Manobras Cirúrgicas Avançadas no Trauma Abdominal
A Arte da Exposição e o Controle do Caos
Autor: Prof. Dr. Ozimo Gama
Categoria: Cirurgia do Trauma / Técnica Cirúrgica / Anatomia Aplicada
Tempo de Leitura: 18 minutos
Introdução
No “teatro de operações” do trauma abdominal grave, o cirurgião enfrenta o maior de todos os inimigos: o tempo. Quando a cavidade é aberta em uma laparotomia de emergência, a “névoa da guerra” — composta por sangue, coágulos e contaminação entérica — obscurece as lesões vitais. Como ensinam Hirshberg e Mattox no clássico Top Knife, “se você não consegue ver a lesão, você não consegue pará-la”. A cirurgia do trauma não é o lugar para dissecções anatômicas delicadas de livro-texto; é o domínio da exposição tática. O domínio das manobras de rotação visceral e mobilização de órgãos sólidos é o que separa o cirurgião hesitante do estrategista capaz de resgatar um paciente da tríade letal (acidose, hipotermia e coagulopatia). Este artigo aprofunda-se nas manobras fundamentais para o controle de danos e tratamento definitivo dos traumas de órgãos específicos.
Manobras de Exposição e Acesso Vascular
Antes de tratar um órgão específico, o cirurgião deve dominar as grandes manobras de rotação, que permitem o acesso ao retroperitônio e aos grandes vasos.
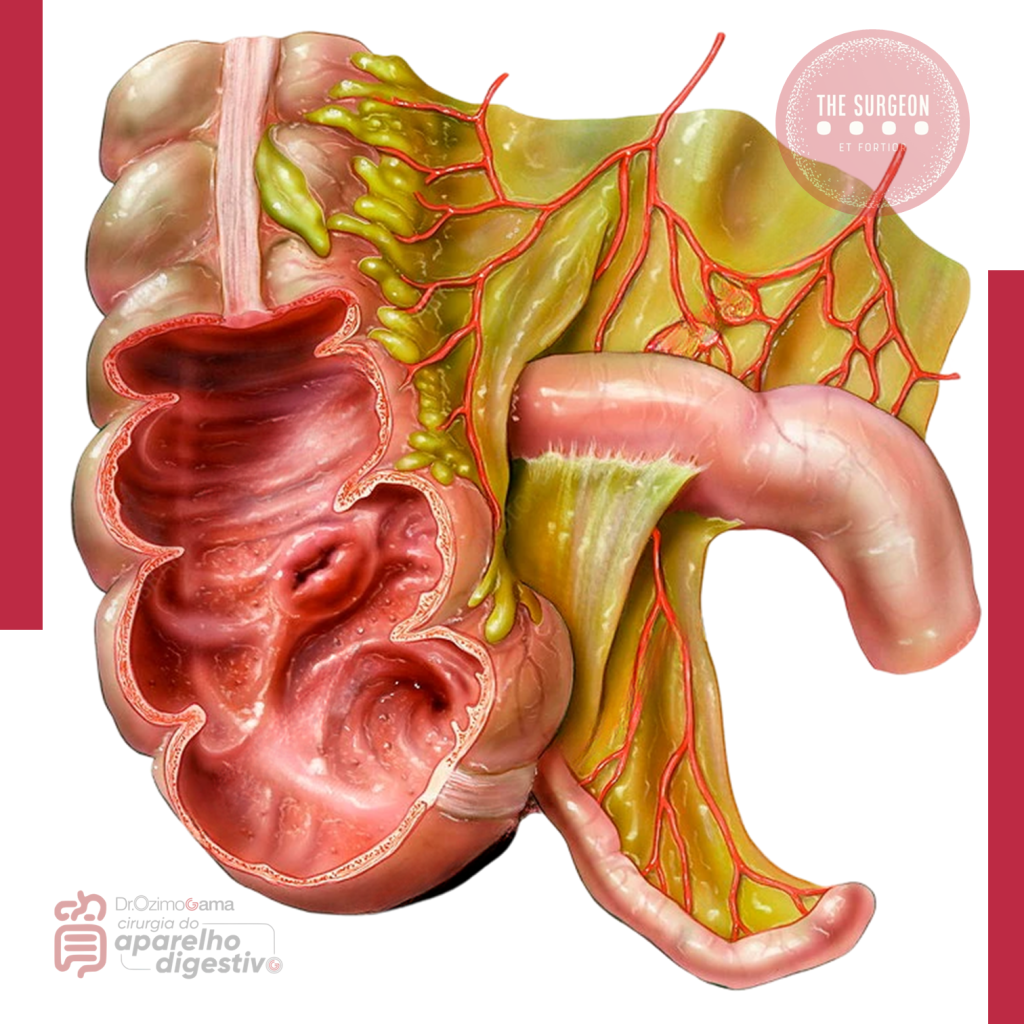
1. Manobra de Mattox (Rotação Visceral Medial Esquerda)
É a “chave mestra” para o acesso à aorta abdominal suprarrenal e aos vasos renais esquerdos.
- Técnica: Inicia-se com a mobilização do cólon esquerdo (linha de Toldt), estendendo a incisão superiormente, atrás do baço e da cauda do pâncreas. Todo o bloco visceral (cólon, rim, pâncreas e baço) é rotacionado medialmente para a direita.
- Aplicação: Essencial para o controle de sangramentos aórticos próximos ao hiato diafragmático ou lesões da artéria mesentérica superior na sua origem.
2. Manobra de Cattell-Braasch (Rotação Visceral Medial Direita)
Permite a visualização completa do retroperitônio infra e parapancreático.
- Técnica: Realiza-se a mobilização do cólon direito e da base do mesentério do intestino delgado até o ligamento de Treitz. O bloco é rotacionado para a esquerda e para cima.
- Aplicação: Exposição da veia cava inferior (VCI) em toda a sua extensão infra-hepática, dos vasos renais direitos e da aorta infrarrenal.
Manobras Específicas por Órgão
1. Fígado: O Gigante da Hemorragia
O trauma hepático é a principal causa de morte por hemorragia intra-abdominal. As manobras visam o controle do influxo e efluxo sanguíneo.
- Manobra de Pringle: Consiste na oclusão do ligamento hepatoduodenal (veia porta, artéria hepática e via biliar). Se o sangramento persistir após o Pringle, a fonte é provavelmente o efluxo (veias hepáticas ou VCI retro-hepática).
- Mobilização Hepática: Secção dos ligamentos falciforme, coronários e triangulares. Isso permite “trazer o fígado para a linha média”, essencial para suturas em lesões posteriores.
- Packing (Tamponamento): A manobra de controle de danos mais eficaz. Envolve o uso de compressas (“The Big Five”) colocadas entre o fígado e a parede abdominal/diafragma, criando compressão direta sobre a lesão.
2. Baço: Preservação vs. Sacrifício
No trauma esplênico, a decisão é binária: salvar ou retirar.
- Mobilização Esplênica: Através da secção do ligamento esplenorenal. O baço é trazido para a ferida cirúrgica, permitindo a inspeção do hilo e da cauda do pâncreas.
- Técnica: Em pacientes instáveis, a esplenectomia rápida é a conduta tática correta. Suturas esplênicas (esplenorrafias) são reservadas para pacientes estáveis e lesões capsulares simples.
3. Complexo Duodeno-Pâncreas
Devido à sua localização retroperitoneal profunda, as lesões aqui são frequentemente ocultas.
- Manobra de Kocher: Abertura da fáscia de coalescência lateral ao duodeno, permitindo a mobilização da segunda porção do duodeno e da cabeça do pâncreas para a linha média.
- Exposição do Corpo/Cauda: Realizada através da abertura do ligamento gastrocólico (acesso à transcavidade dos epíplons).
A Mentalidade de Controle de Danos
Na prática do cirurgião do aparelho digestivo brasileiro, a aplicação dessas manobras deve seguir a lógica da Cirurgia de Controle de Danos (DCS). No trauma, a técnica deve ser “rápida e suja” (quick and dirty), focada na fisiologia e não na anatomia estética. Por exemplo, em uma lesão pancreática grave com sangramento ativo, a manobra de Kocher extensiva é vital para identificar se há envolvimento da VCI ou da aorta. Se houver contaminação entérica maciça por lesão duodenal, o uso de grampeadores para “fechar as pontas” e realizar o packing é superior a tentativas de reconstruções complexas que consomem tempo e levam à exaustão metabólica.
Pontos-Chave para a Estratégia Operacional
- Exposição Antecipada: Não espere o paciente chocar para realizar uma manobra de Mattox ou Cattell-Braasch. Se há hematoma retroperitoneal em expansão, a manobra deve ser imediata.
- Regra do Pringle: Sempre tente o Pringle primeiro em hemorragias hepáticas; é um teste diagnóstico e terapêutico simultâneo.
- Mobilização Ampla: No trauma, “incisões grandes curam grandes cirurgiões”. Não tente operar através de pequenos acessos; a xifopúbica é a via de regra.
- O Packing é uma Manobra Ativa: O tamponamento deve ser firme e direcionado. Mal executado, ele apenas esconde o sangramento; bem executado, ele salva vidas.
- Respeito aos Grandes Vasos: No retroperitônio, cada milímetro de dissecção exige consciência situacional absoluta das variações anatômicas.
Conclusões Aplicadas à Prática
As manobras cirúrgicas no trauma são ferramentas de Estratégia Operacional. Elas permitem que o cirurgião converta um ambiente de caos absoluto em um cenário de controle relativo. Como Professor de Anatomia, reitero que a técnica manual é inútil sem o mapa mental da anatomia cirúrgica. Dominar a rotação visceral medial e as táticas de tamponamento hepático não é um luxo acadêmico, mas a base da sobrevivência no trauma. Na cirurgia digestiva de urgência, a simplicidade e a rapidez nas manobras de exposição são as maiores virtudes que um cirurgião pode possuir. Lembre-se: no trauma, o cirurgião é o estrategista que decide quando lutar por um órgão e quando recuar para salvar a vida.
“Na cirurgia do politrauma as coisas simples funcionam. Técnicas complexas e manobras sofisticadas permitem belas ilustrações, mas muitas vezes não permitem salvar a vida do paciente.” — Asher Hirshberg e Kenneth Mattox.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Hashtags:
#ManobrasCirurgicas #TraumaAbdominal #CirurgiaDeTrauma #TopKnife #AnatomiaCirurgica
Semiologia do Abdome Agudo
A Arte do Diagnóstico Clínico na Era da Tecnologia
Caros acadêmicos e colegas cirurgiões, é com o rigor que a ciência exige e o acolhimento que a medicina impõe que iniciamos esta reflexão. No cenário frenético das emergências brasileiras, a dor abdominal não é apenas uma queixa; é um enigma que responde por 5% a 10% de todas as admissões. Embora vivamos a era da imagem (radiologia), é imperativo recordar que, se 30% dos casos desafiam o diagnóstico inicial, 70% são plenamente resolvíveis apenas com as mãos, os olhos e a percepção clínica. Negligenciar a semiologia em prol da tecnologia não é apenas um erro metodológico; é um risco à vida, dada a morbi-mortalidade associada a diagnósticos tardios.
1. A Fisiopatologia da Dor: O Alerta da Lesão Tecidual
Para compreendermos o abdome agudo, devemos interpretar a dor sob a ótica da quebra da homeostase. Observem bem o raciocínio: a dor é um mecanismo de proteção. Quando um estímulo nocivo atinge o organismo, ele ativa nociceptores localizados no mesentério, nas superfícies peritoneais e na mucosa das vísceras. O ponto fundamental aqui é a dualidade do fenômeno. Existe o componente sensorial discriminativo, que nos permite aferir intensidade e localização, e o componente emocional/afetivo, processado pelo sistema límbico. Por que o exame físico deve começar com um aperto de mão e a palpação do pulso? Não apenas para checar a perfusão, mas para o acolhimento. Ao acolher o emocional do paciente, o cirurgião “limpa” o sinal neural, permitindo que o componente sensorial surja com clareza para a anamnese.
Se analisarmos o Homúnculo de Penfield, notaremos que a representação cortical das vísceras é minúscula se comparada à pele. Vejam a lógica: se a representação no córtex é reduzida, o cérebro tem dificuldade em localizar o sinal. É por essa razão fisiopatológica que a dor visceral é vaga e mal definida, enquanto a dor parietal, vastamente representada no homúnculo, é precisa e cortante.
2. A Trindade da Dor: Visceral, Parietal e Referida
A diferenciação dos padrões de dor é o que separa o diagnóstico brilhante da confusão clínica.
- Dor Visceral: Conduzida por Fibras C (lentas), é a dor da distensão ou torção. Como a inervação visceral é tipicamente bilateral e sua origem é embrionária, ela se manifesta na linha média. O Ligamento de Treitz é o nosso marco: dores do intestino anterior (estômago, duodeno, sistema hepatobiliar) surgem no epigástrio; do intestino médio (do Treitz à flexura hepática, incluindo o apêndice) na região periumbilical; e do intestino posterior no hipogástrio.
- Dor Parietal (Somática): Decorre da irritação do peritônio parietal por processos inflamatórios ou químicos (sangue, pus, bile). Diferente da visceral, é conduzida por fibras aferentes somáticas, sendo bem localizada e indicando, quase invariavelmente, gravidade e potencial cirúrgico.
- Dor Referida: Um fenômeno de convergência medular. As fibras aferentes da víscera e as somáticas da pele entram no corno posterior da medula no mesmo nível. O cérebro, confuso, projeta a dor em uma área distante da origem. Recordem-se do Sinal de Kehr (dor no ombro por irritação diafragmática).
Nota de mestre: Considerem a pelve como uma “Muralha Óssea”. Por ser uma estrutura de difícil palpação, a dor referida é, muitas vezes, o único sinal de alerta para patologias pélvicas graves. Nestes casos, o toque retal e vaginal são as únicas janelas para transpor essa muralha.
3. O Método Osleriano e o Exame Físico
Sir William Osler ensinava: “Cultive o seu poder de observação”. O exame não começa na palpação, mas na fácies de dor, na posição antálgica e na sudorese. O paciente com peritonite está imóvel; o que tem cólica, agita-se. O toque médico deve ser o primeiro ato de tratamento. Ao palpar o pulso e sentir uma taquicardia ou um pulso fino, você já está diagnosticando o choque autonômico antes mesmo de olhar o monitor. O acolhimento reduz a agitação, permitindo que você identifique a defesa abdominal (arco reflexo involuntário) ou a irritação peritoneal (percepção cerebral de dor à descompressão).
4. Diagnóstico Sindrômico: O Norte da Conduta
Antes de buscarmos a etiologia exata, devemos classificar o paciente em um dos cinco grandes grupos:
- Inflamatório: Dor insidiosa que se localiza com o tempo (Ex: Apendicite).
- Perfurativo: Dor súbita, em “facada”, com irritação peritoneal franca. Atenção: processos químicos podem ser lentos se bloqueados pelo mesentério.
- Obstrutivo: Dor em cólica, distensão e parada de eliminação de flatos e fezes.
- Vascular (Isquêmico): Caracteriza-se por uma dor desproporcional ao exame físico. Comum em idosos com comorbidades cardiovasculares.
- Hemorrágico: Dor associada a sinais inequívocos de hipovolemia.
5. Armadilhas e o Triunfo da Clínica
Devemos ter vigilância extrema com os “Pacientes de Alto Risco”: idosos, imunodeprimidos, gestantes e, sobretudo, o paciente que retorna. Qualquer indivíduo que volta à unidade com a mesma queixa é, por definição, de altíssimo risco. Lembrem-se: os exames são complementares. A “rotina de abdome agudo” solicitada sem critério é uma muleta para quem não soube examinar. Como dizemos na cirurgia: “Ausência de evidência não é evidência de ausência.”
6. A Regra de Ouro
O diagnóstico de um abdome agudo deve ser um filme, não uma fotografia. Se o diagnóstico não estiver claro, a observação seriada é o caminho. O prontuário deve ser o registro fiel desse cuidado, contendo a história de vida daquela dor. Encerro com a máxima de Sir Zachary Cope, que deve ecoar em cada plantão:
“Qualquer dor abdominal que perdure por mais de 6 horas em paciente previamente saudável é de importância cirúrgica até prova em contrário.”
O diagnóstico precoce é o que define a fronteira entre a vida e o óbito. Honrem a semiologia.
Gostou ❔ Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e/ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Extra | Epônimos Semiológicos Abdominais
A
- Sinal de Aaron
Dor ou sensação de desconforto no epigástrio/precordial provocada por pressão contínua no ponto de McBurney; classicamente ligado à apendicite.
Descrito por: Charles Dettie Aaron — norte-americano — 1913. - Sinal de Alders
Na dor parietal/aderencial, o ponto doloroso tende a permanecer fixo quando a paciente muda de posição; útil no diferencial de dor abdominal na gestação.
Descrito por: Nicholas Alders — húngaro de origem, naturalizado britânico — 1951.
B
- Sinal de Ballance
Macicez fixa no flanco esquerdo e macicez móvel no flanco direito; sugere ruptura esplênica com hemoperitônio.
Descrito por: Charles Alfred Ballance — britânico/inglês — 1898. - Sinal de Bassler
Dor à compressão do ponto apendicular por trás da linha médio-axilar; descrito em contexto de apendicite crônica.
Descrito por: Anthony Bassler — norte-americano — 1913. - Sinal de Bastedo
Dor em FID desencadeada por insuflação do cólon; empregado na investigação de apendicite.
Descrito por: Walter Bastedo — canadense radicado nos EUA / canadense-americano — 1910. - Sinal de Blumberg
Dor à descompressão brusca; indica irritação peritoneal.
Descrito por: Jacob Moritz Blumberg — alemão — 1907. - Sinal de Boas
Hiperestesia subescapular direita; clássico em colecistite aguda.
Descrito por: Ismar Isidor Boas — alemão — 1894. - Sinal de Bryant
Equimose azulada escrotal por rastreamento de sangue retroperitoneal/hemoperitônio.
Descrito por: John Henry Bryant — britânico/inglês — 1903. - Sinal de Bryan
Dor em FID à tração do útero ou da vagina em gestante; descrito em apendicite na gravidez.
Descrito por: R. Bryan — norte-americano — 1955.
C
- Sinal de Carnett
A dor piora ou não se altera com contração da parede abdominal, favorecendo origem na parede abdominal.
Descrito por: John Berton Carnett — norte-americano — 1926. - Sinal de Chutro
Desvio umbilical para a direita por contratura antálgica; descrito em apendicite aguda.
Descrito por: Pedro Chutro — argentino — 1912. - Sinal do obturador de Cope
Dor hipogástrica/FID com rotação interna da coxa fletida, sugerindo apêndice pélvico inflamado.
Descrito por: Zachary Cope — britânico/inglês — 1919. - Sinal do psoas de Cope
Dor à extensão passiva da coxa direita, associada a apendicite retrocecal.
Descrito por: Zachary Cope — britânico/inglês — 1921. - Sinal de Courvoisier–Terrier
Vesícula biliar palpável, aumentada e indolor em paciente ictérico; sugere obstrução biliar maligna mais que litíase crônica.
Descrito por: Ludwig Georg Courvoisier — suíço — 1890; antecedente histórico associado a Louis-Félix Terrier — francês — 1889. - Sinal de Cullen
Equimose periumbilical, típica de hemorragia intra/retroperitoneal, incluindo pancreatite hemorrágica e gravidez ectópica rota.
Descrito por: Thomas Stephen Cullen — canadense — 1918.
D
- Sinal de Dance
“Vazio” à palpação na fossa ilíaca direita; clássico em intussuscepção.
Descrito por: Jean-Baptiste Hippolyte Dance — francês — 1826. - Sinal de Danforth
Dor no ombro à inspiração por irritação diafragmática; associado a hemoperitônio, especialmente em gravidez ectópica rota.
Descrito por: William C. Danforth — norte-americano — década de 1920. - Sinal de Dunphy
A tosse exacerba a dor abdominal, sugerindo irritação peritoneal localizada.
Descrito por: geralmente atribuído a John Englebert Dunphy — norte-americano — 1953; a atribuição histórica não é totalmente uniforme nas fontes secundárias.
F
- Sinal de Fothergill
Massa da parede abdominal que permanece palpável com contração do reto e não cruza a linha média; típico de hematoma da bainha do reto.
Descrito por: William Edward Fothergill — britânico/inglês — 1926. - Sinal de Fox
Equimose abaixo do ligamento inguinal; indica hemorragia retroperitoneal.
Descrito por: John Adrian Fox — britânico/inglês — 1966.
G
- Sinal de Grey Turner
Equimose em flancos, típica de hemorragia retroperitoneal, sobretudo na pancreatite hemorrágica.
Descrito por: George Grey Turner — britânico/inglês — descrito em 1919 e publicado em 1920.
H
- Sinal de Howship–Romberg
Dor irradiada para a face medial da coxa por compressão do nervo obturatório; clássico de hérnia obturatória.
Descrito por: John Howship — britânico/inglês — 1840; complementado por Moritz Heinrich Romberg — alemão — 1847/1848.
J
- Sinal de Jobert
Desaparecimento da macicez hepática à percussão, sugerindo pneumoperitônio.
Descrito por: Antoine Joseph Jobert de Lamballe — francês — tradicionalmente atribuído à década de 1830; a data primária exata não ficou inequívoca nas fontes abertas consultadas.
K
- Sinal de Kehr
Dor referida no ombro esquerdo por irritação diafragmática, típica de ruptura esplênica ou hemoperitônio.
Descrito por: Hans Kehr — alemão — epônimo consolidado entre o fim do século XIX e início do XX.
L
- Sinal de Lapinsky
Dor em FID agravada por compressão local enquanto o paciente eleva o membro inferior direito estendido; associado à apendicite retrocecal.
Descrito por: Michael Lapinsky — russo — tradicionalmente situado no fim do século XIX; a data exata não foi confirmada com segurança nas fontes abertas consultadas. - Sinal de Lennander
Diferença entre temperatura retal e axilar, usada historicamente em processos inflamatórios abdominais, incluindo apendicite.
Descrito por: Karl Gustaf Lennander — sueco — fim do século XIX. - Sinal de Lockwood
Sensação de gorgolejo ou “borbulhamento” à palpação na FID em apendicite.
Descrito por: Charles Barrett Lockwood — britânico/inglês — epônimo difundido em publicação de 1932, baseada em seu ensino clínico prévio.
M
- Sinal de Mallet-Guy
Dor profunda no hipocôndrio esquerdo/ângulo costovertebral esquerdo, associada à pancreatite crônica.
Descrito por: Pierre Mallet-Guy — francês — 1943. - Sinal de Markle
Dor provocada pela queda sobre os calcanhares (“heel drop test”), sugerindo irritação peritoneal.
Descrito por: George B. Markle IV — norte-americano — 1973. - Sinal de Mayo-Robson
Dor à palpação no ângulo costovertebral esquerdo/“ponto de Mayo-Robson”, associado a doença pancreática.
Descrito por: Arthur William Mayo-Robson — britânico/inglês — início do século XX. - Sinal de McBurney
Dor máxima no ponto de McBurney, típica de apendicite aguda.
Descrito por: Charles Heber McBurney — norte-americano — 1889. - Sinal de Moynihan
Dor/hipersensibilidade no ponto cístico ou sob o rebordo costal direito, em contexto de colecistite.
Descrito por: Berkeley George Andrew Moynihan — britânico/inglês — 1905. - Sinal de Murphy
Interrupção inspiratória por dor à palpação do ponto cístico; clássico em colecistite aguda.
Descrito por: John Benjamin Murphy — norte-americano — 1903.
P
- Sinal de Perman
Dor em FID desencadeada por manobras compressivas à esquerda; historicamente, antecipa o que depois se difundiu como “Rovsing”.
Descrito por: Emil Samuel Perman — sueco — 1904.
R
- Sinal de Rovsing
Hoje descrito como dor em FID à palpação/compressão do lado esquerdo; usado no diagnóstico de apendicite aguda, embora a descrição original de 1907 fosse mecanicamente mais específica.
Descrito por: Niels Thorkild Rovsing — dinamarquês — 1907. - Sinal de Rosenstein / Sitkovskiy
Dor em FID ao decúbito lateral esquerdo; clássico em apendicite.
Descrito por: Piotr Porfiryevich Sitkovskiy — russo — 1922; associação histórica com Paul Rosenstein — alemão.
S
- Triângulo de Sherren
Área triangular de hiperestesia cutânea na FID, descrita na apendicite aguda.
Descrito por: James Sherren — britânico — 1903.
T
- Sinal de Torres Homem
Dor intensa à percussão digital na projeção hepática; classicamente associado a abscesso hepático.
Descrito por: João Vicente Torres Homem — brasileiro — século XIX; a data primária exata não foi localizada de forma confiável nas fontes abertas consultadas.
V
- Sinal de Volkovich–Kocher
Migração da dor do epigástrio/periumbilical para a FID, típica da apendicite aguda.
Descrito por: Nikolay Markianovich Volkovich — ucraniano — epônimo consolidado no início do século XX; frequentemente associado também à tradição clínica de Theodor Kocher — suíço. - Sinal de Voskresensky
Dor em FID durante deslizamento rápido da mão sobre a parede abdominal (“sinal da camisa”); descrito em apendicite.
Descrito por: Vladimir Mikhailovich Voskresensky — russo — 1940.
Qual a função do Apêndice Vermiforme?
Da Teoria Vestigial ao Protagonismo Imunológico e Microbiológico
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 12 minutos)
Introdução
Durante mais de um século, o apêndice vermiforme foi relegado ao status de um mero resquício evolutivo — um órgão vestigial, desprovido de função e célebre apenas pela sua capacidade de inflamar e exigir intervenção cirúrgica de urgência. No entanto, a medicina baseada em evidências e a compreensão profunda do trato gastrointestinal reescreveram esta narrativa. Longe de ser uma “sobra” anatômica, o apêndice é hoje reconhecido como uma estrutura morfofuncional de extrema relevância, atuando ativamente na imunidade mucosal e na regulação do microbioma intestinal. Para o estudante de medicina, o residente e o cirurgião do aparelho digestivo, compreender a anatomia, a fisiologia e o impacto a longo prazo da ressecção deste pequeno órgão tubular é fundamental para aprimorar o raciocínio clínico e refinar as indicações terapêuticas perante a suspeita de um abdome agudo inflamatório.
A Anatomia Cirúrgica do Apêndice
O conhecimento anatômico do apêndice é o mapa que guia o bisturi (ou o trocarte) na sala de operações.
1. Morfologia e Topografia
O apêndice vermiforme é uma projeção tubular cega que se origina na confluência das tênias colônicas, no ceco. Seu comprimento varia de 5 a 25 cm (média de 10 cm). Sua localização típica é na fossa ilíaca direita, topografada na superfície abdominal pelo ponto de McBurney (terço externo da linha imaginária entre a espinha ilíaca anterossuperior direita e a cicatriz umbilical). Contudo, a ponta do apêndice é como o ponteiro de um relógio, assumindo diversas posições:
- Retrocecal (mais comum – aprox. 65%): Oculto atrás do ceco, frequentemente simulando afecções renais ou dificultando o diagnóstico de peritonite precoce.
- Pélvico (aprox. 30%): Pode irritar a bexiga ou anexos uterinos.
- Subcecal, Pré ou Pós-ileal.
- Nota Cirúrgica: Em casos raros de má rotação intestinal ou situs inversus, o apêndice pode estar localizado no quadrante inferior esquerdo.
2. Vascularização e Inervação
A irrigação provém da artéria apendicular, um ramo terminal da artéria ileocólica (ramo da mesentérica superior), que transita na borda livre do mesoapêndice. Por ser uma artéria terminal, a oclusão da luz apendicular (por um fecalito) gera rápido ingurgitamento, trombose, isquemia e, subsequentemente, necrose e perfuração. A inervação, oriunda do plexo mesentérico superior (T10), explica a evolução clássica da dor na apendicite aguda: inicialmente dor visceral, difusa e periumbilical; que, com a progressão da inflamação para o peritônio parietal (inervação somática), migra e se localiza na fossa ilíaca direita.
A Mudança de Paradigma: Funções Funcionais (Microbioma e Imunidade)
A verdadeira revolução na compreensão do apêndice ocorreu no campo da imunologia e da microbiologia.
O “Refúgio Seguríssimo” (Safe House) do Microbioma
A mucosa apendicular é rica em biofilmes e serve como um autêntico “cofre” para as bactérias comensais (benéficas) do intestino. Durante infecções gastrointestinais severas, que resultam em diarreia profusa e “lavagem” da flora cólica, o apêndice mantém um reservatório seguro destas bactérias. Uma vez cessado o quadro agudo, este reservatório é utilizado para repovoar o cólon, restaurando a homeostase do microbioma de forma acelerada.
O Campo de Treinamento Imunológico
O apêndice é um componente vital do GALT (Gut-Associated Lymphoid Tissue). Ele contém uma densa rede de folículos linfoides e células M especializadas. Estas células capturam antígenos luminais e os apresentam aos linfócitos (Células T e B), orquestrando a resposta imune local (produção de IgA) e sistêmica. O apêndice atua como um órgão linfoide primário no desenvolvimento inicial e como um sentinela imune na vida adulta.
Aplicação na Cirurgia Digestiva e Implicações Clínicas
A apendicectomia continua sendo o procedimento cirúrgico de urgência mais realizado no mundo. Contudo, a ablação deste órgão carrega repercussões que a cirurgia moderna não pode mais ignorar.
1. Implicações da Apendicectomia a Longo Prazo
A remoção da “base de treinamento” imune e do reservatório microbiano altera a fisiologia intestinal. Estudos epidemiológicos recentes sugerem que a apendicectomia pode estar associada a um risco aumentado de desenvolvimento de:
- Síndrome do Intestino Irritável (SII): Devido à disbiose crônica (redução de bactérias protetoras, como o Butyricicoccus).
- Doenças Metabólicas e Autoimunes: Modulações na tolerância imunológica têm sido correlacionadas com maior incidência de Diabetes Mellitus tipo 2 e Lúpus.
- Risco Oncológico: Embora ainda exija mais robustez estatística, algumas coortes sugerem que a perda da vigilância imunológica (redução de células T CD3+ e CD8+ induzidas pelo apêndice) possa ter um papel na carcinogênese colorretal.
2. O Debate Contemporâneo: Cirurgia vs. Antibioticoterapia
Dado o seu papel funcional, extirpar o apêndice profilaticamente ou ao menor sinal de dor já não é consenso absoluto. A literatura recente, notavelmente o estudo CODA publicado no New England Journal of Medicine (NEJM, 2020), demonstrou que a antibioticoterapia exclusiva pode ser uma alternativa segura e eficaz em casos selecionados de apendicite aguda não complicada, evitando a cirurgia e preservando o órgão em uma parcela significativa dos pacientes. Cabe ao cirurgião avaliar criteriosamente a presença de fecalitos (que predizem falha do tratamento clínico) e o risco de ruptura.
Pontos-Chave para a Prática Diária
- Não é Vestigial: O apêndice é um órgão linfoide ativo e um modulador essencial do microbioma e da imunidade mucosal.
- A Anatomia Dita a Clínica: A variação na posição da ponta do apêndice (retrocecal, pélvica) é a principal causa de apresentações clínicas atípicas que atrasam o diagnóstico cirúrgico.
- A Isquemia é Precoce: Por ser irrigado por uma artéria terminal (a. apendicular), a obstrução luminal rapidamente evolui para gangrena e perfuração se não diagnosticada.
- Reservatório Protetor: O apêndice atua como um “safe house” bacteriano, acelerando a recuperação do cólon após infecções entéricas severas.
- Apendicectomia tem Preço Fisiológico: A remoção não é isenta de consequências a longo prazo, predispondo a disbiose, SII e possíveis disfunções imunológicas.
- Tratamento Conservador: O manejo não operatório com antibióticos da apendicite não complicada é uma realidade baseada em evidências que deve constar no arsenal terapêutico do cirurgião moderno.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
A apendicectomia — preferencialmente por via laparoscópica — permanecerá como o esteio do tratamento da apendicite aguda, salvando inúmeras vidas de peritonites letais. No entanto, o cirurgião do aparelho digestivo do século XXI deve abandonar a visão mecanicista e adotar uma perspectiva fisiológica. Considerar o tratamento antimicrobiano exclusivo em casos iniciais e não complicados não é uma heresia cirúrgica, mas sim o reconhecimento do valor morfofuncional do apêndice. Além disso, a vigilância clínica a longo prazo dos pacientes apendicectomizados, monitorando o desenvolvimento de disbiose ou patologias colorretais, deve ser encorajada. Conhecer a fundo a biologia daquilo que operamos é o que separa o mero “operador” do verdadeiro médico cirurgião.
A Anatomia Cirúrgica do Apêndice
O conhecimento anatômico do apêndice é o mapa que guia o bisturi (ou o trocarte) na sala de operações.
1. Morfologia e Topografia
O apêndice vermiforme é uma projeção tubular cega que se origina na confluência das tênias colônicas, no ceco. Seu comprimento varia de 5 a 25 cm (média de 10 cm). Sua localização típica é na fossa ilíaca direita, topografada na superfície abdominal pelo ponto de McBurney (terço externo da linha imaginária entre a espinha ilíaca anterossuperior direita e a cicatriz umbilical). Contudo, a ponta do apêndice é como o ponteiro de um relógio, assumindo diversas posições:
- Retrocecal (mais comum – aprox. 65%): Oculto atrás do ceco, frequentemente simulando afecções renais ou dificultando o diagnóstico de peritonite precoce.
- Pélvico (aprox. 30%): Pode irritar a bexiga ou anexos uterinos.
- Subcecal, Pré ou Pós-ileal.
- Nota Cirúrgica: Em casos raros de má rotação intestinal ou situs inversus, o apêndice pode estar localizado no quadrante inferior esquerdo.
2. Vascularização e Inervação
A irrigação provém da artéria apendicular, um ramo terminal da artéria ileocólica (ramo da mesentérica superior), que transita na borda livre do mesoapêndice. Por ser uma artéria terminal, a oclusão da luz apendicular (por um fecalito) gera rápido ingurgitamento, trombose, isquemia e, subsequentemente, necrose e perfuração. A inervação, oriunda do plexo mesentérico superior (T10), explica a evolução clássica da dor na apendicite aguda: inicialmente dor visceral, difusa e periumbilical; que, com a progressão da inflamação para o peritônio parietal (inervação somática), migra e se localiza na fossa ilíaca direita.
3. Resumo Prático da Anatomia Apendicular
Para uma consulta rápida e direcionada à prática cirúrgica, eis a síntese estrutural do apêndice:
- Forma e Dimensões: Estrutura tubular cega, medindo em média 10 cm de comprimento.
- Base Anatômica: Origina-se invariavelmente na confluência das três tênias colônicas no ceco.
- Posição da Ponta: Mais frequentemente retrocecal (65%) ou pélvica (30%).
- Referência de Superfície: Ponto de McBurney (fossa ilíaca direita).
- Irrigação Sanguínea: Artéria apendicular (artéria terminal; o que favorece isquemia precoce em casos de obstrução).
- Inervação Autonômica: Segmento T10, justificando a dor periumbilical referida na fase inicial da inflamação.
“A medicina é uma arte, não uma ciência exata.” — William Osler. A compreensão contínua das funções do apêndice e a evolução das abordagens terapêuticas refletem a natureza dinâmica da prática médica, onde novas evidências moldam as decisões clínicas e cirúrgicas.
Gostou ❔Nos deixe um comentário ✍️, compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Prevention of Bile Duct Injury
Prevention of Bile Duct Injury During Laparoscopic Cholecystectomy
Introduction
Bile duct injury (BDI) during laparoscopic cholecystectomy is a significant surgical complication with profound clinical and medico-legal implications. The incidence of BDI ranges from 0.3% to 0.6%, despite advances in surgical techniques and imaging modalities. The prevalence of BDI remains concerning due to its association with high morbidity and mortality rates. Patients who suffer from BDI often face prolonged hospital stays, multiple surgeries, and long-term complications such as bile leakage, strictures, and secondary biliary cirrhosis. Medico-legally, BDI is one of the most common reasons for litigation against surgeons, often resulting in significant financial settlements and professional repercussions.
Questions and Answers
Question 1: What technique should be used to identify the anatomy during laparoscopic cholecystectomy?
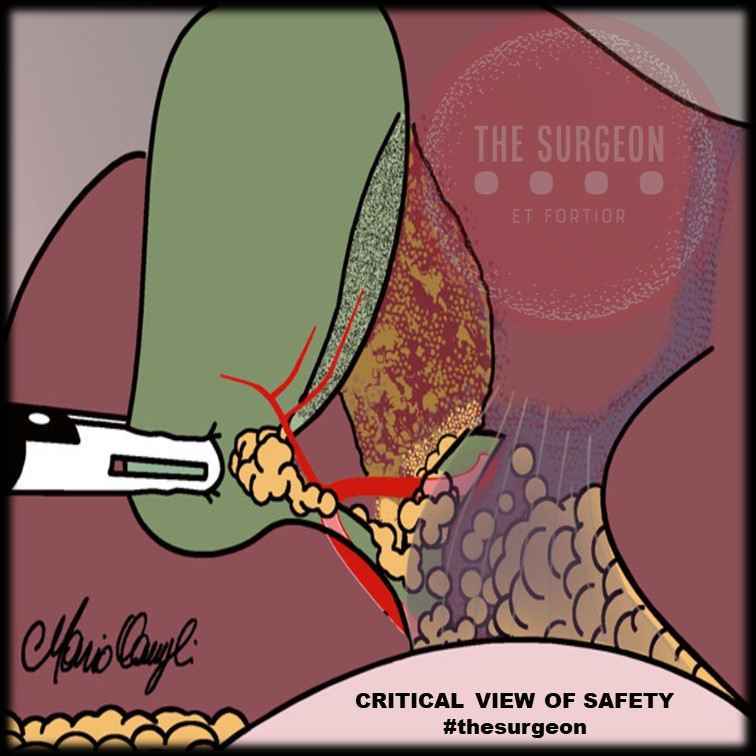
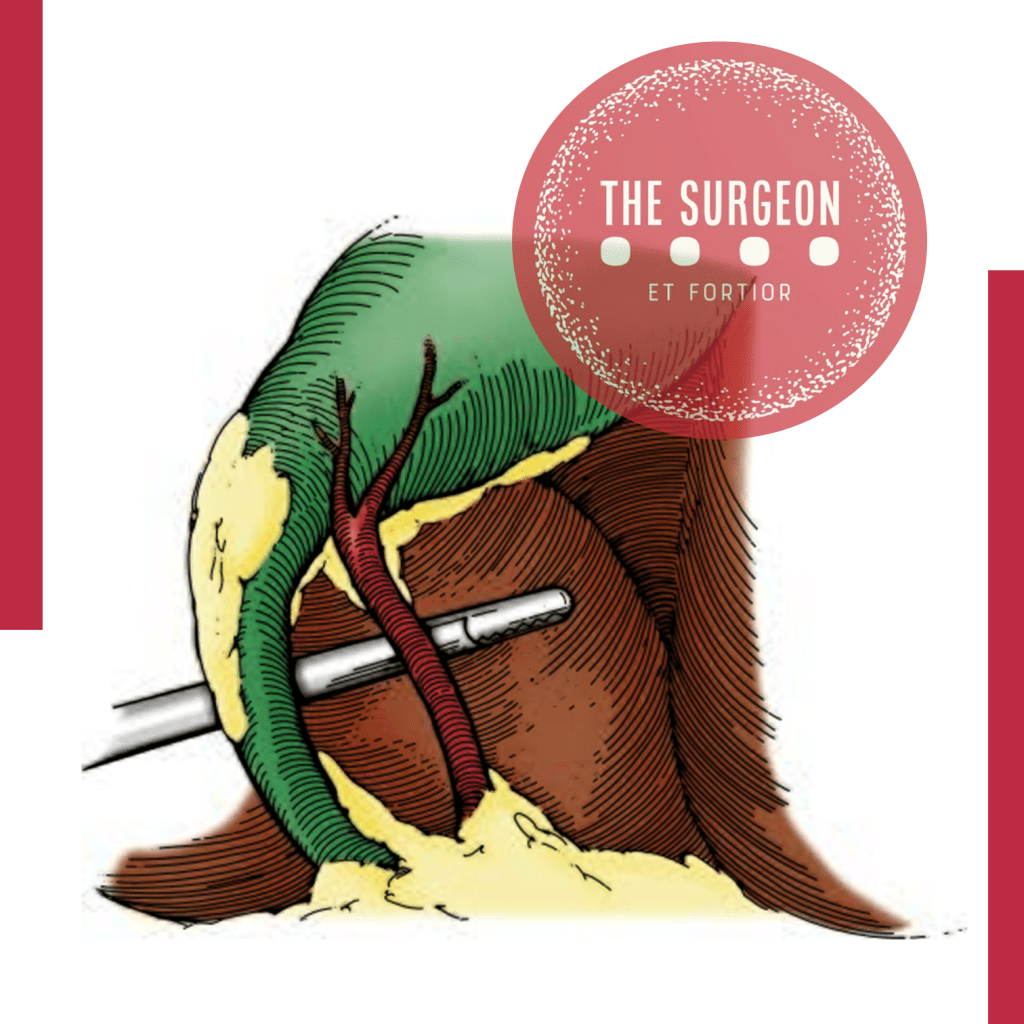
Answer: The Critical View of Safety (CVS) is recommended for identifying the cystic duct and cystic artery.
Key Findings: The incidence of BDI was found to be 2 in one million cases using CVS, compared to 1.5 per 1000 cases with the infundibular technique.
Question 2: When should intraoperative cholangiography (IOC) be used?
Answer: IOC should be used in cases of anatomical uncertainty or suspicion of bile duct injury.
Key Findings: IOC aids in the prevention and immediate management of BDI by providing a precise assessment of biliary anatomy during surgery.
Question 3: What are the recommendations for managing patients with confirmed or suspected bile duct injury?
Answer: Patients with confirmed or suspected BDI should be referred to an experienced surgeon or a multidisciplinary hepatobiliary team.
Key Findings: Early referral to hepatobiliary specialists is associated with better long-term outcomes and lower complication rates.
Question 4: Should the “fundus-first” technique be used when CVS cannot be achieved?
Answer: Yes, the “fundus-first” technique is recommended when CVS cannot be achieved.
Key Findings: This technique is effective for safely dissecting the gallbladder in complex cases where anatomy is unclear.
Question 5: Should CVS be documented during laparoscopic cholecystectomy?
Answer: Yes, documenting CVS with double-static photographs is recommended.
Key Findings: Photographic documentation of CVS ensures correct anatomical identification and serves as a record for later review in case of complications.
Question 6: Should near-infrared biliary imaging be used intraoperatively?
Answer: The evidence for near-infrared biliary imaging is limited; thus, IOC is preferred.
Key Findings: IOC is more widely studied and proven effective in preventing BDI compared to near-infrared imaging.
Question 7: Should surgical risk stratification be used to mitigate the risk of BDI?
Answer: Yes, surgical risk stratification is recommended.
Key Findings: Risk stratification helps identify patients at higher risk of complications, aiding in surgical planning and decision-making.
Question 8: Should the presence of cholecystolithiasis be considered in risk stratification?
Answer: Yes, the presence of cholecystolithiasis should be considered in risk stratification.
Key Findings: Patients with cholecystolithiasis have a higher risk of complications during cholecystectomy, making it important to include this condition in risk assessments.
Question 9: Should immediate cholecystectomy be performed in cases of acute cholecystitis?
Answer: Yes, immediate cholecystectomy within 72 hours is recommended.
Key Findings: Surgery within 72 hours of the onset of acute cholecystitis symptoms is associated with lower complication rates and better patient recovery.
Question 10: Should subtotal cholecystectomy be performed in cases of severe inflammation?
Answer: Yes, subtotal cholecystectomy is recommended in cases of severe inflammation where CVS cannot be obtained.
Key Findings: In severe inflammation scenarios, subtotal cholecystectomy can facilitate the surgery and reduce the risk of BDI.
Question 11: Which approach is preferable, four-port laparoscopic cholecystectomy or reduced-port/single-incision?
Answer: Four-port laparoscopic cholecystectomy is recommended as the standard approach.
Key Findings: The four-port technique is the most studied, showing effectiveness and safety in performing cholecystectomies with lower complication risks.
Question 12: Should interval cholecystectomy be performed following percutaneous cholecystostomy?
Answer: Yes, interval cholecystectomy is recommended after initial stabilization with percutaneous cholecystostomy.
Key Findings: Interval cholecystectomy offers better long-term outcomes and lower risk of recurrent complications compared to no additional treatment.
Question 13: Should laparoscopic cholecystectomy be converted to open in difficult cases?
Answer: Yes, conversion to open surgery is recommended in cases of significant difficulty.
Key Findings: Conversion to open surgery can prevent BDI in situations where laparoscopic dissection is extremely difficult or risky.
Question 14: Should a waiting time be implemented to verify CVS?
Answer: Yes, a waiting time to verify CVS is recommended.
Key Findings: A waiting time allows better anatomical evaluation before proceeding with dissection, reducing the risk of BDI.
Question 15: Should two surgeons be used in complex cases?
Answer: The presence of two surgeons can be beneficial in complex cases, although strong recommendations are not made due to limited evidence.
Key Findings: Some studies suggest that collaboration between two surgeons can improve anatomical identification and reduce complications in difficult cases.
Question 16: Should surgeons receive coaching on CVS to limit the risk or severity of BDI?
Answer: Yes, surgeons should receive coaching on CVS.
Key Findings: Surgeons who receive targeted coaching on CVS show improved anatomical identification and reduced rates of BDI.
Question 17: Should simulation or video-based education be used to train surgeons?
Answer: Yes, simulation or video-based education should be used.
Key Findings: These training methods enhance technical skills, increase surgical precision, and reduce the incidence of BDI during laparoscopic cholecystectomy.
Conclusion
The consensus recommendations provide evidence-based approaches to minimize bile duct injury during laparoscopic cholecystectomy. Practices such as the critical view of safety (CVS), intraoperative cholangiography (IOC), and early referral to specialists can significantly improve surgical outcomes and reduce complications. As famously stated, “The history of surgery is the history of the control of bleeding,” a phrase that underscores the importance of meticulous surgical technique and the prevention of complications like bile duct injuries.
Revisional Surgery for Weight Regain
- Introduction
Revisional bariatric surgery is a weight loss surgery for people who have not lost enough weight or have regained weight after their first bariatric surgery. It is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain.
- Causes of Weight Regain
There are many factors that can contribute to weight regain after bariatric surgery, including:
* Lack of adherence to dietary recommendations
* Hormonal/metabolic imbalance
* Mental health
* Physical inactivity
* Anatomic/surgical factors
* Medications
- Preoperative Evaluation
Before revisional surgery, patients will undergo a thorough evaluation to determine if they are eligible for the procedure. This evaluation will include a medical history and physical exam, as well as blood tests, imaging studies, and a psychological evaluation.
- Selecting the Type of Revisional Surgery
The type of revisional surgery that is best for a patient will depend on a number of factors, including the type of primary surgery they had, the cause of their weight regain, and their overall health. Some of the most common types of revisional surgery include:
* Roux-en-Y gastric bypass (RYGB)
* Sleeve gastrectomy (SG)
* Biliopancreatic diversion with duodenal switch (BPD/DS)
* Single anastomosis gastric bypass (OAGB)
* Sleeve gastrectomy with duodenal switch (SADI-S)
- Weight Loss Following Revisional Surgery
Weight loss after revisional surgery can be significant. In one study, patients who underwent revisional surgery for weight regain lost an average of 50–65.3% of their excess weight after 3 months and 50.1–79.1% of their excess weight after 12 months.
- Complications of Revisional Surgery
Revisional surgery is more complex and technically demanding than primary bariatric surgery, and therefore carries a higher risk of complications. Some of the most common complications of revisional surgery include:
* Hernia
* Anastomotic leak
* Stricture
* Marginal ulcer
* Wound infection
* Hemorrhage
* Perforation
* Obstruction
- Conclusion
Revisional bariatric surgery can be a successful treatment option for patients with weight regain. However, it is important to carefully consider the risks and benefits of the procedure before making a decision. Patients should also work closely with their healthcare team to choose the best type of revisional surgery for them and to prepare for the procedure and its aftermath. Revisional bariatric surgery is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain. It is important to work closely with your healthcare team to determine if you are eligible for the procedure and to choose the best type of surgery for you.
Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
GASTROSTOMY: INDICATIONS, TECHNICAL DETAILS AND POSTOPERATIVE CARE.
Ozimo Pereira Gama Filho
Adjunct Professor at the Federal University of Maranhão
No conflict of interest
- INTRODUCTION
- HISTORICAL ASPECTS
- ANATOMICAL FUNDAMENTALS
- INDICATIONS
- TYPES & TECHNIQUES
- ADVERSE EVENTS
- CUSTO RATIO x EFFECTIVENESS
- CONCLUSIONS
SUMMARY
In the last decade, the use of gastrostomies has been widely indicated as the preferred form of access to the gastrointestinal tract for feeding in chronic conditions and during recovery from acute conditions such as trauma. Together with this increase in indications, new techniques have been developed that have made gastrostomies simpler and less risky. From the classical technique of Stamm performed by laparotomy, two new alternatives that do not require laparotomy emerged: percutaneous endoscopic gastrostomy (PEG) and fluoroscopy gastrostomy. Its main benefit is to avoid a laparotomy, with less associated postoperative pain and earlier return of gastrointestinal function. Although peg is currently widely accepted as the insertion technique of choice due to its simplicity and efficacy, there are patients who are not candidates for an endoscopic approach. In this article we seek to clarify the indications, technical aspects and perioperative care of patients undergoing gastrostomy.
Keywords: Surgical Procedures; Ostomies; Gastrostomy.
Area of Knowledge: General Surgery
- INTRODUCTION
The main indication for enteral or parenteral feeding in the perioperative period is the provision of nutritional support to supply the metabolism of patients with inadequate oral intake. Enteral feeding is the preferred method in relation to parenteral feeding in patients with gastrointestinal dysfunction in the perioperative period due to the inherent risks associated with parenteral nutritional support, such as: infectious complications of the access routes, higher operational cost, and the inability to parenteral nutrition to provide adequate enteral stimulation and subsequent involvement of the intestinal defense barrier [1,2]. In addition, enteral feeding may decrease the risk of bacterial translocation and corresponding bacteremia [3]. Gastric nutritional support is the most common type used. Access to insert the gastrostomy probe can be achieved using endoscopy, interventional radiologia, or surgical techniques (open or laparoscopic). However, since its description in the 1980s [4], percutaneous endoscopic gastrostomy (PEG) is currently considered the method of choice for medium and long-term enteral support.
1.1 Objective: This article reviews the current knowledge about GOSTROSTOMIA in the medical literature, emphasizing the technical and perioperative aspects.
- HISTORICAL ASPECTS
In 876, Verneoil [5] successfully made the first gastrostomy in humans. Since then, several technical modifications have been suggested, such as witzel’s technique in 1891, in which a subseroso tunnel is made on the probe [6]. Stamm, in 1894 [7], described one of the most performed techniques today and in the history of surgical gastrostomy, which consists in the making of suture in a pouch to invaginate the probe inserted into the stomach [8]. In 1980, percutaneous endoscopic gastrostomy was described by Gauderer et al. [4] , which transformed the technique of making gastrostomy.
- ANATOMICAL FUNDAMENTALS
The stomach is a J-shaped dilated cylindrical organ that rests in the left epigastric and hypochodrial region of the abdomen at the level of the first lumbar vertebra. It is previously limited by the left hemidiaphragm, the left lobe of the liver and a triangular portion of the anterior abdominal wall. Subsequently, the pancreas, left kidney and adrenal delimit the stomach. The spleen is posterolaterally and the transverse colon is inferior. It is fixed at two points of continuity: gastroesophageal, superiorly and the duodenal, retroperitoneally. Its ligament attachments also help you in fixation to adjacent organs: gastrophemic (diaphragm), hepatogastric or minor omentum (liver), gastrosplenic or gastrolienal (spleen), and gastrocholic or omentum major (transverse colon). The anatomical regions of the stomach can be distinguished as this: começa superiorly in the continuity of the abdominal part of the esophagus and dthe gastroesophageal junction, the cardiac part of the stomach. Soon below this portion, lies the bottom of the stomach that expands to the left extending above thegastroesophageal junction, forming an acute angle with the distal esophagus known as cardiac notches. The body s andextends as a distensible reservoir and forms a medial edge called the smallest curvature to the right and a side edge called the largest curvature on the left. The gastric den of the stomach is not anatomically distinguishable, but it is estimated to be a region of the angular isis along the distal minor curvature to a point along a lower line to the distal major curvature. It thus ends bymouthing r into the pyloric canal limited by the pyloric sphincter, a palpable thickened ring of muscle that is continuous with the first part of the duodenum.
- INDICATIONS
Gastrostomy is used in the following situations:
- Gastric decompression: can be obtained by means of temporary gastrostomy, occasionally recommended, as a complement to large abdominal operations for which gastric stems, prolonged “adynamic ileus” and digestive fistulas are foreshadowed.
- Nutritional Support: b.1 Temporary; indicated when access to the digestive tract is temporarily impaired for recovery and maintenance of nutritional status (E.g. CEsophageal EC); b.2 Definitive; as palliative therapy in patients with unresectable malignant neoplasia of the head and neck, as wellas n degenerative neurological diseases that lead to irreversible disorders of deglutition.
However, the decision to perform a gastrostomy, as well as its route (surgical, radiological or endoscopic) should be individualizedaccording to the needs, diagnosis, life expectancy of the patient and the available hospital logistics. The objective is not only to optimize perioperative recovery, to improve survival and nutritional status of the patient, but also to promote quality of life, which is not necessarily correlated with nutritional improvement only [9]. Therefore, the appropriate indication, like any other surgical intervention, must be clearly establishedand informed before it is performed. Some of the absolute contraindications of gastrostomy are summarized in Table 1. In addition to absolute contraindication conditions, other situations such as the presence of non-obstructive oromyctological oresophageal malignancy, hepatomegaly, splenomegaly, peritoneal dialysis, portal hypertension with gastric varicose veins, and a history of partial gastrectomy are also considered relative contraindications.
| ABSOLUTE CONTRA – INDICATIONS |
| Coagulopatia Severa (INR > 5, Plaquetas < 50.000 e TPT > 50s) |
| Hemodynamic Instability |
| Septic Shock |
| Refractory Ascites |
| Peritonitis |
| Dermatological infection in the upper abdomen |
| Carcinomatose Peritoneal |
| Interposition of organs that prevent gastric access |
| History of Total Gastrectomy |
| Stenosis or Pyloric Obstruction |
| Severe gastroparesis, in cases of indication for nutritional support |
| Absence of Informed Consent |
- TYPES & TECHNIQUES
Currently there are three techniques for performing gastrostomy: radiological, through percutaneous gastrostomy by fluoroscopy, percutaneous endoscopic gastrostomy (PEG) and surgical gastrostomy. Due to the didactic characteristics of this material, we will focus on endoscopic and surgical gastrostomy , which becomes the main option in the following situations: 1) when the patient will already undergo a laparotomy due to some abdominal condition ; 2) impossibility of performing gastroscopy to perform gastrostomy endoscopic percutaneous (PEG) ; 3) in case of peg technical failure; 4) unavailability of resources for the preparation of PEG or percutaneous gastrostomy by fluoroscopy.
- ENDOSCOPIC PERCUTANEOUS GASTROSTOMY
The informed consent form must be obtained from patients or their legal representatives. Patients should fast for a minimum of 8 hours and receive prophylactic antibiotics one hour before proceeding and intravenous administration of 1-2 g of cefazolin is recommended. The technique introduced by Gauderer et al [4] is the most used technique to insert the PEG gastrostomy probe. In this method, a guide wire is used, inserted in the distal gastric chamber through a needle puncture n to the anterior abdominal wall. This guide wire is then seized endoscopically with a handle and then removed through the esophagus and mouth. Subsequently, the guide wire is fixed to the end of the gastrostomy probe and then pulled from the mouth to the esophagus, stomach and then out to the abdominal wall, where it will be fixed.
- SURGICAL GASTROSTOMY
Surgical gastrostomy can be performed in two ways: 1) via laparotomy – the predominant form; and 2) laparoscopic approach. The preparations are the same as the endoscopic pathway.
5.2.1. GASTROSTOMIA At STAMM
After adequate asepsis and antisepsis, with the patient under anesthesia and in horizontal dorsal decubitus, the technical steps are as follows: 1. Median laparotomy (supraumbilical median incision); 2. Identification of the gastric body; 3. Stomach hold with Babcock tweezers (to evaluate the approach of the stomach to the peritoneum); 4. Suture in pouch (circular area of 2cm) – atraumatic absorbable thread; 5. Section of the stomach wall (0.5cm) – (incision with scalpel or Electrocautery in the center of the suture, of sufficient size, for the placement of a probe with 20 to 26 French); 6. Placement of the Gastrostomy probe in the extension of 5-6 cm, followed by suture closure in a pouch); 7. Tie the suture threads in a pouch around the probe; 8. Apply a second suture in a pouch 1cm above the first (seromuscular stitches); 9. Externalization of the probe by counter opening on the left flank; 10. Fix the stomach wall to the abdominal wall in 4 cardinal points (external ration with the two Kocher tweezers used for grip of the alba line and against traction by means of the index fingers of the wall of the left hipochondrio to approach the parietal peritoneum of the gastric wall); 11. Fixation of the probe to the skin (point with nonabsorbable wire); 12. Closure of the abdominal wall (synthesis of the wall with approximation of the alba thread by continuous suture with monofilament thread 1-0 or 2-0 and of the skin with separate points of nylon 3-0); 13. Dressing.
5.2.1 GASTROSTOMIA EM WITZEL
The initial technical steps from gastrostomy to Witzel are like those of gastrostomy to Stamm, including fixation of the probe to the stomach by a pouch suture. Then, the probe is placed on the gastric wall and a tunnel of 8-10 cm is made by seromuscular suture (continuous or with separate points of absorbable or nonabsorbable thread) covering it and externalization is performed by counteropening.
- ADVERSE EVENTS
According to the literature, the rate of complications for different procedures varies due to the heterogeneity of the samples evaluated. For surgical gastrostomy, the reported complication rates are between 1% and 35%, while for percutaneous radiological gastrostomy it is 3% to 11%, and for percutaneous endoscopic, 17%–32%, the main related adverse event is surgical site infection [10, 11]. Although considered a basic procedure, gastrostomy is associated with an extensive list of related technical complications, care and use of the probe. Serious problems related to the technique include separation of the stomach from the abdominal wall (leading to peritonitis), separation of wounds, hemorrhage, infection, lesion of the posterior gastric wall or other organs, and placement of the tube in an inappropriate place of gastric position. Separation of the stomach from the abdominal wall usually occurs due to inadvertent and premature displacement of the tube, particularly with balloon-like devices, or a rupture during a catheter change. It requires immediate attention, being treated with laparotomy, although in selected cases laparoscopic correction is possible. Most complications can be avoided with the careful choice of the type of procedure, from the appropriate ostomy device, considering it an important intervention and using meticulous technique with the proper approximation of the stomach to the abdominal wall and outflow of the probe through a counter-incision (in conventional procedures), thus avoiding probes in the midline or awfully close to the costal edge.
- COST VS. EFFECTIVENESS
A recent study [12] compared the cost associated with the different gastrostomy techniques, and the results of the evaluation showed variable the benefits of each of the individual percutaneous procedures, indicating that surgical gastrostomy was the onerous mais of the three modalities due to higher costs, complications, and recovery time, as well as the endoscopic technique presenting the cost effectiveness ratio.
- CONCLUSIONS
Despite the technique employed, the decision to performa gastrostomy is not based only on the patient’s survival expectancy, because the adequate indication provides a better quality of life even when the survival of the patient after the procedure is severely limited. Therefore, understanding of techniques, indications, complication rates is essential to guide the surgical team in the scope of multidisciplinary care, as well as the education of patients and their caregivers is vital to ensure the correct maintenance of the devices, thus ensuring adequate nutritional intake of the patient and minimizes complication rates.
References
1 Alverdy J, Chi HS, Sheldon GF. The effect of parenteral nutrition in gastrointestinal immunity. The importance of de estimulação enteral. Ann Surg, 1985; 202: 681-684 [PMID:3935061]
2 Deitch EA, Ma WJ, Ma L, Berg RD, Specian RD. Protein malnutrition predisposes to inflammation-induced intestinal origin septic states. Ann Surg, 1990; 211: 560-567; discussion 560-567 [PMID: 2111125]
3 Deitch EA, Winterton J, Li M, Berg R. The intestine as a portal of entry to bacteremia. Role of protein malnutrition. Ann Surg 1987; 205: 681-692 [PMID: 3592811]
4 Gauderer MW, Ponsky JL, Izant RJ. Gastrostomia sem laparotomy: percutaneous endoscopic technique. J Pediatrician Surg, 1980; 15: 872-875 [PMID: 6780678]
5 Anselmo CB, Tercioti Júnior V, Lopes LR, Coelho Neto JS, Andreollo NA. Surgical gastrostomy: current indications and complications in patients of a university hospital. Rev Col Bras Cir. [Internet journal] 2013;40(6). Available in URL: http://www.scielo.br/rcbc
6 Witzel O. For gastric fistula technique. Chir Zbl. 1891;18:601-4.
7 Stamm M. Gastrostomy: a new method. Med News. 1894;65:324.
8 JP grant. Comparison of percutaneous endoscopic gastrostomy com gastrostomia strain. Ann Surg. 1988;207(5):598-603
9 Bannerman E, Pendlebury J, Phillips F, Ghosh S. Cross-sectional and longitudinal study of health-related quality of life after percutaneous gastrostomy. Eur J Gastroenterol Hepatol 2000; 12: 1101-1109 [PMID: 11057455]
10 Möller P, Lindberg CG, Zilling T. Gastrostomy by various techniques: evaluation of indications, outcome and complications. Scand J Gastroenterol. 1999;34(10):1050-4.
11 Clarke E, Pitts N, Latchford A, Lewis S. A major prospective audit of morbidity and mortality associated with food gastrostomies in the community. Clin Nutr. 2017 Apr;36(2):485-490. DOI: 10.1016/j.clnu.2016.01.008. EPub 2016 January 21. PMID: 26874913.
12 Wollman B, D’Agostino HB, Walus-Wigle JR, Easter DW, Beale A (1995) Radiological, endoscopic and surgical gastrostomy: an institutional evaluation and meta-analysis of the literature. Radiology 197: 699–704.
Specific Competence of Surgical Leadership
Surgeons are uniquely prepared to assume leadership roles because of their position in the operating room (OR). Whether they aspire to the title or not, each and every surgeon is a leader, at least within their surgical team. Their clinical responsibilities offer a rich variety of interpretations that prepare them for a broader role in health care leadership. They deal directly with patients and their families, both in and out of the hospital setting, seeing a perspective that traditional health care administrative leaders rarely experience. They work alongside other direct providers of health care, in varied settings, at night, on weekends, as well as during the typical workday. They understand supply-chain management as something more than lines on a spreadsheet.
The Challenges for a Surgical Leader
Surgeons prefer to lead, not to be led. Surgical training has traditionally emphasized independence, self-reliance, and a well-defined hierarchy as is required in the OR. However, this approach does not work well outside the OR doors. With colleagues, nurses, staff, and patients, they must develop a collaborative approach. Surgeons are entrusted with the responsibility of being the ultimate decision maker in the OR. While great qualities in a surgeon in the OR, it hinders their interactions with others. They have near-absolute authority in the OR, but struggle when switching to a persuasive style while in committees and participating in administrative activities. Most surgeons do not realize they are intimidating to their patients and staff. With patients, a surgeon needs to be empathetic and a good listener. A surgeon needs to slow the pace of the discussion so that the patient can understand and accept the information they are receiving. As perfectionists, surgeons demand a high level of performance of themselves. This sets them up for exhaustion and burnout, becoming actively disengaged, going through the motions, but empty on the inside. Given the many challenges surgeons face, it is difficult for them to understand the leadership role, given its complex demands.
Specific Competencies
Authority
Although teams and all team members provide health care should be allowed input, the team leader makes decisions. The leader must accept the responsibility of making decisions in the presence of all situations. They will have to deal with conflicting opinions and advice from their team, yet they must accept that they will be held accountable for the performance of their team. The surgeon–leader cannot take credit for successes while blaming failures on the team. Good teamwork and excellent communication do not relieve the leader of this responsibility.
Leadership Style
A surgeon often has a position of authority based on their titles or status in an organization that allows them to direct the actions of others. Leadership by this sort of mandate is termed “transactional leadership” and can be successful in accomplishing specific tasks. For example, a surgeon with transactional leadership skills can successfully lead a surgical team through an operation by requesting information and issuing directives. However, a leader will never win the hearts of the team in that manner. The team will not be committed and follow through unless they are empowered and feel they are truly heard. A transformational leader is one who inspires each team member to excel and to take action that supports the entire group. If the leader is successful in creating a genuine atmosphere of cooperation, less time will be spent giving orders and dealing with undercurrents of negativity. This atmosphere can be encouraged by taking the time to listen and understand the history behind its discussion. Blame should be avoided. This will allow the leader to understand the way an individual thinks and the group processes information to facilitate the introduction of change. While leadership style does not guarantee results, the leader’s style sets the stage for a great performance. At the same time, they should be genuine and transparent. This invites the team members to participate, creating an emotional connection. Leaders try to foster an environment where options are sought that meet everyone’s desires.
Conflict Management
Conflict is pervasive, even in healthy, well-run organizations and is not inherently bad. Whether conflict binds an organization together or divides it into factions depends on whether it is constructive or destructive. A good leader needs to know that there are four essential truths about conflict. It is inevitable, it involves costs and risks, the strategies we develop to deal with the conflict can be more damaging than the conflict itself, and conflict can be permanent if not addressed. The leader must recognize the type of conflict that exists and deal with the conflict appropriately. Constructive discussion and debate can result in better decision making by forcing the leader to consider other ideas and perspectives. This dialog is especially helpful when the leader respects the knowledge and opinions of team members with education, experience, and perspective different from the leader’s. Honesty, respect, transparency, communication, and flexibility are all elements that a leader can use to foster cohesion while promoting individual opinion. The leader can create an environment that allows creative thinking, mutual problem solving, and negotiation. These are the hallmarks of a productive conflict. Conflict is viewed as an opportunity, instead of something to be avoided.
Communication Skills
Communication is the primary tool of a successful leader. On important topics, it is incumbent on the leader to be articulate, clear, and compelling. Their influence, power, and credibility come from their ability to communicate. Research has identified the primary skills of an effective communicator. They are set out in the LARSQ model: Listening, Awareness of Emotions, Reframing, Summarizing, and Questions. These are not set in a particular order, but rather should move among each other freely. In a significant or critical conversation, it is important for a leader to listen on multiple levels. The message, body language, and tone of voice all convey meaning. You cannot interrupt or over-talk the other side. They need an opportunity to get their entire message out. Two techniques that enhance listening include pausing and the echo statement. Pausing before speaking allows the other conversant time to process what they have said to make sure the statement is complete and accurate. Echo statements reflect that you have heard what has been said and focuses on a particular aspect needing clarification. Good listening skills assure that the leader can get feedback that is necessary for success.
Vision, Strategy, Tactics, and Goals
One of the major tasks of a leader is to provide a compelling vision, an overarching idea. Vision gives people a sense of belonging. It provides them with a professional identity, attracts commitment, and produces an emotional investment. A leader implements vision by developing strategy that focuses on specific outcomes that move the organization in the direction of the vision. Strategy begins with sorting through the available choices and prioritizing resources. Through clarification, it is possible to set direction. Deficits will become apparent and a leader will want to find new solutions to compensate for those shortfalls. For example, the vision of a hospital is to become a world class health care delivery system. Strategies might include expanding facilities, improving patient satisfaction, giving the highest quality of care, shortening length of hospital stay with minimal readmissions, decreased mortality, and a reduction in the overall costs of health care. Tactics are specific behaviors that support the strategy with the aim to achieve success. Tactics for improving patient satisfaction may include reduced waiting time, spending more time with patients, taking time to communicate in a manner that the patient understands, responding faster to patient calls, etc. These tactics will then allow a leader to develop quantitative goals. Patient satisfaction can be measured. The surgical leader can then construct goals around each tactic, such as increasing satisfaction in specific areas. This information allows a surgical leader to identify barriers and they can take steps to remedy problem areas. This analysis helps a leader find the weakest links in their strategies as they continue toward achieving the vision.
Change Management
The world of health care is in continuous change. The intense rate of political, technical, and administrative change may outpace an individual’s and institution’s ability to adapt. Twenty-first century health care leaders face contradictory demands. They must navigate between competing forces. Leaders must traverse a track record of success with the ability to admit error. They also must maintain visionary ideas with pragmatic results. Individual accountability should be encouraged, while at the same time facilitating teamwork. Most leaders do not understand the change process. There are practical and psychological aspects to change. From an institutional perspective, we know that when 5% of the group begins to change, it affects the entire group. When 20% of a group embraces change, the change is unstoppable.
Succession Planning and Continuous Learning
An often-overlooked area of leadership is planning for human capital movement. As health care professionals retire, take leaves of absences, and move locations, turmoil can erupt in the vacuum. Leaders should regularly be engaging in activities to foster a seamless passing of institutional knowledge to the next generation. They also should seek to maintain continuity to the organization. Ways to accomplish this include senior leaders actively exposing younger colleagues to critical decisions, problem solving, increased authority, and change management. Leaders should identify promising future leaders, give early feedback for areas of improvement, and direct them toward available upward career tracks. Mentoring and coaching help prepare the younger colleagues for the challenges the institution is facing. Teaching success at all levels of leadership helps create sustainable high performance.
Visão Crítica de Segurança (Colecistectomia)
A colecistectomia laparoscópica (CL) é o padrão-ouro para tratamento de cálculos biliares. No entanto, o risco de lesão do ducto biliar (BDI) continua a ser preocupação significativa, uma vez que CL ainda tem taxa de BDI maior do que a via laparotômica, apesar de muitos esforços propostos para aumentar sua segurança. A Visão Crítica da Segurança (CVS) proposta por Strasberg é a técnica para a identificação dos elementos críticos do triângulo de Calot durante a CL. Esta técnica foi adotada em vários programas de ensino e com a proposta de reduzir o risco de lesão acidental da via biliar (LAVB) e o uso da adequado da CVS está associado a menores taxas de LAVB. O objetivo deste #Webinar é abordar a Anatomia Cirúrgica Fundamental para a realização de uma Colecistectomia Laparoscópica.
Intraoperative cholangiography: Selective or Routine?
Intraoperative cholangiography (IOC), described by Mirizzi in 1932, represented a significant advance in the diagnosis of choledocolithiasis during cholecystectomy. The natural history of asymptomatic choledocolithiasis has been investigated in different populations and its therapeutic management continues to be controversial. IOC is traditionally advocated as a procedure to be adopted in all laparoscopic cholecystectomies since it permits to define the anatomy of the biliary tract and to detect common bile duct stones. However, in the laparoscopic era, technological advances in radiologic-endoscopic workup have markedly increased the costs of investigation of patients with suspected choledocolithiasis. The routine use of IOC has raised the question of which cases require the exploration of bile tract anatomy during surgery and whether there are methods to predict preoperatively unsuspected choledocolithiasis. Therefore, the most appropriate management of preoperatively unsuspected choledocolithiasis, i.e., the routine or selective use of IOC, still remains undefined. In this respect, well-defined criteria for the inclusion of patients with possible choledocolithiasis who should be submitted to cholangiography exist in the literature, but there is no safe approach to exclude asymptomatic patients without an indication for contrast examination.
A systematic literature search was performed by KOVACS N, et al (2022) using the following search keys: cholangiogra* and cholecystectomy. The primary outcomes were BDI and retained stone rate. To investigate the differences between the groups (routine IOC vs selective IOC and IOC vs no IOC), they calculated weighted mean differences (WMD) for continuous outcomes and relative risks (RR) for dichotomous outcomes, with 95% confidence intervals (CI). Of the 19,863 articles, 38 were selected and 32 were included in the quantitative synthesis. Routine IOC showed no superiority compared to selective IOC in decreasing BDI (RR = 0.91, 95% CI 0.66; 1.24). Comparing IOC and no IOC, no statistically significant differences were found in the case of BDI, retained stone rate, readmission rate, and length of hospital stay. They found an increased risk of conversion rate to open surgery in the no IOC group (RR = 0.64, CI 0.51; 0.78). The operation time was significantly longer in the IOC group compared to the no IOC group (WMD = 11.25 min, 95% CI 6.57; 15.93). So this data findings suggest that IOC may not be indicated in every case, however, the evidence is very uncertain.
Aderências Pós-Operatórias
Do Manejo Conservador à Decisão Cirúrgica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
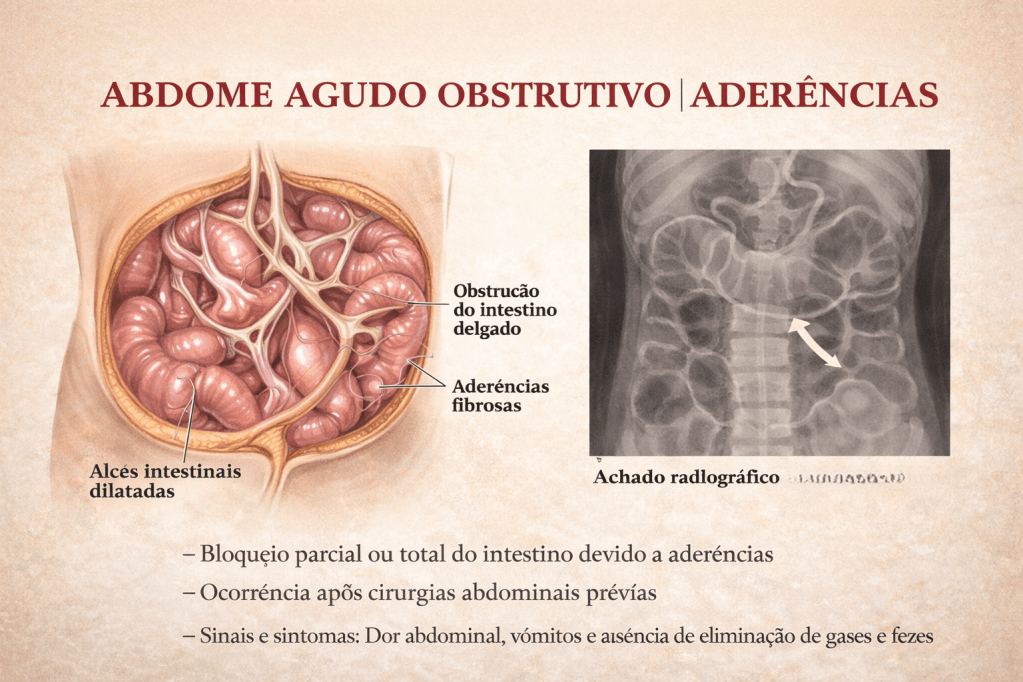
Introdução
A obstrução intestinal é um dos cenários mais desafiadores e frequentes no pronto-socorro cirúrgico. Historicamente, o abdome agudo obstrutivo impunha o dilema imediato da exploração cirúrgica. Hoje, compreendemos que a etiologia dita o compasso da intervenção. Entre todas as causas de obstrução do intestino delgado, as aderências pós-operatórias (bridas) assumem o protagonismo absoluto. Para o estudante de medicina, o residente e o cirurgião do aparelho digestivo, dominar o manejo da obstrução por bridas é essencial. O desafio não reside apenas na técnica operatória da enterólise, mas sim no raciocínio clínico aguçado para responder à pergunta fundamental: quando persistir no tratamento conservador e quando a indicação cirúrgica se torna imperativa?
Cenário Brasileiro
A formação de aderências peritoniais é uma resposta fisiológica quase universal ao trauma cirúrgico (isquemia, manipulação, coágulos), ocorrendo em até 90% dos pacientes submetidos a laparotomias. Contudo, em uma parcela significativa, essa cicatrização aberrante leva à obstrução mecânica. Na prática da cirurgia geral no Brasil, a obstrução por bridas responde por cerca de 60% a 75% das admissões por obstrução do intestino delgado. Dados do Sistema Único de Saúde (DataSUS) indicam que milhares de laparotomias exploradoras são realizadas anualmente no país devido a quadros obstrutivos agudos, gerando elevado custo financeiro e considerável morbimortalidade, além de reinternações frequentes.
Fisiopatologia e Diagnóstico
O peritônio, quando lesado, inicia uma cascata inflamatória rica em fibrina. Em condições normais, a fibrinólise degrada essa matriz em poucos dias. O desequilíbrio nesse sistema — com falha na fibrinólise peritonia — resulta na organização da fibrina em tecido conjuntivo vascularizado: a aderência.
A Tríade Diagnóstica
O diagnóstico é eminentemente clínico e radiológico:
- Clínica: Dor abdominal em cólica, distensão, vômitos (fecaloides em fases tardias) e parada de eliminação de flatus e fezes.
- Laboratório: Hemograma, eletrólitos, lactato e gasometria são cruciais para rastrear sinais de isquemia ou necrose (leucocitose importante, acidose metabólica).
- Imagem: A Tomografia Computadorizada (TC) de abdome com contraste venoso substituiu o raio-X simples como padrão-ouro. A TC identifica o ponto de transição (zona de calibres distintos), o grau de obstrução, a presença de sofrimento de alça (espessamento parietal, pneumatose, gás no sistema porta) e descarta outras etiologias (hérnias internas, neoplasias).
Estratégias de Tratamento
O manejo da obstrução por bridas sofreu uma evolução paradigmática, distanciando-se de intervenções intempestivas em favor de protocolos baseados em evidências.
1. Tratamento Conservador (Não Operatório)
Na ausência de sinais de isquemia intestinal, perfuração ou peritonite localizada, a conduta inicial deve ser conservadora:
- Jejum (NPO) e descompressão com Sonda Nasogástrica (SNG).
- Ressuscitação volêmica vigorosa e correção de distúrbios hidroeletrolíticos.
- Protocolo com Contraste Hidrossolúvel: A administração de contraste hiperosmolar (ex: Gastrografin) via SNG não possui apenas valor diagnóstico (avaliar se o contraste atinge o cólon em 24h), mas também efeito terapêutico. A hiperosmolaridade atrai líquido para a luz intestinal, reduzindo o edema da parede e estimulando o peristaltismo, resolvendo o quadro obstrutivo em uma parcela significativa dos casos.
2. Tratamento Cirúrgico: O Timing Ideal
A falha do tratamento conservador (geralmente avaliada após 48 a 72 horas) ou o surgimento de sinais de alarme (taquicardia, febre, irritação peritoneal, acidose) indicam cirurgia de urgência.
- Laparotomia vs. Laparoscopia: A via aberta continua sendo o padrão em pacientes com múltiplas cirurgias prévias ou grande distensão abdominal. A abordagem laparoscópica é excelente em mãos experientes, mas reserva-se a casos selecionados (ex: suspeita de brida única, distensão moderada), exigindo extrema cautela na introdução do primeiro trocarte (técnica aberta de Hasson) para evitar enterotomias iatrogênicas.
- O Ato Cirúrgico: A lise de aderências deve ser restrita ao necessário para resolver a obstrução. Múltiplas enterólises desnecessárias aumentam o risco de lesões e a formação de novas bridas. Em caso de necrose, a ressecção do segmento isquêmico com anastomose primária ou estomia (a depender da estabilidade hemodinâmica) é imperativa.
Pontos-Chave para a Prática Diária
- Suspeição de Isquemia: Dor refratária a analgésicos, taquicardia inexplicada e acidose metabólica são gritos de socorro de uma alça intestinal isquêmica. O tratamento conservador é contraindicado.
- O Valor da TC: A tomografia não apenas confirma o diagnóstico, mas é o farol que guia a decisão entre a enfermaria e o centro cirúrgico.
- Prevenção Primária: O uso de barreiras antiaderentes ainda carece de evidências universais robustas, mas a técnica cirúrgica apurada, manipulação tecidual delicada e controle rigoroso da hemostasia continuam sendo a melhor profilaxia.
Conclusões Aplicadas
O tratamento da obstrução intestinal por bridas é a quintessência do bom senso cirúrgico. O cirurgião moderno deve possuir a paciência de um clínico para conduzir o tratamento conservador respaldado pelo contraste hidrossolúvel e, simultaneamente, a assertividade de um intervencionista para indicar a sala de cirurgia ao primeiro sinal de estrangulamento.Compreender que “operar a radiografia” é um erro crasso e que a reavaliação clínica seriada (de preferência pelo mesmo examinador) é a bússola do tratamento, molda o profissional de excelência que minimiza ressecções intestinais desnecessárias e reduz a morbidade pós-operatória.
“Na obstrução intestinal aguda, não deixe o sol se pôr ou nascer sobre o paciente sem uma decisão. Mas lembre-se: a cirurgia sem indicação precipita o desastre que buscava evitar.” — Aforismo clássico derivado dos ensinamentos de Sir Zachary Cope, pioneiro no estudo e diagnóstico do abdome agudo.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Managing the “difficult” gallbladder
The gold standard for the surgical treatment of symptomatic cholelithiasis is conventional laparoscopic cholecystectomy (LC). The “difficult gallbladder” is a scenario in which a cholecystectomy turns into an increased surgical risk compared with standard cholecystectomy. The procedure may be difficult due to processes that either obscure normal biliary anatomy (such as acute or chronic inflammation) or operative exposure (obesity or adhesions caused by prior upper abdominal surgery). So, when confronted with a difficult cholecystectomy, the surgeon has a must: to turn the operation into a safe cholecystectomy, which can mean conversion (to an open procedure), cholecystostomy, or partial/ subtotal cholecystectomy. The surgeon should understand that needs to rely on damage control, to prevent more serious complications if choosing to advance and progress to a complete cholecystectomy.
When to Predict a Difficult Laparoscopic Cholecystectomy
A difficult cholecystectomy may be predicted preoperatively based on patient characteristics and ultrasound and laboratory findings. This is probably a very important step in mitigating the high risk associated with a difficult procedure and may serve either to reschedule the procedure or design intraoperative strategies of management to guarantee a safe performance of the surgical procedure.
The following situations are associated with a higher chance of a difficult cholecystectomy:
• Acute cholecystitis (more than 5 days of onset)
• Previous cholecystitis episode
• Male sex
• Obesity
• Cirrhosis
• Sclero-atrophic gallbladder
• Thick walls (>5 mm)
• Previous signs of canalicular dwelling (clinical and laboratory)
Through multivariate analysis, Bourgoin identified these elements of predictive help to identify difficult LC: male sex, previous cholecystitis attack, fibrinogen, neutrophil, and alkaline phosphatase levels. Another important point is the fact of conversion from a laparoscopic procedure to an open and traditional cholecystectomy, usually through a right subcostal incision. Conversion should not be considered as a personal failure, and the surgeon needs to understand the concept of “safety first,” considering that conversion is performed in order to complete the procedure without additional risks and preventing complications and not solving intraoperative complications. It is also useful to define a time threshold to aid in the decision to convert. It is not worth taking an hour and a half and still dissecting adhesions, preventing the correct visualization of the cystic pedicle. This time limit represents a method to prevent inefficiencies in the operating room (OR) schedule as well as additional expenditures.
A smart surgeon should rely to conversion in the following situations:
• Lack of progress in the procedure
• Unclear anatomy/any grade of uncertainty
• CVS not achieved
• Bleeding/vascular injury
• BD injury
• Lack of infrastructure, expertise, and support
Final Remarks
The primary goal of a laparoscopic cholecystectomy in the treatment of symptomatic cholelithiasis is the safe remotion of the gallbladder and the absence of common bile duct injury. Some tips to take into account:
– Never perform a laparoscopic cholecystectomy without a skilled surgeon close by.
– Beware of the easy gallbladder.
– Slow down, take your time.
– Knowledge is power, conversión can be the salvation!
– Do not repair a bile duct injury (unless you have performed at least 25 hepaticojejunostomies).
– Do not ignore postoperative complaints (pain, jaundice, major abdominal discomfort, fever)
Other options when confronted with a difficult laparoscopic cholecystectomy are:
– A percutaneous cholecystostomy, if the risk was identified preoperatively or the patient is a poor surgical candidate;
– An intraoperative cholangiography, which may aid in identifying an injury to the bile duct and solve it, if you are an experienced surgeon;
– A subtotal or partial cholecystectomy;
– Ask for help;
– Conversion to an open procedure;
Vascularização da Árvore Biliar
O Fio da Navalha na Prevenção da Isquemia e Estenose
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 11 minutos)
Introdução
No campo minado que é a cirurgia hepatobiliar, existe um princípio fisiológico que não admite margem para erro: enquanto o parênquima hepático é um órgão privilegiado, nutrido por um duplo suprimento sanguíneo (arterial e venoso portal), a árvore biliar é alimentada exclusivamente pelo sistema arterial. Para o estudante de medicina e o residente de cirurgia, a compreensão desta premissa muda drasticamente a forma como o bisturi e o cautério são manuseados em torno do hilo hepático. A esqueletização excessiva de um ducto biliar não é um sinal de dissecção meticulosa, mas sim a antessala anatómica de uma complicação catastrófica: a isquemia, seguida de necrose e estenose biliar. Neste artigo, dissecaremos a anatomia vascular da árvore biliar, as armadilhas do ducto cístico e os fundamentos anatómicos da cirurgia oncológica da vesícula.
1. A Vesícula Biliar: Topografia e Peculiaridades Histológicas
A vesícula biliar repousa no equador que divide o fígado direito do esquerdo — uma linha imaginária conhecida como Linha de Cantlie (ou linha de Rex-Cantlie), que cursa entre os segmentos IVb e V em direção à veia cava inferior posteriormente.
A vesícula é maioritariamente peritonizada, exceto na sua face posterior, que assenta diretamente na placa cística. No entanto, o seu verdadeiro “Calcanhar de Aquiles” reside na sua histologia: a vesícula biliar não possui muscularis mucosae, não possui submucosa e apresenta uma camada muscular descontínua.
- Implicação Cirúrgica Oncológica: Estas especificidades anatómicas removem as barreiras naturais contra a progressão tumoral, facilitando a invasão direta do cancro da vesícula biliar para o parênquima hepático. É por este motivo que o tratamento cirúrgico curativo exige uma Colecistectomia Radical (que inclui a ressecção em cunha dos segmentos hepáticos IVb e V) sempre que o estadiamento tumoral (T) for superior ou igual a T1b.
2. O Labirinto Anatómico do Ducto Cístico e da Via Biliar Principal
A partir do infundíbulo cónico da vesícula, o ducto cístico estende-se como o limite inferior do triângulo hepatocístico em direção ao hilo, unindo-se ao ducto hepático comum (DHC) para formar o ducto colédoco (VBP). Contudo, na via biliar, a variação anatómica é a regra, não a exceção. O ducto cístico pode correr paralelamente ao DHC, espiralar posteriormente a este e inserir-se na sua face medial. Mais perigoso ainda é quando o cístico se insere no Ducto Setorial Posterior Direito (RPD), uma variação que ocorre em cerca de 4% dos fígados. Esta configuração é notoriamente perigosa, expondo o RPD a um risco altíssimo de secção inadvertida durante uma colecistectomia de rotina. Distalmente, o colédoco entra na cabeça do pâncreas e une-se ao ducto pancreático (Wirsung) para formar a ampola hepatopancreática, controlada pelo Esfíncter de Oddi.
- O Canal Comum Longo: Quando a junção entre o colédoco e o ducto pancreático ocorre de forma anómala (antes do complexo esfincteriano), ocorre o refluxo de enzimas pancreáticas ativadas para a árvore biliar. Este trauma químico crónico é o principal fator de risco para a formação de cistoss do colédoco e para o desenvolvimento de Colangiocarcinoma.
3. O “Tendão de Aquiles”: A Vascularização Arterial da Via Biliar
Como mencionado, a árvore biliar depende de um fluxo arterial ininterrupto. O suprimento segue um padrão de perfusão que ascende e descende ao longo dos ductos:
- O Suprimento Distal (Ascendente): O ducto colédoco recebe a sua irrigação inferior a partir de arteríolas emparelhadas provenientes da Artéria Gastroduodenal (GDA) e da Artéria Pancreatoduodenal Superior Posterior (PSPDA). Esta última representa o suprimento arterial mais importante e constante para o colédoco distal.
- O Suprimento Proximal (Descendente): Proximamente, o colédoco é alimentado por arteríolas originadas da artéria hepática direita (AHD).
- As Artérias Marginais: Estes vasos superiores e inferiores anastomosam-se para formar as artérias marginais, que correm paralelamente ao longo do ducto colédoco, situando-se tipicamente nas posições das 3 e 9 horas (lateral e medialmente). Desnudar (esqueletizar) o colédoco deste suprimento arterial para criar uma anastomose ou durante uma linfadenectomia não regrada acarreta um risco altíssimo de isquemia e estenose cicatricial a longo prazo.
4. O Plexo Epicoledociano Hilar e as Variações Arteriais
No interior do hilo hepático, uma rica rede de capilares liga as artérias hepáticas direita e esquerda. Este leito vascular, denominado Plexo Epicoledociano Hilar, fornece uma circulação colateral crítica que pode manter o suprimento arterial para um lado do fígado caso o vaso ipsilateral seja lesado. A preservação deste plexo e do suprimento sanguíneo arterial para o fígado remanescente é absolutamente crucial na criação de uma anastomose biliodigestiva (hepaticojejunostomia). A isquemia hilar resulta na temível colangiopatia isquémica e no desenvolvimento de abcessos hepáticos intratáveis.
O Papel das Variações Arteriais no Colangiocarcinoma Hilar
Na cirurgia do colangiocarcinoma hilar (Tumor de Klatskin), a complexidade aumenta devido às variações arteriais. A artéria hepática direita cruza posteriormente ao ducto hepático na maioria dos casos, mas em 25% cruza anteriormente. Mais relevante ainda é a presença de uma Artéria Hepática Direita Substituída (ou Acessória), com origem na Artéria Mesentérica Superior (AMS). Ao contrário da anatomia clássica, este vaso cursa lateralmente ao ducto colédoco (e não medialmente), estando extremamente vulnerável à iatrogenia se o cirurgião não o identificar durante a dissecção do ligamento hepatoduodenal. Combinações destas variações com a localização exata do tumor ditarão se uma lesão hilar é ressecável ou irresecável.
Pontos-Chave para a Prática Diária
- A Regra das 3 e 9 Horas: Lembre-se sempre de que o suprimento axial do colédoco viaja nos seus flancos laterais. Evite dissecções circunferenciais extensas da via biliar principal.
- Histologia e Neoplasias: A ausência de submucosa na vesícula biliar dita a necessidade de cirurgia hepática radical (ressecção em cunha do leito) em tumores T1b ou superiores.
- Inserções Anómalas do Cístico: A inserção num ducto setorial direito (RPD) é uma armadilha clássica. A Visão Crítica de Segurança (CVS) e a colangiografia são as únicas formas de evitar a transecção de um ducto principal aberrante.
- O Perigo do Canal Comum: A junção biliopancreática anómala exige vigilância ou tratamento cirúrgico devido ao elevado risco de colangiocarcinoma derivado da inflamação enzimática crónica.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
O respeito pela árvore biliar transcende a mera identificação do tubo que transporta a bílis; exige uma veneração profunda pela sua microvascularização. O cirurgião que descasca a via biliar como se tratasse de um cabo elétrico condena o seu doente a um futuro de estenoses isquémicas, colangites de repetição e reintervenções de extrema complexidade. O domínio da variabilidade anatómica vascular (como as artérias substituídas da AMS) e o conhecimento tático do plexo epicoledociano não são meros detalhes de rodapé dos livros de anatomia; são a linha que separa o triunfo cirúrgico oncológico da catástrofe iatrogénica irreparável.
“A anatomia biliar não segue regras absolutas, apenas tendências. Operar no hilo hepático assumindo a normalidade é o primeiro passo para o desastre. O cirurgião deve dissecar com a expectativa constante da variação.” — Aforismo clássico da cirurgia hepatobiliar.
Gostou ❔Deixe-nos um comentário ✍️, partilhe nas suas redes sociais e|ou envie a sua dúvida pelo 💬 Chat On-line na nossa DM do Instagram.
Critical View Of Safety
“The concept of the critical view was described in 1992 but the term CVS was introduced in 1995 in an analytical review of the emerging problem of biliary injury in laparoscopic cholecystectomy. CVS was conceived not as a way to do laparoscopic cholecystectomy but as a way to avoid biliary injury. To achieve this, what was needed was a secure method of identifying the two tubular structures that are divided in a cholecystectomy, i.e., the cystic duct and the cystic artery. CVS is an adoption of a technique of secure identification in open cholecystectomy in which both cystic structures are putatively identified after which the gallbladder is taken off the cystic plate so that it is hanging free and just attached by the two cystic structures. In laparoscopic surgery complete separation of the body of the gallbladder from the cystic plate makes clipping of the cystic structures difficult so for laparoscopy the requirement was that only the lower part of the gallbladder (about one-third) had to be separated from the cystic plate. The other two requirements are that the hepatocystic triangle is cleared of fat and fibrous tissue and that there are two and only two structures attached to the gallbladder and the latter requirements were the same as in the open technique. Not until all three elements of CVS are attained may the cystic structures be clipped and divided. Intraoperatively CVS should be confirmed in a “time-out” in which the 3 elements of CVS are demonstrated. Note again that CVS is not a method of dissection but a method of target identification akin to concepts used in safe hunting procedures. Several years after the CVS was introduced there did not seem to be a lessening of biliary injuries.
Operative notes of biliary injuries were collected and studied in an attempt to determine if CVS was failing to prevent injury. We found that the method of target identification that was failing was not CVS but the infundibular technique in which the cystic duct is identified by exposing the funnel shape where the infundibulum of the gallbladder joins the cystic duct. This seemed to occur most frequently under conditions of severe acute or chronic inflammation. Inflammatory fusion and contraction may cause juxtaposition or adherence of the common hepatic duct to the side of the gallbladder. When the infundibular technique of identification is used under these conditions a compelling visual deception that the common bile duct is the cystic duct may occur. CVS is much less susceptible to this deception because more exposure is needed to achieve CVS, and either the CVS is attained, by which time the anatomic situation is clarified, or operative conditions prevent attainment of CVS and one of several important “bail-out” strategies is used thus avoiding bile duct injury.
CVS must be considered as part of an overall schema of a culture of safety in cholecystectomy. When CVS cannot be attained there are several bailout strategies such a cholecystostomy or in the case of very severe inflammation discontinuation of the procedure and referral to a tertiary center for care. The most satisfactory bailout procedure is subtotal cholecystectomy of which there are two kinds. Subtotal fenestrating cholecystectomy removes the free wall of the gallbladder and ablates the mucosa but does not close the gallbladder remnant. Subtotal reconstituting cholecystectomy closes the gallbladder making a new smaller gallbladder. Such a gallbladder remnant is undesirable since it may become the site of new gallstone formation and recurrent symptoms . Both types may be done laparoscopically.”
Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 1995;180:101-25.
Medically-Necessary, Time Sensitive: (MeNTS) Score
Operating During The COVID-19 Coronavirus Pandemic
“At the University of Chicago, members of the Department of Surgery decided to investigate this issue more precisely. As stay-at-home restrictions in some states are easing, and as non-emergency medical care is being reconsidered, how does one possibly triage the thousands upon thousands of patients whose surgeries were postponed? Instead of the term “elective,” the University of Chicago’s Department of Surgery chose the phrase “Medically-Necessary, Time Sensitive” (MeNTS). This concept can be utilized to better assess the acuity and safety when determining which patients can get to the operating room in as high benefit/low risk manner as possible. And unlike in any recent time in history, risks to healthcare staff as well as risks to the patient from healthcare staff, are now thrown into the equation. The work was published in the April issue of the Journal of the American College of Surgeons.
On March 17, 2020, the American College of Surgeons recommended that all “elective” surgeries be canceled indefinitely. These guidelines were published, stating that only patients with “high acuity” surgical issues, which would include aggressive cancers and severely symptomatic disease, should proceed. Based on the Elective Surgery Acuity Scale (ESAS), most hospitals were strongly encouraged to cancel any surgery that was not high acuity, including slow-growing cancers, orthopedic and spine surgeries, airway surgeries, and any other surgeries for non-cancerous tumors. Heart surgeries for stable cardiac issues were also put on hold. Patients and surgeons waited. Some patients did, indeed undergo non-Covid-19-related surgeries. But most did not. Redeployment is gradually turning to re-entry.
The re-entry process for non-urgent (yet necessary) surgeries is a complicated one. Decisions and timing, based on a given hospital’s number and severity of Covid-19 patients, combined with a given city or state’s current and projected number of Covid-19 cases, how sick those patients will be, and whether or not a second surge may come, involves a fair amount of guesswork. As we have all seen, data manipulation has become a daily sparring match in many arenas. The authors of the study created an objective surgical risk scoring system, in order to help hospitals across this country, as well as others across the world, better identify appropriate timing regarding which surgeries can go ahead sooner rather than later, and why. They factored several variables into their equation, to account for the multiple potential barriers to care, including health and safety of hospital personnel. They created scoring systems based on three factors: Procedure, Disease and Patient Issues.
CALCULATE MeNTS SCORE HERE
The authors of the study created an objective surgical risk scoring system, in order to help hospitals across this country, as well as others across the world, better identify appropriate timing regarding which surgeries can go ahead sooner rather than later, and why. They factored several variables into their equation, to account for the multiple potential barriers to care, including health and safety of hospital personnel. Each patient would receive an overall conglomerate score, based on all of these factors, with the lower risks giving them more favorable scores to proceed with surgery soon, and the higher risks giving patients a higher score, or higher risk regarding proceeding with surgery, meaning it may be safest, for now, to wait.
Dr. Jeffrey Matthews, senior author of the paper, and Department Chair at the University of Chicago, stated that this model is reproducible across hospital systems, in urban, rural, and academic settings. And in the event of potential unpredictable surges of Covid-19 cases, the scoring system “helps prioritize cases not only from the procedure/disease standpoint but also from the pandemic standpoint with respect to available hospital resources such as PPE, blood, ICU beds, and [regular hospital] beds.”
The scoring system is extremely new, and the coming weeks will reveal how patients, surgeons and hospitals are faring as patients without life-and-death emergencies and/or Covid-19 complications gradually begin filling the operating rooms and hospital beds. In addition, and perhaps just as important, the study authors note that creating systems whereby healthcare resources, safety, and impact on outcomes need to be considered more carefully for each patient intervention, the larger impact of each intervention on public health will be better understood: not only for today’s pandemic, but also in future, as yet unknown, global events.”
Source: Nina Shapiro, 2020
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
Covid-19 and Digestive Surgery
The current world Covid-19 pandemic has been the most discussed topic in the media and scientific journals. Fear, uncertainty, and lack of knowledge about the disease may be the significant factors that justify such reality. It has been known that the disease presents with a rapidly spreading, it is significantly more severe among the elderly, and it has a substantial global socioeconomic impact. Besides the challenges associated with the unknown, there are other factors, such as the deluge of information. In this regard, the high number of scientific publications, encompassing in vitro, case studies, observational and randomized clinical studies, and even systematic reviews add up to the uncertainty. Such a situation is even worse when considering that most healthcare professionals lack adequate knowledge to critically appraise the scientific method, something that has been previously addressed by some authors. Therefore, it is of utmost importance that expert societies supported by data provided by the World Health Organization and the National Health Department take the lead in spreading trustworthy and reliable information.
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
#Medicine #Surgery #GeneralSurgery #DigestiveSurgery
#TheSurgeon #OzimoGama
Laparoscopic JEJUNOSTOMY
Many oncological patients with upper gastrointestinal (GI) tract tumours, apart from other symptoms, are malnourished or cachectic at the time of presentation. In these patients feeding plays a crucial role, including as part of palliative treatment. Many studies have proved the benefits of enteral feeding over parenteral if feasible. Depending on the tumour’s location and clinical stage there are several options of enteral feeding aids available. Since the introduction of percutaneous endoscopic gastrostomy (PEG) and its relatively easy application in most patients, older techniques such as open gastrostomy or jejunostomy have rather few indications.
The majority of non-PEG techniques are used in patients with upper digestive tract, head and neck tumours or trauma that renders the PEG technique unfeasible or unsafe for the patient. In these patients, especially with advanced disease requiring neoadjuvant chemotherapy or palliative treatment, open gastrostomy and jejunostomy were the only options of enteral access. Since the first report of laparoscopic jejunostomy by O’Regan et al. in 1990 there have been several publications presenting techniques and outcomes of laparoscopic feeding jejunostomy. Laparoscopic jejunostomy can accompany staging or diagnostic laparoscopy for upper GI malignancy when the disease appears advanced, hence avoiding additional anaesthesia and an operation in the near future.
In this video the author describe the technique of laparoscopic feeding jejunostomy applied during the staging laparoscopy in patient with advanced upper gastrointestinal tract cancer with co-morbid cachexy, requiring enteral feeding and neoadjuvant chemotherapy.
Welcome and get to know our Social Networks through the following link
Ozimo Gama’s Social Medias
#AnatomiaHumana #CirurgiaGeral #CirurgiaDigestiva #TheSurgeon
General Surgery and COVID19

The American College of Surgeons (ACS) and SAGES has developed COVID-19 and Surgery as an online resource for the surgical community facing the impact of Coronavirus Disease 2019 (COVID-19). Content has been developed or curated under the auspices of ACS and SAGES Regents and Officers to bring surgeons trusted information, including best practices and guidance that specifically target the concerns and challenges surgeons face. As the COVID-19 landscape is rapidly changing, this website is updated several times weekly and houses current and past editions of our electronic newsletter.
American College of Surgeons Bookmark
Soc of Am Gi and End Surg Bookmark (SAGES)
Discover our surgical video channel and lectures associated with the surgeon blog.
Share and Join: https://linktr.ee/TheSurgeon
#Medicine #Surgery #GeneralSurgery #DigestiveSurgery
#TheSurgeon #OzimoGama
Surgical Management of CHRONIC PANCREATITIS
Chronic pancreatitis (CP) is a progressive, destructive, inflammatory process that ends in total destruction of the pâncreas and results in malabsorption, diabetes mellitus, and severe pain. The incidence and prevalence of CP are increasing in the worldwide and incidence is between 1.6 and 23 per 100 000 with increasing prevalence. The treatment of CP is complex; in the majority of cases na interdisciplinary approach is indicated that includes conservative, endoscopic, and surgical therapy. The surgical treatment of CP is based on two main concepts:preservation of tissue via drainage aims to protect against further loss of pancreatic function, and pancreatic resection is performed for nondilated pancreatic ducts, pancreatic head enlargement,or if a pancreatic carcinoma is suspected in the setting of CP.
RESECTIVE PROCEDURES
The vast majority of patients are seen with a ductal obstruction in the pancreatic head, frequently associated with an inflammatory mass. In these patients, pancreatic head resection is the procedure of choice; The partial pancreatoduodenectomy (PD) or Kausch-Whipple procedure, in its classic or pylorus-preserving variant, has been the procedure of choice for pancreatic head resection in CP for many years (Jimenez et al, 2003). The duodenum-preserving pancreatic head resections and its variants—the Beger (1985), Frey (1987), and Bern procedures (Gloor et al, 2001)—represent less invasive, organsparing techniques with equal long-term results. Only very few patients come to medical attention with smallduct disease (diameter of the pancreatic duct ❤ mm) and no mass in the pancreatic head. Possibly, a large majority of those patients from former series had unknown autoimmune pancreatitis. In these cases, a V-shaped excision of the anterior aspect of the pancreas is a safe approach, with effective pain management (Yekebas et al, 2006). In the rare case of a patient seen with segmental CP in the pancreatic body or tail, such as that seen as a result of posttraumatic ductal stenosis, a middle segment pancreatectomy or a pancreatic left resection may be the best approach.
CONCLUSION
The adequate therapy of CP is adjusted to the symptoms of the patient, the stage of the disease, and the morphology of pathologic changes of the pancreas. The surgical technique must be adjusted to the pathomorphologic changes of the pancreas. For patients with CP and an inflammatory mass in the head of the pancreas, the DPPHR is less invasive than a PD and is associated with comparable long-term results. The Bern modification of the DPPHR represents a technical variation that is equally effective but technically less demanding. Whether total pancreatectomy with islet cell transplantation is a viable therapy of CP remains to be proved by further studies. Surgical therapy provides effective long-term pain relief and improvement of quality of life, but it may not stop the decline of endocrine or exocrine pancreatic function. Strategies to improve or maintain endocrine and exocrine function in CP remain an interesting field of research.
Femoral Hernia REPAIR
Femoral hernia is not as common as inguinal hernia. It is often associated with incarceration or strangulation, resulting in peritonitis and mortality.
ANATOMY

The pelvicrural interval (the opening from the abdomen to the thigh) is divided into two spaces: a lateral space, the lacuna musculosa, through which the iliopsoas muscles pass; and a medial space, the lacuna vasculosa, for the femoral vessels. The external iliac vessels run along the anterior surface of the iliopsoas muscle in the pelvis, pass between the iliopubic tract and Cooper’s ligament, and finally course beneath the inguinal ligament to become the femoral vessels. Where the external iliac vessels run down into the lacuna vasculosa, transversalis fascia covers the vessels to form the femoral sheath. It extends approximately 4 cm caudally and ends as the adventitia of the femoral vessels. The medial compartment of the femoral sheath is called the femoral canal, which is ordinarily less than 2 cm in diameter and contains lymphatic vessels and glands. The true opening of the femoral canal is a musculoaponeurotic ring, consisting of Cooper’s ligament inferiorly, the femoral vein laterally, and iliopubic tract superiorly and medially. In the past, the medial border of the femoral ring was for the lacunar ligament. The lacunar ligament is an attachment of the inguinal ligament to the pubic bone, however, and lies in the outer layer of the transversalis fascia.
McVay demonstrated that the medial boundary of the femoral ring is the lateral edge of the aponeurosis of the insertion of the transversus abdominis muscle with transversalis fascia onto the pectin of the pubis, not the lacunar ligament. Condon also demonstrated that the iliopubic tract bridges the femoral canal and then curves posteriorly and inferiorly, its fibers spreading fanwise to insert adjacent to Cooper’s ligament into a broad area of the superior ramus of the pubis. Thus, the true inner ring of the femoral canal is bounded by the iliopubic tract anteriorly and medially, and by Cooper’s ligament posteriorly. If a surgeon incises the inguinal ligament in a tightly incarcerated femoral hernia, he or she will find that the hernia cannot be reduced because of the more deeply placed ring. The distal orifice has a rigid boundary—surrounded by the lacunar ligament medially; the inguinal ligament superiorly; and the fascia of the pectineal muscle—and is usually less than 1 cm in diameter. The rigidity of these structures is the reason why strangulation often occurs in femoral hernias.
ETIOLOGY
Currently, the ‘‘acquired’’ theory is widely accepted; however, the true cause of femoral hernia is not known. McVay demonstrated that the width of the femoral ring, which is determined by the length of the fanwise insertion of the iliopubic tract to Cooper’ ligament, is the main etiologic factor of the femoral hernia. Considering that the femoral hernia is very rare in children and most common in elderly women, however, McVay’s concept cannot be the only reason for the occurrence of femoral hernia. Nyhus noted the presence of a relatively large femoral defect without an accompanying femoral hernia during the preperitoneal approach. This may be caused by the acquired weakness of the transversalis fascia and a consequent predisposition to the development of the femoral hernia.
INCIDENCE
The ratio of femoral hernia relative to all groin hernias is reported to be 2% to 8% in adults . Femoral hernias are very rare in children, and most commonly observed between the ages of 40 and 70. The peak distribution is in the 50s, with a slight decrease in the 60s and 70s. As for sex distribution, femoral hernia is 4 to 5 times more common in female than in male; however, there are some reports that it is more common in men than in women. A right-sided presentation is more common than left, but the reason is not known.
TREATMENT
Finally, femoral hernia is usually thought of as requiring emergency surgical treatment. Only 30% of our cases were treated as emergency operations, however, whereas 70% were elective. Unless patients complain of severe abdominal pain or ileus, surgeons need not perform emergency operations. In summary, the mesh plug hernia repair for femoral hernia has resulted in a reduced recurrence rate, shortened hospital stay, and a low rate of postoperative complications.
How to Make a Good STOMA

INTRODUCTION: Few other surgical procedures adversely affect a patient’s quality of life as much as a poorly functioning stoma. An ideal stoma meets two criteria: (1) The site is optimally matched to a patient’s variability in body form, physical ability and activities. (2) The construction minimises complications that relate to the use of stomal appliances and minimises technical failings such as parastomal hernia or prolapse.
SURGICAL ASPECTS
1.The Skin and Subcutaneous Incision
A circular stomal opening is generally preferred, though for temporary stomata a linear incision minimises skin loss and may improve cosmesis after closure. We favour making a cruciate incision with cutting electrocautery, each quadrant being excised in a curved fashion with electrocautery or curved (Mayo) scissors to prevent charring.
2.Muscle Fascia
A cruciate incision of the muscle fascia is generally used, mirroring that for the skin incision but without excision. It is common practice during laparotomy to align the muscle fasciotomy and skin incision by medial retraction of the rectus sheath using tissue-grasping forceps (e.g. Lanes’). This may reduce angulation of the bowel through the abdominal wall, though is unlikely to affect the duration of paralytic ileus in the post-operative phase and has little effect on eventual function.
3.Muscle
A muscle-splitting incision through rectus abdominis is advocated, though this may simply be a necessary anatomical consequence reflecting the preference for an anterior stoma distant from the umbilicus, iliac crest and midline wounds. Stomal formation lateral to rectus abdominis does not actually seem to increase the risk of para-stomal hernia formation. This is unsurprising, since muscle division and correct closure at apppendicectomy rarely leads to hernia formation.
4.Choice of Bowel for the Construction of a Stoma
The principles of good anastamotic healing apply equally to stomal construction. Attention to tissue handling, vascularity and lack of tension encourage primary healing at the muco-cutaneous junction. Poor technique risks separation of the muco-cutaneous junction and prolonged healing by granulation, leading to stenosis. Tension may worsen stomal or spout retraction and can lead to difficulties in attaching stomal appliances to a concave stoma, particularly if a tight limb of the stoma gives a skin fold crease. Similarly, impaired vascularity can turn stomata a worrying colour, particularly if inotropes are required for a critically ill patient, and although frank necrosis is rare, stenosis may result in the longer term.
“Patients often judge a surgeon’s technical ability by the external appearance of scars, and may also judge a surgeon’s care and precision by the appearance and function of an abdominal stoma.”
Surgical management of gastrointestinal bleeding
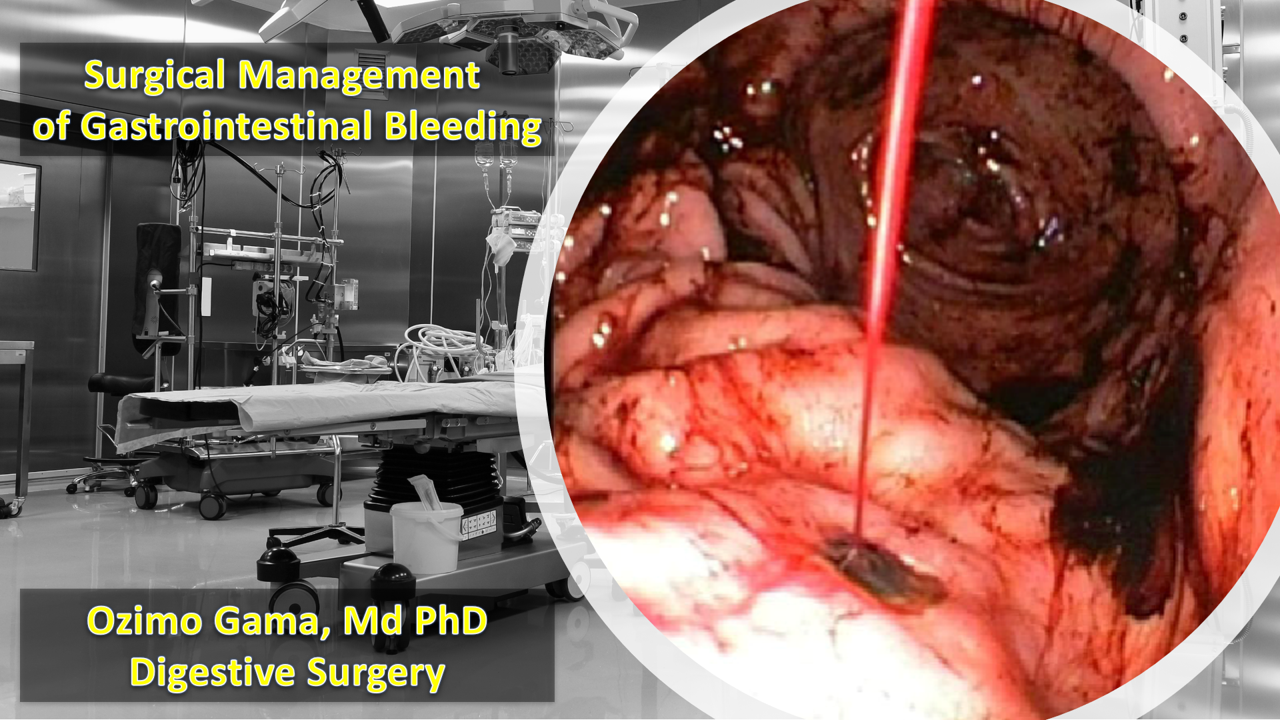
Severe gastrointestinal bleeding has historically been a clinical problem primarily under the purview of the general surgeon. Diagnostic advances made as the result of newer technologies, such as fiberoptic and video endoscopy, selective visceral arteriography, and nuclear scintigraphy, have permitted more accurate and targeted operations. More importantly, they have led to safe, effective nonoperative therapeutic interventions that have obviated the need for surgery in many patients. Today, most gastrointestinal bleeding episodes are initially managed by endoscopic or angiographic control measures. Such interventions are often definitive in obtaining hemostasis. Even temporary cessation or attenuation of massive bleeding in an unstable patient permits a safer, more controlled operative procedure by allowing an adequate period of preoperative resuscitation. Despite the less frequent need for surgical intervention, traditional operative approaches, such as suture ligation, lesion or organ excision, vagotomy, portasystemic anastomosis, and devascularization procedures, continue to be life-saving in many instances. The proliferation of laparoscopic surgery has fostered the application of minimally invasive techniques to highly selected patients with gastrointestinal bleeding. Intraoperative endoscopy has greatly facilitated the accuracy of laparoscopic surgery by endoscopic localization of bleeding lesions requiring excision. It is anticipated that the evolving technologies pertinent to the diagnosis and management of gastrointestinal bleeding will continue to promote collaboration and cooperation between gastroenterologists, radiologists, and surgeons.
Surgical Management
The role of surgery in acute peptic ulcer bleeding has markedly changed over the past two decades. The widespread use of endoscopic treatment has reduced the number of patients requiring surgery. Therefore, the need for routine early surgical consultation in all patients presenting with acute UGIB is now obviated (Gralnek et al., 2008). Emergency surgery should not be delayed, even if the patient is in haemodynamic shock, as this may lead to mortality (Schoenberg, 2001). Failure to stop bleeding with endoscopic haemostasis and/or interventional radiology is the most important and definite indication. The surgical procedures under these circumstances should be limited to achieve haemostasis. The widespread use of PPIs obviated further surgical procedures to reduce acid secretion. Rebleeding tends to necessitate emergency surgery in approximately 60% of cases with an increase in morbidity and mortality (Schoenberg et al.; 2001). The reported mortality rates after emergency surgery range from 2 – 36%. Whether to consider endoscopic retreatment or surgery for bleeding after initial endoscopic control is controversial (Cheung et al., 2009). A second attempt at endoscopic haemostasis is often effective (Cheung et al., 2009), with fewer complications avoiding some surgery without increasing mortality (Lau et al., 1999). Therefore, most patients with evidence of rebleeding can be offered a second attempt at endoscopic haemostasis. This is often effective, may result in fewer complications than surgery, and is the current recommended management approach. Available data suggest that early elective surgery for selected high-risk patients with bleeding peptic ulcer might decrease the overall mortality rate. It is a reasonable approach in ulcers measuring ≥2 cm or patients with hypotension at rebleeding that independently predicts endoscopic retreatment failure (Lau et al., 1999). Early elective surgery in patients presenting with arterial bleeding or a visible vessel of ≥2 mm is superior to endoscopic retreatment and has a relatively low overall mortality rate of 5% (Imhof et al., 1998 & 2003). Additional indications for early elective surgery include age >65 years, previous admission for ulcer plication, blood transfusion of more than 6 units in the first 24 hours and rebleeding within 48 hours (Bender et al., 1994; Mueller et al., 1994). This approach is associated with a low 30–day mortality rate as low as 7%.
Pringle Maneuver

After the first major hepatic resection, a left hepatic resection, carried out in 1888 by Carl Langenbuch, it took another 20 years before the first right hepatectomy was described by Walter Wendel in 1911. Three years before, in 1908, Hogarth Pringle provided the first description of a technique of vascular control, the portal triad clamping, nowadays known as the Pringle maneuver. Liver surgery has progressed rapidly since then. Modern surgical concepts and techniques, together with advances in anesthesiological care, intensive care medicine, perioperative imaging, and interventional radiology, together with multimodal oncological concepts, have resulted in fundamental changes. Perioperative outcome has improved significantly, and even major hepatic resections can be performed with morbidity and mortality rates of less than 45% and 4% respectively in highvolume liver surgery centers. Many liver surgeries performed routinely in specialized centers today were considered to be high-risk or nonresectable by most surgeons less than 1–2 decades ago.Interestingly, operative blood loss remains the most important predictor of postoperative morbidity and mortality, and therefore vascular control remains one of the most important aspects in liver surgery.
“Bleeding control is achieved by vascular control and optimized and careful parenchymal transection during liver surgery, and these two concepts are cross-linked.”
First described by Pringle in 1908, it has proven effective in decreasing haemorrhage during the resection of the liver tissue. It is frequently used, and it consists in temporarily occluding the hepatic artery and the portal vein, thus limiting the flow of blood into the liver, although this also results in an increased venous pressure in the mesenteric territory. Hemodynamic repercussion during the PM is rare because it only diminishes the venous return in 15% of cases. The cardiovascular system slightly increases the systemic vascular resistance as a compensatory response, thereby limiting the drop in the arterial pressure. Through the administration of crystalloids, it is possible to maintain hemodynamic stability.

In the 1990s, the PM was used continuously for 45 min and even up to an hour because the depth of the potential damage that could occur due to hepatic ischemia was not yet known. During the PM, the lack of oxygen affects all liver cells, especially Kupffer cells which represent the largest fixed macrophage mass. When these cells are deprived of oxygen, they are an endless source of production of the tumour necrosis factor (TNF) and interleukins 1, 6, 8 and 10. IL 6 has been described as the cytokine that best correlates to postoperative complications. In order to mitigate the effects of continuous PM, intermittent clamping of the portal pedicle has been developed. This consists of occluding the pedicle for 15 min, removing the clamps for 5 min, and then starting the manoeuvre again. This intermittent passage of the hepatic tissue through ischemia and reperfusion shows the development of hepatic tolerance to the lack of oxygen with decreased cell damage. Greater ischemic tolerance to this intermittent manoeuvre increases the total time it can be used.
Hepatic Surgery: Portal Vein Embolization

INTRODUCTION
Portal vein Embolizations (PVE) is commonly used in the patients requiring extensive liver resection but have insufficient Future Liver Remanescent (FLR) volume on preoperative testing. The procedure involves occluding portal venous flow to the side of the liver with the lesion thereby redirecting portal flow to the contralateral side, in an attempt to cause hypertrophy and increase the volume of the FLR prior to hepatectomy.
PVE was first described by Kinoshita and later reported by Makuuchi as a technique to facilitate hepatic resection of hilar cholangiocarcinoma. The technique is now widely used by surgeons all over the world to optimize FLR volume before major liver resections.
PHYSIOPATHOLOGY
PVE works because the extrahepatic factors that induce liver hypertrophy are carried primarily by the portal vein and not the hepatic artery. The increase in FLR size seen after PVE is due to both clonal expansion and cellular hypertrophy, and the extent of post-embolization liver growth is generally proportional to the degree of portal flow diversion. The mechanism of liver regeneration after PVE is a complex phenomenon and is not fully understood. Although the exact trigger of liver regeneration remains unknown, several studies have identified periportal inflammation in the embolized liver as an important predictor of liver regeneration.

THECNICAL ASPECTS
PVE is technically feasible in 99% of the patients with low risk of complications. Studies have shown the FLR to increase by a median of 40–62% after a median of 34–37 days after PVE, and 72.2–80% of the patients are able to undergo resection as planned. It is generally indicated for patients being considered for right or extended right hepatectomy in the setting of a relatively small FLR. It is rarely required before extended left hepatectomy or left trisectionectomy, since the right posterior section (segments 6 and 7) comprises about 30% of total liver volume.
PVE is usually performed through percutaneous transhepatic access to the portal venous system, but there is considerable variability in technique between centers. The access route can be ipsilateral (portal access at the same side being resected) with retrograde embolization or contralateral (portal access through FLR) with antegrade embolization. The type of approach selected depends on a number of factors including operator preference, anatomic variability, type of resection planned, extent of embolization, and type of embolic agent used. Many authors prefer ipsilateral approach especially for right-sided tumors as this technique allows easy catheterization of segment 4 branches when they must be embolized and also minimizes the theoretic risk of injuring the FLR vasculature or bile ducts through a contralateral approach and potentially making a patient ineligible for surgery.
However, majority of the studies on contralateral PVE show it to be a safe technique with low complication rate. Di Stefano et al. reported a large series of contralateral PVE in 188 patients and described 12 complications (6.4%) only 6 of which could be related to access route and none precluded liver resection. Site of portal vein access can also change depending on the choice of embolic material selected which can include glue, Gelfoam, n-butyl-cyanoacrylate (NBC), different types and sizes of beads, alcohol, and nitinol plus. All agents have similar efficacy and there are no official recommendations for a particular type of agent.
RESULTS
Proponents of PVE believe that there should be very little or no tumor progression during the 4–6 week wait period for regeneration after PVE. Rapid growth of the FLR can be expected within the first 3–4 weeks after PVE and can continue till 6–8 weeks. Results from multiple studies suggest that 8–30% hypertrophy over 2–6 weeks can be expected with slower rates in cirrhotic patients. Most studies comparing outcomes after major hepatectomy with and without preoperative PVE report superior outcomes with PVE. Farges et al. demonstrated significantly less risk of postoperative complications, duration of intensive care unit, and hospital stay in patients with cirrhosis who underwent right hepatectomy after PVE compared to those who did not have preoperative PVE. The authors also reported no benefit of PVE in patients with a normal liver and FLR >30%. Abulkhir et al. reported results from a meta-analysis of 1088 patients undergoing PVE and showed a markedly lower incidence of Post Hepatectomy Liver Failure (PHLF) and death compared to series reporting outcomes after major hepatectomy in patients who did not undergo PVE. All patients had FLR volume increase, and 85% went on to have liver resection after PVE with a PHLF incidence of 2.5% and a surgical mortality of 0.8%. Several studies looking at the effect of systemic neoadjuvant chemotherapy on the degree of hypertrophy after PVE show no significant impact on liver regeneration and growth.
VOLUMETRIC RESPONSE

The volumetric response to PVE is also a very important factor in understanding the regenerative capacity of a patient’s liver and when used together with FLR volume can help identify patients at risk of poor postsurgical outcome. Ribero et al. demonstrated that the risk of PHLF was significantly higher not only in patients with FLR ≤ 20% but also in patients with normal liver who demonstrated ≤5% of FLR hypertrophy after PVE. The authors concluded that the degree of hypertrophy >10% in patients with severe underlying liver disease and >5% in patients with normal liver predicts a low risk of PHLF and post-resection mortality. Many authors do not routinely offer resection to patients with borderline FLR who demonstrate ≤5% hypertrophy after PVE.
Predicting LIVER REMNANT Function
Careful analysis of outcome based on liver remnant volume stratified by underlying liver disease has led to recommendations regarding the safe limits of resection. The liver remnant to be left after resection is termed the future liver remnant (FLR). For patients with normal underlying liver, complications, extended hospital stay, admission to the intensive care unit, and hepatic insufficiency are rare when the standardized FLR is >20% of the TLV. For patients with tumor-related cholestasis or marked underlying liver disease, a 40% liver remnant is necessary to avoid cholestasis, fluid retention, and liver failure. Among patients who have been treated with preoperative systemic chemotherapy for more than 12 weeks, FLR >30% reduces the rate of postoperative liver insufficiency and subsequent mortality.
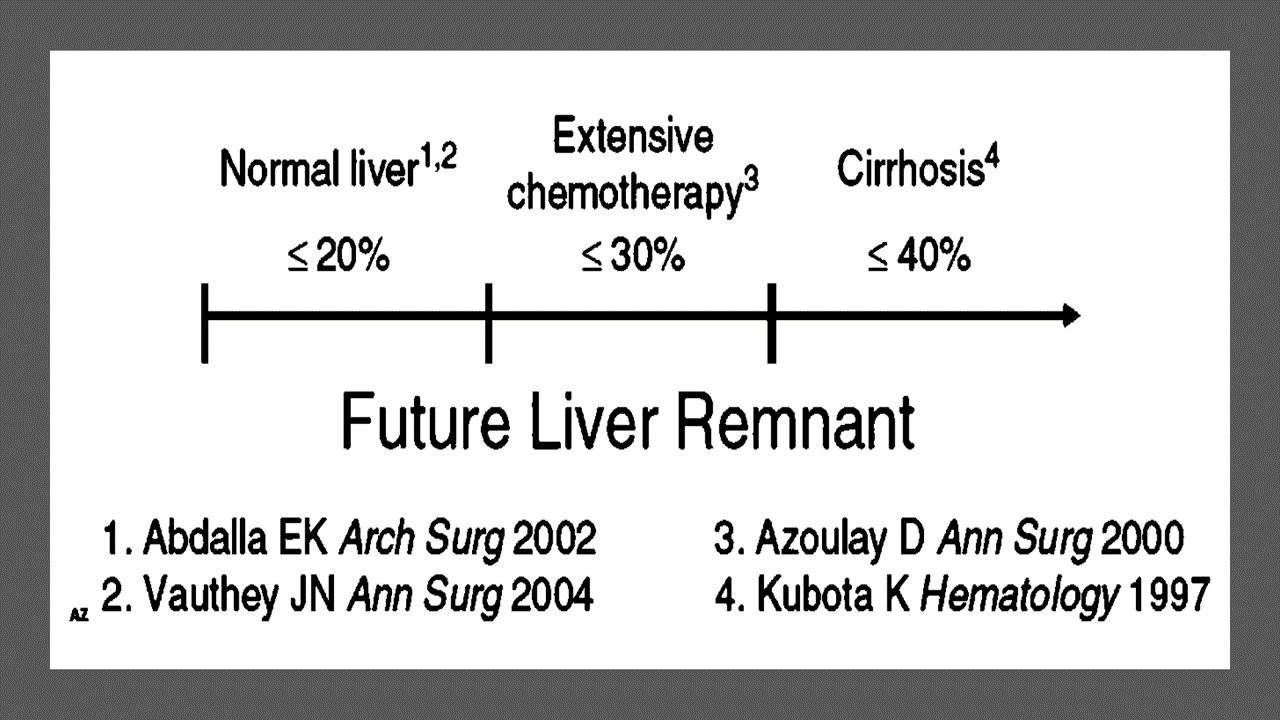
When the liver remnant is normal or has only mild disease, the volume of liver remnant can be measured directly and accurately with threedimensional computed tomography (CT) volumetry. However, inaccuracy may arise because the liver to be resected is often diseased, particularly in patients with cirrhosis or biliary obstruction. When multiple or large tumors occupy a large volume of the liver to be resected, subtracting tumor volumes from liver volume further decreases accuracy of CT volumetry. The calculated TLV, which has been derived from the association between body surface area (BSA) and liver size, provides a standard estimate of the TLV. The following formula is used:
TLV (cm3) = –794.41 + 1267.28 × BSA (square meters)
Thus, the standardized FLR (sFLR) volume calculation uses the measured FLR volume from CT volumetry as the numerator and the calculated TLV as the denominator: Standardized FLR (sFLR) = measured FLR volume/TLV Calculating the standardized TLV corrects the actual liver volume to the individual patient’s size and provides an individualized estimate of that patient’s postresection liver function. In the event of an inadequate FLR prior to major hepatectomy, preoperative liver preparation may include portal vein embolization (PVE).
Classroom: Principles of Hepatic Surgery
Videos of Surgical Procedures

This page provides links to prerecorded webcasts of surgical procedures. These are actual operations performed at medical centers in the Brazil. Please note that you cannot send in questions by email, though the webcast may say that you can, because you are not seeing these videos live. The videos open in a second window. If you have a pop-up blocker, you will need to disable it to view the programs.
Videos of Surgical Procedures
Strangulation in GROIN HERNIAS
Importance
Declining Mortality Rates
In both the UK and the USA, the annual death rate due to inguinal and femoral hernias has significantly decreased over the past two to three decades. In the UK, deaths from these hernias declined by 22% to 55% between 1975 and 1990. Similarly, in the USA, the annual deaths per 100,000 population for patients with hernia and intestinal obstruction decreased from 5.1 in 1968 to 3.0 in 1988. For patients with obstructed inguinal hernias, 88% underwent surgery, with a remarkably low mortality rate of 0.05%. These improvements suggest that elective groin hernia surgery has played a crucial role in reducing overall mortality rates.
Elective Surgery and Strangulation Rates
Supporting this observation, the USA has lower rates of strangulation compared to the UK, possibly due to the threefold higher rate of elective hernia surgeries in the USA. Nevertheless, statistics indicate that the rate of elective hernia surgeries in the USA per 100,000 population decreased from 358 to 220 between 1975 and 1990, although this may be an artifact of data collection rather than a genuine decline.
Mortality Analysis from UK and Denmark Studies
During 1991–1992, the UK National Confidential Enquiry Into Perioperative Deaths investigated 210 deaths following inguinal hernia repair and 120 deaths following femoral hernia repair. This inquiry, which focuses on the quality of surgery, anesthesia, and perioperative care, found that many patients were elderly (45 were aged 80–89 years) and significantly infirm; 24 were ASA grade III and 21 ASA grade IV. The majority of postoperative mortality was attributed to preexisting cardiorespiratory issues.
A nationwide study in Denmark of 158 patients who died after acute groin hernia repair by Kjaergaard et al. also found that these patients were old (median age 83 years) and frail (>80% with significant comorbidity), with frequent delays in diagnosis and treatment. These findings highlight the need for high-quality care by experienced surgeons and anesthetists, especially for patients with high ASA grades.
Postoperative Care Recommendations
Postoperative care for these patients should occur in a high-dependency unit or intensive therapy unit. This might necessitate transferring selected patients to appropriate hospitals and facilities. Decisions about interventional surgery should be made in consultation with the relatives of extremely elderly, frail, or moribund patients, adopting a humane approach that may rule out surgery.
Emergency Admissions and Prioritization
Forty percent of patients with femoral hernias are admitted as emergency cases with strangulation or incarceration, while only 3% of patients with direct inguinal hernias present with strangulation. This disparity has implications for prioritizing patients on waiting lists when these hernias present electively in outpatient clinics.
Risk of Strangulation
A groin hernia is at its greatest risk of strangulation within three months of onset. For inguinal hernias, the cumulative probability of strangulation is 2.8% at three months after presentation, rising to 4.5% after two years. The risk is much higher for femoral hernias, with a 22% probability of strangulation at three months, rising to 45% at 21 months. Right-sided hernias have a higher strangulation rate than left-sided hernias, potentially due to anatomical differences in mesenteric attachment. The decline in hernia-related mortality in both the UK and USA underscores the importance of elective hernia surgery. Ensuring timely surgery, especially for high-risk femoral hernias, and providing high-quality perioperative care for elderly and frail patients are crucial steps in further reducing mortality and improving patient outcomes.
Evidence-Based Medicine
In a randomized trial, evaluating an expectative approach to minimally symptomatic inguinal hernias, Fitzgibbons et al. in the group of patients randomized to watchful waiting found a risk of an acute hernia episode of 1.8 in 1,000 patient years. In another trial, O’Dwyer and colleagues, randomizing patients with painless inguinal hernias to observation or operation, found two acute episodes in 80 patients randomized to observation. In both studies, a large percentage of patients randomized to nonoperative care were eventually operated due to symptoms. Neuhauser, who studied a population in Columbia where elective herniorrhaphy was virtually unobtainable, found an annual rate of strangulation of 0.29% for inguinal hernias.
Management of Strangulation
The diagnosis of hernias is primarily based on clinical symptoms and signs, supplemented by imaging studies when necessary. Pain at the hernia site is a constant symptom. In cases of obstruction with intestinal strangulation, patients may present with colicky abdominal pain, distension, vomiting, and constipation. Physical examination may reveal signs of dehydration, with or without central nervous system depression, especially in elderly patients with uremia, along with abdominal signs of intestinal obstruction.
Femoral hernias can be easily missed, particularly in obese women, making a thorough physical examination essential for an accurate diagnosis. However, physical examination alone is often insufficient to confirm the presence of a strangulated femoral hernia versus lymphadenopathy or a lymph node abscess. In such cases, urgent radiographic studies, such as ultrasound or CT scan, may be necessary.
The choice of incision depends on the type of hernia if the diagnosis is clear. When there is doubt, a half Pfannenstiel incision, 2 cm above the pubic ramus extending laterally, provides adequate access to all types of femoral or inguinal hernias. The fundus of the hernia sac is exposed, and an incision is made to assess the viability of its contents. If nonviability is detected, the transverse incision should be converted into a laparotomy incision, followed by the release of the constricting hernia ring, reduction of the sac’s contents, resection, and reanastomosis. Precautions must be taken to avoid contamination of the general peritoneal cavity by gangrenous bowel or intestinal contents.
In most cases, once the constriction of the hernia ring is released, circulation to the intestine is restored, and viability returns. The intestine that initially appears dusky or non-peristaltic may regain color with a short period of warming with damp packs. If viability is doubtful, resection should be performed. Resection rates are highest for femoral or recurrent inguinal hernias and lowest for simple inguinal hernias. Other organs, such as the bladder or omentum, should be resected as needed.
After peritoneal lavage and formal closure of the laparotomy incision, specific repair of the hernia should be performed. Prosthetic mesh should not be used in a contaminated operative field due to the high risk of wound infection. Hernia repair should follow the general principles of elective hernia repair. It is important to remember that in this predominantly frail and elderly patient group with a high postoperative mortality risk, the primary objective of the operation is to stop the vicious cycle of strangulation, with hernia repair being a secondary objective.
Key Point
The risk of an acute groin hernia episode is of particular relevance, when discussing indication for operation of painless or minimally symptomatic hernias. A sensible approach in groin hernias would be, in accordance with the guidelines from the European Hernia Society to advise a male patient, that the risk of an acute operation, with an easily reducible (“disappears when lying down”) inguinal hernia with little or no symptoms, is low and that the indication for operation in this instance is not absolute, but also inform, that usually the hernia after some time will cause symptoms, eventually leading to an operation. In contrast, female patients with a groin hernia, due to the high frequency of femoral hernias and a relatively high risk of acute hernia episodes, should usually be recommended an operation.
Minimally Invasive Approach to Choledocholithiasis
Introduction
The incidence of choledocholithiasis in patients undergoing cholecystectomy is estimated to be 10 %. The presence of common bile duct stones is associated with several known complications including cholangitis, gallstone pancreatitis, obstructive jaundice, and hepatic abscess. Making the diagnosis early and prompt management is crucial. Traditionally, when choledocholithiasis is identified with intraoperative cholangiography during the cholecystectomy, it has been managed surgically by open choledochotomy and place- ment of a T-tube. This open surgical approach has a morbidity rate of 10–15 %, mortality rate of <1 %, with a <6 % incidence of retained stones. Patients who fail endoscopic retrieval of CBD stones, as well as cases in which an endoscopic approach is not appropriate, should be explored surgically.
Clinical Manifestation
Acute obstruction of the bile duct by a stone causes a rapid distension of the biliary tree and activation of local pain fibers. Pain is the most common presenting symptom for choledocholithiasis and is localized to either the right upper quadrant or to the epigastrium. The obstruction will also cause bile stasis which is a risk factor for bacterial over- growth. The bacteria may originate from the duodenum or the stone itself. The combination of biliary obstruction and colo- nization of the biliary tree will lead to the development of fevers, the second most common presenting symptom of cho- ledocholithiasis. Biliary obstruction, if unrelieved, will lead to jaundice. When these three symptoms (pain, fever, and jaundice) are found simultaneously, it is known as Charcot’s triad. This triad suggests the diagnosis of acute ascending cholangitis, a potentially life-threatening condition. If not treated promptly, this can lead to hypotension and decreased metal status, both signs of severe sepsis. When combined with Charcot’s triad, this constellation of symptoms is commonly referred to as Reynolds pentad.
Laparoscopic common bile duct exploration
Laparoscopic common bile duct exploration (LCBDE) allows for single stage treatment of gallstone disease, reducing overall hospital stay, improving safety and cost-effectiveness when compared to the two-stage approach of ERCP and laparoscopic cholecystectomy. Bile duct clearance can be confirmed by direct visualization with a choledochoscope. But, before the advent of choledochoscope, bile duct clearance was uncertain, and blind instrumentation of the duct resulted in accentuated edema and inflammation. Due to advancement in instruments, optical magnification, and direct visualization, laparoscopic exploration of the CBD results in fewer traumas to the bile duct. This has led to an increasing tendency to close the duct primarily, reducing the need for placement of T-tubes. Still, laparoscopic bile duct exploration is being done in only a few centers. Apart from the need for special instruments, there is also a significant learning curve to acquire expertise to be able to perform a laparoscopic bile duct surgery.
Morbidity and mortality rates of laparoscopic exploration are comparable to ERCP (2–17 and 1–5 %), and there is no clear difference in primary success rates between the two approaches. However, the endoscopic approach may be preferable for elderly and frail patients, who are at higher risk with surgery. Patients older than 70–80 years of age have a 4–10 % mortality rate with open duct exploration. It may be as high as 20 % in elderly patients undergoing urgent procedures. In comparison, advanced age and comor- bidities do not have a significant impact on overall complication rates for ERCP. A success rate of over 90 % has been reported with laparoscopic CBD exploration. Availability of surgical expertise and appropriate equipment affect the success rate of laparoscopic exploration, as does the size, number of the CBD stones, as well as biliary anatomy. Over the years, laparoscopic exploration has become efficient, safe, and cost effective. Complications include CBD laceration, stricture formation, bile leak, abscess, pancreatitis, and retained stones.
In cases of failure of laparoscopic CBD exploration, a guidewire or stent can be passed through the cystic duct, common bile duct, and through the ampulla into the duodenum followed by cholecystectomy. This makes the identification and cannulation of the ampulla easier during the post- operative ERCP. Laparoscopic common bile duct exploration is traditionally performed through a transcystic or transductal approach. The transcystic approach is appropriate under certain circumstances. These include a small stone (<10 mm) located in the CBD, presence of small common bile duct (<6 mm), or if there is poor access to the common duct. The transductal approach is preferable in cases of large stones, stones in proximal ducts (hepatic ducts), large occluding stones in a large duct, presence of multiple stones, or if the cystic duct is small (<4 mm) or tortuous. Contraindications for laparoscopic approach include lack of training, and severe inflammation in the porta hepatis making the exploration difficult and risky.
Key Points
With advancement in imaging technology, laparoscopic and endoscopic techniques, management of common bile duct stone has changed drasti- cally in recent years. This has made the treatment of this condition safe and more efficient. Many options are now available to manage this condition, and any particular modality for treatment should be chosen carefully based on the patient related factors, institutional protocol, available expertise, resources, and cost-effectiveness.
Classroom: M.I.A. of Choledocholithiasis
Management of Complicated Appendicitis: Open or Laparoscopic Surgery?
Patients with acute appendicitis can present at different stages of the disease process, ranging from mild mucosal inflammation to frank perforation with abscess formation. The reported overall incidence of acute appendicitis varies with age, gender, and geographical differences. Interestingly, while the incidence of non-perforated appendicitis in the United States decreased between 1970 and 2004, no significant decline in the rate of perforated appendicitis was observed despite the increasng use of computed tomography (CT) and fewer negative appendectomies.
Of 32,683 appendectomies sampled from the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) hospitals between 2005 and 2008, 5,405 patients (16.5%) had a preoperative diagnosis of acute appendicitis with peritonitis/abscess.
The definition of complicated appendicitis varies slightly in the literature. Clinicopathological diagnoses (gangrenous, perforated, appendiceal abscess/phlegmon) of acute appendicitis are commonly used for its definition. Classically, patients at the extremes of age are more likely to present with complicated appendicitis. Similarly, pre-morbid conditions including diabetes and type of medical insurance are significantly associated with the risk of perforation.
The importance of early appendectomy has also been emphasized to prevent perforation of the appendix and the sub- sequent negative impact on patient outcomes. However, more recent meta-analysis data supports the safety of a relatively short (12–24 h) delay before appendectomy, which was not significantly associated with increased rate of complicated appendicitis. Teixeira et al. also showed that the time to appendectomy was not a significant risk factor for perforated appendicitis but did result in a significantly increased risk of surgical site infection.
The outcome of patients with complicated appendicitis is significantly worse than patients with uncomplicated appendicitis. A population-based study from Sweden showed that, in a risk-adjusted model, patients with perforated appendicitis were 2.34 times more likely to die after appendectomy than non- perforated appendicitis patients. Because of its higher mortality and morbidity in patients with complicated appendicitis, the management of complicated appendicitis has evolved significantly over the last few decades.
Open or Laparoscopic Surgery
Since the first laparoscopic appendectomy was described by Semm in 1983, multiple studies have compared operative time, complication rates, length of hospital stay, hospital cost, and other outcomes between open and laparoscopic appendectomy for acute appendicitis. The most recent Cochrane review included 67 studies showing that laparoscopic appendectomy was associated with a lower incidence of wound infection, reduced postoperative pain, shorter postoperative length of hospital stay, and faster recovery to daily activity. In contrast, reduced risk of intra-abdominal abscesses and shorter operative time were found as the advantages of open appendectomy.
Due to increased surgeon experience in uncomplicated appendicitis, laparoscopic appendectomy is more frequently attempted even in complicated appendicitis cases as an alternative approach to open appendectomy. Although the general surgical steps for complicated appendicitis are similar to those for uncomplicated appendicitis, the laparoscopic procedure can be more technically demanding. Therefore, conversion from laparoscopic appendectomy to open appendectomy can be expected.
Despite these concerns, the laparoscopic approach in patients with com- plicated appendicitis has been proven to be safe and comparable to open appendectomy. Retrospective studies using a large database in the United States uniformly showed more favorable clinical outcomes (mortality, morbidity, length of hospital stay, readmission rate) and hospital costs in patients who underwent laparoscopic appendectomy when compared to open appendectomy. The real risk of developing an intra- abdominal abscess after laparoscopic appendectomy remains unclear. A meta-analysis by Markides et al. found no significant difference in the intra-abdominal abscess rate between laparoscopic and open appendectomy for complicated appendicitis, whereas Ingraham et al. showed a higher likelihood of developing an organ-space surgical site infection in patients undergoing laparoscopic appendectomy.
Non Operative Management of SPLENIC TRAUMA

The spleen, an important component of the reticuloendothelial system in normal adults, is a highly vascular solid organ that arises as a mass of differentiated mesenchymal tissue during early embryonic development. The normal adult spleen weighs between 75 and 100 g and receives an average blood flow of 300 mL/min. It functions as the primary filter of the reticuloendothelial system by sequestering and removing antigens, bacteria, and senescent or damaged cellular elements from the circulation. In addition, the spleen has an important role in humoral immunity because it produces immunoglobulin M and opsonins for the complement activation system.
The increased availability of high-resolution CT scan and advances in arterial angiography and embolization techniques have contributed to the success of nonoperative management of splenic injuries.
The hemodynamically stable patient with blunt splenic trauma can be adequately managed with bed rest, serial abdominal exams, and hemoglobin and hematocrit monitoring. This approach, in combination with occasional angiography, especially for grade III and IV injuries, confers a splenic salvage rate of up to 95%.
In the setting of expectant management, indications for angiography have been delineated by several studies and include the following CT scan features: contrast extravasation, the presence of a pseudoaneurysm, significant hemoperitoneum, high-grade injury, and evidence of a vascular injury. The goal of angiography is to localize bleeding and embolize the source with coils or a gelatin foam product. Embolization can occur either at the main splenic artery just distal to the dorsal pancreatic portion of the vessel—known as proximal embolization—or selectively at the distal branch of the injured vessel. The goal behind the former technique is to decrease the perfusion pressure to the spleen to encourage hemostasis. The disadvantage to this technique is global splenic ischemia, and many have questioned the spleen’s immunocompetence following proximal embolization.
Malhotra et al. examined the effects of angioembolization on splenic function by examining serum levels of a particular T-cell line. T-cell proportions between patients who had undergone splenic embolization with asplenic patients and healthy controls were similar suggesting some degree of splenic immunocompetency was maintained. A Norwegian study comparing blood samples from patients who had undergone angioembolization with healthy controls demonstrated that the study samples had similar levels of pneumococcal immunoglobulins and no Howell-Jolly bodies, suggesting normal splenic function. Although these preliminary studies remain encouraging, there is no definitive evidence that splenic immunocompetency is fully maintained following angio-embolization.
There is no question that advancements in interventional techniques have contributed to the successful nonoperative management of splenic injuries. This has certainly changed the strategy, but it has not completely replaced operative intervention. The challenge now remains predicting those patients who will ultimately require splenectomy.
Many groups have studied potential predictors of nonoperative failure. Earlier studies found that a higher injury grade, increased transfusion requirement, and hypotension on initial presentation consistently predicted failure of nonoperative management. More recent literature reflects the use of advanced imaging techniques for predicting which patients will ultimately require splenectomy. Haan looked at the overall outcomes of patients admitted with blunt splenic trauma and reported several radiographic findings that were prevalent among patients requiring splenectomy after angioembolization:
- contrast extravasation,
- pseudoaneurysm,
- significant hemoperitoneum,
- and arteriovenous fistula.
Among these characteristics, an arteriovenous fistula had the highest rate of nonoperative failure at 40%. Nonradiographic features associated with significant risk of nonoperative failure include age greater than 40, injury severity score of 25 or greater, or presence of large-volume hemoperitoneum.
Aside from radiographic findings, some groups have also examined the mechanism of injury and its association with nonoperative failure. Plurad et al. conducted a retrospective review over a 15-year period and found that patients who were victims of blunt assault were more likely to fail nonoperative management: 36% of these patients required splenectomy versus 11.5% of patients from all other mechanisms combined. These findings suggest that regardless of overall injury severity, individuals who sustain a direct transfer of injury to the left torso are more likely to require splenectomy.
Currently, the accepted standard of care for most splenic trauma is expectant management with close observation. Operative intervention is reserved for the hemodynamically labile patient who shows signs of active hemorrhage and who does not respond appropriately to fluid resuscitation. Although these clinical scenarios seem straightforward, it is often the condition of the patient who falls in between the two ends of the spectrum that can be the most challenging to manage. In the setting of advanced imaging techniques and interventional radiology, the trauma surgeon has more diagnostic information and more treatment options for the patient with splenic trauma.
Perioperative Medicine

Surgery and anesthesia profoundly alter the normal physiologic and metabolic states. Estimating the patient’s ability to respond to these stresses in the postoperative period is the task of the preoperative evaluation. Perioperative complications are often the result of failure, in the preoperative period, to identify underlying medical conditions, maximize the patient’s preoperative health, or accurately assess perioperative risk. Sophisticated laboratory studies and specialized testing are no substitute for a thoughtful and careful history and physical examination. Sophisticated technology has merit primarily in confirming clinical suspicion.
Classroom: Perioperative Medicine
How to Choose a Mesh in Hernia Repair

Since the introduction of polypropylene (PP) mesh for hernia repair, surgeons continue to discuss the use of mesh in a variety of settings for one of the most common operations performed by general surgeons—hernia repair. This discussion has involved raw materials, cost, and outcomes and for many years referred to only a few products, as manufacturing was limited. Nowadays, with multiple permanent, absorbable, biologic, and hybrid products on the market, the choice of mesh for a hernia repair can be daunting. Increasing clinical complexity further emphasizes the need for individualizing care, but more frequently, hospital supply chain personnel institute product procurement procedures for cost control, limiting mesh choice for surgeons. This can force surgeons into a “one-size-fits-all” practice regarding mesh choice, which may not be ideal for some patients. Conversely, current literature lacks definitive evidence supporting the use of one mesh over another, a fact that has not escaped the radar screen of the hospital supply chain and mesh industry, both of which attempt to limit vendor and mesh choice for financial gain. It is unlikely that this type of “proof” will ever come to fruition. This leaves us with choosing a mesh based on an algorithm that is centered on the patient and the patient’s unique clinical scenario.
Article: Mesh in Hernia Repair
A “PROFISSÃO” CIRÚRGICA
 “A arte de curar vem do coração e da mente mais do que das mãos.” – Hipócrates
“A arte de curar vem do coração e da mente mais do que das mãos.” – Hipócrates
Na complexa tapeçaria da sociedade moderna, as profissões desempenham papéis fundamentais na organização dos serviços necessários ao bem-estar coletivo. Definida pelo American College of Surgeons, uma profissão é um campo onde a maestria de um corpo complexo de conhecimento e habilidades é essencial. É uma vocação em que o conhecimento científico ou a prática de uma arte, fundamentada nesse conhecimento, é empregada em benefício dos outros. O compromisso com a competência, a integridade e a moralidade forma a base de um contrato social entre a profissão e a sociedade, que concede à profissão um monopólio sobre o uso de seu conhecimento, considerável autonomia na prática e o privilégio da auto-regulação. Em troca, a profissão deve prestar contas a quem serve e à sociedade como um todo.
Os Elementos Essenciais da Profissão
No cerne de toda profissão estão quatro elementos fundamentais:
- Monopólio do Conhecimento Especializado: Profissionais detêm o direito exclusivo de utilizar conhecimentos e habilidades especializados, o que lhes confere uma posição única na sociedade.
- Autonomia e Auto-Regulação: Em troca deste monopólio, profissionais desfrutam de uma relativa autonomia na prática e são responsáveis pela sua própria regulação.
- Serviço Altruísta: A profissão deve servir tanto indivíduos quanto a sociedade de forma altruísta, colocando o bem-estar do paciente acima de outros interesses.
- Responsabilidade pela Manutenção e Expansão do Conhecimento: Profissionais são responsáveis por atualizar e expandir continuamente seu conhecimento e habilidades.
O Que é Profissionalismo?
Profissionalismo descreve as qualidades cognitivas, morais e colegiais de um profissional. É o conjunto de razões pelas quais um pai se orgulha de dizer que seu filho é um médico e cirurgião. Profissionalismo é mais do que apenas conhecimento técnico; é uma combinação de ética, respeito e dedicação ao ofício e ao paciente.
Por Que Precisamos de um Código de Conduta Profissional?
A confiança é o alicerce da prática cirúrgica. O Código de Conduta Profissional esclarece a relação entre a profissão cirúrgica e a sociedade que serve, frequentemente referido como contrato social. Para os pacientes, o código cristaliza o compromisso da comunidade cirúrgica em relação aos indivíduos e suas comunidades. A confiança é construída, tijolo por tijolo.
O Código de Conduta Profissional
O Código de Conduta Profissional aplica os princípios gerais do profissionalismo à prática cirúrgica e serve como a fundação sobre a qual os privilégios profissionais e a confiança dos pacientes e do público são conquistados. Durante o cuidado pré-operatório, intraoperatório e pós-operatório, os cirurgiões têm a responsabilidade de:
- Advogar Eficazmente pelos interesses dos pacientes.
- Divulgar Opções Terapêuticas incluindo seus riscos e benefícios.
- Divulgar e Resolver Conflitos de Interesse que possam influenciar as decisões de cuidado.
- Ser Sensível e Respeitoso com os pacientes, compreendendo sua vulnerabilidade durante o período perioperatório.
- Divulgar Completamente Eventos Adversos e Erros Médicos.
- Reconhecer Necessidades Psicológicas, Sociais, Culturais e Espirituais dos pacientes.
- Incorporar Cuidados Especiais para Pacientes Terminais.
- Reconhecer e Apoiar as Necessidades das Famílias dos Pacientes.
- Respeitar o Conhecimento, Dignidade e Perspectiva de outros profissionais de saúde.
A Necessidade do Código de Profissionalismo para Cirurgiões
Procedimentos cirúrgicos são experiências extremas que impactam os pacientes fisiológica, psicológica e socialmente. Quando os pacientes se submetem a uma experiência cirúrgica, devem confiar que o cirurgião colocará seu bem-estar acima de todas as outras considerações. O código escrito ajuda a reforçar esses valores, garantindo que a confiança e o compromisso sejam mantidos.
Princípios Fundamentais do Código de Conduta Profissional
- Primazia do Bem-Estar do Paciente: Os interesses do paciente sempre devem vir em primeiro lugar. O altruísmo é central para esse conceito, e é o altruísmo do cirurgião que fomenta a confiança na relação médico-paciente.
- Autonomia do Paciente: Pacientes devem entender e tomar suas próprias decisões informadas sobre o tratamento. Os médicos devem ser honestos para que os pacientes façam escolhas educadas, garantindo que essas decisões estejam alinhadas com práticas éticas.
- Justiça Social: Como médicos, devemos advogar pelos pacientes individuais enquanto promovemos a saúde do sistema de saúde como um todo. Precisamos equilibrar as necessidades dos pacientes (autonomia) sem desviar recursos escassos que beneficiariam a sociedade (justiça social).
“Não há maior coisa a ser conquistada do que a confiança dos pacientes e da sociedade, pois ela é a base sobre a qual construímos nossas práticas e nossa profissão.” – William Osler
FERIDA PÓS-OPERATÓRIA

A avaliação e os cuidados de feridas pós-operatórias deve ser do domínio de todos os profissionais que atuam na clínica cirúrgica. O conhecimento a cerca dos processos relacionados a cicatrização tecidual é importante tanto nos cuidados como na prevenção de complicações, tais como: infecções e deiscência. Como tal, todos os profissionais médicos, sendo eles cirurgiões ou de outras especialidades, que participam do manejo clínico dos pacientes no período perioperatório devem apreciar a fisiologia da cicatrização de feridas e os princípios de tratamento de feridas pós-operatório. O objetivo deste artigo é atualizar os profissionais médicos de outras especialidades sobre os aspectos importantes do tratamento de feridas pós-operatório através de uma revisão da fisiologia da cicatrização de feridas, os métodos de limpeza e curativo, bem como um guia sobre complicações de feridas pós-operatórias mais prevalentes e como devem ser manejados nesta situação.
Causas de conversão da VIDEOCOLECISTECTOMIA

Atualmente, a colecistectomia laparoscópica é a abordagem preferida para o tratamento da litíase biliar, representando cerca de 90% dos procedimentos realizados, uma marca alcançada nos Estados Unidos em 1992. A popularidade dessa técnica se deve a suas vantagens evidentes: menos dor no pós-operatório, recuperação mais rápida, redução dos dias de trabalho perdidos e menor tempo de hospitalização. Apesar de ser considerada o padrão-ouro na cirurgia biliar, a colecistectomia laparoscópica não está isenta de desafios. Entre 2% e 15% dos casos podem exigir a conversão para cirurgia convencional. Os motivos mais comuns para essa conversão incluem dificuldades na identificação da anatomia, suspeita de lesão da árvore biliar e controle de sangramentos. Identificar os fatores que contribuem para uma maior taxa de conversão é essencial para a equipe cirúrgica. Isso não apenas permite uma avaliação mais precisa da complexidade do procedimento, mas também ajuda na preparação do paciente para possíveis riscos e na mobilização de cirurgiões mais experientes quando necessário. Em um cenário onde a precisão e a segurança são cruciais, a compreensão dos desafios e a preparação adequada podem fazer toda a diferença no resultado da cirurgia.
Relacionados ao Paciente: 1. Obesidade (IMC > 35), 2. Sexo Masculino, 3. Idade > 65 anos, 4. Diabetes Mellitus e 5. ASA > 2.
Relacionadas a Doença: 1. Colecistite Aguda, 2. Líquido Pericolecístico, 3. Pós – CPRE, 4. Síndrome de Mirizzi e 5. Edema da parede da vesícula > 5 mm.
Relacionadas a Cirurgia: 1. Hemorragia, 2. Aderências firmes, 3. Anatomia obscura, 4. Fístulas internas e 5. Cirurgia abdominal prévia.
Review of POSTGASTRECTOMY SYNDROMES

The first postgastrectomy syndrome was noted not long after the first gastrectomy was performed: Billroth reported a case of epigastric pain associated with bilious vomiting as a sequel of gastric surgery in 1885. Several classic treatises exist on the subject; we cannot improve on them and merely provide a few references for the interested reader. Surgical procedures on the stomach, performed for reasons such as peptic ulcer disease, cancer, obesity, or gastroesophageal reflux disease, can result in various post-gastrectomy syndromes. These syndromes include chronic symptoms that range from mild discomfort to life-altering conditions. This guide covers the most common syndromes and their characteristics.
GASTRECTOMY VIDEO SURGERY
Dumping Syndrome
Dumping Syndrome is characterized by gastrointestinal and vasomotor symptoms that occur after food intake due to rapid gastric emptying. This syndrome can occur after surgeries that alter the regulation of gastric emptying or gastric compliance, such as gastrectomy, proximal vagotomy, sleeve gastrectomy, fundoplication, pyloroplasty, and gastrojejunostomy (GJ). Depending on the speed of emptying and the osmolarity of gastric contents, symptoms can vary.
- Early Dumping: Occurs within 30 minutes after food intake and is characterized by palpitations, tachycardia, fatigue, a need to lie down after meals, flushing or pallor, sweating, dizziness, hypotension, headache, and possibly syncope. Abdominal symptoms include early satiety, epigastric fullness, abdominal pain, bloating, hypermotility, and splenic blood pooling.
- Late Dumping: Appears 1 to 3 hours after eating, due to reactive hypoglycemia caused by an initially high glucose load leading to an inappropriately high insulin response. Symptoms include sweating, faintness, difficulty concentrating, and altered levels of consciousness.
Diagnosis is confirmed through an oral glucose tolerance test or a gastric emptying scintigraphy study.
Post-Vagotomy Diarrhea
Post-vagotomy diarrhea is a common complication after vagotomy, characterized by frequent episodes of watery diarrhea. It can be attributed to changes in intestinal motility and bile secretion.
Gastric Stasis
Gastric stasis or delayed gastric emptying can occur due to disruption of normal gastric motility. Symptoms include nausea, vomiting, and a feeling of fullness. Diagnosis is confirmed through gastric emptying studies.
Bile Reflux Gastritis
Bile reflux gastritis is caused by the reflux of bile into the stomach, resulting in epigastric pain and bilious vomiting. Diagnosis can be confirmed through upper endoscopy and gastric pH monitoring.
Afferent and Efferent Loop Syndromes
Afferent loop syndrome occurs after Billroth II reconstruction and is characterized by abdominal pain, bilious vomiting, and distention. Efferent loop syndrome occurs when there is an obstruction of the efferent loop, leading to similar symptoms.
Roux Syndrome
Roux syndrome is a complication of Roux-en-Y procedures, characterized by postprandial abdominal pain and vomiting. Diagnosis is made through a contrast gastrointestinal transit study.
Therapeutic Approach
Management of post-gastrectomy syndromes includes dietary modifications, such as eating small frequent meals, separating liquids and solids, increasing protein and fat intake, and reducing simple sugars. In some cases, additional pharmacological or surgical interventions may be necessary. Understanding these syndromes and their therapeutic approaches is crucial to providing effective care and improving the quality of life for post-gastrectomy patients.
This article focuses on the small proportion of patients with severe, debilitating symptoms; these symptoms can challenge the acumen of the surgeon who is providing the patient’s long-term follow-up and care.
POSTGASTRECTOMY_SYNDROMES_REVIEW_ARTICLE
Abdominal Hernia Surgical EMERGENCIES
A hernia is a weakness or disruption of the fibromuscular tissues through which an internal organ (or part of the organ) protrudes or slides through. Collectively, inguinal and femoral hernias are often lumped together into groin hernias. Surgery remains the only effective treatment, but the optimal timing and method of repair remain controversial. Although strangulation rates of 3% at 3 months have been reported by some investigators, the largest prospective randomized trial of (watchful waiting) men with minimally symptomatic inguinal hernias showed that watchful waiting is safe. Frequency of strangulation was only 2.4% in patients followed up for as long as 11.5 years. Long-term follow-up shows that more than two-thirds of men using a strategy of watchful waiting cross over to surgical repair, with pain being the most common reasons. This risk of crossover is higher in patients older than 65 years. Once an inguinal hernia becomes symptomatic, surgical repair is clearly indicated. Femoral hernias are more likely to present with strangulation and require emergency surgery and are thus repaired even when asymptomatic. Because this article focuses on incarcerated hernias, nonoperative options are not discussed.
PRINCIPLES OF OSTOMY MANAGEMENT

The creation of a stoma is a technical exercise. Like most undertakings, if done correctly, the stoma will usually function well with minimal complications for the remainder of the ostomate’s life. Conversely, if created poorly, stoma complications are common and can lead to years of misery. Intestinal stomas are in fact enterocutaneous anastomoses and all the principles that apply to creation of any anastomosis (i.e., using healthy intestine, avoiding ischemia and undue tension) are important in stoma creation.
PREVENTION COMPLICATIONS OF COLON SURGERY
COMPLICATIONS OF COLON SURGERY_REVIEW ARTICLE

Colon surgery represents a high number of patients treated at a department of gastrointestinal surgery and is not limited to colon cancer. It includes other non-neoplastic pathologies such as inflammatory bowel disease, diverticular disease or colonic volvulus. As with any major procedure, colon surgery patients may present serious or even fatal complications. The incidence of postoperative complications from colon surgery has been estimated at between 10% and 30% according to selected series. Preventive measures against surgical complications include selection of an appropriate procedure for the patient as well as good preoperative care, appropriate surgical technique and good postoperative management. When diagnosis has been established, risks for patient should be assessed according to patient’s health conditions and type of surgery accomplished. When the patient meets the surgical requirements, an appropriate course of preoperative care should be carried out including colon wash antibiotics and antithrombotic prophylaxis. Postoperative period will be equivalent to any major abdominal surgery. Typically, it was considered appropriate to wait a few days before initiating feeding in order to protect anastomosis; however, some authors agree that an early oral diet hours after intervention is not associated with a higher risk of anastomotic dehiscence and other complications.
Laparoscopic Colorectal Surgery
With the introduction of laparoscopic colectomy nearly 20 years ago, a relatively slow adoption of laparoscopic colorectal surgery into surgical practice has taken place. It is estimated that 10% to 25% of all colorectal resections are performed utilizing laparoscopy. The persistent steep learning curve, the lack of high-volume colorectal surgery by general surgeons (who perform the bulk of colonic resection in the United States), and the modest advantages reported are but a few of the reasons that the percentage of laparoscopic colorectal procedures has not dramatically risen. With the publication of several large, prospective randomized trials for colon cancer, along with the interest in single-port surgery and natural orifice surgery, there appears to be a renewed interest in minimally invasive procedures for the colon and rectum. This chapter will provide an overview of these issues and offer a current assessment of the common diseases to which minimally invasive techniques have been applied.
Learning Curve
Numerous previous studies have evaluated the learning curve involved in laparoscopic colectomy. It is estimated by conventional laparoscopic techniques that the learning curve for laparoscopic colectomy is at least 20 cases but more likely 50 cases. The need to work in multiple quadrants of the abdomen, the need for a skilled laparoscopic assistant, and the lack of yearly volume has kept the learning curve relatively steep. The surgeon may also need to work in reverse angles to the camera. All of these combined add to the complexity of the procedure and result in the need to perform a number of cases before the surgeon and surgical team become proficient. More recent publications have suggested the learning curve is more than 20 cases. In a prospective randomized study of colorectal cancer in the United Kingdom, the CLASICC trial, surgeons had to perform at least 20 laparoscopic resections before they were allowed to enter the study. The study began in July 1996 and was completed in July 2002. Despite the surgeons’ prior experience, the rate of conversion dropped from 38% to 16% over the course of the study, suggesting that a minimum of 20 cases may not be enough to overcome the learning curve. In the COLOR trial from Europe, another prospective randomized study for colon cancer that required a prerequisite experience in laparoscopic colon resection before surgeons could enter patients in the study, surgeon and hospital volume were directly related to a number of operative and postoperative outcomes. The median operative time for high-volume hospitals (>10 cases/year) was 188 minutes, compared to 241 minutes for low-volume hospitals (<5 cases/year); likewise, conversion rates were 9% versus 24% for the two groups. High-volume groups also had more lymph nodes in the resected specimens, fewer complications, and shortened hospital stays. These two relatively recent multicenter studies suggest that the learning curve is clearly greater than 20 cases and that surgeons need to perform a minimum yearly number of procedures to maintain their skills.
Outcomes
There may not be another area in recent surgical history that has been more heavily scrutinized than laparoscopic colorectal surgery. The plethora of accumulated data allows a careful assessment of all outcome measures for nearly every colorectal disease and procedure. In comparison to conventional colorectal surgery, the benefits of laparoscopy for colorectal procedures compared to open techniques include a reduction in postoperative ileus, postoperative pain, and a concomitant reduction in the need for analgesics; an earlier tolerance of diet; a shortened hospital stay; a quicker resumption of normal activities; improved cosmesis; and possibly preservation of immune function. This is offset by a prolongation in operative time, the cost of laparoscopic equipment, and the learning curve for these technically challenging procedures. When reporting the outcomes of laparoscopic colectomy, a natural selection bias applies when comparing conventional and laparoscopic cases. The most complex cases are generally not suitable for a laparoscopic approach and therefore are performed via an open approach. Also, in many series the results of the successfully completed laparoscopic cases are compared to conventional cases, and the cases converted from a laparoscopic to a conventional procedure may be analyzed separately. Few studies, with the exception of the larger prospective randomized studies, leave the converted cases in the laparoscopic group as part of the “intention to treat” laparoscopic group. This clearly introduces selection bias.
Although the results of prospective randomized trials are available for almost every disease process requiring colorectal resection, the majority of studies of laparoscopic colectomy are retrospective case-control series or noncomparative reports. The conclusions regarding patient outcomes must therefore come from the repetitiveness of the results rather than the superiority of the study design. For any one study, the evidence may be weak; but collectively, because of the reproducibility of results by a large number of institutions, even with different operative techniques and postoperative management parameters, the preponderance of evidence favors a minimally invasive approach with respect to postoperative outcomes.
Operative Time
Nearly all the comparative studies provide information regarding operative times. The definition of the operative time may vary with each series, and there may be different groups of surgeons performing the laparoscopic and conventional procedures. With the exception of a few reports, nearly all studies demonstrated a prolonged operative time associated with laparoscopic procedures. In prospective randomized trials, the procedure was roughly 40 to 60 minutes longer in the laparoscopic groups. As the surgeon and team gain experience with laparoscopic colectomy, the operating times do reliably fall, but rarely do they return to the comparable time for a conventional approach.
Return of Bowel Activity and Resumption of Diet
Reduction in postoperative ileus is one of the proposed major advantages of minimally invasive surgery. Nearly all of the retrospective and prospective studies comparing open and laparoscopic colectomy have shown a statistically significant reduction in the time to passage of flatus and stool. Most series demonstrate a 1- to 2-day advantage for the laparoscopic group. Whether the reduction of ileus relates to less bowel manipulation or less intestinal exposure to air during minimally invasive surgery remains unknown. With the reduction in postoperative ileus, the tolerance by the patient of both liquids and solid foods is quicker following laparoscopic resection. The time to resumption of diet varies from 2 days to 7 days, but in the majority of comparative studies, this is still 1 to 2 days sooner than in patients undergoing conventional surgery. Again, the physician and patient were not blinded in nearly all studies, which may have altered patient expectations. However, the overwhelming reproducible data reported in both retrospective and prospective studies of laparoscopic procedures does likely favor a reduction of postoperative ileus and tolerance of liquid and solid diets.
Postoperative Pain
To measure postoperative pain, a variety of assessments have been performed to demonstrate a significant reduction in pain following minimally invasive surgery; some studies utilize an analog pain scale, and others measure narcotic requirements. Physician bias and psychologic conditioning of patients may interfere with the evaluation of postoperative pain. There are also cultural variations in the response to pain. Three of the early prospective randomized trials have evaluated pain postoperatively, and all three have found a reduction in narcotic requirements in patients undergoing laparoscopic colectomy. In the COST study, the need both for intravenous and oral analgesics was less in patients undergoing successfully completed laparoscopic resections. Numerous other nonrandomized studies have shown a reduction in postoperative pain and narcotic usage.
Length of Stay
The quicker resolution of ileus, earlier resumption of diet, and reduced postoperative pain has resulted in a shortened length of stay for patients after laparoscopic resection when compared to traditional procedures. Recovery after conventional surgery has also been shortened, but in the absence of minimally invasive techniques, it would seem unlikely that the length of stay could be further reduced. In nearly all comparative studies, the length of hospitalization was 1 to 6 days less for the laparoscopic group. Although psychological conditioning of the patient cannot be helped and likely has a desirable effect, the benefits of minimally invasive procedures on the overall length of stay cannot be discounted. The benefit, however, is more likely a 1 to 2 day advantage only. The more recent introduction of clinical pathways, both in conventional and laparoscopic surgery, has also narrowed the gap but appears to be more reliable in patients undergoing a minimally invasive approach.
Hospital Costs
One of the disadvantages of laparoscopy is the higher cost related to longer operative times and increased expenditures in disposable equipment. Whether the total cost of the hospitalization (operative and hospital costs) is higher following laparoscopic colectomy is debatable. A case-control study from the Mayo Clinic looked at total costs following laparoscopic and open ileocolic resection for Crohn’s disease (CD). In this study, 66 patients underwent laparoscopic or conventional ileocolic resection and were well matched. Patients in the laparoscopic group had less postoperative pain, tolerated a regular diet 1 to 2 days sooner, and had a shorter length of stay (4 vs. 7 days). In the cost analysis, despite higher operative costs, the overall mean cost was $3273 less in the laparoscopic group. The procedures were performed by different groups of surgeons at the institution, and although the surgeons may have introduced biases, this study was undertaken during the current era of cost containment, in which all physicians are encouraged to reduce hospital stays. The results are similar for elective sigmoid diverticular resection with a mean cost savings of $700 to $800. Clearly, if operative times and equipment expenditure are minimized, the overall cost of a laparoscopic resection should not exceed a conventional approach.
Retromuscular Prefascial Mesh Hernia Repair (Jean RIVES – René STOPPA)
An incisional hernia is usually defined as a chronic postoperative defect of the abdominal wall through which intra-abdominal viscera protrude. Progress in surgical techniques, even with laparoscopic surgery, has not led to the elimination of incisional hernias. On the contrary, the incidence of this complication seems to be increasing as more major and lengthy operations are being performed, especially in elderly patients with concomitant organic disease. The incidence of this condition has been reported to be as high as 11% of all laparotomies. Surgical repair is difficult in the patient with a large abdominal wall defect, especially if the herniated viscera has “lost its right of domain” in the abdominal cavity. It must be remembered that surgical repair of an incisional hernia is not the same thing as closure of a laparotomy. Weakening of the abdominal wall and the consequences of decreased abdominal pressure on diaphragmatic mobility and respiratory function must also be considered. Placement of a prosthetic mesh is essential because without mesh, the recurrence rate is prohibitive, varying from 30% to 60%. The which is the subject of this article, was popularized by Jean Rives and has been used in our department since 1966.
RIVES_STOPPA_HERNIA_REPAIR
TRATAMENTO DA TROMBOSE HEMORROIDÁRIA
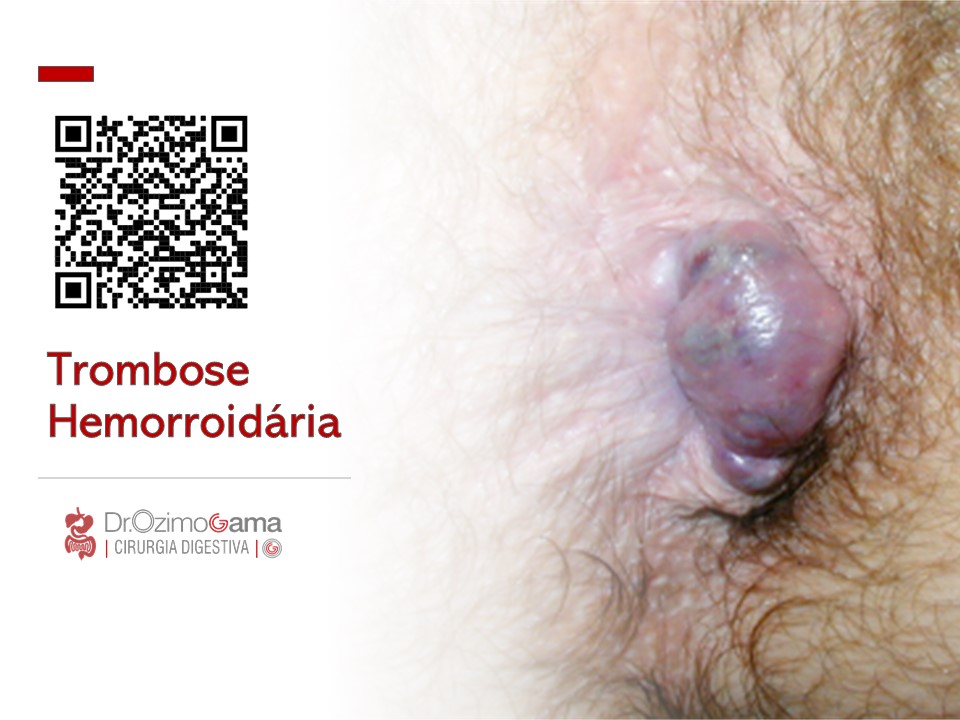
A trombose hemorroidária é uma condição dolorosa e desconfortável que ocorre quando um coágulo de sangue se forma dentro de uma hemorróida. Essa condição é mais comum em gestantes, pessoas com constipação intestinal crônica, e em situações que aumentam a pressão intra-abdominal, como exercícios físicos intensos e levantamento de peso. Neste artigo, exploraremos as causas, sintomas e opções de tratamento para a trombose hemorroidária, ajudando você a entender melhor essa condição e a buscar a melhor abordagem para o seu caso.
Causas da Trombose Hemorroidária
A trombose hemorroidária geralmente está relacionada a fatores de estilo de vida e hábitos pessoais. Os principais gatilhos incluem:
- Obstipação (prisão de ventre): O esforço excessivo para evacuar aumenta a pressão nas veias do ânus, e as fezes endurecidas podem causar traumatismo no tecido anal.
- Gravidez: A pressão adicional no abdômen durante a gravidez pode contribuir para o desenvolvimento da trombose.
- Esforços prolongados e levantamento de peso: Atividades que aumentam a pressão intra-abdominal são fatores de risco.
- Higiene inadequada: A falta de cuidados apropriados na região anal pode exacerbar a condição.
- Fatores adicionais: Permanecer sentado por longos períodos, consumo excessivo de alimentos picantes e bebidas alcoólicas, e prática de sexo anal.
Sintomas da Trombose Hemorroidária
Os principais sinais de trombose hemorroidária incluem:
- Dor intensa na região anal: A dor é geralmente súbita e pode ser bastante severa.
- Sangramento: Frequentemente observado durante a evacuação.
- Inchaço e aumento de volume: Um nódulo na região anal pode se tornar arroxeado ou preto, indicando a presença de um trombo.
Tratamentos Indicados
O tratamento da trombose hemorroidária varia conforme a gravidade da condição. Entre as abordagens recomendadas estão:
- Uso de analgésicos e pomadas anestésicas: Para alívio da dor e desconforto.
- Banhos de assento: Utilizar água morna para aliviar os sintomas e reduzir o inchaço.
- Correção dos hábitos alimentares: Aumentar a ingestão de fibras e líquidos para prevenir a obstipação.
- Tratamento cirúrgico: Em casos mais graves, pode ser necessário realizar uma cirurgia para remover a hemorróida e o trombo.
Se você apresenta sintomas semelhantes, é crucial consultar um médico para uma avaliação adequada e receber o tratamento adequado.
Gostou ❔ Nos deixe um comentário ✍️, ou mande sua dúvida pelo 💬 Chat On-line.
Lembre-se: A informação aqui fornecida é para orientação geral. Sempre consulte seu médico para aconselhamento específico sobre sua situação.
Não Deixe Sua Saúde Esperar! Entre em Contato Conosco Agora!
☎️ CHAT ONLINE 📞
Entre em contato conosco através da nossa central de atendimento dedicada. Nossos especialistas estão prontos para fornecer orientações personalizadas e responder a todas as suas perguntas. Juntos, podemos trabalhar para garantir o melhor para a sua saúde.
🩺(98)991304454 & (98)988664454📲
ATENDIMENTO REMOTO (TELEMEDICINA)
🤳🏻 Agende com os nossos #especialistas pelo WhatsApp ou Telefone
📲 👩🏽⚕️ Clínico: (98) 9 9130 4454
📲 👨🏻⚕️ Cirúrgico: (98) 9 8866 4454
#TromboseHemorroidária #SaúdeDigestiva #TratamentoHemorroidário #BemEstar #CuidadosComASaúde
VEJA AQUI ONDE FAZER UMA AVALIAÇÃO ESPECIALIZADA E RECEBER UMA SEGUNDA OPINIÃO
BILIARY-ENTERIC ANASTOMOSIS
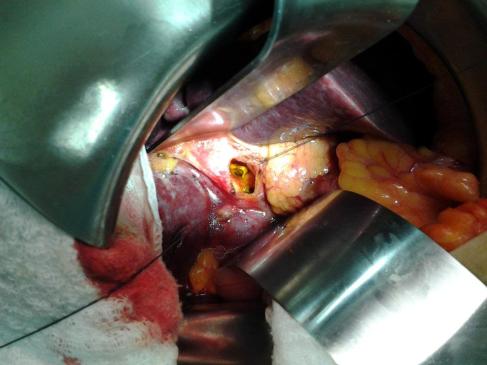 The operative conduct of the biliary-enteric anastomosis centers around three technical steps: 1) identification of healthy bile duct mucosa proximal to the site of obstruction; 2) preparation of a segment of alimentary tract, most often a Roux-en-Y jejunal limb; and 3) construction of a direct mucosa-to-mucosa anastomosis between these two. Selection of the proper anastomosis is dictated by the indication for biliary decompression and the anatomic location of the biliary obstruction. A right subcostal incision with or without an upper midline extension or left subcostal extension provides adequate exposure for construction of the biliary-enteric anastomosis. Use of retractors capable of upward elevation and cephalad retraction of the costal edges are quite valuable for optimizing visual exposure of the relevant hilar anatomy.
The operative conduct of the biliary-enteric anastomosis centers around three technical steps: 1) identification of healthy bile duct mucosa proximal to the site of obstruction; 2) preparation of a segment of alimentary tract, most often a Roux-en-Y jejunal limb; and 3) construction of a direct mucosa-to-mucosa anastomosis between these two. Selection of the proper anastomosis is dictated by the indication for biliary decompression and the anatomic location of the biliary obstruction. A right subcostal incision with or without an upper midline extension or left subcostal extension provides adequate exposure for construction of the biliary-enteric anastomosis. Use of retractors capable of upward elevation and cephalad retraction of the costal edges are quite valuable for optimizing visual exposure of the relevant hilar anatomy.

Division of the ligamentum teres and mobilization of the falciform ligament off the anterior surface of the liver also facilitate operative exposure; anterocephalad retraction of the ligamentum teres and division of the bridge of tissue overlying the umbilical fissure are critical for optimal visualization of the vascular inflow and biliary drainage of segments II, III, and IV. Cholecystectomy also exposes the cystic plate, which runs in continuity with the hilar plate. Lowering of the hilar plate permits exposure of the left hepatic duct as it courses along the base of segment IVb. In cases of unilateral hepatic atrophy as a result of long-standing biliary obstruction or preoperative portal vein embolization, it is critical to understand that the normal anatomic relationships of the portal structures are altered. In the more common circumstance of right-sided atrophy, the portal and hilar structures are rotated posteriorly and to the right; as a result, the portal vein, which is typically most posterior, is often encountered first; meticulous dissection is necessary to identify the common bile duct and hepatic duct deep within the porta hepatis.
BILIARY-ENTERIC ANASTOMOSIS_THECNICAL ASPECTS
Telas de Baixa Gramatura: PHS e UHS
EVOLUÇÃO HISTÓRICA DA HERNIORRAFIA INGUINAL
As hérnias não diminuem com o tempo, muito pelo contrário – o estado de saúde do paciente só tende a piorar, aumentando o risco cirúrgico – e não existe medicação para tratá-las. Os primeiros registros do tratamento cirúrgico das hérnias abdominais datam de 1500 a.C, mencionadas no Papiro de Ebers. Em 100 d.C. Celso, realizando os primórdios da cirurgia convencional, extirpava o saco herniário e deixava intactos o cordão e o testículo. Já em 700 d.C. , numa conduta mais agressiva, Pablo de Egina, sacrificava o testículo. Utilizando o princípio da cauterização dos tecidos pelo calor, Albucasis em 1000 d.C., expunha o saco herniário e o cauterizava. Com o advento da técnica asséptica registra-se, em 1869, a realização por Lister da primeira hérnia estrangulada em caráter de urgência com princípios antissépticos. O primeiro esboço de padronização da técnica operatória das hérnias abdominais ocorre a partir da publicação dos estudos de Edoardo Bassini, em 1887, primeiro estudo de reparo de hérnias com suturas. A técnica se propaga pelo mundo mas a deficiente comunicação e as múltiplas modificações levam a resultados negativos. Já em 1940, Earle Shouldice, usando quatro planos de reforço, revoluciona a técnica com tensão, principalmente após 1950 com um melhor entendimento da anatomia, porém observam-se ainda recorrências altas, acima de 10%. Em 1967, René Stoppa e Jean Rives, utilizam a técnica pré-peritoneal com a introdução de um novo conceito: a prótese. Uma tela gigante recobrindo todos os possíveis orifícios herniários, obtendo excelente taxa de recidiva, às custas da necessidade de incisão mediana e descolamento amplo pré-peritoneal.
PADRÃO OURO
Somente em 1984 Irving Lichtenstein, por via anterior, utiliza uma tela de polipropileno. Esta técnica foi considerada uma grande evolução e passou, nos anos 90, a ser considerado o Padrão Ouro nos reparos das hérnias inguinais possibilitando, pela primeira vez, o reparo ambulatorial com anestesia local e sedação. Com o inicio da cirurgia videolaparoscópica no início dos anos 90, desenvolveram-se diversas técnicas utilizando-se a tela pela técnica laparoscópica. Devido aos altos custos e complexidade do procedimento cirúrgico e anestésico em comparação à técnica por via aberta, estas não se estabeleceram como melhor opção. Finalmente, em 1997, Arthur Gilbert, inicia o sistema PHS (“Prolene Hernia System”) que reúne as vantagens quanto a recidiva do reparo pré-peritoneal, a simplicidade de fixação da técnica de plug e eficácia da técnica de tela plana através de um reparo tridimensional com uma tela dupla colocada pela via anterior. Dois anos mais tarde, ocorre a publicação dos primeiros resultados do PHS – `Hernia Institute of Florida – USA, firmando o aspecto inovador e a eficácia da tela dupla, otimizando o conforto e segurança do paciente e proporcionando retorno mais precoce às atividades laborativas e utilizando facilmente anestesia local e sedação.
Veja AQUI COMO ESCOLHER A PRÓTESE para seu paciente com Hérnia Abdominal.
Lesão de Vias Biliares na Colecistectomia: Prevenção e Tratamento
A via laparoscópica tem sido reconhecida como padrão de excelência para a colecistectomias. Phillipe Mouret foi quem primeiro a realizou em 1987, mas outros procedimentos já haviam sido realizados por laparoscopia e foram descritos por ginecologistas. Desenvolvida no final da década de 80 e início dos anos 90, a videolaparoscopia mudou os conceitos de acesso cirúrgico e campo operatório, introduzindo a concepção de “cirurgia minimamente invasiva”.A colecistectomia é um dos procedimentos cirúrgicos mais realizados no mundo. Com o advento da videolaparoscopia, tornou-se uma cirurgia menos traumática, mais estética, com períodos mais curtos de internação. Em contrapartida, observou-se o aumento da incidência de lesões de via biliar extra-hepática quando comparado ao procedimento aberto, fato preocupante devido à morbidade elevada desse tipo de lesão, cuja mortalidade não é desprezível.
Apendicectomia Convencional X Laparoscópica : Existe Vantagem?

A apendicite aguda é uma doença freqüente que acomete em sua maioria homens com uma idade média ao redor de 20 anos. O seu tratamento é cirúrgico e está bem estabelecido em sua abordagem convencional. O surgimento e o desenvolvimento da videolaparoscopia abriu uma nova opção para a abordagem cirúrgica dessa patologia, permitindo uma abordagem minimamente invasiva com todas as vantagens dessa técnica.
A primeira apendicectomia videolaparoscópica foi realizada há pouco mais de 20 anos. Nessas duas décadas muito se discutiu, e ainda discute-se, a respeito deste procedimento. Mesmo encontrando com freqüência vários estudos bem realizados na literatura, ainda não há um consenso a respeito das indicações precisas para a realização da apendicectomia laparoscópica; ou mesmo sobre qual método seria superior – o convencional ou o laparoscópico. No entanto, uma revisão atual da literatura nos permite observar que os novos estudos mostram a apendicectomia laparoscópica como um procedimento seguro e eficaz, que pode ser utilizado no tratamento da apendicite complicada em qualquer faixa etária e quando o diagnóstico é duvidoso. Por estes motivos, essa opção cirúrgica está ganhando cada vez mais aceitação sendo que vários trabalhos recentes apontam-na como procedimento de escolha no tratamento da apendicite aguda.
ARTIGO DE METANÁLISE
Úlcera Péptica Perfurada: Sutura ou Ressecção?
A perfuração tem sido a complicação da úlcera péptica mais operada nos últimos anos . Devido à grande eficiência dos novos medicamentos para o tratamento clínico das úlceras gastroduodenais, a cirurgia para o tratamento dessa doença tem ficado apenas para o caso de algumas complicações, principalmente a estenose e a perfuração. Também devido ao pequeno número de gastrectomias para o tratamento das úlceras pépticas, o treinamento dos jovens cirurgiões ficou prejudicado, sendo que esses têm pouca familiaridade com o procedimento. Devido a todos esses fatores, o tratamento cirúrgico da úlcera perfurada traz grandes dilemas ao cirurgião. A úlcera aguda perfurada , isto é , aquela que não apresenta o calo fibroso ao redor da perfuração, deve ser tratada com a simples sutura da lesão. Já a úlcera crônica perfurada deve ser tratada sempre que possível pela gastrectomia. Uma revisão da literatura mostrou que, em algumas situações, é prudente se evitar a ressecção gástrica e optar pela sutura. Essas situações podem ser resumidas em: inexperiência do cirurgião na realização de gastrectomia apropriada, cavidade abdominal muito contaminada, paciente em mau estado geral em que o prolongamento do ato operatório irá piorar o quadro clínico, pacientes com mais de 60 anos, mais de 24 horas de perfuração e perfuração gástrica ou duodenal maior que 5 milímetros. A sutura tem elevadas taxas de recidiva ulcerosa (+ de 50%) e baixas taxas de mortalidade, a vagotomia associada a alguma técnica de drenagem apresenta baixa mortalidade porém a taxa de recidiva ainda é alta (10%) e, finalmente, a gastrectomia parcial tem taxa de mortalidade pouco maior que a vagotomia porém a recidiva ulcerosa é extremamente baixa (<1%). A gastrectomia parcial é o procedimento por nós preferido quando a ressecção gástrica está indicada devido à sua segurança e às baixas taxas de recidiva. Podemos concluir, portanto, que o melhor tratamento para a úlcera péptica perfurada é aquele que leva em conta o tipo de perfuração, as condições clínicas do doente, as condições locais da cavidade abdominal e a experiência do cirurgião na realização da gastrectomia.
AVALIAÇÃO PRÉ-OPERATÓRIA
MEDICINA BASEADA EM EVIDÊNCIA_AVALIAÇÃO PRÉ_OPERATÓRIA

O médico CIRURGIÃO realiza a avaliação pré-operatória e define a necessidade de avaliação complementar, considerando a otimização das condições clínicas do paciente e a realização de exames complementares. Pacientes hígidos, com idade inferior a 40 anos, sem fatores de risco detectados na anamnese e no exame físico, a serem submetidos a cirurgias de pequeno porte, após a avaliação clínica básica poderão ser encaminhados à cirurgia após avaliação do Anestesiologista. Porém, nos casos em que o paciente tenha alguma comorbidade, idade maior de 60 anos, estado físico ASA II ou acima, obesidade, dependência funcional ou cirurgia de médio ou grande porte, estará indicado avaliação pré-operatória mais pormenorizada .