a dissecção.

Anatomia dos Ductos Hepáticos Direitos
Na configuração mais comum, o ducto hepático seccional anterior direito (segmentos 5 e 8) e o ducto hepático seccional posterior direito (segmentos 6 e 7) unem-se para formar o ducto hepático direito (DHD), que se junta ao ducto hepático esquerdo para formar o DHC. Essa anatomia ocorre em 57-72% dos indivíduos. Em 15-30% dos casos, os ductos seccionais direito anterior e posterior não se unem, cada um tendo uma junção separada com o DHC ou o ducto hepático esquerdo, aumentando o risco de lesão durante a cirurgia.

Ductos Subvesicais
Os ductos subvesicais, localizados superficialmente no leito da vesícula biliar, são a fonte mais comum de vazamento biliar pós-operatório. Observados em cerca de um terço dos espécimes humanos, esses ductos, geralmente originários do segmento 5, drenam para o ducto hepático direito, o DHD ou o DHC. A dissecção deve ser mantida fora do fígado sempre que possível para evitar lesões.
Anatomia do Ducto Cístico
O ducto cístico geralmente se junta ao DHC, com a localização da junção variando do hilo hepático à área pré-ampular. Configurações paralelas e espirais do ducto cístico são comuns e representam fatores de risco significativos para lesões durante a colecistectomia. Fusão inflamatória e contração do ducto cístico são situações de alto risco.
A variabilidade anatômica dos ductos biliares extra-hepáticos requer uma abordagem cuidadosa durante a colecistectomia laparoscópica para evitar lesões. O conhecimento detalhado das possíveis variações e a identificação precisa dos marcos anatômicos são fundamentais para o sucesso cirúrgico. As variações anatômicas dos ductos biliares são frequentes e devem ser esperadas pelos cirurgiões. A terminologia adequada e o reconhecimento dessas variações são cruciais para prevenir lesões durante a colecistectomia laparoscópica. Como afirmou Lahey: “A variação anatômica é a regra, não a exceção.”
Referências
- Michels, NA. “Every duct drains a definite segmental area of the liver.”
- Strasberg, SM. “Anatomic and historical basis of variations in bile ducts.”
- Bismuth, H. “Variations in the anatomy of the extrahepatic bile ducts.”
Diferenciação Morfológica do Ducto Cístico e do Ducto Colédoco no Contexto da Colecistectomia Laparoscópica
Introdução
Na cirurgia laparoscópica, a precisão na identificação e dissecação das estruturas anatômicas é fundamental para a prevenção de lesões iatrogênicas da via biliar. O conhecimento detalhado das variações anatômicas do ducto cístico e do ducto colédoco é essencial para evitar complicações graves. Conforme disse Lahey em 1954: “Cholecystectomy is a dangerous operation unless one realizes that variations are very common.” Assim, a terminologia anatômica deve ser correta, consistente e padronizada para evitar mal-entendidos.
Anatomia Normal
O ducto cístico conecta a vesícula biliar ao ducto hepático comum, formando o ducto colédoco. Este último é a principal via de passagem da bile do fígado para o duodeno. A correta identificação dessas estruturas é crucial durante a colecistectomia para evitar lesões.
Dimensões e Diâmetro
O ducto cístico geralmente tem um comprimento de 2-4 cm e um diâmetro de 1-5 mm. Já o ducto colédoco mede aproximadamente 6-8 cm de comprimento e tem um diâmetro que varia de 4-10 mm.
Trajeto e Topografia
O ducto cístico apresenta um trajeto sinuoso, que pode variar significativamente entre os pacientes. Ele se une ao ducto hepático comum para formar o ducto colédoco, que desce posteriormente ao duodeno até a ampola de Vater. A topografia dessas estruturas é fundamental para a correta dissecação durante a cirurgia.
Vascularização
O ducto cístico é irrigado principalmente pela artéria cística, um ramo da artéria hepática direita. O ducto colédoco, por sua vez, recebe irrigação de pequenos ramos axiais das artérias hepática direita, gástrica direita e pancreatoduodenal onde 60% é inferior e 40% é superior.
Epônimos e Terminologia Correta
Historicamente, a anatomia hepato-biliar tem sido confundida por diferentes nomenclaturas e epônimos imprecisos. O termo “triângulo de Calot”, descrito por Jean-Francois Calot em 1890, refere-se à área limitada pelo ducto cístico, o ducto hepático comum e a artéria cística. No entanto, o triângulo de Calot não é uma definição anatomicamente precisa, sendo preferível o termo “triângulo hepatocístico”, que é delimitado pelo ducto cístico e a vesícula biliar, o ducto hepático comum e a borda do fígado. Esta é uma área crítica onde ductos hepáticos direitos e a artéria hepática direita podem ser encontrados, sendo comumente obscurecida por inflamação aguda ou crônica.
Principais Variações Anatômicas e Suas Incidências
As variações anatômicas são comuns e devem ser antecipadas. Cerca de 20% dos ductos císticos seguem um trajeto paralelo ao ducto hepático comum, compartilhando uma bainha comum de tecido conjuntivo. Outros 5-10% têm um curso espiralado, cruzando o ducto hepático comum de forma ventral ou dorsal. As variações no trajeto e na união dos ductos são fatores de risco significativos para lesões durante a cirurgia.
Idiotopia em Relação ao Duodeno e Holotipia em Relação à Tríade Portal
O ducto colédoco passa diretamente atrás do duodeno, uma característica que o diferencia do ducto cístico durante a dissecção. Em relação à tríade portal, o ducto cístico e o ducto colédoco são componentes críticos, juntamente com a artéria hepática e a veia porta, sendo importantes marcos anatômicos para a orientação cirúrgica.
Sintopia Entre Eles
A relação espacial entre o ducto cístico e o ducto colédoco é complexa e varia com a inflamação e a dissecção cirúrgica. Durante a colecistectomia, a visualização clara do “triângulo hepatocístico” é fundamental para identificar corretamente o ducto cístico e evitar danos ao ducto colédoco.
Conclusão
O entendimento detalhado das variações anatômicas do ducto cístico e do ducto colédoco é crucial para a realização segura da colecistectomia laparoscópica. A aplicação do conceito da “vista crítica de segurança” ajuda a minimizar os riscos de lesões. Portanto, a terminologia precisa e o conhecimento das variações anatômicas são fundamentais para a prática cirúrgica segura.
Does routine performance of IOC prevent common bile duct injuries?
The main goals of IOC are to identify bile duct stones, clarify biliary anatomy, and prevent bile duct injuries. Indications for IOC during laparoscopic cholecystectomy may include jaundice or a history of jaundice, a history of pancreatitis particularly related to gallstone pancreatitis, elevated liver function tests, a common bile duct larger than 5-7mm in diameter, a cystic duct larger than 3mm in diameter, multiple small gallbladder stones, unclear anatomy, common bile duct stones visualized on preoperative ultrasound, possible bile duct injury or leak, and a short cystic duct.

The SAGES guidelines for the clinical application of laparoscopic biliary tract surgery recommended that IOC may decrease the risk of bile duct injury when used routinely and can allow access to the biliary tree for therapeutic intervention (Level II evidence, grade B recommendation). In a more recent guideline from the European Association for Endoscopic Surgery (EAES) regarding the prevention and treatment of bile duct injuries during laparoscopic cholecystectomy, the authors commented that the routine use of IOC remained controversial and that routine IOC could not be recommend based on the available literature. This guideline, however, indicated that IOC allows forearly identification of bile duct injuries as long as they are correctly interpreted.
Recommendation
Surgeons should use IOC liberally, be familiar with its indications, and become facile with the technique and interpretation of cholangiogram images. While IOC may decrease the risk of bile duct injury its routine use remains controversial; further high quality evidence is needed before routine IOC can be recommended. (Quality of evidence: +++, weak)
Safe Cholecystectomy | Bailout Procedures : When and How
Cholecystectomy is a common surgical procedure, with over 750,000 performed annually in the United States and 200,000 in Brazil. Popularized in the early 1990s, laparoscopic cholecystectomy (LC) is now considered the gold standard for routine cases of benign gallbladder and biliary pathology. LC has clear advantages over the traditional open approach, such as lower morbidity, less pain, and faster recovery. However, it is associated with a three to five times increase in bile duct injury (BDI). Major BDI can be a catastrophic complication, significantly increasing mortality. Additionally, patients who suffer a BDI often require further interventions, have a higher risk of additional complications, and experience a reduced quality of life. BDI is a common cause of legal litigation and remains one of the most frequent reasons for monetary compensation.
Correct Anatomical Identification The “classic injury” to the bile duct occurs when the common bile duct is mistaken for the cystic duct. This typically happens in the setting of severe acute or chronic inflammation, where the gallbladder may fuse to the lateral wall of the common hepatic duct, predisposing the surgeon to misidentify the biliary anatomy. This can result in a major BDI, where a segment of the common hepatic duct and bile duct is removed. Beyond this classic injury, other injuries to the biliary system can occur, such as sectional or segmental ducts disconnected from the liver with or without bile leakage, bile leakage from the cystic duct stump, long-term strictures due to thermal or iatrogenic damage, or combined vasculobiliary injuries.
Critical View of Safety (CVS) The Critical View of Safety (CVS), introduced by Strasberg et al. in 1995, is a method of safe anatomical identification that serves as a set of criteria to ensure the proper identification of the appropriate anatomy before ductal structures are ligated. These criteria include separating the lower end of the gallbladder from the liver to expose at least the lower third of the cystic plate, cleaning all fibrous and fatty tissue within the hepatocystic triangle, and seeing only two structures entering the gallbladder. The CVS mirrors the safe identification that occurs in traditional open cholecystectomy. While there are no level 1 data to support its use (due to the large sample size required to discriminate between an injury that occurs at a relatively low incidence), there is a body of literature of over 6000 cases where CVS was achieved without any major BDI.
Culture of Safety in Cholecystectomy (COSIC) Strict adherence to CVS is crucial to reducing BDI, but it is only part of the Culture of Safety in Cholecystectomy (COSIC), which requires that safety be at the forefront. Besides achieving CVS in total cholecystectomy cases, COSIC also requires proper patient selection and evaluation, adjustment of surgical technique in non-routine cases, use of bailout procedures, and avoiding complex cases when appropriate expertise is unavailable. The American Society of Gastrointestinal and Endoscopic Surgeons (SAGES) has developed a six-step program to enhance cholecystectomy safety:
- Understanding CVS and using it for identifying the cystic duct and artery.
- Considering an intraoperative pause before clipping or cutting any structure.
- Understanding aberrant anatomy.
- Liberal use of cholangiography or other intraoperative imaging means of the biliary system.
- Recognizing when dissection is approaching a significant danger zone and terminating the operation by a safe method, other than cholecystectomy, if the conditions around the gallbladder are too dangerous.
- Seeking assistance from another surgeon when conditions are difficult.
Bailout Procedures: When and How to Opt Deciding when to stop dissection of the hepatocystic triangle and opt for a bailout procedure rather than total cholecystectomy can be challenging. To make this decision before any biliary or vascular injury occurs, the surgeon must constantly ask: “Is it possible to safely achieve CVS?” When the answer is “No” or “I am not sure,” we recommend considering a bailout procedure. Early adoption of a bailout procedure is believed to reduce the difficulty of making this decision and avoid inadvertent injuries to the biliary system while trying to dissect in difficult and obstructed planes. It is essential always to remember that this operation is performed for benign pathology.
There are three clear bailout options in difficult cases:
- Stop the Operation (Stop, Drain, and Refer): Stopping the operation may conflict with the surgeon’s goal of “solving a problem,” but it should be considered and is a viable and safe option to avoid BDI. The patient should continue with a short course of antibiotics or even undergo postoperative drain placement and/or percutaneous cholecystostomy. A second attempt at cholecystectomy can be considered in 2–3 months.
- Surgical Cholecystostomy: The gallbladder’s fundus can be opened after placing a purse-string suture, the contents are aspirated, and a drainage catheter is placed in the gallbladder lumen. This method works as a temporary measure since definitive cholecystectomy will likely be necessary in 2–3 months.
- Subtotal “Fenestrating” Cholecystectomy: Subtotal cholecystectomy has been a surgical option for over 100 years. In 2016, an attempt was made to define subtotal cholecystectomy into two distinct subtypes to allow for improved study and dissemination of the technique. When a new gallbladder remnant is created, this is called “reconstituting” subtotal cholecystectomy. When the gallbladder is left open with a remaining portion, this is called “fenestrating” subtotal cholecystectomy. Recent systematic reviews have demonstrated the safety of these procedures. Fenestrating subtotal cholecystectomy is recommended as the most definitive bailout procedure.
Once the decision is made to proceed with fenestrating subtotal cholecystectomy, the surgeon should consider their experience and either convert to an open procedure or continue laparoscopically. This procedure can be safely performed laparoscopically with minimal “advanced” laparoscopic maneuvers, but it can also be easily performed using an open technique.
Fenestrating Subtotal Cholecystectomy Procedure The first step involves incising the anterior (peritonealized) wall of the gallbladder at the fundus. By initially leaving the gallbladder body intact, its contents can be evacuated more easily. It may be advisable to place a surgical sponge or “endobag” under the gallbladder to facilitate catching any stones that might spill upon opening. The incision should continue towards the infundibulum, removing most of the anterior wall of the gallbladder. A very important consideration of this technique involves leaving a portion of the anterior infundibulum wall intact to avoid inadvertent entry into the hepatoduodenal ligament. Once most of the anterior wall is removed and the gallbladder contents, including all stones, are evacuated, the internal aspect of the gallbladder can be examined. It is essential to identify whether continuous biliary drainage from the gallbladder is present. In most “difficult” gallbladders requiring fenestrating subtotal cholecystectomy, the cystic duct is obliterated and does not require formal ligation. However, in the rare instances where the duct is patent and bile continues to drain from it, the internal orifice of the cystic duct should be closed with non-permanent sutures from the internal aspect of the gallbladder. At no point should external ligation of the cystic duct be attempted, which could potentially injure the bile duct. A drain should be left in the hepatorenal recess. No drain is needed inside the gallbladder lumen. The drain should be monitored for biliary drainage. Although generally rare, if a postoperative biliary fistula occurs, standard management should proceed. Routine postoperative endoscopic sphincterotomy is not recommended unless the biliary fistula is persistent, as most of them are self-limiting.

The main goal of laparoscopic cholecystectomy is “safety first, total cholecystectomy second.” While most laparoscopic cholecystectomies are straightforward, the surgeon must always keep this safety culture at the forefront and remain vigilant to anticipate dangerous situations. COSIC will help minimize (or eliminate) BDI and assist the surgeon in managing difficult operating conditions or clinical scenarios. Safe management of the difficult gallbladder is possible with operational adjustments and liberal use of bailout procedures, specifically fenestrating subtotal cholecystectomy.
Colecistectomia Segura: Quando e Como realizar procedimentos de Resgate | Colecistectomia Subtotal
A colecistectomia é um procedimento cirúrgico comum, com mais de 750.000 realizadas anualmente nos Estados Unidos e 200.000 no Brasil. Popularizada no início dos anos 1990, a colecistectomia laparoscópica (CL) é agora considerada o padrão-ouro para casos rotineiros de patologia benigna da vesícula biliar e biliar. A CL tem vantagens claras sobre a abordagem aberta tradicional, como menor morbidade, menos dor e recuperação mais rápida. No entanto, está associada a um aumento de três a cinco vezes na lesão do ducto biliar (LDB). A LDB maior pode ser uma complicação catastrófica, aumentando claramente a mortalidade. Além disso, pacientes que sofrem uma LDB geralmente precisam de intervenções adicionais, têm maior risco de complicações adicionais e sofrem redução na qualidade de vida. A LDB é uma causa comum de litígios legais e continua sendo uma das razões mais comuns para indenizações monetários.
Identificação Anatômica Correta
A “lesão clássica” do ducto biliar ocorre quando o ducto biliar comum é confundido com o ducto cístico. Geralmente, isso ocorre em um cenário de inflamação aguda ou crônica severa, onde a vesícula biliar pode se fundir à parede lateral do ducto hepático comum, predispondo o cirurgião a identificar erroneamente a anatomia biliar. Isso pode resultar em uma LDB maior, onde um segmento do ducto hepático comum e do ducto biliar é removido. Além dessa lesão clássica, outras lesões do sistema biliar podem ocorrer, como ductos seccionais ou segmentares desconectados do fígado com ou sem vazamento biliar, vazamento biliar do coto do ducto cístico, estenoses de longo prazo por danos térmicos ou iatrogênicos, ou lesões vasculobiliares combinadas.
Visão Crítica de Segurança (CVS)
A visão crítica de segurança (CVS), introduzida por Strasberg et al. em 1995, é um método de identificação anatômica segura que serve como um conjunto de critérios para garantir a identificação adequada da anatomia apropriada antes da ligadura de estruturas ductais. Esses critérios incluem a separação da extremidade inferior da vesícula biliar do fígado para expor pelo menos o terço inferior da placa cística, todos os tecidos fibrosos e adiposos limpos dentro do triângulo hepatocístico e apenas duas estruturas vistas entrando na vesícula biliar. A CVS imita a identificação segura que ocorre na colecistectomia aberta tradicional. Embora não existam dados de nível 1 para apoiar seu uso (devido ao grande tamanho da amostra necessário para discriminar entre uma lesão que ocorre com uma incidência relativamente baixa), há um corpo de literatura de mais de 6000 casos onde a CVS foi alcançada sem nenhuma LDB maior.
Cultura de Segurança em Colecistectomia (COSIC)
Embora a adesão estrita à CVS seja importante para diminuir a LDB, ela é apenas uma parte da Cultura de Segurança em Colecistectomia (COSIC), que exige que a segurança esteja na vanguarda. Além de alcançar a CVS em casos de colecistectomia total, a COSIC também requer seleção e avaliação adequadas do paciente, ajuste da técnica cirúrgica em casos não rotineiros, uso de procedimentos de resgate e evitar casos complexos quando a experiência apropriada não estiver disponível. A Sociedade Americana de Cirurgiões Gastrointestinais e Endoscópicos (SAGES) desenvolveu um programa de seis etapas para aumentar a segurança da colecistectomia:
- Compreender a CVS e usá-la para identificação do ducto cístico e da artéria.
- Considerar uma pausa intraoperatória antes de prender ou cortar qualquer estrutura.
- Compreender a anatomia aberrante.
- Uso liberal de colangiografia ou outros meios de imagem intraoperatória do sistema biliar.
- Reconhecer quando a dissecção está se aproximando de uma zona de risco significativo e terminar a operação por um método seguro, além da colecistectomia, se as condições ao redor da vesícula biliar forem muito perigosas.
- Buscar ajuda de outro cirurgião quando as condições forem difíceis.
Procedimentos de Resgate: Quando e Como Optar
Decidir quando interromper a dissecção do triângulo hepatocístico e optar por um procedimento de resgate em vez da colecistectomia total pode ser desafiador. Para tomar essa decisão antes que ocorra qualquer lesão biliar ou vascular, o cirurgião precisa constantemente se perguntar: “É possível alcançar a CVS com segurança?” Quando a resposta for “Não” ou “Não tenho certeza”, recomendamos considerar um procedimento de resgate. Acreditamos que a adoção precoce de um procedimento de resgate diminuirá a dificuldade de tomar essa decisão e evitará lesões inadvertidas ao sistema biliar ao tentar dissecar em planos difíceis e obstruídos. É importante sempre lembrar que esta operação é feita para patologia benigna.
Existem três opções claras de resgate em casos difíceis:
- Interromper a Operação (Parar, Drenar e Encaminhar):
- Parar a operação pode atuar em tensão com o objetivo do cirurgião de “resolver um problema”, mas deve ser considerado e é uma opção viável e segura para evitar a LDB. O paciente deve continuar com um curso curto de antibióticos ou até mesmo ser submetido à colocação de um dreno e/ou de colecistostomia percutânea pós-operatória. Uma segunda tentativa de colecistectomia pode ser considerada em 2–3 meses.
- Colecistostomia Cirúrgica:
- O fundo da vesícula biliar pode ser aberto após a colocação de uma sutura em “bolsa de tabaco”, o conteúdo é aspirado e um cateter de drenagem é colocado na luz da vesícula biliar. Este método funciona como uma medida temporária, pois a colecistectomia definitiva provavelmente será necessária em 2–3 meses.
- Colecistectomia Subtotal “Fenestrante”:
- A colecistectomia subtotal tem sido uma opção cirúrgica por mais de 100 anos. Em 2016, foi feita uma tentativa de definir a colecistectomia subtotal em dois subtipos distintos para permitir um estudo e disseminação melhorados da técnica. Quando um novo remanescente da vesícula biliar é criado, isso é chamado de colecistectomia subtotal “reconstituinte”. Quando a vesícula biliar é deixada aberta com uma porção restante, isso é chamado de colecistectomia subtotal “fenestrante”. Recentes revisões sistemáticas demonstraram a segurança desses procedimentos. Recomenda-se a colecistectomia subtotal fenestrante como o procedimento de resgate mais definitivo.
Uma vez que a decisão é tomada para proceder com a colecistectomia subtotal fenestrante, o cirurgião deve considerar sua experiência e se converter para um procedimento aberto ou continuar laparoscopicamente. Acreditamos que esse procedimento pode ser realizado com segurança laparoscopicamente com manobras laparoscópicas mínimas “avançadas”; no entanto, também pode ser realizado facilmente usando uma técnica aberta.
Procedimento de Colecistectomia Subtotal “Fenestrante”
O primeiro passo envolve a incisão da parede anterior (peritonealizada) da vesícula biliar no fundo. Ao deixar o corpo da vesícula biliar intacto inicialmente, o conteúdo da vesícula biliar pode ser evacuado mais facilmente. Pode ser aconselhável colocar uma esponja cirúrgica ou “endobag” sob a vesícula biliar para facilitar a captura de quaisquer pedras que possam ser derramadas ao abri-la. A incisão deve ser continuada em direção ao infundíbulo, removendo a maior parte da parede anterior da vesícula biliar. Uma consideração muito importante dessa técnica envolve deixar uma porção da parede anterior do infundíbulo intacta para evitar a entrada inadvertida no ligamento hepatoduodenal. Uma vez que a maior parte da parede anterior é removida e o conteúdo da vesícula biliar, incluindo todas as pedras, são evacuados, o aspecto interno da vesícula biliar pode ser examinado. É importante identificar se há ou não drenagem biliar contínua da vesícula biliar. Observa-se que na maioria das vesículas biliares “difíceis” que requerem colecistectomia subtotal fenestrante, o ducto cístico está obliterado e não requer ligadura formal. No entanto, nas raras instâncias em que o ducto está patente e a bile continua a drenar dele, o orifício interno do ducto cístico deve ser fechado com sutura não permanente do aspecto interno da vesícula biliar. Em nenhum momento deve ser tentada a ligadura externa do ducto cístico, o que poderia potencialmente lesionar o ducto biliar. Um dreno deve ser deixado no recessus hepatorenal. Nenhum dreno é necessário dentro da luz da vesícula biliar. O dreno deve ser monitorado para drenagem biliar. Embora geralmente rara, se ocorrer uma fístula biliar pós-operatória, o manejo padrão deve prosseguir. Não se recomenda a esfincterotomia endoscópica pós-operatória de rotina, a menos que a fístula biliar seja persistente, pois a maioria delas é autolimitada.

A meta principal da colecistectomia laparoscópica é “segurança em primeiro lugar, colecistectomia total em segundo”. Embora a maioria das colecistectomias laparoscópicas realizadas sejam diretas, o cirurgião deve sempre manter essa cultura de segurança na vanguarda e permanecer vigilante para se antecipar a situações perigosas. A COSIC ajudará a minimizar (ou eliminar) a LDB e ajudará o cirurgião a manejar condições operatórias difíceis ou cenários clínicos. O manejo seguro da vesícula biliar difícil é possível com ajustes operacionais e uso liberal de procedimentos de resgate, especificamente a colecistectomia subtotal fenestrante.
SAFE laparoscopic cholecystectomy
The CVS technique, which was first described by Strasberg et al. in 1995, was introduced to reduce the risk of bile duct injury. A recent Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) expert Delphi consensus deemed the CVS as being the most important factor for overall safety.
Nowadays, the CVS technique is the gold standard to perform a safe cholecystectomy with identification of the vital structures such as the cystic duct.The reviewed literature suggests that judicious establishment of CVS could decrease bile duct injury rate, from an average 0.4% to nearly 0%.
To establish CVS, two windows need to be created during dissection of Calot’s triangle: one window between the cystic artery, cystic duct, and gallbladder, and another one between the cystic artery, gallbladder, and liver. The CVS technique is aimed especially at mobilizing the gallbladder neck from the liver in the appropriate cystic plate to obtain a circumferential identification of the cystic duct and its transition into the gallbladder.
The guiding structure for dissection should be the wall of the gallbladder. Proper retraction of the fundus cephalad and of the infundibulum posteriorly and laterally is necessary, and tenting by excessive lateral pulling on the gallbladder should be avoided. Cephalad traction on the fundus compresses Calot’s triangle, while lateral traction on Hartmann’s pouch tents up the CBD, which may then be mistaken for the cystic duct, especially when that duct is very short. The cystic duct should be dissected in a retrograde fashion, starting at gallbladder proceeding with the identification of the cystic duct–gallbladder junction on both sides and the visualization of the cystic duct–common bile duct junction prior to clipping.
Calot’s triangle should be dissected from all fibrous and fatty tissues. At the end of the dissection, only the cystic duct and artery cystica should enter the gallbladder and the bottom of the liver bed should be visible. The CBD is not necessary to be exposed. Failure to achieve the CVS is an absolute indication for conversion or additional bile duct imaging. The CVS should be described in the operative report.
Connor et al. and Wakabayashi et al. elegantly describe five key initial steps in performing safe laparoscopic cholecystectomy: (1) retract the gallbladder laterally to a 10 o’clock position relative to the principle plane of the liver (Cantlie’s line); (2) confirm Hartmann’s pouch is retracted up and towards segment IV; (3) identify Rouviere’s sulcus which marks the level of the right posterior portal pedicle and is identifiable in >80% of the patients. An imaginary line (R4U) drawn along the sulcus and carried across to the base of segment IV shows the level ventral to which dissection is “safe” and dorsal to which it is not; (4) dissect the posterior peritoneum of the hepatobiliary or hepatocystic triangle; and (5) confirm the critical view is obtained.
Preventive strategies and safe surgery are of utmost importance to minimize BDI during laparoscopic cholecystectomy. Although many methods used in the prevention of BDI have demonstrated promising results, there is no consensus regarding a systematic reporting system of BDI. Currently, CVS seems to be the cornerstone for a safe laparoscopic cholecystectomy. In difficult cases, a sufficient attention to alternative techniques should be apprehended. In such cases, intraoperative imaging may delineate the biliary anatomy.
Prevenção de Lesão da Via Biliar durante a Colecistectomia
A colecistectomia laparoscópica tornou-se o padrão ouro para o tratamento da colelitíase sintomática e outras doenças da vesícula biliar. No entanto, a incidência de lesões da via biliar (LVB) aumentou de 0,2–0,3% na era da colecistectomia aberta convencional para 0,5–0,8% na era da colecistectomia laparoscópica. Essas lesões são frequentemente associadas a significativa morbidade e mortalidade pós-operatória, reduzida qualidade de vida a longo prazo e representam um desafio cirúrgico considerável. A prevenção dessas lesões deve ser o principal objetivo, exigindo adesão a princípios rigorosos de dissecação meticulosa e segura das estruturas identificadas.
Causas e Fatores de Risco para Lesões da Via Biliar
As lesões da via biliar podem ocorrer devido à dissecação incorreta ou incompleta do triângulo de Calot, especialmente em casos de inflamação significativa no local cirúrgico ou anatomia aberrante do ducto biliar. Outros fatores que aumentam a incidência de LVB incluem:
- Inexperiência do Cirurgião: A “curva de aprendizado” inicial contribuiu para a alta taxa de lesões biliares nos primeiros relatos.
- Inflamação e Cicatrizes: Inflamação aguda ou crônica e cicatrizes densas podem obscurecer a anatomia normal e aumentar a dificuldade do procedimento.
- Obesidade: A presença de tecido adiposo abundante ao redor do ligamento hepatoduodenal aumenta a dificuldade cirúrgica e promove lesões biliares.
- Anatomia Aberrante: Variações anatômicas, como um ducto hepático direito aberrante, podem ser erroneamente identificadas como ducto cístico e ligadas ou cortadas.
- Uso Excessivo de Energia: O uso excessivo de dispositivos de energia, como eletrocautério, pode levar a lesões térmicas e isquemia do ducto biliar.
- Fatores Adversos: Idade avançada, gênero masculino, obesidade mórbida, longa duração dos sintomas antes da cirurgia, cirurgias abdominais superiores prévias e inflamação aguda contribuem para o aumento do risco.
Princípios Técnicos para Prevenção de Lesões da Via Biliar
Para prevenir lesões durante a colecistectomia laparoscópica, os cirurgiões devem seguir princípios técnicos essenciais:
- Visão Crítica de Segurança (CVS): A técnica CVS, descrita por Strasberg et al., é fundamental para a identificação segura das estruturas vitais, como o ducto cístico. Dois espaços devem ser criados durante a dissecação do triângulo de Calot para identificar circunferencialmente o ducto cístico e sua transição para a vesícula biliar.
- Dissecação Meticulosa do Triângulo de Calot: A limpeza do triângulo hepatocístico de gordura e tecidos fibrosos, e a separação do terço inferior da vesícula biliar do fígado para expor a placa cística são passos cruciais.
- Uso Judicioso de Dispositivos de Energia: Os dispositivos de energia devem ser usados com cautela no triângulo de Calot, com configurações de baixa potência e coagulação de pequenos pedaços de tecido por vez.
- Reconhecimento de Dissecção Insegura: O cirurgião deve ser capaz de reconhecer quando a dissecção se torna insegura, com alto potencial para LVB, e estar preparado para buscar alternativas técnicas, pedir uma segunda opinião ou converter para uma colecistectomia aberta.
- Imagens Intraoperatórias: Técnicas como colangiografia intraoperatória, ultrassonografia laparoscópica e colangiografia por fluorescência de infravermelho próximo podem ser usadas para avaliar a anatomia biliar.
Procedimentos de Resgate e Conversão
Em casos onde a CVS não pode ser alcançada e as estratégias de resgate não podem ser implementadas, não hesite em converter para uma colecistectomia aberta. Procedimentos de resgate, como colecistectomia subtotal ou colecistectomia iniciada pelo fundo, podem ser necessários em casos de inflamação severa e/ou incapacidade de realizar a CVS.
Avaliação Pré-operatória e Planejamento
A avaliação pré-operatória de preditores de colecistectomia difícil é crucial. Fatores como gênero masculino, obesidade, idade avançada, histórico de ataques de cólica biliar, intervalo prolongado entre o início e a apresentação dos sintomas, cirurgias abdominais superiores prévias e cirrose devem ser identificados. A seleção cuidadosa de pacientes e a preparação para a possibilidade de conversão para cirurgia aberta são essenciais para a segurança do procedimento.
Conclusão
A prevenção de lesões da via biliar durante a colecistectomia laparoscópica exige uma abordagem meticulosa, conhecimento anatômico detalhado e a aplicação de técnicas cirúrgicas refinadas. A adesão à técnica CVS, uso cauteloso de dispositivos de energia e a prontidão para procedimentos de resgate ou conversão são fundamentais para minimizar riscos e melhorar os resultados dos pacientes. A experiência do cirurgião e a colaboração da equipe cirúrgica são fatores críticos para a execução segura desta operação comum, mas potencialmente desafiadora.
Mechanisms of Bile Duct Injury
Mechanism of Bile Duct Injury: Understanding the Risks in Laparoscopic Cholecystectomy
Introduction
Laparoscopic cholecystectomy, the “gold standard” for treating symptomatic gallbladder disease, has transformed surgical practice since its introduction in the early 1990s. Despite its widespread adoption and the improved safety profile over time, the procedure remains fraught with risks, particularly bile duct injuries (BDIs). The incidence of BDIs during laparoscopic cholecystectomy has declined from its peak, but this complication still represents a significant challenge in digestive surgery, leading to substantial morbidity, mortality, and legal consequences. In Brazil, where an estimated 300,000 cholecystectomies are performed annually, BDIs continue to be a significant concern. This article delves into the mechanisms of bile duct injuries during laparoscopic cholecystectomy, exploring the factors that contribute to these adverse events and their implications for surgical practice.
Development of the Theme
The advent of laparoscopic cholecystectomy marked a turning point in the management of gallbladder disease, offering patients reduced postoperative pain, shorter hospital stays, and faster recovery times. However, the initial enthusiasm for this minimally invasive approach was tempered by a notable increase in bile duct injuries. As surgeons adapted to the new technique, the incidence of BDIs spiked, with early reports indicating injury rates as high as 0.7%. Today, with increased experience and refined techniques, the incidence has decreased to approximately 0.1% to 0.2%. Despite these improvements, the risk remains significant, with estimates suggesting that one in three general surgeons will cause a bile duct injury at some point in their careers.
Iatrogenic bile duct injuries are most often the result of perceptual errors in identifying biliary anatomy during surgery. The most common injury involves a complete transection of the common bile duct, which is also the most difficult to manage. Typically, excessive cephalad retraction of the gallbladder fundus or insufficient lateral retraction on the infundibulum leads to an alignment of the cystic and common bile ducts, causing the common bile duct to be mistaken for the cystic duct. This misidentification results in clipping and transecting the common bile duct—a scenario that can lead to devastating outcomes if not promptly recognized and appropriately managed.
Inflammatory conditions, such as acute or chronic cholecystitis, further complicate the surgical landscape. Thickened and friable tissue, along with adhesions, can obscure normal anatomical landmarks, increasing the difficulty of the procedure. Aberrant biliary anatomy, such as a low-lying right hepatic duct, poses additional risks, as these anatomical variations can be easily overlooked during surgery, leading to unintended ductal injury.
Energy sources used for hemostasis, such as electrocautery, introduce another layer of complexity. Excessive or inappropriate use of these tools can damage the bile duct or its blood supply, resulting in stricture formation or bile leaks. Another common mechanism of injury occurs when a clip is inadvertently placed across the common bile duct, often in a hurried attempt to control bleeding from the hilum without a clear view of the anatomy.
In the context of laparoscopic cholecystectomy, BDIs are particularly perilous when the common bile duct is mistaken for the cystic duct. This classical injury pattern, first described by Davidoff and colleagues, typically involves clipping and dividing the common bile duct, with further proximal dissection leading to injury of the right hepatic artery and more proximal ductal structures, including the common hepatic duct and intrahepatic ducts. Poor visualization due to inadequate illumination, excessive smoke, or intraoperative bleeding exacerbates these risks, making meticulous surgical technique and optimal visualization crucial to avoiding these injuries.
Key Points
- Perceptual Errors: The primary mechanism of bile duct injury during laparoscopic cholecystectomy is the misidentification of biliary anatomy, particularly the confusion between the cystic and common bile ducts.
- Risk Factors: Inflammatory conditions, aberrant anatomy, and excessive use of energy devices significantly increase the risk of bile duct injuries.
- Incidence: Despite advancements in technique, bile duct injuries remain a significant concern, with a 0.1% to 0.2% incidence in laparoscopic cholecystectomy. In Brazil, the annual rate of cholecystectomies underscores the importance of vigilance in preventing these injuries.
- Complications: Bile duct injuries can lead to severe complications, including biliary stricture, leakage, infection, and even death. The financial and legal implications further highlight the need for preventive measures.
Conclusion
Understanding the mechanisms of bile duct injury during laparoscopic cholecystectomy is crucial for improving surgical outcomes and minimizing patient morbidity. Surgeons must remain vigilant in identifying biliary anatomy, particularly in the presence of risk factors such as inflammation and aberrant anatomy. Enhanced visualization techniques, careful dissection, and judicious use of energy devices are essential strategies to reduce the incidence of BDIs. As the field of minimally invasive surgery continues to evolve, ongoing education and training in these areas are paramount to ensuring patient safety and improving the quality of care.
In the words of Alexis Carrel, “There is no such thing as minor surgery, but there are many minor surgeons”. This sentiment is particularly relevant to the surgical treatment of biliary diseases, where the combination of skill, experience, and compassion is vital to patient outcomes.
Did you like it? Leave us a comment ✍️, share on your social networks, and | or send your question via 💬 Online Chat in our Instagram DM.
LaparoscopicCholecystectomy #BileDuctInjury #SurgicalComplications #DigestiveSurgery #SurgicalEducation
Prevention of Bile Duct Injury
Prevention of Bile Duct Injury During Laparoscopic Cholecystectomy
Introduction
Bile duct injury (BDI) during laparoscopic cholecystectomy is a significant surgical complication with profound clinical and medico-legal implications. The incidence of BDI ranges from 0.3% to 0.6%, despite advances in surgical techniques and imaging modalities. The prevalence of BDI remains concerning due to its association with high morbidity and mortality rates. Patients who suffer from BDI often face prolonged hospital stays, multiple surgeries, and long-term complications such as bile leakage, strictures, and secondary biliary cirrhosis. Medico-legally, BDI is one of the most common reasons for litigation against surgeons, often resulting in significant financial settlements and professional repercussions.
Questions and Answers
Question 1: What technique should be used to identify the anatomy during laparoscopic cholecystectomy?
Answer: The Critical View of Safety (CVS) is recommended for identifying the cystic duct and cystic artery.
Key Findings: The incidence of BDI was found to be 2 in one million cases using CVS, compared to 1.5 per 1000 cases with the infundibular technique.
Question 2: When should intraoperative cholangiography (IOC) be used?
Answer: IOC should be used in cases of anatomical uncertainty or suspicion of bile duct injury.
Key Findings: IOC aids in the prevention and immediate management of BDI by providing a precise assessment of biliary anatomy during surgery.
Question 3: What are the recommendations for managing patients with confirmed or suspected bile duct injury?
Answer: Patients with confirmed or suspected BDI should be referred to an experienced surgeon or a multidisciplinary hepatobiliary team.
Key Findings: Early referral to hepatobiliary specialists is associated with better long-term outcomes and lower complication rates.
Question 4: Should the “fundus-first” technique be used when CVS cannot be achieved?
Answer: Yes, the “fundus-first” technique is recommended when CVS cannot be achieved.
Key Findings: This technique is effective for safely dissecting the gallbladder in complex cases where anatomy is unclear.
Question 5: Should CVS be documented during laparoscopic cholecystectomy?
Answer: Yes, documenting CVS with double-static photographs is recommended.
Key Findings: Photographic documentation of CVS ensures correct anatomical identification and serves as a record for later review in case of complications.
Question 6: Should near-infrared biliary imaging be used intraoperatively?
Answer: The evidence for near-infrared biliary imaging is limited; thus, IOC is preferred.
Key Findings: IOC is more widely studied and proven effective in preventing BDI compared to near-infrared imaging.
Question 7: Should surgical risk stratification be used to mitigate the risk of BDI?
Answer: Yes, surgical risk stratification is recommended.
Key Findings: Risk stratification helps identify patients at higher risk of complications, aiding in surgical planning and decision-making.
Question 8: Should the presence of cholecystolithiasis be considered in risk stratification?
Answer: Yes, the presence of cholecystolithiasis should be considered in risk stratification.
Key Findings: Patients with cholecystolithiasis have a higher risk of complications during cholecystectomy, making it important to include this condition in risk assessments.
Question 9: Should immediate cholecystectomy be performed in cases of acute cholecystitis?
Answer: Yes, immediate cholecystectomy within 72 hours is recommended.
Key Findings: Surgery within 72 hours of the onset of acute cholecystitis symptoms is associated with lower complication rates and better patient recovery.
Question 10: Should subtotal cholecystectomy be performed in cases of severe inflammation?
Answer: Yes, subtotal cholecystectomy is recommended in cases of severe inflammation where CVS cannot be obtained.
Key Findings: In severe inflammation scenarios, subtotal cholecystectomy can facilitate the surgery and reduce the risk of BDI.
Question 11: Which approach is preferable, four-port laparoscopic cholecystectomy or reduced-port/single-incision?
Answer: Four-port laparoscopic cholecystectomy is recommended as the standard approach.
Key Findings: The four-port technique is the most studied, showing effectiveness and safety in performing cholecystectomies with lower complication risks.
Question 12: Should interval cholecystectomy be performed following percutaneous cholecystostomy?
Answer: Yes, interval cholecystectomy is recommended after initial stabilization with percutaneous cholecystostomy.
Key Findings: Interval cholecystectomy offers better long-term outcomes and lower risk of recurrent complications compared to no additional treatment.
Question 13: Should laparoscopic cholecystectomy be converted to open in difficult cases?
Answer: Yes, conversion to open surgery is recommended in cases of significant difficulty.
Key Findings: Conversion to open surgery can prevent BDI in situations where laparoscopic dissection is extremely difficult or risky.
Question 14: Should a waiting time be implemented to verify CVS?
Answer: Yes, a waiting time to verify CVS is recommended.
Key Findings: A waiting time allows better anatomical evaluation before proceeding with dissection, reducing the risk of BDI.
Question 15: Should two surgeons be used in complex cases?
Answer: The presence of two surgeons can be beneficial in complex cases, although strong recommendations are not made due to limited evidence.
Key Findings: Some studies suggest that collaboration between two surgeons can improve anatomical identification and reduce complications in difficult cases.
Question 16: Should surgeons receive coaching on CVS to limit the risk or severity of BDI?
Answer: Yes, surgeons should receive coaching on CVS.
Key Findings: Surgeons who receive targeted coaching on CVS show improved anatomical identification and reduced rates of BDI.
Question 17: Should simulation or video-based education be used to train surgeons?
Answer: Yes, simulation or video-based education should be used.
Key Findings: These training methods enhance technical skills, increase surgical precision, and reduce the incidence of BDI during laparoscopic cholecystectomy.
Conclusion
The consensus recommendations provide evidence-based approaches to minimize bile duct injury during laparoscopic cholecystectomy. Practices such as the critical view of safety (CVS), intraoperative cholangiography (IOC), and early referral to specialists can significantly improve surgical outcomes and reduce complications. As famously stated, “The history of surgery is the history of the control of bleeding,” a phrase that underscores the importance of meticulous surgical technique and the prevention of complications like bile duct injuries.
Mondino de Luzzi (1270-1326) e o surgimento do MONITOR DE ANATOMIA
Mondino, oriundo de Bolonha, nasceu e concluiu seus estudos em sua cidade natal, obtendo sua graduação por volta do ano de 1290. A partir de 1306, tornou-se membro do corpo docente da universidade local. Ele recebeu instrução de Tadeu, compartilhando a mesma época de estudo com Mondeville, e dedicou-se de maneira sistemática à Anatomia, realizando dissecações públicas do corpo humano. Mondino é reconhecido como o pioneiro na “restauração” da Anatomia. Em 1316, publicou o tratado intitulado “Anothomia”, considerado o primeiro trabalho “moderno” na área, distinguindo-se por sua abordagem prática e original, diferenciando-se de simples traduções de textos clássicos.
A obra de Mondino apresenta desafios, conforme apontado por Singer (1996), destacando-se a nomenclatura confusa e as condições peculiares da dissecação naquela época. A ausência de conservantes apropriados, apesar do conhecimento acumulado pelos egípcios em técnicas de embalsamamento, tornava a dissecação um processo extenuante, preferencialmente realizado no inverno e em até quatro dias específicos para cada região do corpo.
Apesar de imprecisões anatômicas, como apontado por Friedman e Friedman (2001), Mondino desempenhou um papel crucial na instituição da dissecação como componente essencial do estudo anatômico. Essa prática foi posteriormente integrada ao currículo médico da Universidade de Bolonha, permitindo, até o final do século XVI, que as execuções de criminosos fossem realizadas de maneira que não comprometesse o trabalho anatômico, representando um avanço no uso do corpo humano na construção do conhecimento.
A contribuição de Mondino foi duradoura, pois sua obra foi uma das principais fontes de conhecimento em Anatomia humana por mais de duzentos anos, até o advento da obra de Vesalius no século XVI. Ao assumir a cátedra da disciplina, Mondino introduziu uma nova dinâmica nas aulas de Anatomia, afastando-se da dissecação e inserindo o ostensor (aluno) e o demonstrator ou incisore (técnico) para conduzirem os procedimentos, enquanto os alunos observavam.
As técnicas predominantes, como dissecação a fresco, maceração e preparações secas ao sol, eram utilizadas por Mondino e seus contemporâneos. Apesar de suas reservas quanto à maceração, essa técnica continuou a ser praticada, como confirmado pelos textos de Guido de Vigevano em 1345, representando a persistência do uso da dissecação para fins educacionais em Bolonha.
Mondino não expandiu significativamente o conhecimento anatômico existente, mas contribuiu para a formação de anatomistas que perpetuaram a tradição da disciplina em Bolonha, Pádua e em outros países. Notáveis estudiosos, como Gabrielle de Gerbi e Alessandro Achillini, aprimoraram e ampliaram as descrições anatômicas de Mondino em suas próprias contribuições.
A anatomia permanece como alicerce fundamental para o desenvolvimento da medicina ao oferecer conhecimentos cruciais para o adequada exercício profissional. O legado de anatomistas como Mondino, que enfrentaram desafios significativos em suas dissecações pioneiras, ecoa nas salas de aula modernas. A persistência do estudo anatômico é essencial para a formação médica, e os monitores de anatomia desempenham um papel vital nesse processo educativo. Atuando como elo entre a teoria e a prática, esses monitores, herdeiros contemporâneos do ostensor de Mondino, desempenham um papel crucial ao auxiliar na orientação dos alunos nas complexidades da dissecação e na compreensão da anatomia humana. Sua contribuição atual é inseparável do legado histórico, garantindo que o conhecimento anatômico continue a florescer, moldando as futuras gerações de profissionais de saúde e consolidando a anatomia como um pilar indispensável no edifício da medicina.
ODE AOS MESTRES ANATÔMICOS
Ser mestre é perpetuar juventude, Afrontando o inexorável fio do tempo, Desdobrando-se, multiplicando-se, Nas almas dos discípulos, criando ensejo.
Escolas germinam quando a maturidade, Se entrelaça à força do nobre sentimento, O mestre, sábio, fala à mente e coração, Exemplo luminoso, toque profundo, alento.
Na odisseia do saber, mestre é guia, Navegando oceanos de experiência viva, Com luz, desbrava trilhas no pensamento.
O mestre, como sol em seu zênite, Aquece a jornada do aprendizado, Conservando-se jovem, eternamente, erudito.
Assim, na sala de aula, é o comandante, Que com alma e sabedoria encanta, O mestre, semeando luz e meta.
Gastrointestinal Anastomosis
Navigating the Gastrointestinal Anastomosis: A Surgical Odyssey
Unveiling the Historical Tapestry 🕰️
The creation of gastrointestinal anastomoses, an art in general surgery, has evolved over centuries. In delving into this surgical saga, fundamental principles stand tall, guiding the surgeon’s hands. This chapter unfolds the historical nuances, general tenets for successful anastomosis creation, and delves into pivotal technical considerations amidst current controversies.
The Dance of Healing and Anatomy 🩹🔍
Understanding the physiological waltz of gastrointestinal wound healing and the intricacies of intestinal wall anatomy sets the stage. An enterotomy’s inception triggers a symphony of vasoconstriction, vasodilation, and capillary changes, orchestrating the ballet of tissue healing. Granulation tissue emerges, heralding the proliferative phase, where collagen undergoes a dance of lysis and synthesis.
Layers of the Gastrointestinal Tapestry 🧵
The intestinal wall, a multilayered tapestry, unravels its secrets. The serosa, a connective tissue cloak veiled by mesothelial lining, demands precise apposition to thwart leakage risks. The submucosa, the stronghold of tensile strength, anchors the sutures knitting the anastomosis. Intestinal mucosa seals the deal, driven by epithelial cell migration and hyperplasia, crafting a watertight barrier.
Local Factors: Paving the Path to Healing ⚒️🩹
The local factors influencing this symphony include intrinsic blood supply and tension management. Adequate blood supply, a lifeline for tissue oxygenation, hinges on meticulous surgical technique. Tension, a delicate partner in this dance, demands finesse; too much jeopardizes perfusion, too little invites inflammatory infiltrates. The colon, in particular, demands a surgeon’s nuanced touch.
Systemic Harmony: The Ripples of Patient Factors 🔄🌊
Systemic factors contribute their ripples to this surgical pond. Hypotension, hypovolemia, and sepsis compose a dissonant note affecting blood flow and oxygen delivery. Patient-specific variables — malnutrition, immunosuppression, and medication use (hello, steroids and NSAIDs) — compose a subplot, influencing the narrative of wound healing.
As the surgeon steps into this intricate ballet of anastomosis creation, history, physiology, and patient-specific factors converge. Each suture, each decision, shapes the narrative of healing. The gastrointestinal odyssey continues, blending tradition with innovation, as surgeons embark on the timeless quest for successful anastomoses. 🌐🔍🩺
Tubes and Drains
Unlocking the World of Tubes and Drains in Medical Practice 🩹
Understanding the diverse array of tubes and drains is crucial for any medical practitioner, and it all begins with appreciating the French size system, where the outer diameter of a catheter is denoted. A quick calculation (French size multiplied by 0.33) reveals the catheter’s outer diameter in millimeters.
Gastrointestinal Tract Tubes 🍽️
Starting with nasogastric tubes designed to evacuate gastric contents, these are frequently employed in patients facing ileus or obstruction. Modern nasogastric tubes often incorporate a sump function, preventing suction locks and enhancing efficiency. Nonsump tubes, though less common, may be used for intermittent suction. Nasogastric tubes also serve in feeding, with soft, fine-bore tubes being preferred for this purpose. Nasoenteric tubes, intended for feeding, require careful attention to safety during instillation.
Nasobiliary tubes, often placed endoscopically, aid in biliary drainage in cases of obstruction or fistula. T-tubes within the common bile duct ensure closed gravity drainage. Gastrostomy tubes, placed surgically or via percutaneous endoscopic gastrostomy (PEG), find utility in drainage or feeding. Jejunostomy tubes, inserted surgically or endoscopically, are vital for long-term nutritional access.
Respiratory Tract Tubes 🫁
Chest tubes play a pivotal role in pleural cavity drainage, addressing issues like pneumothorax, hemothorax, or effusion. The three-bottle system facilitates constant suction, drainage, and prevention of air entry, crucial for maintaining a water seal.
Endotracheal tubes, cuffed for a secure tracheal seal, cater to short-term mechanical ventilation needs in adults. Tracheotomy tubes, directly inserted into the trachea through the neck, become essential for prolonged mechanical ventilation or when maintaining a patent airway is challenging.
Urinary Tract Tubes 🚰
Bladder catheters, commonly known as “Foley” catheters, serve to straight drain urine. Nephrostomy tubes, placed in the renal pelvis, drain urine above obstructions or delicate ureteral anastomoses. Percutaneously placed tubes, often pigtail catheters, assist in draining abscesses, typically guided by interventional radiologists.
Surgical Drains 🌡️
Closed suction drains, such as Jackson-Pratt and Hemovac, prove invaluable for evacuating fluid collections during surgery. Sump suction drains, like Davol drains, are larger and designed for continuous suction in scenarios with thick or particulate drainage. Passive tubes, exemplified by Penrose drains, offer a pathway for fluid without applied suction, serving as a two-way conduit for bacteria. Understanding these various tubes and drains is pivotal for medical practitioners navigating complex clinical scenarios. 💉💊
Revisional Surgery for Weight Regain
- Introduction
Revisional bariatric surgery is a weight loss surgery for people who have not lost enough weight or have regained weight after their first bariatric surgery. It is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain.
- Causes of Weight Regain
There are many factors that can contribute to weight regain after bariatric surgery, including:
* Lack of adherence to dietary recommendations
* Hormonal/metabolic imbalance
* Mental health
* Physical inactivity
* Anatomic/surgical factors
* Medications
- Preoperative Evaluation
Before revisional surgery, patients will undergo a thorough evaluation to determine if they are eligible for the procedure. This evaluation will include a medical history and physical exam, as well as blood tests, imaging studies, and a psychological evaluation.
- Selecting the Type of Revisional Surgery
The type of revisional surgery that is best for a patient will depend on a number of factors, including the type of primary surgery they had, the cause of their weight regain, and their overall health. Some of the most common types of revisional surgery include:
* Roux-en-Y gastric bypass (RYGB)
* Sleeve gastrectomy (SG)
* Biliopancreatic diversion with duodenal switch (BPD/DS)
* Single anastomosis gastric bypass (OAGB)
* Sleeve gastrectomy with duodenal switch (SADI-S)
- Weight Loss Following Revisional Surgery
Weight loss after revisional surgery can be significant. In one study, patients who underwent revisional surgery for weight regain lost an average of 50–65.3% of their excess weight after 3 months and 50.1–79.1% of their excess weight after 12 months.
- Complications of Revisional Surgery
Revisional surgery is more complex and technically demanding than primary bariatric surgery, and therefore carries a higher risk of complications. Some of the most common complications of revisional surgery include:
* Hernia
* Anastomotic leak
* Stricture
* Marginal ulcer
* Wound infection
* Hemorrhage
* Perforation
* Obstruction
- Conclusion
Revisional bariatric surgery can be a successful treatment option for patients with weight regain. However, it is important to carefully consider the risks and benefits of the procedure before making a decision. Patients should also work closely with their healthcare team to choose the best type of revisional surgery for them and to prepare for the procedure and its aftermath. Revisional bariatric surgery is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain. It is important to work closely with your healthcare team to determine if you are eligible for the procedure and to choose the best type of surgery for you.
10 Anatomical Aspects for Prevention the Bile Duct Injury
Essential aspects to visualize and interpret the anatomy during a cholecystectomy:
1. Have the necessary instruments for the procedure, with adequate positioning of the trocars and a 30-degree optic.
2. Cephalic traction of the gallbladder fundus and lateral traction (pointing to the patient’s right shoulder), to reduce redundancy of the infundibulum.
3. Puncture and evacuation of the gallbladder to improve its retraction, in cases where traction cannot be performed easily (acute cholecystitis).
4. Lateral and caudal traction of the infundibulum, for correct exposure of Calot’s triangle, exposing the CD and artery.
5. “Critical view of Safety” to avoid misidentification of the bile ducts, ensuring that only two structures (CD and artery) are attached to the gallbladder. For this, they must be dissected separately, and the proximal third of the gallbladder must be moved from its fossa, to ensure that there is no anatomical variant there.
6. Systematic use of intraoperative cholangiography. Ideally by transcystic route or possibly by a puncture of the gallbladder.
7. Ligation of the cystic duct with knots (“endoloop”) to prevent migration of metallic clips that could condition a postoperative leak.
8. In case of severe inflammation of the gallbladder pedicle, with its retraction or lack of recognition of cystic structures, a subtotal cholecystectomy might be indicated.
9. In case of hemorrhage, avoid indiscriminate clip placement and or blind cautery. Opt for compressive maneuvers and, once the bleeding site has been identified, evaluate the best method of hemostasis.
10. If the surgeon is not able to resolve the injury caused, it is always better to ask for help from a colleague, and if necessary, to refer the patient to a specialized center.
#SafetyFirst
The main goal in the postoperative management of BDI is to control sepsis in the first instance and to convert an uncontrolled biliary leak into a controlled external biliary fistula to achieve optimal local and systemic control. Definitive treatment to re-establish biliary continuity will be deferred once this primary goal is achieved and should not be obsessively pursued in the acute phase. The factors that will determine the initial presentation of a patient with a BDI in the postoperative stage are related to the time elapsed since the primary surgery, the type of injury, the mechanism of injury, and the overall general condition of the patient.
Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
GASTROSTOMY: INDICATIONS, TECHNICAL DETAILS AND POSTOPERATIVE CARE.
Ozimo Pereira Gama Filho
Adjunct Professor at the Federal University of Maranhão
No conflict of interest
- INTRODUCTION
- HISTORICAL ASPECTS
- ANATOMICAL FUNDAMENTALS
- INDICATIONS
- TYPES & TECHNIQUES
- ADVERSE EVENTS
- CUSTO RATIO x EFFECTIVENESS
- CONCLUSIONS
SUMMARY
In the last decade, the use of gastrostomies has been widely indicated as the preferred form of access to the gastrointestinal tract for feeding in chronic conditions and during recovery from acute conditions such as trauma. Together with this increase in indications, new techniques have been developed that have made gastrostomies simpler and less risky. From the classical technique of Stamm performed by laparotomy, two new alternatives that do not require laparotomy emerged: percutaneous endoscopic gastrostomy (PEG) and fluoroscopy gastrostomy. Its main benefit is to avoid a laparotomy, with less associated postoperative pain and earlier return of gastrointestinal function. Although peg is currently widely accepted as the insertion technique of choice due to its simplicity and efficacy, there are patients who are not candidates for an endoscopic approach. In this article we seek to clarify the indications, technical aspects and perioperative care of patients undergoing gastrostomy.
Keywords: Surgical Procedures; Ostomies; Gastrostomy.
Area of Knowledge: General Surgery
- INTRODUCTION
The main indication for enteral or parenteral feeding in the perioperative period is the provision of nutritional support to supply the metabolism of patients with inadequate oral intake. Enteral feeding is the preferred method in relation to parenteral feeding in patients with gastrointestinal dysfunction in the perioperative period due to the inherent risks associated with parenteral nutritional support, such as: infectious complications of the access routes, higher operational cost, and the inability to parenteral nutrition to provide adequate enteral stimulation and subsequent involvement of the intestinal defense barrier [1,2]. In addition, enteral feeding may decrease the risk of bacterial translocation and corresponding bacteremia [3]. Gastric nutritional support is the most common type used. Access to insert the gastrostomy probe can be achieved using endoscopy, interventional radiologia, or surgical techniques (open or laparoscopic). However, since its description in the 1980s [4], percutaneous endoscopic gastrostomy (PEG) is currently considered the method of choice for medium and long-term enteral support.
1.1 Objective: This article reviews the current knowledge about GOSTROSTOMIA in the medical literature, emphasizing the technical and perioperative aspects.
- HISTORICAL ASPECTS
In 876, Verneoil [5] successfully made the first gastrostomy in humans. Since then, several technical modifications have been suggested, such as witzel’s technique in 1891, in which a subseroso tunnel is made on the probe [6]. Stamm, in 1894 [7], described one of the most performed techniques today and in the history of surgical gastrostomy, which consists in the making of suture in a pouch to invaginate the probe inserted into the stomach [8]. In 1980, percutaneous endoscopic gastrostomy was described by Gauderer et al. [4] , which transformed the technique of making gastrostomy.
- ANATOMICAL FUNDAMENTALS
The stomach is a J-shaped dilated cylindrical organ that rests in the left epigastric and hypochodrial region of the abdomen at the level of the first lumbar vertebra. It is previously limited by the left hemidiaphragm, the left lobe of the liver and a triangular portion of the anterior abdominal wall. Subsequently, the pancreas, left kidney and adrenal delimit the stomach. The spleen is posterolaterally and the transverse colon is inferior. It is fixed at two points of continuity: gastroesophageal, superiorly and the duodenal, retroperitoneally. Its ligament attachments also help you in fixation to adjacent organs: gastrophemic (diaphragm), hepatogastric or minor omentum (liver), gastrosplenic or gastrolienal (spleen), and gastrocholic or omentum major (transverse colon). The anatomical regions of the stomach can be distinguished as this: começa superiorly in the continuity of the abdominal part of the esophagus and dthe gastroesophageal junction, the cardiac part of the stomach. Soon below this portion, lies the bottom of the stomach that expands to the left extending above thegastroesophageal junction, forming an acute angle with the distal esophagus known as cardiac notches. The body s andextends as a distensible reservoir and forms a medial edge called the smallest curvature to the right and a side edge called the largest curvature on the left. The gastric den of the stomach is not anatomically distinguishable, but it is estimated to be a region of the angular isis along the distal minor curvature to a point along a lower line to the distal major curvature. It thus ends bymouthing r into the pyloric canal limited by the pyloric sphincter, a palpable thickened ring of muscle that is continuous with the first part of the duodenum.
- INDICATIONS
Gastrostomy is used in the following situations:
- Gastric decompression: can be obtained by means of temporary gastrostomy, occasionally recommended, as a complement to large abdominal operations for which gastric stems, prolonged “adynamic ileus” and digestive fistulas are foreshadowed.
- Nutritional Support: b.1 Temporary; indicated when access to the digestive tract is temporarily impaired for recovery and maintenance of nutritional status (E.g. CEsophageal EC); b.2 Definitive; as palliative therapy in patients with unresectable malignant neoplasia of the head and neck, as wellas n degenerative neurological diseases that lead to irreversible disorders of deglutition.
However, the decision to perform a gastrostomy, as well as its route (surgical, radiological or endoscopic) should be individualizedaccording to the needs, diagnosis, life expectancy of the patient and the available hospital logistics. The objective is not only to optimize perioperative recovery, to improve survival and nutritional status of the patient, but also to promote quality of life, which is not necessarily correlated with nutritional improvement only [9]. Therefore, the appropriate indication, like any other surgical intervention, must be clearly establishedand informed before it is performed. Some of the absolute contraindications of gastrostomy are summarized in Table 1. In addition to absolute contraindication conditions, other situations such as the presence of non-obstructive oromyctological oresophageal malignancy, hepatomegaly, splenomegaly, peritoneal dialysis, portal hypertension with gastric varicose veins, and a history of partial gastrectomy are also considered relative contraindications.
| ABSOLUTE CONTRA – INDICATIONS |
| Coagulopatia Severa (INR > 5, Plaquetas < 50.000 e TPT > 50s) |
| Hemodynamic Instability |
| Septic Shock |
| Refractory Ascites |
| Peritonitis |
| Dermatological infection in the upper abdomen |
| Carcinomatose Peritoneal |
| Interposition of organs that prevent gastric access |
| History of Total Gastrectomy |
| Stenosis or Pyloric Obstruction |
| Severe gastroparesis, in cases of indication for nutritional support |
| Absence of Informed Consent |
- TYPES & TECHNIQUES
Currently there are three techniques for performing gastrostomy: radiological, through percutaneous gastrostomy by fluoroscopy, percutaneous endoscopic gastrostomy (PEG) and surgical gastrostomy. Due to the didactic characteristics of this material, we will focus on endoscopic and surgical gastrostomy , which becomes the main option in the following situations: 1) when the patient will already undergo a laparotomy due to some abdominal condition ; 2) impossibility of performing gastroscopy to perform gastrostomy endoscopic percutaneous (PEG) ; 3) in case of peg technical failure; 4) unavailability of resources for the preparation of PEG or percutaneous gastrostomy by fluoroscopy.
- ENDOSCOPIC PERCUTANEOUS GASTROSTOMY
The informed consent form must be obtained from patients or their legal representatives. Patients should fast for a minimum of 8 hours and receive prophylactic antibiotics one hour before proceeding and intravenous administration of 1-2 g of cefazolin is recommended. The technique introduced by Gauderer et al [4] is the most used technique to insert the PEG gastrostomy probe. In this method, a guide wire is used, inserted in the distal gastric chamber through a needle puncture n to the anterior abdominal wall. This guide wire is then seized endoscopically with a handle and then removed through the esophagus and mouth. Subsequently, the guide wire is fixed to the end of the gastrostomy probe and then pulled from the mouth to the esophagus, stomach and then out to the abdominal wall, where it will be fixed.
- SURGICAL GASTROSTOMY
Surgical gastrostomy can be performed in two ways: 1) via laparotomy – the predominant form; and 2) laparoscopic approach. The preparations are the same as the endoscopic pathway.
5.2.1. GASTROSTOMIA At STAMM
After adequate asepsis and antisepsis, with the patient under anesthesia and in horizontal dorsal decubitus, the technical steps are as follows: 1. Median laparotomy (supraumbilical median incision); 2. Identification of the gastric body; 3. Stomach hold with Babcock tweezers (to evaluate the approach of the stomach to the peritoneum); 4. Suture in pouch (circular area of 2cm) – atraumatic absorbable thread; 5. Section of the stomach wall (0.5cm) – (incision with scalpel or Electrocautery in the center of the suture, of sufficient size, for the placement of a probe with 20 to 26 French); 6. Placement of the Gastrostomy probe in the extension of 5-6 cm, followed by suture closure in a pouch); 7. Tie the suture threads in a pouch around the probe; 8. Apply a second suture in a pouch 1cm above the first (seromuscular stitches); 9. Externalization of the probe by counter opening on the left flank; 10. Fix the stomach wall to the abdominal wall in 4 cardinal points (external ration with the two Kocher tweezers used for grip of the alba line and against traction by means of the index fingers of the wall of the left hipochondrio to approach the parietal peritoneum of the gastric wall); 11. Fixation of the probe to the skin (point with nonabsorbable wire); 12. Closure of the abdominal wall (synthesis of the wall with approximation of the alba thread by continuous suture with monofilament thread 1-0 or 2-0 and of the skin with separate points of nylon 3-0); 13. Dressing.
5.2.1 GASTROSTOMIA EM WITZEL
The initial technical steps from gastrostomy to Witzel are like those of gastrostomy to Stamm, including fixation of the probe to the stomach by a pouch suture. Then, the probe is placed on the gastric wall and a tunnel of 8-10 cm is made by seromuscular suture (continuous or with separate points of absorbable or nonabsorbable thread) covering it and externalization is performed by counteropening.
- ADVERSE EVENTS
According to the literature, the rate of complications for different procedures varies due to the heterogeneity of the samples evaluated. For surgical gastrostomy, the reported complication rates are between 1% and 35%, while for percutaneous radiological gastrostomy it is 3% to 11%, and for percutaneous endoscopic, 17%–32%, the main related adverse event is surgical site infection [10, 11]. Although considered a basic procedure, gastrostomy is associated with an extensive list of related technical complications, care and use of the probe. Serious problems related to the technique include separation of the stomach from the abdominal wall (leading to peritonitis), separation of wounds, hemorrhage, infection, lesion of the posterior gastric wall or other organs, and placement of the tube in an inappropriate place of gastric position. Separation of the stomach from the abdominal wall usually occurs due to inadvertent and premature displacement of the tube, particularly with balloon-like devices, or a rupture during a catheter change. It requires immediate attention, being treated with laparotomy, although in selected cases laparoscopic correction is possible. Most complications can be avoided with the careful choice of the type of procedure, from the appropriate ostomy device, considering it an important intervention and using meticulous technique with the proper approximation of the stomach to the abdominal wall and outflow of the probe through a counter-incision (in conventional procedures), thus avoiding probes in the midline or awfully close to the costal edge.
- COST VS. EFFECTIVENESS
A recent study [12] compared the cost associated with the different gastrostomy techniques, and the results of the evaluation showed variable the benefits of each of the individual percutaneous procedures, indicating that surgical gastrostomy was the onerous mais of the three modalities due to higher costs, complications, and recovery time, as well as the endoscopic technique presenting the cost effectiveness ratio.
- CONCLUSIONS
Despite the technique employed, the decision to performa gastrostomy is not based only on the patient’s survival expectancy, because the adequate indication provides a better quality of life even when the survival of the patient after the procedure is severely limited. Therefore, understanding of techniques, indications, complication rates is essential to guide the surgical team in the scope of multidisciplinary care, as well as the education of patients and their caregivers is vital to ensure the correct maintenance of the devices, thus ensuring adequate nutritional intake of the patient and minimizes complication rates.
References
1 Alverdy J, Chi HS, Sheldon GF. The effect of parenteral nutrition in gastrointestinal immunity. The importance of de estimulação enteral. Ann Surg, 1985; 202: 681-684 [PMID:3935061]
2 Deitch EA, Ma WJ, Ma L, Berg RD, Specian RD. Protein malnutrition predisposes to inflammation-induced intestinal origin septic states. Ann Surg, 1990; 211: 560-567; discussion 560-567 [PMID: 2111125]
3 Deitch EA, Winterton J, Li M, Berg R. The intestine as a portal of entry to bacteremia. Role of protein malnutrition. Ann Surg 1987; 205: 681-692 [PMID: 3592811]
4 Gauderer MW, Ponsky JL, Izant RJ. Gastrostomia sem laparotomy: percutaneous endoscopic technique. J Pediatrician Surg, 1980; 15: 872-875 [PMID: 6780678]
5 Anselmo CB, Tercioti Júnior V, Lopes LR, Coelho Neto JS, Andreollo NA. Surgical gastrostomy: current indications and complications in patients of a university hospital. Rev Col Bras Cir. [Internet journal] 2013;40(6). Available in URL: http://www.scielo.br/rcbc
6 Witzel O. For gastric fistula technique. Chir Zbl. 1891;18:601-4.
7 Stamm M. Gastrostomy: a new method. Med News. 1894;65:324.
8 JP grant. Comparison of percutaneous endoscopic gastrostomy com gastrostomia strain. Ann Surg. 1988;207(5):598-603
9 Bannerman E, Pendlebury J, Phillips F, Ghosh S. Cross-sectional and longitudinal study of health-related quality of life after percutaneous gastrostomy. Eur J Gastroenterol Hepatol 2000; 12: 1101-1109 [PMID: 11057455]
10 Möller P, Lindberg CG, Zilling T. Gastrostomy by various techniques: evaluation of indications, outcome and complications. Scand J Gastroenterol. 1999;34(10):1050-4.
11 Clarke E, Pitts N, Latchford A, Lewis S. A major prospective audit of morbidity and mortality associated with food gastrostomies in the community. Clin Nutr. 2017 Apr;36(2):485-490. DOI: 10.1016/j.clnu.2016.01.008. EPub 2016 January 21. PMID: 26874913.
12 Wollman B, D’Agostino HB, Walus-Wigle JR, Easter DW, Beale A (1995) Radiological, endoscopic and surgical gastrostomy: an institutional evaluation and meta-analysis of the literature. Radiology 197: 699–704.
Michelangelo Buonarroti
5 Segredos de Anatomia Escondidos na Capela Sistina: A Neuroanatomia Clandestina de Michelangelo
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
Onde a Fé e o Bisturi se Encontram
Na formação médica contemporânea, o estudo da anatomia topográfica num laboratório de dissecação é o rito de passagem fundamental para qualquer cirurgião. No Brasil, apesar dos desafios atuais na captação de cadáveres para estudo nas faculdades de medicina, a dissecação continua a ser a base insubstituível da técnica cirúrgica. Contudo, no século XVI, o estudo do corpo humano além da superfície era um tabu severo, vigiado de perto pelas leis civis e canónicas. Michelangelo Buonarroti, celebrado mundialmente pela sua terribilità — aquela força emocional e técnica avassaladora —, viveu um paradoxo perigoso. O artista profundamente religioso era também um anatomista clandestino. Desafiando os dogmas de sua época, ele dedicou inúmeras noites à dissecação de cadáveres, buscando nos tendões, fáscias e vísceras a geometria da perfeição biológica. O que a humanidade demorou 500 anos para perceber é que Michelangelo não utilizou este conhecimento apenas para conferir realismo hipertrófico às suas figuras; ele codificou verdadeiras lições de neuroanatomia diretamente no teto da Capela Sistina. O santuário do conclave papal guarda um mapa do intelecto humano, sugerindo que a verdadeira centelha divina reside na intrincada arquitetura do sistema nervoso central. Abaixo, dissecamos os 5 segredos anatómicos ocultos nesta obra-prima.
A Anatomia Oculta nos Afrescos
1. O Cérebro como a “Faísca Divina” em A Criação de Adão
Em 1990, o médico norte-americano Frank Meshberger publicou uma descoberta no prestigiado Journal of the American Medical Association (JAMA) que alterou o curso da história da arte e da medicina. Ele demonstrou que a figura de Deus, envolta num manto avermelhado e cercada por anjos, compõe um corte sagital milimetricamente exato de um cérebro humano. A genialidade reside nos detalhes: o contorno inferior do manto segue com precisão o sulco lateral (Fissura de Sylvius). O braço estendido do Criador atravessa exatamente o giro do cíngulo. Um “anjo” posicionado inferiormente desenha o trajeto da artéria basilar, enquanto o pé bifurcado de um querubim representa a glândula pituitária (hipófise). Para Michelangelo, o que Adão recebe no toque não é apenas o fôlego biológico, mas o intelletto — a razão suprema.
2. O Tronco Encefálico no Pescoço de Deus
A análise de A Criação de Adão revela o cérebro macroscópico, mas o painel central A Separação da Luz das Trevas aprofunda-se em estruturas neurais complexas. Em 2010, os investigadores Ian Suk e Rafael Tamargo, neurocirurgiões da Universidade Johns Hopkins, identificaram um detalhe audacioso. O pescoço de Deus exibe uma anatomia irregular que foge à hipertrofia muscular renascentista padrão. Utilizando um escorço dramático (técnica de perspetiva que encurta a figura), Michelangelo camuflou uma visão ventral do tronco encefálico humano. Estão lá, representados com rigor de atlas cirúrgico, o bulbo, a ponte e a estrutura em forma de “Y” característica dos nervos óticos e do quiasma ótico.
3. Arquitetura Neural: Ventrículos e o Corpo Caloso
A investigação ganhou novos contornos em 2015, quando Ciurea et al. identificaram como o mestre utilizou a técnica do chiaroscuro (luz e sombra) aplicando o princípio da Gestalt para ocultar formas. Na mesma cena da Separação da Luz das Trevas, os olhos treinados de um neuroanatomista conseguem completar as figuras:
- Área A: O quarto ventrículo perfeitamente delineado.
- Área B: A ponte de Varólio, formada pela curvatura da coxa direita da divindade, exibindo a oliva pontina e o sulco colateral anterior.
- Área C: O manto superior que emula uma imagem espelhada do corpo caloso (a via de comunicação entre os hemisférios cerebrais). A inversão da imagem serviu para manter o fluxo estético sem perder a mensagem anatómica.
4. O Anatomista Clandestino de Santo Spirito
Para que um artista atingisse tal nível de detalhe no século XVI — superando as obras de Galeno (que se baseavam em dissecações de macacos e porcos) —, a observação clínica superficial não bastaria. Aos 18 anos, Michelangelo firmou um acordo secreto com o prior da Igreja de Santo Spirito, em Florença. Em troca da escultura de um crucifixo de madeira, o prior permitia-lhe dissecar cadáveres humanos no hospital da igreja durante a madrugada. armado com um bisturi primitivo, ele antecipou descobertas que a medicina oficial, com Andreas Vesalius, só documentaria décadas mais tarde.
5. Uma Mensagem de Protesto e Subversão (A Costela Oculta)
O anatomista era também um livre-pensador influenciado pelo Neoplatonismo florentino. O investigador Deivis Campos (2019) identificou uma “costela extra” no lado esquerdo do torso de Adão. Ao invés de pintar uma costela a ser removida para a criação de Eva (como ditava o dogma), Michelangelo pintou uma costela extra e oculta. Esta representação anatómica subversiva sugere que as essências feminina e masculina coexistem na natureza humana original, uma alusão ao misticismo da Cabala e um protesto velado contra a interpretação literal das escrituras.
Pontos-Chave para a Formação Médica
- A Observação é a Base da Medicina: O génio de Michelangelo prova que a capacidade de observar minuciosamente a anatomia é o que separa o conhecimento superficial da verdadeira maestria.
- Integração Arte e Ciência: As estruturas neurais (fissura de Sylvius, tronco encefálico, quiasma ótico) pintadas no século XVI mostram que a medicina e a arte nasceram da mesma curiosidade intrínseca sobre o milagre da vida.
- O Cérebro como Sede da Consciência: Ao colocar o sistema nervoso central no epicentro do ato da Criação, Michelangelo antecipou em séculos a premissa da neurociência moderna: somos o nosso cérebro.
Conclusões Aplicadas à Prática do Cirurgião
Para o cirurgião do aparelho digestivo, ou de qualquer outra especialidade, a história de Michelangelo traz uma lição de humildade e reverência. Quando entramos no centro cirúrgico e realizamos uma incisão, não estamos apenas a manipular tecidos e órgãos; estamos a interagir com a mais sublime e complexa arquitetura do universo conhecido. Se o maior segredo do Renascimento — o mapeamento do nosso intelecto na morada do papado — permaneceu oculto por cinco séculos à vista de todos, devemos questionar-nos diariamente: o que mais estamos a falhar em enxergar nos nossos doentes devido à pressa ou à desatenção clínica? A medicina de excelência exige o olhar crítico do cientista e a sensibilidade meticulosa do artista.
“Onde o espírito não conduz a mão do artista, não há arte. E onde o conhecimento da anatomia não conduz a mão do médico, não há cura. A estrutura do corpo humano é a obra-prima inquestionável do Criador.” — Adaptado dos pensamentos do Renascimento Médico.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Gallstone Ileus
INTRODUCTION
Gallstone ileus is a misnomer: this condition is not a physiologic ileus at all, but a mechanical obstruction of the intestinal lumen (most commonly the small bowel) by a large gallstone that has passed through a cholecystoenteric fistula. Cholecystoenteric fistulae may occur from the gallbladder to the adjacent luminal viscera-duodenum (most common), stomach, or colon. Gallstone obstruction of the stomach at the pylorus is known as Bouveret’s syndrome. Cholecystocolic fistula is less common. Colonic obstruction in this situation typically occurs at the sigmoid colon. Most common is cholecystoduodenal fistula, with a large (usually >2 cm) gallstone passing through the small bowel and becoming lodged in the terminal ileum. Cholecystoenteric fistula is felt to be caused by a combination of pressure, necrosis, and inflammation with chronic longstanding gallstone disease. Up to 25% of patients who develop gallstone ileus will harbor multiple stones in the alimentary tract; therefore, a close inspection of the entire intestine is important at the time of operation.
CLINICAL PRESENTATION
Over the past few years, however, the incidence of gallstone ileus has been shown to be greater than previously thought. Several recent large population-based series have found that gallstone ileus accounts for approximately 0.1% of all small bowel obstructions. The disease usually affects women (70%) and those in the seventh or eighth decade of life. Most patients present with bloating, crampy abdominal pain, and vomiting, symptoms typical of mechanical small bowel obstruction. A careful history may reveal earlier episodic colicky right upper quadrant abdominal pain consistent with gallstone disease. The classic finding on plain abdominal radiograph is that of Rigler’s triad (pneumobilia, dilated small bowel loops with air-fluid levels, and a large, calcified gallstone in the lumen of the small bowel). Currently, computed tomography (CT) is used ubiquitously. CT has 99% accuracy for diagnosing gallstone ileus. Typical CT findings include pneumobilia, dilated loops of small bowel with air-fluid levels consistent with small bowel obstruction, and transition point with the ectopic stone always visible radiologically.
MANAGEMENT
Operation is required for all patients with gallstone ileus, as spontaneous passage of these large stones is rare once the patient has become symptomatic. It is crucial to optimize the patient physiologically as much as possible in this semi urgent situation, with the understanding that most gallstone ileus patients are elderly and commonly have numerous medical comorbidities. Two contemporary series of registry data have expanded our understanding of gallstone ileus. This condition was once thought to be relatively rare; however, the National Inpatient Sample study identified 3268 gallstone ileus patients, which accounts for approximately 0.1% of all patients admitted to the hospital with mechanical small bowel obstruction during this time period. In this series, overall hospital mortality was substantial at 6.7%. Mortality was significantly higher in patients who underwent cholecystectomy and closure of the biliary fistula compared to those who simply had small bowel obstruction addressed by cholecystolithotomy. Overall, 77% of the 3268 patients had small bowel obstruction pathology treated and the remaining 23% had biliary fistula closed and cholecystectomy at the same operation. An interesting finding was the substantial incidence of postoperative renal insufficiency, or approximately 30% in the entire group of patients. The latter finding highlights the need for preoperative resuscitation and close attention to postoperative fluid management. The surgeon must consider carefully feasibility of same operation intervention to repair biliary pathology: It is safe to defer biliary operation to a later date with a second staged operation. When this strategy of two stage operation is selected, surgeons should consider and counsel their patient regarding the substantial incidence of recurrent biliary symptoms.
Tratamento Cirúrgico da ACALASIA ESOFÁGICA
A acalasia, definida como a falha ou relaxamento incompleto do esfíncter esofágico inferior (EEI), acompanhada de aperistalse do corpo esofágico na ausência de obstrução mecânica, é o tipo mais comum de distúrbio da motilidade esofágica. Tem uma incidência de 1 em 100.000 pessoas, com uma prevalência de 10 em 100.000. Não há diferença na prevalência de gênero entre as idades de 30 e 60 anos. A causa primária da acalasia ainda é indeterminada, mas acredita-se que surja da degeneração das células ganglionares inibitórias no plexo miontérico do EEI e corpo esofágico. Fatores associados a um risco aumentado de acalasia incluem distúrbios virais/neurodegenerativos, síndrome de Down, diabetes mellitus tipo 1, hipotireoidismo e condições autoimunes, como a síndrome de Sjögren, o lúpus eritematoso sistêmico e a uveíte. Os casos familiares são raros.
O diagnóstico de acalasia deve ser suspeitado em pacientes com disfagia para sólidos e líquidos que não melhora com o uso de inibidores da bomba de prótons. Se não for tratada, a acalasia é uma doença progressiva que pode evoluir para megaesôfago e está associada a um aumento do risco de carcinoma de células escamosas do esôfago. Embora não existam critérios padronizados para determinar a gravidade da doença, o diâmetro e a confirmação do esôfago dentro da cavidade torácica são geralmente considerados os dois principais fatores. A escala de Eckardt é uma escala frequentemente usada para avaliar a gravidade da doença e a eficácia da terapia. Qualitativamente, a acalasia grave é definida como um diâmetro esofágico maior que 6 cm; a acalasia em estágio avançado inclui a angulação distal, um esôfago sigmóide/tortuoso com diâmetro maior que 6 cm ou um megaesôfago com diâmetro maior que 10 cm. Aproximadamente 5% a 15% das pessoas com acalasia evoluem para acalasia em estágio avançado, são geralmente resistentes aos tratamentos endoscópicos e cirúrgicos iniciais e, em última instância, requerem uma esofagectomia.
MIOTOMIA Á HELLER
A miotomia laparoscópica de Heller (LHM) com fundoplicatura parcial, desenvolvida como uma alternativa minimamente invasiva à miotomia anterior aberta tradicional e posteriormente à miotomia toracoscópica, tem sido o padrão ouro para o tratamento da acalasia nas últimas três décadas. O objetivo da miotomia é abrir completamente o EEI e aliviar a disfagia. A LHM proporciona alívio sintomático inicial da disfagia em cerca de 90% dos pacientes com tipos I e II de acalasia e em 50% dos pacientes com tipo III de acalasia, enquanto diminui as taxas de refluxo pós-operatório. Em comparação com os procedimentos de miotomia aberta, a LHM está associada a menor dor pós-operatória, menor tempo de internação hospitalar e retorno mais precoce à função. A LHM é indicada como tratamento de primeira linha para todos os candidatos à cirurgia com acalasia que estejam dispostos a se submeter à cirurgia ou para aqueles que falharam na dilatação endoscópica. A operação consiste em dividir os músculos do EEI, seguida por uma fundoplicatura para diminuir o refluxo pós-operatório. A fundoplicatura parcial é favorecida em relação à fundoplicatura total porque reduz a falha do tratamento. As duas principais complicações da cirurgia são perfuração da mucosa e DRGE.
Surgical Management of Chronic Pancreatitis
Chronic pancreatitis is a progressive, destructive inflammatory process that ends in destruction of the pancreatic parenchyma resulting in malabsorption, diabetes mellitus, and severe pain. The etiology of chronic pancreatitis is multifactorial. About 65–70% of patients have a history of alcohol abuse, the remaining patients are classified as idiopathic chronic pancreatitis (20–25%), including tropical pancreatitis, a major cause of childhood chronic pancreatitis in tropical regions, or unusual causes including hereditary pancreatitis, cystic fibrosis, and chronic pancreatitis-associated metabolic and congenital factors. Current evidence suggests that a combination of predisposing factors, including environmental, toxic, and genetic, are involved in most patients rather than one single factor. The best-known hypotheses about the pathogenesis of chronic pancreatitis include necrosis-fibrosis, toxic-metabolic, oxidative stress, plug and stone formation with duct obstruction, and primary duct obstruction. Repeated episodes of inflammation initiated by autodigestion, one or more episodes of severe pancreatitis, oxidative stress, and/or toxic-metabolic factors lead to activation and continued stimulation of parenchymal pancreatic stellate cells. These stellate cells cause the fibrosis characteristic of chronic pancreatitis. Nevertheless, multiple hypotheses exist to explain the pathophysiology in the various subgroups of patients with chronic pancreatitis, but to date there is no single unifying theory.
OPERATIVE TREATMENT
There are several different concepts for the operative treatment of chronic pancreatitis. The concept of preservation of functioning pancreatic parenchyma (drainage operations) would be the goal for protection against further loss of pancreatic function. The second main concept is based on resective procedures either in the situation where there is no dilation of the pancreatic duct, if the pancreatic head is enlarged, or if a pancreatic carcinoma is suspected in the setting of chronic pancreatitis. These two concepts involve different operative procedures.
Drainage procedures
Sphincterotomy of the pancreatic duct was one of the first operative procedures proposed for patients with a dilated pancreatic duct in chronic pancreatitis with presumed obstruction or stenosis at the papilla Vater. Unfortunately, this procedure was associated with minimal lasting success for the amelioration of pain, indicating that a stenosis at the papilla of Vater is not the cause of pain in chronic pancreatitis nor the cause of ductal dilation. In contrast, direct ductal-enteric drainage by the original Puestow procedure or its modification by Partington and Rochelle is more successful in patients with chronic pancreatitis and a dilated pancreatic duct. The original Puestow procedure included resection of the tail of the pancreas with filleting open the pancreatic duct proximally along the body of the pancreas with anastomosis to a Roux-en-Y loop of jejunum. Partington and Rochelle modified the Puestow procedure by eliminating the resection of the pancreatic tail. A recent procedure involves a wedge-shaped opening of the pancreatic duct (even when the duct is <5 mm) with a subsequent pancreatico-jejunostomy. The preservation of functional tissue and reduction of operative mortality to less than 1% and morbidity to less than 10% are the goals and benefits of this operation. Unfortunately, large series have reported persistence or recurrence of pain at long-term follow up (>5 year) in 30–50% of patients; in addition, patients with a dominant mass in the head of the pancreas and a non-dilated pancreatic duct do not appear to profit from a drainage procedure at all. A recent randomized controlled trial demonstrated that operative drainage in selected patients with a large duct was more effective than endoscopic treatment in patients with obstruction of the pancreatic duct.
Pancreatic Resections
Pancreatoduodenectomy (Kausch-Whipple procedure). For many surgeons, a pancreatoduodenectomy is the gold standard for patients with the pain of chronic pancreatitis, although the newer, duodenum-preserving procedures are good (and possibly better) alternatives as well (see below). The approach of resection of the proximal gland is based on Longmire’s tenet that the ‘‘pacemaker’’ of pain is in the head of the pancreas. The indications for pancreatoduodenectomy in patients with chronic pancreatitis and pain are:
(1) a non-dilated pancreatic duct (diameter < 6 mm measured in the body of the gland),
(2) an enlarged head of the pancreas, often containing cysts and calcifications,
(3) a previous, ineffective ductal drainage procedure, and/or
(4) when there is the possibility of malignancy in the head of the gland.
This latter subgroup comprises up to 6–10% of patients undergoing operative intervention for chronic pancreatitis. After pancreatoduodenectomy, > 80% of patients have permanent pain relief, which is greater than after a drainage operation. In experienced centers, a pancreatoduodenectomy can be performed with a low operative mortality rate (< 2%), and a morbidity of 40%. Although the classic pancreatoduodenectomy has these advantages, there is some long-term morbidity in chronic pancreatitis patients, especially regarding quality of life. In addition to development of diabetes, patients experience postoperative digestive dysfunction, including dumping, diarrhea, peptic ulcer, and dyspeptic complaints. To address these effects of the classic pancreatoduodenectomy which involved a distal gastrectomy, ‘‘organ-preserving’’ operations like the pylorus-preserving pancreatoduodenectomy. Symptoms of dumping and bile-reflux gastritis can be decreased by preserving the stomach, the pylorus, and the first part of the duodenum. In addition, regarding quality of life, a pylorus-preserving technique provides better results than the classic pancreatoduodenectomy procedure; weight gain occurs in 90% of the patients postoperatively while still leading to long-lasting pain relief in 85–90% of the patients. Pylorus-preserving resections, however, appear to have a greater incidence of transient delayed gastric emptying postoperatively (20–30% of the patients) as well as the risk of cholangitis and the long-term occurrence of exocrine and endocrine pancreatic insufficiency (seen in >45% of patients), representing the possible drawbacks of this operation in chronic pancreatitis patients. The relevant studies (level I and II) comparing classic with pylorus preserving pancreatoduodenectomy could not demonstrate a clear advantage for either resection. One should remember, however, that pancreatoduodenectomy was originally introduced to treat malignant pancreatic or periampullary disease by an oncologic resection. Therefore, for a benign disorder such as chronic pancreatitis, there is no reason – other than the occasional inability to exclude pancreatic cancer definitely – to remove peripancreatic organs (the distal stomach, the duodenum, and the extrahepatic bile ducts), which are involved only secondarily in chronic pancreatitis. This concept stimulated the development of organ-preserving pancreatic resections.
Duodenum-preserving pancreatic head resection (DPPHR).
This procedure addresses patients with a dominant mass in the head of the pancreas with or without a dilated main pancreatic duct. The duodenum-preserving resection (Beger procedure) includes a ventral dissection and dorsal mobilization of the head of the pancreas. After division of the pancreas anterior to the porto-mesenteric vein (as with a pancreatoduodenectomy), the resection is carried out toward the papilla of Vater. A subtotal resection of the pancreatic head is carried out leaving a small margin of pancreatic tissue associated with the duodenum containing the common bile duct; a small rim of pancreatic tissue toward the vena cava should be preserved as well during removal of most all of the uncinate process. In most patients, it is possible to free the bile duct from the surrounding scarring without disrupting continuity with the ampulla of Vater, thereby avoiding the need for a bilio-digestive anastomosis.
In some patients (20%), the common bile duct is obstructed and should be opened, so that the bile will drain into the cavity of the resected pancreatic head which is drained into a Roux-en-Y limb of jejunum. The standard reconstruction consists of a Roux-en- pancreaticojejunostomy to the distal pancreatic remnant (body and tail of pancreas) and a pancreatojejunostomy to the rim of pancreas at the duodenum (including the opened bile duct if needed). In up to 10% of patients, this DPPHR procedure is combined with a lateral pancreaticojejunostomy to drain multiple stenoses of the main pancreatic duct. The mortality rate is low (1%), and the morbidity rate is around 15%, less than after pancreatoduodenectomy.
When compared with pancreatoduodenectomy in patients with chronic pancreatitis, the DPPHR offers the advantage of preserving the duodenum and extrahepatic biliary tree, and its superiority over even the pylorus-preserving resection has been shown in prospective studies. Patients who underwent the DPPHR had greater weight gain, a better glucose tolerance, and a higher capacity for insulin secretion. In long-term follow-up, about 20% of the patients developed new onset of diabetes mellitus, like the incidence of diabetes after pancreatoduodenectomy. There is some evidence that endocrine function may be better preserved after DPPHR when compared with patients with chronic pancreatitis not undergoing operation, secondary to the relief of pancreatic ductal obstruction/hypertension. Regarding pain status, 90% of patients after DPPHR have long-term relief of pain. Regarding quality of life, 69% of the patients in one study were rehabilitated professionally, 26% retired, and only 5% of the patients were unimproved. Considering the better pain status, a lesser frequency of acute episodes of chronic pancreatitis, especially in those patients with an enlarged pancreatic head, marked decrease in the need for further hospitalization, low early and late mortality rate, and the restoration of a better quality of life, evidence suggests that DPPHR may delay the natural course of the disease of chronic pancreatitis.
The DPPHR was modified by Frey and colleagues to include a longitudinal pancreatico-jejunostomy combined with a local ‘‘coring out’’ of the pancreatic head without the need for an extensive dissection near the superior mesenteric vessels as with the DPPHR. The Frey and DPPHR have undergone evaluation in multiple comparative trials, confirming their effectiveness as operative procedures for chronic pancreatitis. A modified technique (Bern procedure) of the Beger and Frey procedures has been described recently in patients with chronic pancreatitis. This extended Frey procedure combines the advantages of the Beger and Frey procedure by maintaining a non-anatomic, subtotal central pancreatic head resection but without the need for transsection of the gland over the superior mesenteric vein (SMV), the most tedious part of the DPPHR procedure which was the major advantage offered by the Frey procedure. This modified technique reduces the risk of intraoperative bleeding which is especially increased in the presence of portal hypertension.
Left-sided pancreatic resection (distal pancreatectomy)
Most surgeons believe that the pancreatic head is the pacemaker in chronic pancreatitis, and therefore, pancreatic head resections should be the target for most patients with chronic pancreatitis affecting the entire gland. There is, however, a small and carefully selected group of patients in whom a left-sided pancreatic resection is the appropriate treatment. This subgroup is selected by imaging techniques, including CT, ERCP, or MRI outlining inflammatory complications, such as pseudocysts, fistula, and pancreatic duct stenosis, involving only (or primarily) the body and/or tail region of the pancreas. A good example is the patient who develops a mid-ductal stricture after an episode of necrotizing pancreatitis secondary to gallstone pancreatitis. Similarly, suspicion of a neoplasm or recurrent acute pancreatitis believed secondary to an isolated, mid-ductal stricture may be justification for a left-sided pancreatic resection. Overall, about 10% of all patients who undergo operative intervention for chronic pancreatitis may be candidates for a distal pancreatectomy. These distal pancreatectomies for benign disease can be performed without splenectomy, but conservation of the splenic artery and vein can be difficult and is time-consuming. Nevertheless, the advantage of avoiding the possibility of overwhelming postsplenectomy sepsis should be taken into consideration as well as the importance of the spleen for maintenance of the host defense system. Thus, preservation of the spleen is desirable if there is no clear indication for splenectomy, such as perisplenic pseudocyst or inflammatory/fibrotic encasement of the splenic vessels.
Central pancreatectomy (middle segmentectomy).
Benign lesions of the neck and proximal body of the pancreas, such as the exceedingly rare focal chronic pancreatitis or post-traumatic pancreatitis, pose an interesting operative challenge. If the lesions are not amenable to simple enucleation, surgeons may be faced with the choice of performing a right-sided resection (pancreatoduodenectomy) or a left-sided resection (distal pancreatectomy) to include the lesion, resulting in resection of a substantial amount of otherwise functioning pancreatic parenchyma. Central pancreatic resections have been reported primarily for benign or low-grade neoplasms with Roux-en-Y pancreatojejunostomy reconstruction. Central pancreatectomy affords the possibility of saving functional pancreatic tissue in attempt to avoid the complications of pancreatic insufficiency. Further studies, however, must prove the effectiveness of such an operation for patients with chronic pancreatitis. Central resections in patients with chronic pancreatitis must be viewed with caution and considered only in highly selected cases.
Pancreatic resection with islet cell autotransplantation.
Because of the concerns of pancreatic endocrine insufficiency after any pancreatic resection for chronic pancreatitis, renewed interest has focused on the possibility of performing a total pancreatectomy, isolating the islets, and reinfusing (autotransplanting) the islets into the liver. Improvements in islet cell harvesting and preservation for islet cell allotransplantation for diabetics have allowed new enthusiasm in this technique for patients with chronic pancreatitis. Results to date are encouraging, but the inability to harvest reliably an adequate number of islets and to prove successful engraftment within the liver remain current limitations. This approach may be more effective early in the disease when islets have not been depleted.
OUTCOMES
In summary, definite evidence for the best operative method for treating painful chronic pancreatitis is still not fully accepted. The study designs in the few randomized controlled trials (evidencebased Level I data) available today have some limitations in design and reporting of morbidity and include only small numbers of patients. Nevertheless, the different variations of the DPPHR, Beger, Frey, and Bern procedures appear to be as equally successful in achieving long-term pain control as pancreatoduodenectomy, but they have fewer postoperative complications and appear to be superior with regard to preservation of pancreatic function and quality of life.
Visão Crítica de Segurança (Colecistectomia)
A colecistectomia laparoscópica (CL) é o padrão-ouro para tratamento de cálculos biliares. No entanto, o risco de lesão do ducto biliar (BDI) continua a ser preocupação significativa, uma vez que CL ainda tem taxa de BDI maior do que a via laparotômica, apesar de muitos esforços propostos para aumentar sua segurança. A Visão Crítica da Segurança (CVS) proposta por Strasberg é a técnica para a identificação dos elementos críticos do triângulo de Calot durante a CL. Esta técnica foi adotada em vários programas de ensino e com a proposta de reduzir o risco de lesão acidental da via biliar (LAVB) e o uso da adequado da CVS está associado a menores taxas de LAVB. O objetivo deste #Webinar é abordar a Anatomia Cirúrgica Fundamental para a realização de uma Colecistectomia Laparoscópica.
Intraoperative cholangiography: Selective or Routine?
Intraoperative cholangiography (IOC), described by Mirizzi in 1932, represented a significant advance in the diagnosis of choledocolithiasis during cholecystectomy. The natural history of asymptomatic choledocolithiasis has been investigated in different populations and its therapeutic management continues to be controversial. IOC is traditionally advocated as a procedure to be adopted in all laparoscopic cholecystectomies since it permits to define the anatomy of the biliary tract and to detect common bile duct stones. However, in the laparoscopic era, technological advances in radiologic-endoscopic workup have markedly increased the costs of investigation of patients with suspected choledocolithiasis. The routine use of IOC has raised the question of which cases require the exploration of bile tract anatomy during surgery and whether there are methods to predict preoperatively unsuspected choledocolithiasis. Therefore, the most appropriate management of preoperatively unsuspected choledocolithiasis, i.e., the routine or selective use of IOC, still remains undefined. In this respect, well-defined criteria for the inclusion of patients with possible choledocolithiasis who should be submitted to cholangiography exist in the literature, but there is no safe approach to exclude asymptomatic patients without an indication for contrast examination.
A systematic literature search was performed by KOVACS N, et al (2022) using the following search keys: cholangiogra* and cholecystectomy. The primary outcomes were BDI and retained stone rate. To investigate the differences between the groups (routine IOC vs selective IOC and IOC vs no IOC), they calculated weighted mean differences (WMD) for continuous outcomes and relative risks (RR) for dichotomous outcomes, with 95% confidence intervals (CI). Of the 19,863 articles, 38 were selected and 32 were included in the quantitative synthesis. Routine IOC showed no superiority compared to selective IOC in decreasing BDI (RR = 0.91, 95% CI 0.66; 1.24). Comparing IOC and no IOC, no statistically significant differences were found in the case of BDI, retained stone rate, readmission rate, and length of hospital stay. They found an increased risk of conversion rate to open surgery in the no IOC group (RR = 0.64, CI 0.51; 0.78). The operation time was significantly longer in the IOC group compared to the no IOC group (WMD = 11.25 min, 95% CI 6.57; 15.93). So this data findings suggest that IOC may not be indicated in every case, however, the evidence is very uncertain.
Postpancreatectomy Hemorrhage
Postoperative complications represent one of the most debated topics in pancreatic surgery. Indeed, the rate of complications following pancreatectomy is among the highest in abdominal surgery, with morbidity ranging between 30 and 60%. They are often characterized by elevated clinical burden, with a consequent challenging postoperative management. Mortality rates can exceed 5%. The impact on patient recovery and hospital stay eventually leads to massive utilization of resources and increases costs for the health system. The International Study Group of Pancreatic Surgery (ISGPS) provides standardized definitions and clinically based classifications for the most common complications after pancreatectomy, including postoperative pancreatic fistula (POPF), post pancreatectomy hemorrhage (PPH), delayed gastric emptying (DGE), bile leakage, and chyle leak.
Post Pancreatectomy Hemorrhage
Despite its lower incidence compared with POPF with reported rates of 3–10% after pancreatectomy, PPH remains one of the major of postoperative complications, with mortality rates ranging from 30 to 50%. According to current ISGPS recommendations, PPH is classified in three grades (A, B, and C) based on two main criteria: timing of the hemorrhage and severity of the bleeding. The timing is dichotomized as early, occurring within 24 hours from the index surgery, and late, when it happens afterwards.
Management is tailored according to the clinical picture, the timing of onset, and the presumptive location. Early hemorrhage is generally due to either unsuccessful intraoperative hemostasis or to an underlying coagulopathy. The vast majority of patients are basically asymptomatic, with PPH having no influence on the postoperative course. However, when the bleeding is severe, re-laparotomy is recommended, with the aim of finding and controlling the source of bleeding. This approach usually guarantees an uneventful subsequent course. Late PPH is often challenging and the pathogenesis is diverse. Vascular erosion secondary to POPF or intraabdominal abscess, late failure of intraoperative hemostatic devices, arterial pseudoaneurysm, and intraluminal ulceration are some of the most common causes. In this setting, surgical access to the source of bleeding may be challenging. Angiography (if extraluminal) and endoscopy (if intraluminal) represent the primary approaches to treatment. Surgery is reserved for hemodynamically unstable patients and for those who present with deteriorating condition, multiorgan failure or sepsis. Given these assumptions, except for early mild events, contrast-enhanced abdominal CT is crucial in all cases of PPH, possibly allowing identification of the source and planning the management accordingly. Also, it should be noted that late massive hemorrhages may be preceded by mild self-limiting sentinel bleeds. A prompt abdominal CT scan aimed at excluding vascular lesions is therefore strongly recommended in these cases.
The incidence of complications following pancreatic resection remains high. The ISGPS established standardized definitions and clinical grading systems for POPF, PPH, DGE, and biliary and chyle leak. These classification systems have enabled unbiased comparisons of intraoperative techniques and management decisions. However, the management policies of these complications are most often driven by a patient’s condition and local surgical expertise and is not always based on the available high-level evidence. The development of high-volume specialized units with appropriate resources and multidisciplinary experience in complication management might further improve the evidence and the outcomes.
Subtotal cholecystectomy for difficult acute cholecystitis
Laparoscopic cholecystectomy is considered the gold standard for treatment of benign gallbladder diseases. Cholecystectomy using this method can be completed in 90% of elective cholecystectomies and 70% of emergency cholecystectomies. Acute cholecystitis, especially if difficult, can change the above paradigm, resulting in open conversion or change of technique. The conditions that define a difficult cholecystectomy are as follows: necessity of conversion from laparoscopic to open surgery; duration of procedure greater than 180 min; blood loss greater than 300 ml; and urgent need for involvement of a more experienced surgeon. One of the “rescue” procedures to complete the surgery safely (both for the surgeons and patients) is subtotal cholecystectomy (STC). Open and laparoscopic subtotal cholecystectomy have been reported. For many surgeons, this is considered a bail out technique, and the timing of decision making is crucial to avoid catastrophic complications. Te capability to perform STC in laparoscopy is increasingly requested during difficult laparoscopic cholecystectomy. Difficult LC has a risk of BDI from 3 to 5 times higher in laparoscopy than open surgery. In case of operative difficulties of young surgeons mostly trained in laparoscopy the help of senior surgeons is strongly recommended. The purpose of the present study is to clarify how laparoscopic subtotal cholecystectomy may be used to complete a difficult cholecystectomy for acute cholecystitis without serious complications.

Biliary leakage represents the most frequent complication of incomplete resection of the gallbladder wall in cases of difficult acute cholecystectomy treated with subtotal cholecystectomy. This complication is rarely fatal but requires correct treatment. If bile leakage does not stop spontaneously seven days postoperatively, the possible treatments are endoscopic biliary sphincterotomy, endoscopic plastic stent, and a fully covered self-expanding metal stent. When performing closure of the gallbladder stump, suturing the anterior residual of both anterior and posterior wall represents the best method to have fewer complications. Complications, if not lethal, decrease the patient’s quality of life. Intraoperatively, it is of utmost importance to carefully expose the gallbladder stump to avoid left-in-place stones, wash the entire cavity and drain the abdomen. Bile duct injuries can be a significant complication in this type of surgery. Prevention of the lesions with conversion from laparoscopic to open, or the opinion of older surgeon in case of difficulties is strongly recommended. Mortality is a very rare complication. The limitations of our study are given by the heterogeneity of the techniques used for LSC and the lack of a long-term follow-up analyzing the related complications.
Aderências Pós-Operatórias
Do Manejo Conservador à Decisão Cirúrgica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
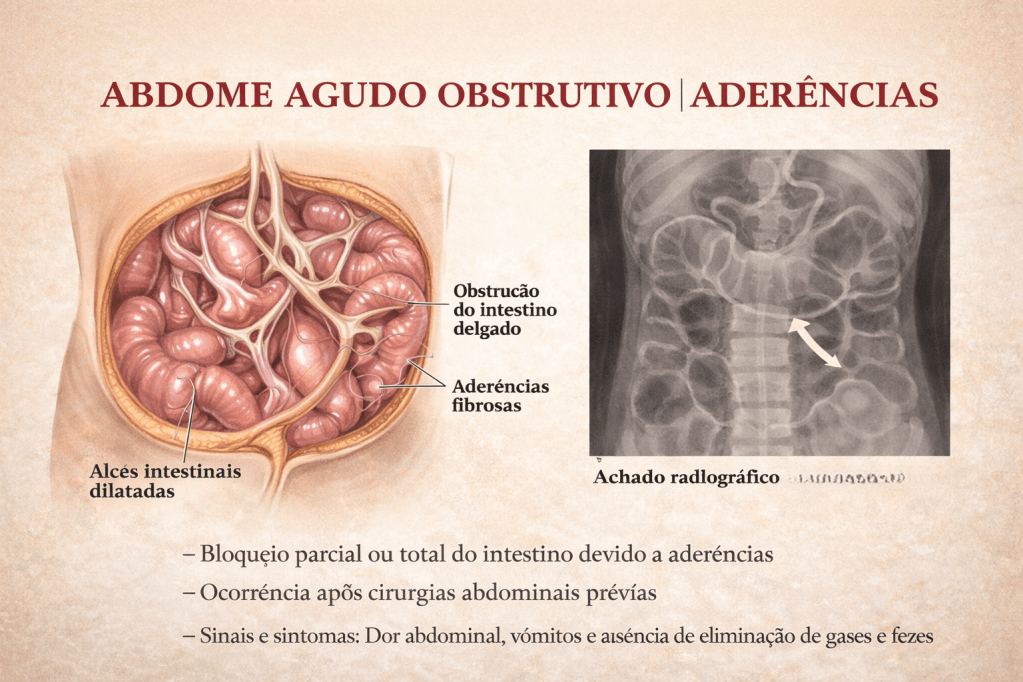
Introdução
A obstrução intestinal é um dos cenários mais desafiadores e frequentes no pronto-socorro cirúrgico. Historicamente, o abdome agudo obstrutivo impunha o dilema imediato da exploração cirúrgica. Hoje, compreendemos que a etiologia dita o compasso da intervenção. Entre todas as causas de obstrução do intestino delgado, as aderências pós-operatórias (bridas) assumem o protagonismo absoluto. Para o estudante de medicina, o residente e o cirurgião do aparelho digestivo, dominar o manejo da obstrução por bridas é essencial. O desafio não reside apenas na técnica operatória da enterólise, mas sim no raciocínio clínico aguçado para responder à pergunta fundamental: quando persistir no tratamento conservador e quando a indicação cirúrgica se torna imperativa?
Cenário Brasileiro
A formação de aderências peritoniais é uma resposta fisiológica quase universal ao trauma cirúrgico (isquemia, manipulação, coágulos), ocorrendo em até 90% dos pacientes submetidos a laparotomias. Contudo, em uma parcela significativa, essa cicatrização aberrante leva à obstrução mecânica. Na prática da cirurgia geral no Brasil, a obstrução por bridas responde por cerca de 60% a 75% das admissões por obstrução do intestino delgado. Dados do Sistema Único de Saúde (DataSUS) indicam que milhares de laparotomias exploradoras são realizadas anualmente no país devido a quadros obstrutivos agudos, gerando elevado custo financeiro e considerável morbimortalidade, além de reinternações frequentes.
Fisiopatologia e Diagnóstico
O peritônio, quando lesado, inicia uma cascata inflamatória rica em fibrina. Em condições normais, a fibrinólise degrada essa matriz em poucos dias. O desequilíbrio nesse sistema — com falha na fibrinólise peritonia — resulta na organização da fibrina em tecido conjuntivo vascularizado: a aderência.
A Tríade Diagnóstica
O diagnóstico é eminentemente clínico e radiológico:
- Clínica: Dor abdominal em cólica, distensão, vômitos (fecaloides em fases tardias) e parada de eliminação de flatus e fezes.
- Laboratório: Hemograma, eletrólitos, lactato e gasometria são cruciais para rastrear sinais de isquemia ou necrose (leucocitose importante, acidose metabólica).
- Imagem: A Tomografia Computadorizada (TC) de abdome com contraste venoso substituiu o raio-X simples como padrão-ouro. A TC identifica o ponto de transição (zona de calibres distintos), o grau de obstrução, a presença de sofrimento de alça (espessamento parietal, pneumatose, gás no sistema porta) e descarta outras etiologias (hérnias internas, neoplasias).
Estratégias de Tratamento
O manejo da obstrução por bridas sofreu uma evolução paradigmática, distanciando-se de intervenções intempestivas em favor de protocolos baseados em evidências.
1. Tratamento Conservador (Não Operatório)
Na ausência de sinais de isquemia intestinal, perfuração ou peritonite localizada, a conduta inicial deve ser conservadora:
- Jejum (NPO) e descompressão com Sonda Nasogástrica (SNG).
- Ressuscitação volêmica vigorosa e correção de distúrbios hidroeletrolíticos.
- Protocolo com Contraste Hidrossolúvel: A administração de contraste hiperosmolar (ex: Gastrografin) via SNG não possui apenas valor diagnóstico (avaliar se o contraste atinge o cólon em 24h), mas também efeito terapêutico. A hiperosmolaridade atrai líquido para a luz intestinal, reduzindo o edema da parede e estimulando o peristaltismo, resolvendo o quadro obstrutivo em uma parcela significativa dos casos.
2. Tratamento Cirúrgico: O Timing Ideal
A falha do tratamento conservador (geralmente avaliada após 48 a 72 horas) ou o surgimento de sinais de alarme (taquicardia, febre, irritação peritoneal, acidose) indicam cirurgia de urgência.
- Laparotomia vs. Laparoscopia: A via aberta continua sendo o padrão em pacientes com múltiplas cirurgias prévias ou grande distensão abdominal. A abordagem laparoscópica é excelente em mãos experientes, mas reserva-se a casos selecionados (ex: suspeita de brida única, distensão moderada), exigindo extrema cautela na introdução do primeiro trocarte (técnica aberta de Hasson) para evitar enterotomias iatrogênicas.
- O Ato Cirúrgico: A lise de aderências deve ser restrita ao necessário para resolver a obstrução. Múltiplas enterólises desnecessárias aumentam o risco de lesões e a formação de novas bridas. Em caso de necrose, a ressecção do segmento isquêmico com anastomose primária ou estomia (a depender da estabilidade hemodinâmica) é imperativa.
Pontos-Chave para a Prática Diária
- Suspeição de Isquemia: Dor refratária a analgésicos, taquicardia inexplicada e acidose metabólica são gritos de socorro de uma alça intestinal isquêmica. O tratamento conservador é contraindicado.
- O Valor da TC: A tomografia não apenas confirma o diagnóstico, mas é o farol que guia a decisão entre a enfermaria e o centro cirúrgico.
- Prevenção Primária: O uso de barreiras antiaderentes ainda carece de evidências universais robustas, mas a técnica cirúrgica apurada, manipulação tecidual delicada e controle rigoroso da hemostasia continuam sendo a melhor profilaxia.
Conclusões Aplicadas
O tratamento da obstrução intestinal por bridas é a quintessência do bom senso cirúrgico. O cirurgião moderno deve possuir a paciência de um clínico para conduzir o tratamento conservador respaldado pelo contraste hidrossolúvel e, simultaneamente, a assertividade de um intervencionista para indicar a sala de cirurgia ao primeiro sinal de estrangulamento.Compreender que “operar a radiografia” é um erro crasso e que a reavaliação clínica seriada (de preferência pelo mesmo examinador) é a bússola do tratamento, molda o profissional de excelência que minimiza ressecções intestinais desnecessárias e reduz a morbidade pós-operatória.
“Na obstrução intestinal aguda, não deixe o sol se pôr ou nascer sobre o paciente sem uma decisão. Mas lembre-se: a cirurgia sem indicação precipita o desastre que buscava evitar.” — Aforismo clássico derivado dos ensinamentos de Sir Zachary Cope, pioneiro no estudo e diagnóstico do abdome agudo.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.