The Surgical Coach
Surgical Wisdom Unveiled: A Top Ten List and Commandments
Reflections on a Surgical Journey 🌟
Life’s journey is a mosaic woven with threads of guidance from parents, siblings, and mentors. This chapter transcends the mundane, embracing philosophy and personal testimony on sculpting a triumphant surgical career. Dr. Richard C. Thirlby, in the spirit of David Letterman, unfurls a top ten list that serves as a compass for aspiring surgeons.
Dr. Thirlby’s Top Ten Surgical Tenets 📜🌐
- Training is Fun (You’ll Never Forget It): A nod to lifelong learning, acknowledging the perpetual metamorphosis in surgical careers.
- Job Security: General surgeons, vital and in demand, find positions across diverse landscapes, from bustling urban centers to the serene rural expanses.
- The Pay is Not Bad: Comfortable compensation, soaring above societal averages, promises financial stability.
- Your Mother Will Be Proud of You: A familial pride resonates, extending beyond mothers to fathers, aunts, and a tapestry of family members.
- Surgeons Have Panache: Embracing the surgical personality and the unique culture that envelopes surgical realms.
- You Will Have Heroes; You Will Be a Hero: Surgeons, sculpted by influencers, reciprocate by becoming beacons of hope for grateful patients.
- There is Spirituality if You Want It: The inexplicable recoveries, the miraculous moments that defy statistical norms.
- You Will Change Patients’ Lives: A profound personal satisfaction derived from the tangible impact on patients’ destinies.
- Patients Will Change Your Life: Daily lessons from patients foster humility, nonjudgmentalism, and a continuous journey towards becoming a better human being.
- I Love to Cut: A poetic reflection of the joy derived from the meticulous artistry of surgical procedures, executed with precision for the greater good.
The Commandments of Surgical Living 🌌📜
Adding depth to the narrative, akin to timeless commandments, Dr. James D. Hardy contributes a list transcending millennia, etched in the New King James Version of the Holy Bible.
- Know Your Higher Power: An homage to the spiritual facet of life and the sanctity of the Sabbath day.
- Respect Your Roots: An acknowledgment of the significance of parents and the importance of familial bonds.
- Do No Harm: An ancient ethos resonates through the prohibition of actions such as murder, adultery, theft, lying, and coveting others’ belongings.
- Strive for Excellence: An unending pursuit of personal and professional growth, embodying efficiency, excellence, and the preservation of integrity.
- Prepare for Leadership: A call to groom leaders, emphasizing the importance of educational and professional growth.
- Nourish Professional Relationships: Recognizing the value of mentors, preserving the wisdom passed down through generations.
- Remember Your Roots: An echo from Dr. Hardy’s personal ten commandments, urging individuals to honor their origin and represent it with pride.
- Cherish Family: A gentle reminder to spend quality time with family, recognizing the profound impact of love on children.
- Spend Time Alone: Advocating for moments of solitude, fostering creative thinking and personal reflection.
- Find Joy in Your Work: A profound truth encapsulated in the sustenance derived from the daily pursuit of meaningful work one genuinely enjoys.
In this amalgamation of Dr. Thirlby’s top ten and Dr. Hardy’s commandments, a roadmap unfolds — a guide not just for a surgical career but for a fulfilling and purpose-driven life. 🌈🔍🔬
Gastrointestinal Anastomosis
Navigating the Gastrointestinal Anastomosis: A Surgical Odyssey
Unveiling the Historical Tapestry 🕰️
The creation of gastrointestinal anastomoses, an art in general surgery, has evolved over centuries. In delving into this surgical saga, fundamental principles stand tall, guiding the surgeon’s hands. This chapter unfolds the historical nuances, general tenets for successful anastomosis creation, and delves into pivotal technical considerations amidst current controversies.
The Dance of Healing and Anatomy 🩹🔍
Understanding the physiological waltz of gastrointestinal wound healing and the intricacies of intestinal wall anatomy sets the stage. An enterotomy’s inception triggers a symphony of vasoconstriction, vasodilation, and capillary changes, orchestrating the ballet of tissue healing. Granulation tissue emerges, heralding the proliferative phase, where collagen undergoes a dance of lysis and synthesis.
Layers of the Gastrointestinal Tapestry 🧵
The intestinal wall, a multilayered tapestry, unravels its secrets. The serosa, a connective tissue cloak veiled by mesothelial lining, demands precise apposition to thwart leakage risks. The submucosa, the stronghold of tensile strength, anchors the sutures knitting the anastomosis. Intestinal mucosa seals the deal, driven by epithelial cell migration and hyperplasia, crafting a watertight barrier.
Local Factors: Paving the Path to Healing ⚒️🩹
The local factors influencing this symphony include intrinsic blood supply and tension management. Adequate blood supply, a lifeline for tissue oxygenation, hinges on meticulous surgical technique. Tension, a delicate partner in this dance, demands finesse; too much jeopardizes perfusion, too little invites inflammatory infiltrates. The colon, in particular, demands a surgeon’s nuanced touch.
Systemic Harmony: The Ripples of Patient Factors 🔄🌊
Systemic factors contribute their ripples to this surgical pond. Hypotension, hypovolemia, and sepsis compose a dissonant note affecting blood flow and oxygen delivery. Patient-specific variables — malnutrition, immunosuppression, and medication use (hello, steroids and NSAIDs) — compose a subplot, influencing the narrative of wound healing.
As the surgeon steps into this intricate ballet of anastomosis creation, history, physiology, and patient-specific factors converge. Each suture, each decision, shapes the narrative of healing. The gastrointestinal odyssey continues, blending tradition with innovation, as surgeons embark on the timeless quest for successful anastomoses. 🌐🔍🩺
Pre Surgical Evaluation of BLEEDING
Unraveling Bleeding Risks: A Surgical Odyssey
In the realm of surgical care, meticulous assessment of bleeding risk is paramount. The age of onset of bleeding and the specific sites affected offer crucial clues, helping differentiate between inherited and acquired bleeding disorders. Inherited disorders, often manifesting in childhood, may lurk beneath the surface, surfacing during surgical trauma in adulthood.
Decoding the History 🕰️
Interrogating the patient’s history unveils key insights. Medication usage, both prescription and over-the-counter, unfurls potential contributors to bleeding events. Family history provides a roadmap for inheritance patterns, crucial in diagnosing disorders like hemophilia. The severity of past bleeding incidents serves as a yardstick, guiding expectations during surgical challenges. Comorbidities, especially liver and kidney dysfunction, loom large in magnifying bleeding risks.
The Physical Symphony 🩺🎶
While the physical exam plays a supportive role, it may hint at platelet disorders through findings like petechiae and ecchymoses. Platelet function issues or deficiencies may manifest similarly, emphasizing the importance of a comprehensive history. Single-site bleeding tends to be non-indicative of a bleeding disorder, while multisite bleeding raises red flags.
Laboratory Pilgrimage 🧪
A pilgrimage through laboratory tests offers a comprehensive snapshot of hemostatic competence. Assessing platelet count, complete blood count (CBC), platelet function, aPTT, PT, and fibrinogen levels becomes the map for surgical decisions.
Unmasking Causes of Excessive Surgical Bleeding 🚩
Most patients enter the operating room with normal hemostasis. However, certain surgeries, like liver transplants or trauma interventions, may trigger consumptive coagulopathy. Preexisting hemostatic defects, especially congenital bleeding disorders like hemophilia and von Willebrand disease, require keen suspicion.
Hunting the Culprit: Acquired Bleeding Disorders 🎯
Liver disease emerges as a common instigator of coagulation abnormalities, while anticoagulant therapies like Coumadin and heparin cast shadows on surgical hemostasis. Acquired thrombocytopenia, often linked to splenomegaly or medications, and platelet function disorders, especially induced by aspirin and clopidogrel, populate the landscape of surgical challenges.
Navigating Intraoperative Waters ⚓🔍
Intraoperative bleeding may cascade from shock, massive transfusions, or acute hemolytic reactions. Hemostatic agents, from gelatin sponge to topical thrombin, stand as stalwart navigators through these turbulent waters.
Postoperative Chessboard: A Risky Endgame ♟️🩹
Postoperative bleeding, often stemming from inadequate hemostasis, unveils additional players. Residual heparin, altered liver function, and acquired clotting factor deficiencies post-hepatectomy amplify the stakes. Fibrinolysis disorders may also cast shadows post-surgery.
Dancing with Disseminated Intravascular Coagulation (DIC) 🩸🎭
DIC, a theatrical presentation of intravascular coagulation gone awry, demands a spot on the stage. Prompt recognition and addressing precipitating factors are pivotal, with cryoprecipitate and platelet transfusions standing as protagonists.
Fibrinolytic Fantasia: When Clotting Goes Amiss 🌪️🩹
Primary and secondary fibrinolysis emerge as culprits in postsurgical bleeding, often linked to lytic therapy, severe liver failure, or DIC. Managing fibrinolytic storms necessitates tailored interventions.
Hypercoagulable Waltz in Surgical Limelight 💃🕺
A careful dance with thromboembolism risks follows, accentuating the importance of patient history in unraveling congenital and acquired hypercoagulable states. A familial narrative often unravels the genetic predispositions steering this intricate choreography.
In the surgical arena, every patient’s hemostatic tale unfolds uniquely. Through history, examination, and laboratory revelations, surgeons navigate the delicate balance between bleeding and clotting, ensuring a symphony of healing amidst the surgical odyssey. 🌐🔍🩺
Nutritional Surgical Care
Navigating the Nutritional Maze in Surgical Care 🌐🔍
Surgeons bear the responsibility of caring for patients whose nutritional status may be compromised, influencing their ability to heal optimally. The challenges encompass an array of issues, including anorexia, inanition, gluconeogenesis acceleration, hyperglycemia, insulin resistance, and electrolyte and hormonal imbalances. These factors intricately impact surgical responses and a patient’s healing capacity. Let’s delve into the complex world of digestive tract, esophageal, gastric, intestinal, and other surgeries, exploring how they interplay with nutritional considerations.
Digestive Tract Surgery 🍽️
The digestive tract, a bustling center of metabolic activity, plays a pivotal role in nutrient digestion, absorption, and metabolism. Surgical interventions involving the gastrointestinal (GI) tract can lead to malabsorption and maldigestion, causing nutritional deficiencies. Understanding the site of nutrient absorption aids in identifying potential postoperative deficiencies. Enhancing nutritional status before surgery becomes crucial for a smoother postoperative recovery.
Esophageal Surgery 🥄
Various conditions affecting the esophagus, from corrosive injuries to obstruction, necessitate surgical intervention. Procedures involve replacing the esophagus with the stomach or intestine, each carrying unique considerations. Nutritional support, including nasoenteric feeding tubes or parenteral nutrition (PN), may be necessary preoperatively for obstructed esophagi, with additional intraoperative measures for optimal postoperative outcomes.
Gastric Surgery 🥢
Gastric surgical procedures, while addressing specific issues, can potentially lead to malnutrition. Patients may experience dumping syndrome, requiring dietary modifications and cautious fluid intake. Anemia and metabolic bone diseases are common consequences, demanding periodic injections and calcium-vitamin D supplementation. Understanding postgastrectomy dietary modifications and careful fluid management becomes paramount.
Intestinal Surgery 🍴
Resection of excessive lengths of the intestine, especially in short bowel syndrome, can result in severe malabsorption and malnutrition. Long-term PN might be necessary to maintain nutritional balance. Pancreaticoduodenectomy, a complex surgery, requires postoperative monitoring for complications like delayed gastric emptying, diabetes mellitus, and malabsorption, influencing nutrient guidelines.
Ileostomy and Colostomy 🚽
Procedures like ileostomy or colostomy, creating artificial anuses, are employed for various intestinal issues. Patients with ostomies generally follow regular diets, with adjustments based on stoma output. High-output ostomies necessitate specific dietary precautions to manage fluid levels. Nutritional assessment’s crucial role in surgical outcomes emphasizes the growing interest in tailored preoperative nutritional support and the potential resurgence of parenteral nutrition.
Conclusion 🩺💡
Understanding the intricate dance between surgical interventions and nutritional considerations is paramount for surgeons and medical practitioners. As regulatory scrutiny intensifies, the role of nutrition in preventing complications and improving outcomes will likely take center stage, emphasizing the importance of personalized nutritional strategies in the surgical journey. 🌟💪
Tubes and Drains
Unlocking the World of Tubes and Drains in Medical Practice 🩹
Understanding the diverse array of tubes and drains is crucial for any medical practitioner, and it all begins with appreciating the French size system, where the outer diameter of a catheter is denoted. A quick calculation (French size multiplied by 0.33) reveals the catheter’s outer diameter in millimeters.
Gastrointestinal Tract Tubes 🍽️
Starting with nasogastric tubes designed to evacuate gastric contents, these are frequently employed in patients facing ileus or obstruction. Modern nasogastric tubes often incorporate a sump function, preventing suction locks and enhancing efficiency. Nonsump tubes, though less common, may be used for intermittent suction. Nasogastric tubes also serve in feeding, with soft, fine-bore tubes being preferred for this purpose. Nasoenteric tubes, intended for feeding, require careful attention to safety during instillation.
Nasobiliary tubes, often placed endoscopically, aid in biliary drainage in cases of obstruction or fistula. T-tubes within the common bile duct ensure closed gravity drainage. Gastrostomy tubes, placed surgically or via percutaneous endoscopic gastrostomy (PEG), find utility in drainage or feeding. Jejunostomy tubes, inserted surgically or endoscopically, are vital for long-term nutritional access.
Respiratory Tract Tubes 🫁
Chest tubes play a pivotal role in pleural cavity drainage, addressing issues like pneumothorax, hemothorax, or effusion. The three-bottle system facilitates constant suction, drainage, and prevention of air entry, crucial for maintaining a water seal.
Endotracheal tubes, cuffed for a secure tracheal seal, cater to short-term mechanical ventilation needs in adults. Tracheotomy tubes, directly inserted into the trachea through the neck, become essential for prolonged mechanical ventilation or when maintaining a patent airway is challenging.
Urinary Tract Tubes 🚰
Bladder catheters, commonly known as “Foley” catheters, serve to straight drain urine. Nephrostomy tubes, placed in the renal pelvis, drain urine above obstructions or delicate ureteral anastomoses. Percutaneously placed tubes, often pigtail catheters, assist in draining abscesses, typically guided by interventional radiologists.
Surgical Drains 🌡️
Closed suction drains, such as Jackson-Pratt and Hemovac, prove invaluable for evacuating fluid collections during surgery. Sump suction drains, like Davol drains, are larger and designed for continuous suction in scenarios with thick or particulate drainage. Passive tubes, exemplified by Penrose drains, offer a pathway for fluid without applied suction, serving as a two-way conduit for bacteria. Understanding these various tubes and drains is pivotal for medical practitioners navigating complex clinical scenarios. 💉💊
The Geriatric Patient
Navigating Surgical Challenges in an Aging Population: A Delicate Balance 🌐
The ongoing aging process within the American population brings forth a set of unique challenges that surgeons must adeptly navigate for decades to come. Elderly individuals, compared to their younger counterparts, often exhibit diminished physiological reserves. Their health is frequently influenced by medications that can alter normal physiological responses, such as β-blockers, or impact surgical outcomes, like warfarin or platelet aggregation–inhibiting agents. Additionally, baseline impairments, ranging from sensory issues to difficulties in ambulation or dementia, may complicate their ability to engage in everyday activities.
One perplexing dilemma faced by surgeons when caring for elderly patients revolves around the decision to pursue an aggressive intervention plan. Transparent communication between the patient and physician is paramount in determining the appropriate level of aggressiveness in the patient’s best interest. This conversation takes on heightened significance in the elderly population. Engaging in repeated discussions with patients and their families, starting before surgery and extending into the postoperative phase, is crucial. Generally, patients express a desire for aggressive medical care as long as there remains a reasonable chance for meaningful survival.
While these discussions may be uncomfortable, they are as integral to the patient’s care as any aspect of their medical history. It is imperative to recognize that surgical care is provided by individuals who genuinely care about the patient’s overall well-being. In certain situations, medical care may prioritize alleviating pain over prolonging life. Ideally, these conversations should occur in a serene and comfortable setting, free from distractions.
Moreover, it is essential to underscore that discussions about end-of-life matters are not legal proceedings. No forms need to be signed. These discussions are akin to any other conversation between a doctor and a patient regarding their care. The dialogue involves a careful consideration of the strengths and weaknesses of different approaches until a collaborative plan of action is determined. The only distinction lies in the profound nature of end-of-life discussions, offering patients the best opportunity to shape their destinies. Consequently, these discussions should be approached with the utmost reverence, acknowledging the gravity of the subject matter. 🤝💙
Estabelecendo Conexões Essenciais 💬
Técnicas de Entrevista na Medicina: Estabelecendo Conexões Essenciais 💬
A relação médico-paciente é uma parte vital do cuidado cirúrgico. O vínculo entre o cirurgião e o paciente deve ser construído, mantido e valorizado. Boas técnicas de entrevista são fundamentais para estabelecer essa relação. O alicerce para uma boa entrevista advém de uma preocupação genuína com as pessoas. Embora existam habilidades de entrevista que podem ser aprendidas, a qualidade da interação pode ser aprimorada. Estudantes de medicina devem reconhecer seu papel especial no cuidado do paciente, não se envergonhando de sua posição e compreendendo que são membros eficazes da equipe. Pacientes frequentemente veem os estudantes de medicina como acessíveis, compartilhando detalhes que poderiam ocultar de membros mais seniores da equipe. O papel do estudante é descobrir as queixas médicas principais do paciente, realizar uma história e exame físico focalizados e apresentar os achados à equipe. Uma entrevista eficaz requer a comunicação sobre quem você é e como se encaixa na equipe. 🏥
Desafios da Entrevista na Medicina: Adaptando-se a Diferentes Ambientes 🌐
Entrevistar bem pode ser desafiador devido à variedade de ambientes, como sala de cirurgia, unidade de terapia intensiva, consultório particular, leito hospitalar, sala de emergência e ambulatório. Cada ambiente apresenta desafios únicos à comunicação eficaz. Para construir boas relações médico-paciente, cirurgiões ajustam seus estilos a cada ambiente e à personalidade e necessidades de cada paciente. Algumas regras básicas são comuns a todas as entrevistas profissionais. A primeira regra é deixar claro ao paciente que, durante a história e o exame, nada além de uma emergência de vida ou morte terá maior importância do que a interação entre o cirurgião e o paciente naquele momento. Este é o primeiro e melhor momento para conectar-se com o paciente. É crucial que o paciente compreenda que um cirurgião cuidadoso, conhecedor e dedicado será seu parceiro na jornada pelo tratamento de doenças cirúrgicas. O cirurgião deve observar outras regras, incluindo dar atenção adequada à aparência pessoal para transmitir uma imagem profissional que inspire confiança, estabelecer contato visual, comunicar interesse, calor e compreensão, ouvir de forma não julgadora, aceitar o paciente como pessoa, ouvir a descrição do problema do paciente e ajudar o paciente a se sentir confortável na comunicação. 👩⚕️👨⚕️
Primeiros Minutos: Estabelecendo uma Base Sólida 🤝
Ao receber o paciente em um ambiente ambulatorial, os primeiros minutos são dedicados a cumprimentar o paciente pelo nome formal, apertar as mãos, se apresentar e explicar o papel do cirurgião. A atenção à privacidade do paciente, ajustando o estilo de conversação e o vocabulário às necessidades do paciente, descobrindo a atitude do paciente em relação à clínica, conhecendo a ocupação do paciente e entendendo o que o paciente sabe sobre sua condição são passos fundamentais. A seguir, ocorre a exploração do problema, movendo-se de perguntas abertas para perguntas fechadas. Técnicas importantes incluem o uso de transições, fazer perguntas específicas e claras e reformular o problema para verificação. É crucial determinar se o paciente tem alguma pergunta. No final da entrevista, o cirurgião explica quais serão os próximos passos e que realizará um exame no paciente. Por fim, verifica-se se o paciente está confortável. 🌟
Diferenças nos Ambientes de Atendimento: Adaptação e Compreensão 🏨
As técnicas utilizadas em ambientes ambulatoriais são igualmente adequadas para encontros em ambientes hospitalares e de pronto-socorro. Geralmente, mais tempo é dedicado ao paciente nas entrevistas iniciais e subsequentes do que em um ambiente ambulatorial. Na entrevista inicial, os pacientes podem estar com dor, preocupados com problemas financeiros e ansiosos com a falta de privacidade ou dietas desagradáveis. Eles também podem ter dificuldade para dormir, sentir medo do tratamento ou se sentir impotentes. É importante comunicar gentil e confiantemente o propósito da entrevista e quanto tempo levará. O paciente não apenas escuta, mas também observa o comportamento e a vestimenta do médico. O ambiente também afeta a entrevista. Por exemplo, um ambiente apertado, barulhento e lotado pode afetar a qualidade da comunicação. Pacientes podem ter sentimentos negativos devido a insensibilidades por parte do médico ou de outras pessoas. É fundamental evitar falar com o paciente na entrada do quarto, dar ou receber informações pessoais em um ambiente lotado, falar sobre um paciente em um elevador ou em outro espaço público, ou falar com um paciente sem fechar a cortina em uma enfermaria. 🌆
Conclusão: Construindo Relações de Confiança na Medicina 🌐
Em resumo, dominar as técnicas de entrevista na medicina é essencial para construir relações sólidas entre médico e paciente. Os cirurgiões habilmente ajustam suas abordagens a diferentes ambientes e personalidades dos pacientes, aplicando regras básicas universais. Os primeiros minutos são cruciais para estabelecer uma base sólida, enquanto a exploração cuidadosa do problema requer uma transição de perguntas abertas para fechadas. Adaptações ao ambiente e compreensão das preocupações do paciente são vitais para uma comunicação eficaz. Ao incorporar essas técnicas, os médicos podem garantir que a interação médico-paciente seja centrada no paciente, transmitindo confiança e dedicação à jornada conjunta pelo tratamento. 👨⚕️🤝👩⚕️
Navigating Your Clinical Experience in Surgery
Transitioning to the Role as a Junior Member of the Surgical Health Care Team: Navigating Your Clinical Experience in Surgery
Embarking on your surgical clerkship is an immersive clinical journey that extends beyond aspiring surgeons. This experience is a valuable opportunity to refine essential clinical skills applicable to any medical specialty. As you progress in your medical career, encounters with patients requiring surgical intervention will be inevitable. The insights gained during your surgery clerkship will empower you to identify surgical diseases, understand the need for surgical consultations, and develop empathy for the emotional, physiological, and logistical aspects patients and their families may face during operations or consultations.
Approaching Your Role: A Shift in Learning Dynamics
As a seasoned learner in medical school, your entry into the health care team during your 3rd year marks a profound transition. The focus is no longer solely on memorization; it now involves understanding patients’ complaints and diseases. Balancing voluminous information, time constraints, and determining the depth of knowledge required for patient care present unique challenges. Successful clinical performance is influenced by various factors:
- Preparatory Coursework and Experience: Constructing new knowledge relies on existing foundations. Deep knowledge, especially in anatomy, facilitates a seamless connection between new information and prior understanding.
- Quality of Study Methods: Active learning demands taking responsibility for your education. Maintaining a disciplined study plan aligned with your learning style is essential.
- Organizational Skills: Successful learners adeptly manage time and priorities to avoid last-minute cramming, reducing stress.
- Motivation and Emotion: Enthusiasm and positive feelings toward content, team, and environment significantly impact the clerkship experience and how you are perceived by patients and the team.
- Physical Health: The link between physical well-being and effective learning underscores the importance of attending to personal health needs.
- Distractibility and Concentration Skills: Active engagement in learning, whether through reading or listening, is crucial for processing and translating information into meaningful knowledge.
Maximizing Your Learning: Strategies for Success
1. Prepare, Practice, and Review:
- Preparation: Activate prior knowledge by pre-reading about upcoming topics, enhancing comprehension, and fostering long-term memory.
- Practice: Actively engage in learning, take notes, and generate questions. Studies show that self-generated notes enhance retention.
- Review: Ongoing review, coupled with self-assessment using test questions, patient management problems, and creating personal tests, reinforces information.
2. Organize Your Knowledge:
- Get the Big Picture: Familiarize yourself with learning objectives, chapter headings, and subheadings before reading. Listing questions beforehand guides focused reading.
- Review Charts and Diagrams: Key information in charts and diagrams aids study. Testing yourself on missing information enhances understanding.
- Emphasize Integration: Relate new information to patient encounters, lectures, or images. Create mind maps to organize information and identify patterns.
3. Know Expectations and Thyself:
- Clarify your role and responsibilities by reviewing syllabus materials and seeking guidance from experienced peers, residents, or faculty.
- Define personal learning goals and learning style, being proactive in your approach to studying.
4. Ask! Ask! Ask!:
- Be persistent and assertive in seeking clarification, feedback, or assistance.
- Utilize the wealth of expertise within the surgical team, including nurses, physician assistants, pharmacists, social workers, and technicians.
Maximizing Your Emotional Intelligence: Strategies for Well-Being
1. Focus Forward with a Positive Attitude:
- Make decisions with a positive end in mind, maintaining situational awareness in varied scenarios.
- Cultivate open-mindedness, positive energy, and enthusiasm to contribute constructively to patient care and the team.
2. Set Goals and Celebrate Successes:
- Define short- and long-range goals, including personal, financial, and relationship goals.
- Document achievements, reinforcing a sense of accomplishment and self-confidence.
3. Promote a Supportive Learning Environment:
- Take responsibility for mistakes, learn from them, and foster trust within the team.
- Avoid negativity by steering clear of individuals with persistent negative attitudes or behaviors.
In conclusion, approach your surgery clerkship with a proactive mindset. Maximize both your intellectual capabilities and emotional intelligence to derive the utmost benefit from this transformative experience. Keep in mind the wisdom of John Wooden, emphasizing the importance of being the best version of yourself. This principle not only ensures success in your surgery clerkship but resonates throughout your medical journey.
REVISIONAL BARIATRIC SURGERY
- INTRODUCTION
Morbid obesity is a global chronic disease affecting 13% of people worldwide. Weight loss surgery has been proven to be effective in addressing this chronic disease and its associated comorbidities. In 2016, over 200,000 procedures were performed in the United States, and the volume continues to grow. Cases analyzed between 2015 and 2018 indicate an overall growth rate of 21.9%. The most common surgeries performed in the United States are Roux-en-Y gastric bypass (RYGB), sleeve gastrectomy (SG), and biliopancreatic diversion with duodenal switch (BPD/DS). Estimated mean weight loss is 33% of the initial body weight. Unfortunately, it is estimated that up to 25% of patients will have weight regain after primary surgery. Weight regains or recidivism has emerged as a clinical entity and important public health issue given its association with re-emergence of obesity related comorbidities, worsening quality of life, and increased healthcare costs. With the increased number of primary bariatric surgery performed worldwide, revisional surgery has also increased, and it has been shown to be the fastest-growing category of bariatric procedures, currently representing 7 to 15% of all bariatric operations and long-term rates of revisional surgery have been estimated to be as high as 56%.
2. CAUSES
Weight regain is estimated in up to 25% of patients following primary bariatric surgery. In addition, it is estimated that more than 80% of the weight regain happens within the first 6 years following primary surgery. A major factor contributing to weight regain is lack of adherence to recommended followup visits, observed in approximately 60% of patients 4 years after primary surgery. The etiology of weight regain has been attributed to:
• Noncompliance with dietary recommendations.
• Hormonal/metabolic imbalance.
• Mental health.
• Physical inactivity.
• Anatomic/surgical factors.
• Medications.
3. PREOPERATIVE EVALUATION
A multidisciplinary evaluation is essential prior to recommending revisional surgery to patients presenting with weight gain. As with primary surgery, a nutritional evaluation, behavioral/psychological assessment, and endoscopic and contrast series studies should be obtained. The latter will not only aid in establishing an anatomic etiology for weight regain if present, but it will also aid in choosing the type of revisional surgery.
4. SELECTING THE TYPE OF REVISIONAL SURGERY
There are several revisional procedures following primary bariatric surgery. The choice of revisional surgery is tailored according to initial surgery, cause of failure, and surgeon’s experience. Multiple revisional surgeries have been described for all primary bariatric surgeries, but no standardized guidelines have been established. In June 2019, 70 experts from 27 countries formed a committee and created the first consensus on revisional bariatric surgery. An agreement of 70% or more was considered consensus. Consensus was achieved in several points including but not limited to:
(1) RBS is justified in some patients;
(2) RBS is more technically challenging than the respective primary bariatric surgery;
(3) second or third RBS can be justified in some patients;
(4) candidates should undergo a nutritional assessment, psychological evaluation, endoscopy, and a contrast series;
(5) RYGB, one anastomosis gastric bypass (OAGB), and SADI-S are options after gastric banding; and
(6) OAGB, BPD/DS, and SADI-S are options after sleeve gastrectomy. Regarding revision for primary RYGB, the only consensus obtained was lengthening of the biliopancreatic limb as RBS option for RYGB or OAGB.
Roux-En-Y Gastric Bypass
Roux-en-Y gastric bypass is one of the most common weight loss procedures performed worldwide and is considered by many to be the gold standard. Unfortunately, approximately 10–34% of patients experience inadequate weight loss or weight gain and may ultimately require revision. The most common etiology of weight regain is pouch dilation. Other reported etiologies include enlarged gastric pouch greater than 5 cm in diameter, wide gastro-jejunal anastomosis (GJA), anastomosis greater than 1 cm, GJA > 1.5 cm in diameter, dilated GJA greater than or equal to 2 cm, pouch >30 mL, pouch dilation >120 mL, weight recidivism with or without gastric fistula, gastric fistula, short-limb bypass, and hyperphagic behavior. Multiple revisional surgeries have been described. In a recent systemic review and meta-analysis, distal Roux-en-Y gastric bypass (DRGB) alone showed the highest decrease in BMI at 1-year follow-up versus biliopancreatic diversion with duodenal switch (BPD/DS) or single anastomosis duodeno-ileal bypass and sleeve gastrectomy (SADI-S) at 3-year follow-up. Overall, they found maximal BMI decrease in DRGB alone, followed by BPD/DS or SADI-S, laparoscopic pouch and/or GJA resizing, and endoscopic pouch and/or GJA resizing.
Sleeve Gastrectomy
Sleeve gastrectomy (SG) is currently the most commonly performed bariatric procedure worldwide. Its relatively simple technique and low complication rate contribute to it being preferred over some other procedures. Revision is estimated in up to 30% of cases for multiple etiologies, including weight regain. Loss of restriction is one of the main anatomic factors contributing to weight regain. Although revision to RYGB or DS has been recommended as the standard of care, some studies have described revision with re-sleeve for dilation of the residual stomach as the cause. The overall %EWL following re-sleeve can be up to 57% at 12 months and up to 60% at 20 months. A retrospective study analyzed conversion from SG to either RYGB or SADI for insufficient weight loss or weight regain. Out of 140 patients, 66 patients underwent SG to SADI, and 74 patients underwent SG to RYGB. SADI was found to achieve 8.7%, 12.4%, and 19.4% more total body weight loss at 6, 12, and 24 months compared to RYGB for weight regain alone. RYGB is preferred when symptoms of reflux accompany weight regain. For patients with super morbid obesity and weight regain after SG, in the absence of reflux symptoms, conversion to biliopancreatic diversion with duodenal switch (BPD/DS) is recommended for maximal weight loss, with %EWL ranging from 70 to 80% at 2 years.
5. WEIGHT LOSS FOLLOWING REVISIONAL SURGERY
Weight loss after revisional bariatric surgery leads to significant weight loss in the long term, rates varying per procedure performed. A single-center retrospective study for patients who underwent revisional surgery for weight regain (52.4%) S. Ardila et al. analyzed weight loss at 3-, 6-, 9-, and 12-month intervals. Patients with a primary restrictive procedure and reflux symptoms underwent conversion to either RYGB or BPD/DS and experienced 50–65.3%EWL at 3 months and 50.1–79.1%EWL at 12 months. Patients with initial RYGB underwent GJ revision for pouch or GJ abnormalities. For those without anatomic abnormalities, they underwent conversion to distal bypass. At 3 months, %EWL was 36.6 for GJ revision and 37.5% for distal revision.
6. Complications of Revisional Surgery
Revisional bariatric surgery is complex, is technically demanding, and is therefore associated with higher morbidity and mortality. Compared to primary surgery, revisional surgery has been associated with higher rates of postoperative complications, longer operative times, longer hospital stay, conversion to open surgery, readmission, and unplanned admission to the critical care unit. In comparing primary versus revisional RYGB, revisional surgery was associated with higher rates of leak, hemorrhage, wound infection, stricture, ulcer, perforation, and hernia. A single-center retrospective study analyzed complications after revisional surgeries performed at their center for weight regain between 2012 and 2015. Of 84 patients, 43 presented for weight regain (52.4%). Complications included incarcerated ventral hernia following AGB conversion to SG, anastomotic leak and recurrent intussusception following gastro-jejunostomy revision, and stricture and marginal ulcer following AGB conversion to RYGB . In summary, reported complications of revisional bariatric surgery are:
• Hernia.
• Anastomotic leak.
• Stricture.
• Marginal ulcer.
• Wound infection.
• Hemorrhage.
• Perforation.
• Obstruction.
Conclusion
Weight regain after primary bariatric surgery is multifactorial. It is imperative to establish guidelines for classifying weight gain in order to guide subsequent intervention and thus to aid bariatric teams internationally in the management of this clinical entity. Revisional surgery has been shown to be a successful treatment option for patients presenting with weight gain, with rates of weight loss nearing those seen after primary surgery. Furthermore, the importance of revisional surgery lies in its ability to readdress many of the obesity-related comorbidities which prompted the primary procedure. When indicated, it is important to tailor the type of revisional surgery to each patient. Although Roux-en-Y gastric bypass remains the most common type of revisional surgery after primary surgery of any type, duodenal switch is emerging as the revision procedure of choice for superobese patients and patients with failed sleeve gastrectomy secondary to weight regain in the absence of reflux symptoms.
Revisional Surgery for Weight Regain
- Introduction
Revisional bariatric surgery is a weight loss surgery for people who have not lost enough weight or have regained weight after their first bariatric surgery. It is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain.
- Causes of Weight Regain
There are many factors that can contribute to weight regain after bariatric surgery, including:
* Lack of adherence to dietary recommendations
* Hormonal/metabolic imbalance
* Mental health
* Physical inactivity
* Anatomic/surgical factors
* Medications
- Preoperative Evaluation
Before revisional surgery, patients will undergo a thorough evaluation to determine if they are eligible for the procedure. This evaluation will include a medical history and physical exam, as well as blood tests, imaging studies, and a psychological evaluation.
- Selecting the Type of Revisional Surgery
The type of revisional surgery that is best for a patient will depend on a number of factors, including the type of primary surgery they had, the cause of their weight regain, and their overall health. Some of the most common types of revisional surgery include:
* Roux-en-Y gastric bypass (RYGB)
* Sleeve gastrectomy (SG)
* Biliopancreatic diversion with duodenal switch (BPD/DS)
* Single anastomosis gastric bypass (OAGB)
* Sleeve gastrectomy with duodenal switch (SADI-S)
- Weight Loss Following Revisional Surgery
Weight loss after revisional surgery can be significant. In one study, patients who underwent revisional surgery for weight regain lost an average of 50–65.3% of their excess weight after 3 months and 50.1–79.1% of their excess weight after 12 months.
- Complications of Revisional Surgery
Revisional surgery is more complex and technically demanding than primary bariatric surgery, and therefore carries a higher risk of complications. Some of the most common complications of revisional surgery include:
* Hernia
* Anastomotic leak
* Stricture
* Marginal ulcer
* Wound infection
* Hemorrhage
* Perforation
* Obstruction
- Conclusion
Revisional bariatric surgery can be a successful treatment option for patients with weight regain. However, it is important to carefully consider the risks and benefits of the procedure before making a decision. Patients should also work closely with their healthcare team to choose the best type of revisional surgery for them and to prepare for the procedure and its aftermath. Revisional bariatric surgery is a complex and technically demanding procedure, but it can be a successful treatment option for patients with weight regain. It is important to work closely with your healthcare team to determine if you are eligible for the procedure and to choose the best type of surgery for you.
The Stoic Surgeon: Drawing Inspiration from Marcus Aurelius in Medical Practice
Introduction
The practice of medicine and surgery, while profoundly rewarding, can also be challenging and emotionally demanding. In these moments, many professionals seek guidance and inspiration to navigate the inherent difficulties in human healthcare. A rich source of timeless wisdom can be found in the words of the philosopher-emperor Marcus Aurelius, whose Stoic teachings resonate powerfully in the context of contemporary medicine. Let’s explore how Marcus Aurelius’s ideas can illuminate and fortify medical and surgical practice.
Development
- “What we control is what we calmly accept.” – In moments of uncertainty and pressure in the operating room, the serene acceptance of circumstances is crucial for maintaining composure and focus.
- “Do not expect the doctors to do everything for you.” – Marcus Aurelius reminds us of the importance of recognizing our own limitations. Collaboration between doctor and patient is essential for effective treatment.
- “Pain is inevitable. Suffering is optional.” – When dealing with patients in painful situations, it’s vital to remember that our role extends beyond physical treatment to emotional support and alleviating suffering.
- “Doing what needs to be done when it needs to be done, to the best of our ability, is already enough.” – In a profession as demanding as medicine, it’s vital to remember that every effort dedicated to patient care is valuable and meaningful.
- “What is morally right is more important than what is legally right.” – In some situations, it may be necessary to make decisions that go beyond protocol, aiming for the best interest of the patient.
- “To wait is harmful. Not to wait is necessary.” – Readiness and agility in making medical decisions can be crucial in emergency situations.
- “Time is a river, and facts are currents; we cannot escape, we cannot choose where we will go, nor when, nor how.” – This reflection reminds us of life’s impermanence and the importance of cherishing every moment in medical practice.
- “Man’s health is our treasure.” – Marcus Aurelius emphasizes the valuable responsibility doctors have in preserving and restoring individuals’ health.
- “Every day that dawns brings some evil, which we must accept, for no day will be without it.” – Recognizing that challenges are part of the medical journey allows professionals to face them with courage and determination.
- “Where is the good, if not in doing good to those in need?” – The practice of medicine is a living testament to compassion and altruism. Through it, doctors have the opportunity to demonstrate the true essence of human kindness.
Conclusion
Just as Marcus Aurelius faced the challenges of the Roman Empire with serenity and wisdom, the modern surgeon finds in his words a guide to practicing medicine with virtue and humanity. By uniting technical skill with Stoic philosophy, the surgeon becomes not only a master of their craft but also an example of Stoic activity for the entire medical community. May each operating room be a temple of virtue and care, where the legacy of Marcus Aurelius lives on through the hands of Stoic surgeons.
The Art of Healing: Wisdom from Opus Dei for Medical Professionals
1. “Work is a path to holiness.” – Just as in Opus Dei, where work is considered a means to grow closer to God, in the field of medicine, every interaction with a patient, every diagnosis, and every surgery is an opportunity to serve and make a positive impact.
2. “In your daily work, offer up small sacrifices for your patients.” – Much like Opus Dei encourages offering up small sacrifices for spiritual growth, in medicine, dedicating extra time, attention, or effort for a patient’s well-being can be a powerful form of compassion.
3. “Strive for excellence in your field; it is a reflection of your dedication to your patients.” – Opus Dei emphasizes the pursuit of excellence in one’s profession as a form of service to God. Similarly, in medicine, continuous learning and improvement directly benefit the quality of care provided to patients.
4. “Treat each patient with dignity, respect, and kindness.” – This fundamental principle aligns with Opus Dei’s emphasis on valuing every individual. In medicine, showing compassion and empathy is as important as clinical expertise.
5. “Pray for guidance in making difficult decisions.” – Just as Opus Dei encourages seeking spiritual guidance in challenging situations, in medicine, turning to one’s faith for moral and ethical dilemmas can provide clarity and a sense of purpose.
6. “Foster a culture of trust and collaboration among colleagues.” – Opus Dei emphasizes unity and mutual support among its members. Similarly, in the medical field, teamwork and open communication are vital for providing the best possible care for patients.
7. “Never underestimate the power of a kind word or gesture.” – Small acts of kindness, like those encouraged in Opus Dei, can have a profound impact on a patient’s experience and recovery in the medical setting.
8. “Cultivate a spirit of gratitude for the opportunity to serve others.” – Recognizing the privilege of being able to heal and alleviate suffering is a perspective shared by Opus Dei and medical professionals alike.
9. “Strive for balance between professional and personal life.” – Opus Dei emphasizes the importance of a balanced life. In medicine, maintaining a healthy work-life balance is crucial for sustaining a long and fulfilling career.
10. “Embrace the challenges of medicine as opportunities for growth and service.” – Opus Dei encourages embracing life’s challenges as a means of spiritual growth. Likewise, in medicine, facing the complexities and difficulties of healthcare with dedication and compassion can lead to profound personal and professional development.
Incorporating these principles from Opus Dei into the practice of medicine can not only enhance the quality of care provided but also contribute to a more compassionate and fulfilling healthcare experience for both practitioners and patients alike.
Wasteland in the Operating Room: Reflections on Medical Practice
“April is the cruellest month” – This T.S. Eliot verse may, in a way, resonate in the hallways of hospitals. April, for many, signifies the onset of spring, but in the operating room, it can symbolize the start of intense challenges.
“Mixing memory and desire” – As surgeons, we are constantly challenged to balance past experiences with future aspirations. The memory of previous cases shapes our desire to refine techniques and achieve better outcomes for our patients.
“Spring is dead, buried is hope” – Post-operative desolation often mirrors this verse. Surgery is at times a painful and grueling process, but it is in these moments that our hope for recovery and healing remains steadfast.
“Here is no water but only rock” – The operating room, with its sterile white surfaces, may seem devoid of life. Yet, it is here that life is preserved and restored, where the line between mortality and survival is drawn with surgical precision.
“Shaking the bitter foam of past experience” – As healthcare professionals, we are compelled to learn from each procedure. Every case is an opportunity for refinement, a chance to perfect our skills and grasp the intricacies of medicine.
“I cannot help but think/ This that we believe to be the end is only the beginning” – When the scalpel glides, when the sutures are closed, it is crucial to remember that the conclusion of a surgery is just the start of the patient’s recovery process. Hope is renewed in the promise of a healthier tomorrow.
“In the waiting room, gazing at the sky” – Even in moments of pre-operative anxiety, we look to the future with optimism. We know that medicine is a journey of challenges and triumphs, and each patient is a star in the sky of our professional experience.
“Here we are, contemplating the horizon” – In our medical careers, we face each day with determination and resilience. We are always keeping an eye on the horizon, seeking new techniques, advancements, and opportunities to enhance patient care.
“Shantih shantih shantih” – At the end of each procedure, when silence fills the operating room, it is a tranquil reminder that, even amidst complexity and challenges, medicine is ultimately an act of compassion and healing.
Just as in T.S. Eliot’s “The Waste Land,” in medicine and surgery, desolation and hope intertwine. Each procedure is an opportunity for learning and renewal. Through dedication and a commitment to patient well-being, we find our “shantih,” the peace we seek throughout this medical journey.

Liderança Inspiradora de Lord Nelson: Lições para Cirurgiões Contemporâneos
A história nos presenteou com inúmeros líderes notáveis, cujas habilidades e características têm inspirado gerações ao longo dos séculos. Um desses ícones é Lord Nelson, o lendário almirante britânico do século XVIII, conhecido por sua coragem e maestria tática nas batalhas navais. Surpreendentemente, as características de liderança de Lord Nelson têm aplicações valiosas mesmo na prática contemporânea da cirurgia. Neste artigo, exploraremos algumas dessas qualidades e como nós cirurgiões podemos aplicá-las para alcançar o sucesso em nossas batalhas cirúrgicas.
“Eu sou inabalável em minha determinação, como nunca estive em minha vida, desde que coloquei o pé no convés de um navio.”
1. Determinação Inabalável: Lord Nelson era conhecido por sua determinação inabalável em alcançar seus objetivos. Ele enfrentou inúmeras adversidades e desafios durante suas batalhas navais, mas nunca desistiu diante das dificuldades. Os cirurgiões contemporâneos podemos aprender com essa qualidade, mantendo-se resilientes diante de obstáculos, buscando soluções mesmo nas situações mais desafiadoras e garantindo o melhor atendimento possível para nossos pacientes. Durante a Batalha do Nilo, em 1798, Nelson enfrentou condições adversas quando a frota britânica ficou encalhada em bancos de areia. Enfrentando uma situação aparentemente desesperadora, ele permaneceu determinado a superar os obstáculos para alcançar a vitória. Com habilidades táticas brilhantes, Nelson conseguiu finalmente manobrar sua frota para fora dos bancos de areia e alcançar uma vitória decisiva sobre a frota francesa.
“Eu sempre fui um covarde em tudo, exceto nesta profissão.”
2. Coragem sob Pressão: Durante a Batalha de Copenhague, em 1801, Nelson foi ordenado a se retirar pelo comandante em chefe, mas ele ignorou a ordem e ergueu o telescópio em seu olho cego para afirmar que não conseguia ver o sinal. Ele então liderou seu esquadrão em um ataque corajoso, superando uma poderosa defesa dinamarquesa. Sua determinação e coragem sob pressão foram fundamentais para a vitória britânica. Em meio ao caos e perigo das batalhas navais, Lord Nelson demonstrou coragem excepcional. Seu exemplo inspirador pode nos lembrar da importância de permanecermos calmos e focados, mesmo durante procedimentos complexos ou emergências médicas. A coragem para tomar decisões rápidas e precisas pode fazer a diferença entre a vida e a morte em uma sala de cirurgia.
“A vontade não pode ser chamada de coragem, mas dever, o resultado da força da mente; e qualquer um que possua esse poder, quando chega a hora de agir, encontrará a coragem que necessita.”
3. Liderança Carismática: Lord Nelson conquistou o respeito e a admiração de sua tripulação com sua liderança carismática. Durante a Batalha do Nilo, quando o perigo era iminente e a tripulação estava nervosa, Nelson subiu ao convés e enfrentou corajosamente o inimigo, encorajando seus marinheiros a seguirem seu exemplo. Sua presença inspiradora foi fundamental para manter o moral elevado e motivar sua equipe a lutar com determinação. Lord Nelson era conhecido por sua liderança carismática e inspiradora. Ele conquistou o respeito e a lealdade de sua tripulação com sua empatia e capacidade de se conectar emocionalmente com seus marinheiros. Os cirurgiões contemporâneos também podem se beneficiar ao desenvolver uma liderança que inspire confiança e motivação em suas equipes médicas, promovendo um ambiente colaborativo e eficiente.
“Eu planejo que cada homem atue de acordo com o que vê melhor, mas qualquer homem que venha entre as duas frotas, não pode ser errado, portanto, não pode ser chamado de insubordinação, mas uma obediência em que cada oficial julga por si mesmo o que é melhor a ser feito.”
4. Capacidade de Adaptação: Durante a Batalha de Trafalgar, a estratégia inicial de Nelson era enfrentar a frota inimiga de maneira mais tradicional. No entanto, quando percebeu que a formação inimiga era mais forte do que o esperado, ele rapidamente adaptou sua tática e implementou a “manobra de Nelson”, cortando a linha inimiga e concentrando seu poder de fogo para alcançar a vitória. Durante suas batalhas, Lord Nelson enfrentou situações imprevistas e mudanças de planos. Sua capacidade de adaptação e flexibilidade permitiu-lhe ajustar suas estratégias conforme necessário. Na cirurgia contemporânea, os profissionais também devem ser ágeis e adaptáveis, prontos para responder a mudanças repentinas durante procedimentos cirúrgicos complexos ou emergências médicas.
“Seja qual for a estação em que você estiver, carregue consigo o pensamento de que todos os olhos estão voltados para você. Comande a si mesmo sempre que for o exemplo de todos os outros.”
5. Comunicação Efetiva: Lord Nelson era conhecido por sua habilidade de comunicação clara e direta com sua equipe. Antes da Batalha do Nilo, ele escreveu uma carta aos marinheiros enfatizando a importância da vitória e oferecendo palavras de encorajamento. Sua mensagem sincera e motivadora demonstrava sua preocupação com o bem-estar de sua tripulação e ajudou a criar um senso de propósito comum entre seus homens. A comunicação efetiva era uma das principais características de liderança de Lord Nelson. Ele sabia como transmitir suas ordens com clareza e também valorizava o feedback de sua equipe. Cirurgiões contemporâneos podem seguir esse exemplo, enfatizando a comunicação transparente com seus colegas e pacientes para garantir que todos estejam alinhados quanto aos procedimentos e tratamentos.
Em suma, a história de Lord Nelson nos ensina que as características de liderança transcendem as épocas e têm aplicação valiosa em diversas áreas, incluindo a medicina. Ao incorporar a determinação, coragem, carisma, adaptação e comunicação efetiva de Lord Nelson, podemos nos inspirar e tornar líderes excepcionais em nossas práticas médicas, promovendo um atendimento de qualidade e impactando positivamente a vida dos pacientes. A frase ‘A Inglaterra espera que cada um faça sua parte’ tornou-se um lema inspirador e atemporal, capturando a essência da liderança de Lord Nelson na Batalha de Trafalgar. Essas palavras ecoam através dos séculos, lembrando-nos da importância da responsabilidade individual e do trabalho em equipe para alcançar o sucesso em qualquer empreendimento. Assim como os marinheiros da frota britânica foram impulsionados por esse chamado à ação, também podemos aplicar essa mensagem em nossas próprias vidas e carreiras. Como cirurgiões, somos lembrados da nossa obrigação de desempenhar cada procedimento com diligência e dedicação, e ao mesmo tempo, liderar nossas equipes com empatia e coragem. A mensagem de Lord Nelson nos inspira a superar desafios, enfrentar pressões e trabalhar juntos para alcançar os melhores resultados possíveis em nossas práticas médicas. Ao seguir esse exemplo atemporal de liderança podemos oferecer o melhor atendimento possível aos nossos pacientes e à sociedade.
Ortodoxia Cirúrgica
Embora “Ortodoxia” seja uma obra filosófica e teológica escrita por G.K. Chesterton, suas ideias podem ter algumas aplicações interessantes e reflexivas no cotidiano da cirurgia. Claro que a relação direta pode não ser evidente, mas certos princípios filosóficos podem fornecer perspectivas valiosas para os cirurgiões. Aqui estão algumas maneiras pelas quais as ideias de “Ortodoxia” podem ser aplicadas no contexto cirúrgico:
- Valorizar o pensamento paradoxal: A cirurgia é uma disciplina complexa e muitas vezes ambígua, onde os médicos devem tomar decisões cruciais em situações desafiadoras. Valorizar o pensamento paradoxal pode ajudar os cirurgiões a considerar opções diversas e até opostas antes de tomar decisões importantes.
- Reconhecimento da complexidade humana: Chesterton destaca a importância de compreender a natureza complexa da realidade. No contexto cirúrgico, isso se traduz em tratar cada paciente como um indivíduo único, com suas próprias circunstâncias médicas, emocionais e sociais. Isso pode ajudar os cirurgiões a abordar cada caso com uma mente aberta e livre de preconceitos.
- Equilíbrio entre tradição e inovação: Assim como Chesterton valoriza a tradição cultural, os cirurgiões podem se beneficiar de uma abordagem equilibrada entre as técnicas tradicionais e as inovações médicas. Combinar o conhecimento estabelecido com as mais recentes pesquisas e tecnologias pode levar a melhores resultados para os pacientes.
- Enfrentar a incerteza: A cirurgia pode ser imprevisível, e os resultados nem sempre são garantidos. A ortodoxia de Chesterton nos encoraja a aceitar a incerteza e a enfrentar os desafios com coragem e confiança. Essa mentalidade pode nos ajudar a enfrentar situações complicadas e se adaptar a cenários imprevistos.
- Importância da ética e moralidade: Chesterton enfatiza a importância da moralidade e da virtude. Na cirurgia, esses princípios são essenciais para garantir a melhor qualidade de atendimento ao paciente, respeitando sempre a dignidade e os direitos humanos.
- Valorizar a imaginação: A imaginação é uma parte essencial do trabalho cirúrgico, permitindo aos médicos visualizar procedimentos, simular situações e pensar em soluções criativas. A capacidade de imaginar possibilidades pode ajudar os cirurgiões a planejar cuidadosamente cada intervenção.
Embora “Ortodoxia” não tenha sido escrito com o objetivo específico de se aplicar à cirurgia, as ideias e princípios contidos na obra podem inspirar uma reflexão mais profunda e nos guiar de forma mais consciente, sensível e equilibrada.
As virtudes cardinais cirúrgicas
A prática cirúrgica é uma forma de arte que exige habilidade técnica e precisão, mas vai além disso. Os cirurgiões não apenas dominam as técnicas e procedimentos, mas também são desafiados a aplicar virtudes cardinais em cada etapa do ato operatório. A diérese, exérese, hemostasia e síntese, as quatro fases cruciais da cirurgia, podem ser vistas como um reflexo das virtudes cardinais: prudência, justiça, fortaleza e temperança. Vamos explorar como essas virtudes se manifestam na rotina de um cirurgião comprometido com o bem-estar dos pacientes.
Diérese (A Prudência como Guia) : A primeira etapa da cirurgia, a diérese, é o momento em que o cirurgião realiza uma incisão precisa para acessar o local a ser tratado. A prudência, virtude da sabedoria prática, entra em cena através do domínio da anatomia. O cirurgião deve avaliar cuidadosamente cada caso, analisar os riscos e tomar decisões fundamentadas. A prudência orienta a escolha das melhores abordagens cirúrgicas, levando em consideração a saúde geral do paciente, suas necessidades individuais e o objetivo final da intervenção.
Exérese (A Justiça na Busca pelo Equilíbrio): Na fase de exérese, o cirurgião remove tecidos ou estruturas comprometidas pela doença. Aqui, a justiça desempenha um papel essencial. O cirurgião deve agir com equidade, buscando remover apenas o que é necessário, sem excessos ou negligências. A justiça implica em tratar cada paciente com equidade, respeito e imparcialidade, levando em consideração os melhores interesses do indivíduo e buscando o bem comum. É um compromisso em garantir que o procedimento cirúrgico seja realizado com integridade e sempre em benefício do paciente.
Hemostasia (A Fortaleza para Enfrentar Desafios) : Durante a fase de hemostasia, o cirurgião aplica técnicas para controlar o sangramento e garantir um campo cirúrgico claro. Nesse momento, a fortaleza se faz presente. A cirurgia pode apresentar situações imprevistas, complicações ou momentos de grande pressão. A fortaleza permite ao cirurgião manter-se firme, agir com coragem diante de adversidades e tomar decisões rápidas, mas sábias, para proteger a vida e o bem-estar do paciente. A fortaleza é a virtude que impulsiona o cirurgião a enfrentar desafios com firmeza e superar obstáculos através de uma alma inabalável, mantendo durante todo o procedimento uma determinação com o melhor prognóstico do paciente.
Síntese (A Temperança na Busca do Equilíbrio Final) : A última etapa, a síntese, envolve a restauração da integridade do tecido por meio de suturas ou outros meios. Nesse momento, a temperança se revela. A temperança é a virtude que permite ao cirurgião exercer controle e moderação, evitando excessos e buscando a harmonia. A escolha adequada do material de sutura, a técnica precisa e o cuidado meticuloso são fundamentais. A temperança assegura que a finalização do ato operatório seja feita com prudência, justiça e fortaleza, considerando o bem-estar a longo prazo do paciente.
Logo, podemos concluir que a prática da cirurgia transcende a habilidade técnica e exige o cultivo das virtudes cardinais. A prudência, a justiça, a fortaleza e a temperança tornam-se guias éticos para o cirurgião comprometido com o cuidado ao paciente. A diérese, exérese, hemostasia e síntese, cada uma refletindo uma virtude cardinal, são etapas cruciais em busca da excelência médica. Quando o cirurgião incorpora essas virtudes em sua rotina, ele se torna não apenas um técnico habilidoso, mas um verdadeiro médico que busca o bem-estar e a cura integral do paciente. A arte da cirurgia, assim, se revela não apenas como uma expressão de destreza manual, mas como uma expressão do cuidado compassivo e virtuoso que o médico oferece ao paciente.
10 Anatomical Aspects for Prevention the Bile Duct Injury
Essential aspects to visualize and interpret the anatomy during a cholecystectomy:
1. Have the necessary instruments for the procedure, with adequate positioning of the trocars and a 30-degree optic.
2. Cephalic traction of the gallbladder fundus and lateral traction (pointing to the patient’s right shoulder), to reduce redundancy of the infundibulum.
3. Puncture and evacuation of the gallbladder to improve its retraction, in cases where traction cannot be performed easily (acute cholecystitis).
4. Lateral and caudal traction of the infundibulum, for correct exposure of Calot’s triangle, exposing the CD and artery.
5. “Critical view of Safety” to avoid misidentification of the bile ducts, ensuring that only two structures (CD and artery) are attached to the gallbladder. For this, they must be dissected separately, and the proximal third of the gallbladder must be moved from its fossa, to ensure that there is no anatomical variant there.
6. Systematic use of intraoperative cholangiography. Ideally by transcystic route or possibly by a puncture of the gallbladder.
7. Ligation of the cystic duct with knots (“endoloop”) to prevent migration of metallic clips that could condition a postoperative leak.
8. In case of severe inflammation of the gallbladder pedicle, with its retraction or lack of recognition of cystic structures, a subtotal cholecystectomy might be indicated.
9. In case of hemorrhage, avoid indiscriminate clip placement and or blind cautery. Opt for compressive maneuvers and, once the bleeding site has been identified, evaluate the best method of hemostasis.
10. If the surgeon is not able to resolve the injury caused, it is always better to ask for help from a colleague, and if necessary, to refer the patient to a specialized center.
#SafetyFirst
The main goal in the postoperative management of BDI is to control sepsis in the first instance and to convert an uncontrolled biliary leak into a controlled external biliary fistula to achieve optimal local and systemic control. Definitive treatment to re-establish biliary continuity will be deferred once this primary goal is achieved and should not be obsessively pursued in the acute phase. The factors that will determine the initial presentation of a patient with a BDI in the postoperative stage are related to the time elapsed since the primary surgery, the type of injury, the mechanism of injury, and the overall general condition of the patient.
Sobre o FUTURO
Aos cinco anos, o que você queria ser quando crescesse?
Médico.
Tudo começou quando eu tinha por volta de 4 – 5 anos e após um acidente domiciliar, precisei passar por uma cirurgia na mão. A forma como aquele profissional que nos atendeu acalmou a angústia dos meus pais e tratou com habilidade o ferimento me marcou profundamente. Apesar de não ter ideia do que isso significaria na minha jornada futura, aquele sentimento de ação e resolução se tornou uma paixão que me acompanha até hoje.

Anatomia Cirúrgica da REGIÃO INGUINAL
A hérnia inguinal é uma condição comum que ocorre quando um órgão abdominal protraí através de uma fraqueza na parede abdominal na região abdominal. O orifício miopectineal é a principal área de fraqueza na parede abdominal onde a hérnia inguinal pode se desenvolver. O conhecimento da anatomia da parede abdominal é importante para entender a patofisiologia da hérnia inguinal e para ajudar no diagnóstico e tratamento dessa condição médica comum.
GASTROSTOMY: INDICATIONS, TECHNICAL DETAILS AND POSTOPERATIVE CARE.
Ozimo Pereira Gama Filho
Adjunct Professor at the Federal University of Maranhão
No conflict of interest
- INTRODUCTION
- HISTORICAL ASPECTS
- ANATOMICAL FUNDAMENTALS
- INDICATIONS
- TYPES & TECHNIQUES
- ADVERSE EVENTS
- CUSTO RATIO x EFFECTIVENESS
- CONCLUSIONS
SUMMARY
In the last decade, the use of gastrostomies has been widely indicated as the preferred form of access to the gastrointestinal tract for feeding in chronic conditions and during recovery from acute conditions such as trauma. Together with this increase in indications, new techniques have been developed that have made gastrostomies simpler and less risky. From the classical technique of Stamm performed by laparotomy, two new alternatives that do not require laparotomy emerged: percutaneous endoscopic gastrostomy (PEG) and fluoroscopy gastrostomy. Its main benefit is to avoid a laparotomy, with less associated postoperative pain and earlier return of gastrointestinal function. Although peg is currently widely accepted as the insertion technique of choice due to its simplicity and efficacy, there are patients who are not candidates for an endoscopic approach. In this article we seek to clarify the indications, technical aspects and perioperative care of patients undergoing gastrostomy.
Keywords: Surgical Procedures; Ostomies; Gastrostomy.
Area of Knowledge: General Surgery
- INTRODUCTION
The main indication for enteral or parenteral feeding in the perioperative period is the provision of nutritional support to supply the metabolism of patients with inadequate oral intake. Enteral feeding is the preferred method in relation to parenteral feeding in patients with gastrointestinal dysfunction in the perioperative period due to the inherent risks associated with parenteral nutritional support, such as: infectious complications of the access routes, higher operational cost, and the inability to parenteral nutrition to provide adequate enteral stimulation and subsequent involvement of the intestinal defense barrier [1,2]. In addition, enteral feeding may decrease the risk of bacterial translocation and corresponding bacteremia [3]. Gastric nutritional support is the most common type used. Access to insert the gastrostomy probe can be achieved using endoscopy, interventional radiologia, or surgical techniques (open or laparoscopic). However, since its description in the 1980s [4], percutaneous endoscopic gastrostomy (PEG) is currently considered the method of choice for medium and long-term enteral support.
1.1 Objective: This article reviews the current knowledge about GOSTROSTOMIA in the medical literature, emphasizing the technical and perioperative aspects.
- HISTORICAL ASPECTS
In 876, Verneoil [5] successfully made the first gastrostomy in humans. Since then, several technical modifications have been suggested, such as witzel’s technique in 1891, in which a subseroso tunnel is made on the probe [6]. Stamm, in 1894 [7], described one of the most performed techniques today and in the history of surgical gastrostomy, which consists in the making of suture in a pouch to invaginate the probe inserted into the stomach [8]. In 1980, percutaneous endoscopic gastrostomy was described by Gauderer et al. [4] , which transformed the technique of making gastrostomy.
- ANATOMICAL FUNDAMENTALS
The stomach is a J-shaped dilated cylindrical organ that rests in the left epigastric and hypochodrial region of the abdomen at the level of the first lumbar vertebra. It is previously limited by the left hemidiaphragm, the left lobe of the liver and a triangular portion of the anterior abdominal wall. Subsequently, the pancreas, left kidney and adrenal delimit the stomach. The spleen is posterolaterally and the transverse colon is inferior. It is fixed at two points of continuity: gastroesophageal, superiorly and the duodenal, retroperitoneally. Its ligament attachments also help you in fixation to adjacent organs: gastrophemic (diaphragm), hepatogastric or minor omentum (liver), gastrosplenic or gastrolienal (spleen), and gastrocholic or omentum major (transverse colon). The anatomical regions of the stomach can be distinguished as this: começa superiorly in the continuity of the abdominal part of the esophagus and dthe gastroesophageal junction, the cardiac part of the stomach. Soon below this portion, lies the bottom of the stomach that expands to the left extending above thegastroesophageal junction, forming an acute angle with the distal esophagus known as cardiac notches. The body s andextends as a distensible reservoir and forms a medial edge called the smallest curvature to the right and a side edge called the largest curvature on the left. The gastric den of the stomach is not anatomically distinguishable, but it is estimated to be a region of the angular isis along the distal minor curvature to a point along a lower line to the distal major curvature. It thus ends bymouthing r into the pyloric canal limited by the pyloric sphincter, a palpable thickened ring of muscle that is continuous with the first part of the duodenum.
- INDICATIONS
Gastrostomy is used in the following situations:
- Gastric decompression: can be obtained by means of temporary gastrostomy, occasionally recommended, as a complement to large abdominal operations for which gastric stems, prolonged “adynamic ileus” and digestive fistulas are foreshadowed.
- Nutritional Support: b.1 Temporary; indicated when access to the digestive tract is temporarily impaired for recovery and maintenance of nutritional status (E.g. CEsophageal EC); b.2 Definitive; as palliative therapy in patients with unresectable malignant neoplasia of the head and neck, as wellas n degenerative neurological diseases that lead to irreversible disorders of deglutition.
However, the decision to perform a gastrostomy, as well as its route (surgical, radiological or endoscopic) should be individualizedaccording to the needs, diagnosis, life expectancy of the patient and the available hospital logistics. The objective is not only to optimize perioperative recovery, to improve survival and nutritional status of the patient, but also to promote quality of life, which is not necessarily correlated with nutritional improvement only [9]. Therefore, the appropriate indication, like any other surgical intervention, must be clearly establishedand informed before it is performed. Some of the absolute contraindications of gastrostomy are summarized in Table 1. In addition to absolute contraindication conditions, other situations such as the presence of non-obstructive oromyctological oresophageal malignancy, hepatomegaly, splenomegaly, peritoneal dialysis, portal hypertension with gastric varicose veins, and a history of partial gastrectomy are also considered relative contraindications.
| ABSOLUTE CONTRA – INDICATIONS |
| Coagulopatia Severa (INR > 5, Plaquetas < 50.000 e TPT > 50s) |
| Hemodynamic Instability |
| Septic Shock |
| Refractory Ascites |
| Peritonitis |
| Dermatological infection in the upper abdomen |
| Carcinomatose Peritoneal |
| Interposition of organs that prevent gastric access |
| History of Total Gastrectomy |
| Stenosis or Pyloric Obstruction |
| Severe gastroparesis, in cases of indication for nutritional support |
| Absence of Informed Consent |
- TYPES & TECHNIQUES
Currently there are three techniques for performing gastrostomy: radiological, through percutaneous gastrostomy by fluoroscopy, percutaneous endoscopic gastrostomy (PEG) and surgical gastrostomy. Due to the didactic characteristics of this material, we will focus on endoscopic and surgical gastrostomy , which becomes the main option in the following situations: 1) when the patient will already undergo a laparotomy due to some abdominal condition ; 2) impossibility of performing gastroscopy to perform gastrostomy endoscopic percutaneous (PEG) ; 3) in case of peg technical failure; 4) unavailability of resources for the preparation of PEG or percutaneous gastrostomy by fluoroscopy.
- ENDOSCOPIC PERCUTANEOUS GASTROSTOMY
The informed consent form must be obtained from patients or their legal representatives. Patients should fast for a minimum of 8 hours and receive prophylactic antibiotics one hour before proceeding and intravenous administration of 1-2 g of cefazolin is recommended. The technique introduced by Gauderer et al [4] is the most used technique to insert the PEG gastrostomy probe. In this method, a guide wire is used, inserted in the distal gastric chamber through a needle puncture n to the anterior abdominal wall. This guide wire is then seized endoscopically with a handle and then removed through the esophagus and mouth. Subsequently, the guide wire is fixed to the end of the gastrostomy probe and then pulled from the mouth to the esophagus, stomach and then out to the abdominal wall, where it will be fixed.
- SURGICAL GASTROSTOMY
Surgical gastrostomy can be performed in two ways: 1) via laparotomy – the predominant form; and 2) laparoscopic approach. The preparations are the same as the endoscopic pathway.
5.2.1. GASTROSTOMIA At STAMM
After adequate asepsis and antisepsis, with the patient under anesthesia and in horizontal dorsal decubitus, the technical steps are as follows: 1. Median laparotomy (supraumbilical median incision); 2. Identification of the gastric body; 3. Stomach hold with Babcock tweezers (to evaluate the approach of the stomach to the peritoneum); 4. Suture in pouch (circular area of 2cm) – atraumatic absorbable thread; 5. Section of the stomach wall (0.5cm) – (incision with scalpel or Electrocautery in the center of the suture, of sufficient size, for the placement of a probe with 20 to 26 French); 6. Placement of the Gastrostomy probe in the extension of 5-6 cm, followed by suture closure in a pouch); 7. Tie the suture threads in a pouch around the probe; 8. Apply a second suture in a pouch 1cm above the first (seromuscular stitches); 9. Externalization of the probe by counter opening on the left flank; 10. Fix the stomach wall to the abdominal wall in 4 cardinal points (external ration with the two Kocher tweezers used for grip of the alba line and against traction by means of the index fingers of the wall of the left hipochondrio to approach the parietal peritoneum of the gastric wall); 11. Fixation of the probe to the skin (point with nonabsorbable wire); 12. Closure of the abdominal wall (synthesis of the wall with approximation of the alba thread by continuous suture with monofilament thread 1-0 or 2-0 and of the skin with separate points of nylon 3-0); 13. Dressing.
5.2.1 GASTROSTOMIA EM WITZEL
The initial technical steps from gastrostomy to Witzel are like those of gastrostomy to Stamm, including fixation of the probe to the stomach by a pouch suture. Then, the probe is placed on the gastric wall and a tunnel of 8-10 cm is made by seromuscular suture (continuous or with separate points of absorbable or nonabsorbable thread) covering it and externalization is performed by counteropening.
- ADVERSE EVENTS
According to the literature, the rate of complications for different procedures varies due to the heterogeneity of the samples evaluated. For surgical gastrostomy, the reported complication rates are between 1% and 35%, while for percutaneous radiological gastrostomy it is 3% to 11%, and for percutaneous endoscopic, 17%–32%, the main related adverse event is surgical site infection [10, 11]. Although considered a basic procedure, gastrostomy is associated with an extensive list of related technical complications, care and use of the probe. Serious problems related to the technique include separation of the stomach from the abdominal wall (leading to peritonitis), separation of wounds, hemorrhage, infection, lesion of the posterior gastric wall or other organs, and placement of the tube in an inappropriate place of gastric position. Separation of the stomach from the abdominal wall usually occurs due to inadvertent and premature displacement of the tube, particularly with balloon-like devices, or a rupture during a catheter change. It requires immediate attention, being treated with laparotomy, although in selected cases laparoscopic correction is possible. Most complications can be avoided with the careful choice of the type of procedure, from the appropriate ostomy device, considering it an important intervention and using meticulous technique with the proper approximation of the stomach to the abdominal wall and outflow of the probe through a counter-incision (in conventional procedures), thus avoiding probes in the midline or awfully close to the costal edge.
- COST VS. EFFECTIVENESS
A recent study [12] compared the cost associated with the different gastrostomy techniques, and the results of the evaluation showed variable the benefits of each of the individual percutaneous procedures, indicating that surgical gastrostomy was the onerous mais of the three modalities due to higher costs, complications, and recovery time, as well as the endoscopic technique presenting the cost effectiveness ratio.
- CONCLUSIONS
Despite the technique employed, the decision to performa gastrostomy is not based only on the patient’s survival expectancy, because the adequate indication provides a better quality of life even when the survival of the patient after the procedure is severely limited. Therefore, understanding of techniques, indications, complication rates is essential to guide the surgical team in the scope of multidisciplinary care, as well as the education of patients and their caregivers is vital to ensure the correct maintenance of the devices, thus ensuring adequate nutritional intake of the patient and minimizes complication rates.
References
1 Alverdy J, Chi HS, Sheldon GF. The effect of parenteral nutrition in gastrointestinal immunity. The importance of de estimulação enteral. Ann Surg, 1985; 202: 681-684 [PMID:3935061]
2 Deitch EA, Ma WJ, Ma L, Berg RD, Specian RD. Protein malnutrition predisposes to inflammation-induced intestinal origin septic states. Ann Surg, 1990; 211: 560-567; discussion 560-567 [PMID: 2111125]
3 Deitch EA, Winterton J, Li M, Berg R. The intestine as a portal of entry to bacteremia. Role of protein malnutrition. Ann Surg 1987; 205: 681-692 [PMID: 3592811]
4 Gauderer MW, Ponsky JL, Izant RJ. Gastrostomia sem laparotomy: percutaneous endoscopic technique. J Pediatrician Surg, 1980; 15: 872-875 [PMID: 6780678]
5 Anselmo CB, Tercioti Júnior V, Lopes LR, Coelho Neto JS, Andreollo NA. Surgical gastrostomy: current indications and complications in patients of a university hospital. Rev Col Bras Cir. [Internet journal] 2013;40(6). Available in URL: http://www.scielo.br/rcbc
6 Witzel O. For gastric fistula technique. Chir Zbl. 1891;18:601-4.
7 Stamm M. Gastrostomy: a new method. Med News. 1894;65:324.
8 JP grant. Comparison of percutaneous endoscopic gastrostomy com gastrostomia strain. Ann Surg. 1988;207(5):598-603
9 Bannerman E, Pendlebury J, Phillips F, Ghosh S. Cross-sectional and longitudinal study of health-related quality of life after percutaneous gastrostomy. Eur J Gastroenterol Hepatol 2000; 12: 1101-1109 [PMID: 11057455]
10 Möller P, Lindberg CG, Zilling T. Gastrostomy by various techniques: evaluation of indications, outcome and complications. Scand J Gastroenterol. 1999;34(10):1050-4.
11 Clarke E, Pitts N, Latchford A, Lewis S. A major prospective audit of morbidity and mortality associated with food gastrostomies in the community. Clin Nutr. 2017 Apr;36(2):485-490. DOI: 10.1016/j.clnu.2016.01.008. EPub 2016 January 21. PMID: 26874913.
12 Wollman B, D’Agostino HB, Walus-Wigle JR, Easter DW, Beale A (1995) Radiological, endoscopic and surgical gastrostomy: an institutional evaluation and meta-analysis of the literature. Radiology 197: 699–704.
A Arte da Anatomia (EBook)
Desde a Antiguidade, os médicos, anatomistas e artistas se dedicaram a descrever e representar o corpo humano, por meio de desenhos, pinturas, esculturas e outras formas de representação artística. Com o passar dos séculos, houve uma evolução significativa na forma como as ilustrações anatômicas eram produzidas, desde as primeiras representações rudimentares até as ilustrações altamente detalhadas e realistas que temos hoje.
Boa Leitura!!!
Michelangelo Buonarroti
5 Segredos de Anatomia Escondidos na Capela Sistina: A Neuroanatomia Clandestina de Michelangelo
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
Onde a Fé e o Bisturi se Encontram
Na formação médica contemporânea, o estudo da anatomia topográfica num laboratório de dissecação é o rito de passagem fundamental para qualquer cirurgião. No Brasil, apesar dos desafios atuais na captação de cadáveres para estudo nas faculdades de medicina, a dissecação continua a ser a base insubstituível da técnica cirúrgica. Contudo, no século XVI, o estudo do corpo humano além da superfície era um tabu severo, vigiado de perto pelas leis civis e canónicas. Michelangelo Buonarroti, celebrado mundialmente pela sua terribilità — aquela força emocional e técnica avassaladora —, viveu um paradoxo perigoso. O artista profundamente religioso era também um anatomista clandestino. Desafiando os dogmas de sua época, ele dedicou inúmeras noites à dissecação de cadáveres, buscando nos tendões, fáscias e vísceras a geometria da perfeição biológica. O que a humanidade demorou 500 anos para perceber é que Michelangelo não utilizou este conhecimento apenas para conferir realismo hipertrófico às suas figuras; ele codificou verdadeiras lições de neuroanatomia diretamente no teto da Capela Sistina. O santuário do conclave papal guarda um mapa do intelecto humano, sugerindo que a verdadeira centelha divina reside na intrincada arquitetura do sistema nervoso central. Abaixo, dissecamos os 5 segredos anatómicos ocultos nesta obra-prima.
A Anatomia Oculta nos Afrescos
1. O Cérebro como a “Faísca Divina” em A Criação de Adão
Em 1990, o médico norte-americano Frank Meshberger publicou uma descoberta no prestigiado Journal of the American Medical Association (JAMA) que alterou o curso da história da arte e da medicina. Ele demonstrou que a figura de Deus, envolta num manto avermelhado e cercada por anjos, compõe um corte sagital milimetricamente exato de um cérebro humano. A genialidade reside nos detalhes: o contorno inferior do manto segue com precisão o sulco lateral (Fissura de Sylvius). O braço estendido do Criador atravessa exatamente o giro do cíngulo. Um “anjo” posicionado inferiormente desenha o trajeto da artéria basilar, enquanto o pé bifurcado de um querubim representa a glândula pituitária (hipófise). Para Michelangelo, o que Adão recebe no toque não é apenas o fôlego biológico, mas o intelletto — a razão suprema.
2. O Tronco Encefálico no Pescoço de Deus
A análise de A Criação de Adão revela o cérebro macroscópico, mas o painel central A Separação da Luz das Trevas aprofunda-se em estruturas neurais complexas. Em 2010, os investigadores Ian Suk e Rafael Tamargo, neurocirurgiões da Universidade Johns Hopkins, identificaram um detalhe audacioso. O pescoço de Deus exibe uma anatomia irregular que foge à hipertrofia muscular renascentista padrão. Utilizando um escorço dramático (técnica de perspetiva que encurta a figura), Michelangelo camuflou uma visão ventral do tronco encefálico humano. Estão lá, representados com rigor de atlas cirúrgico, o bulbo, a ponte e a estrutura em forma de “Y” característica dos nervos óticos e do quiasma ótico.
3. Arquitetura Neural: Ventrículos e o Corpo Caloso
A investigação ganhou novos contornos em 2015, quando Ciurea et al. identificaram como o mestre utilizou a técnica do chiaroscuro (luz e sombra) aplicando o princípio da Gestalt para ocultar formas. Na mesma cena da Separação da Luz das Trevas, os olhos treinados de um neuroanatomista conseguem completar as figuras:
- Área A: O quarto ventrículo perfeitamente delineado.
- Área B: A ponte de Varólio, formada pela curvatura da coxa direita da divindade, exibindo a oliva pontina e o sulco colateral anterior.
- Área C: O manto superior que emula uma imagem espelhada do corpo caloso (a via de comunicação entre os hemisférios cerebrais). A inversão da imagem serviu para manter o fluxo estético sem perder a mensagem anatómica.
4. O Anatomista Clandestino de Santo Spirito
Para que um artista atingisse tal nível de detalhe no século XVI — superando as obras de Galeno (que se baseavam em dissecações de macacos e porcos) —, a observação clínica superficial não bastaria. Aos 18 anos, Michelangelo firmou um acordo secreto com o prior da Igreja de Santo Spirito, em Florença. Em troca da escultura de um crucifixo de madeira, o prior permitia-lhe dissecar cadáveres humanos no hospital da igreja durante a madrugada. armado com um bisturi primitivo, ele antecipou descobertas que a medicina oficial, com Andreas Vesalius, só documentaria décadas mais tarde.
5. Uma Mensagem de Protesto e Subversão (A Costela Oculta)
O anatomista era também um livre-pensador influenciado pelo Neoplatonismo florentino. O investigador Deivis Campos (2019) identificou uma “costela extra” no lado esquerdo do torso de Adão. Ao invés de pintar uma costela a ser removida para a criação de Eva (como ditava o dogma), Michelangelo pintou uma costela extra e oculta. Esta representação anatómica subversiva sugere que as essências feminina e masculina coexistem na natureza humana original, uma alusão ao misticismo da Cabala e um protesto velado contra a interpretação literal das escrituras.
Pontos-Chave para a Formação Médica
- A Observação é a Base da Medicina: O génio de Michelangelo prova que a capacidade de observar minuciosamente a anatomia é o que separa o conhecimento superficial da verdadeira maestria.
- Integração Arte e Ciência: As estruturas neurais (fissura de Sylvius, tronco encefálico, quiasma ótico) pintadas no século XVI mostram que a medicina e a arte nasceram da mesma curiosidade intrínseca sobre o milagre da vida.
- O Cérebro como Sede da Consciência: Ao colocar o sistema nervoso central no epicentro do ato da Criação, Michelangelo antecipou em séculos a premissa da neurociência moderna: somos o nosso cérebro.
Conclusões Aplicadas à Prática do Cirurgião
Para o cirurgião do aparelho digestivo, ou de qualquer outra especialidade, a história de Michelangelo traz uma lição de humildade e reverência. Quando entramos no centro cirúrgico e realizamos uma incisão, não estamos apenas a manipular tecidos e órgãos; estamos a interagir com a mais sublime e complexa arquitetura do universo conhecido. Se o maior segredo do Renascimento — o mapeamento do nosso intelecto na morada do papado — permaneceu oculto por cinco séculos à vista de todos, devemos questionar-nos diariamente: o que mais estamos a falhar em enxergar nos nossos doentes devido à pressa ou à desatenção clínica? A medicina de excelência exige o olhar crítico do cientista e a sensibilidade meticulosa do artista.
“Onde o espírito não conduz a mão do artista, não há arte. E onde o conhecimento da anatomia não conduz a mão do médico, não há cura. A estrutura do corpo humano é a obra-prima inquestionável do Criador.” — Adaptado dos pensamentos do Renascimento Médico.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
Specific Competence of Surgical Leadership
Surgeons are uniquely prepared to assume leadership roles because of their position in the operating room (OR). Whether they aspire to the title or not, each and every surgeon is a leader, at least within their surgical team. Their clinical responsibilities offer a rich variety of interpretations that prepare them for a broader role in health care leadership. They deal directly with patients and their families, both in and out of the hospital setting, seeing a perspective that traditional health care administrative leaders rarely experience. They work alongside other direct providers of health care, in varied settings, at night, on weekends, as well as during the typical workday. They understand supply-chain management as something more than lines on a spreadsheet.
The Challenges for a Surgical Leader
Surgeons prefer to lead, not to be led. Surgical training has traditionally emphasized independence, self-reliance, and a well-defined hierarchy as is required in the OR. However, this approach does not work well outside the OR doors. With colleagues, nurses, staff, and patients, they must develop a collaborative approach. Surgeons are entrusted with the responsibility of being the ultimate decision maker in the OR. While great qualities in a surgeon in the OR, it hinders their interactions with others. They have near-absolute authority in the OR, but struggle when switching to a persuasive style while in committees and participating in administrative activities. Most surgeons do not realize they are intimidating to their patients and staff. With patients, a surgeon needs to be empathetic and a good listener. A surgeon needs to slow the pace of the discussion so that the patient can understand and accept the information they are receiving. As perfectionists, surgeons demand a high level of performance of themselves. This sets them up for exhaustion and burnout, becoming actively disengaged, going through the motions, but empty on the inside. Given the many challenges surgeons face, it is difficult for them to understand the leadership role, given its complex demands.
Specific Competencies
Authority
Although teams and all team members provide health care should be allowed input, the team leader makes decisions. The leader must accept the responsibility of making decisions in the presence of all situations. They will have to deal with conflicting opinions and advice from their team, yet they must accept that they will be held accountable for the performance of their team. The surgeon–leader cannot take credit for successes while blaming failures on the team. Good teamwork and excellent communication do not relieve the leader of this responsibility.
Leadership Style
A surgeon often has a position of authority based on their titles or status in an organization that allows them to direct the actions of others. Leadership by this sort of mandate is termed “transactional leadership” and can be successful in accomplishing specific tasks. For example, a surgeon with transactional leadership skills can successfully lead a surgical team through an operation by requesting information and issuing directives. However, a leader will never win the hearts of the team in that manner. The team will not be committed and follow through unless they are empowered and feel they are truly heard. A transformational leader is one who inspires each team member to excel and to take action that supports the entire group. If the leader is successful in creating a genuine atmosphere of cooperation, less time will be spent giving orders and dealing with undercurrents of negativity. This atmosphere can be encouraged by taking the time to listen and understand the history behind its discussion. Blame should be avoided. This will allow the leader to understand the way an individual thinks and the group processes information to facilitate the introduction of change. While leadership style does not guarantee results, the leader’s style sets the stage for a great performance. At the same time, they should be genuine and transparent. This invites the team members to participate, creating an emotional connection. Leaders try to foster an environment where options are sought that meet everyone’s desires.
Conflict Management
Conflict is pervasive, even in healthy, well-run organizations and is not inherently bad. Whether conflict binds an organization together or divides it into factions depends on whether it is constructive or destructive. A good leader needs to know that there are four essential truths about conflict. It is inevitable, it involves costs and risks, the strategies we develop to deal with the conflict can be more damaging than the conflict itself, and conflict can be permanent if not addressed. The leader must recognize the type of conflict that exists and deal with the conflict appropriately. Constructive discussion and debate can result in better decision making by forcing the leader to consider other ideas and perspectives. This dialog is especially helpful when the leader respects the knowledge and opinions of team members with education, experience, and perspective different from the leader’s. Honesty, respect, transparency, communication, and flexibility are all elements that a leader can use to foster cohesion while promoting individual opinion. The leader can create an environment that allows creative thinking, mutual problem solving, and negotiation. These are the hallmarks of a productive conflict. Conflict is viewed as an opportunity, instead of something to be avoided.
Communication Skills
Communication is the primary tool of a successful leader. On important topics, it is incumbent on the leader to be articulate, clear, and compelling. Their influence, power, and credibility come from their ability to communicate. Research has identified the primary skills of an effective communicator. They are set out in the LARSQ model: Listening, Awareness of Emotions, Reframing, Summarizing, and Questions. These are not set in a particular order, but rather should move among each other freely. In a significant or critical conversation, it is important for a leader to listen on multiple levels. The message, body language, and tone of voice all convey meaning. You cannot interrupt or over-talk the other side. They need an opportunity to get their entire message out. Two techniques that enhance listening include pausing and the echo statement. Pausing before speaking allows the other conversant time to process what they have said to make sure the statement is complete and accurate. Echo statements reflect that you have heard what has been said and focuses on a particular aspect needing clarification. Good listening skills assure that the leader can get feedback that is necessary for success.
Vision, Strategy, Tactics, and Goals
One of the major tasks of a leader is to provide a compelling vision, an overarching idea. Vision gives people a sense of belonging. It provides them with a professional identity, attracts commitment, and produces an emotional investment. A leader implements vision by developing strategy that focuses on specific outcomes that move the organization in the direction of the vision. Strategy begins with sorting through the available choices and prioritizing resources. Through clarification, it is possible to set direction. Deficits will become apparent and a leader will want to find new solutions to compensate for those shortfalls. For example, the vision of a hospital is to become a world class health care delivery system. Strategies might include expanding facilities, improving patient satisfaction, giving the highest quality of care, shortening length of hospital stay with minimal readmissions, decreased mortality, and a reduction in the overall costs of health care. Tactics are specific behaviors that support the strategy with the aim to achieve success. Tactics for improving patient satisfaction may include reduced waiting time, spending more time with patients, taking time to communicate in a manner that the patient understands, responding faster to patient calls, etc. These tactics will then allow a leader to develop quantitative goals. Patient satisfaction can be measured. The surgical leader can then construct goals around each tactic, such as increasing satisfaction in specific areas. This information allows a surgical leader to identify barriers and they can take steps to remedy problem areas. This analysis helps a leader find the weakest links in their strategies as they continue toward achieving the vision.
Change Management
The world of health care is in continuous change. The intense rate of political, technical, and administrative change may outpace an individual’s and institution’s ability to adapt. Twenty-first century health care leaders face contradictory demands. They must navigate between competing forces. Leaders must traverse a track record of success with the ability to admit error. They also must maintain visionary ideas with pragmatic results. Individual accountability should be encouraged, while at the same time facilitating teamwork. Most leaders do not understand the change process. There are practical and psychological aspects to change. From an institutional perspective, we know that when 5% of the group begins to change, it affects the entire group. When 20% of a group embraces change, the change is unstoppable.
Succession Planning and Continuous Learning
An often-overlooked area of leadership is planning for human capital movement. As health care professionals retire, take leaves of absences, and move locations, turmoil can erupt in the vacuum. Leaders should regularly be engaging in activities to foster a seamless passing of institutional knowledge to the next generation. They also should seek to maintain continuity to the organization. Ways to accomplish this include senior leaders actively exposing younger colleagues to critical decisions, problem solving, increased authority, and change management. Leaders should identify promising future leaders, give early feedback for areas of improvement, and direct them toward available upward career tracks. Mentoring and coaching help prepare the younger colleagues for the challenges the institution is facing. Teaching success at all levels of leadership helps create sustainable high performance.
Gallstone Ileus
INTRODUCTION
Gallstone ileus is a misnomer: this condition is not a physiologic ileus at all, but a mechanical obstruction of the intestinal lumen (most commonly the small bowel) by a large gallstone that has passed through a cholecystoenteric fistula. Cholecystoenteric fistulae may occur from the gallbladder to the adjacent luminal viscera-duodenum (most common), stomach, or colon. Gallstone obstruction of the stomach at the pylorus is known as Bouveret’s syndrome. Cholecystocolic fistula is less common. Colonic obstruction in this situation typically occurs at the sigmoid colon. Most common is cholecystoduodenal fistula, with a large (usually >2 cm) gallstone passing through the small bowel and becoming lodged in the terminal ileum. Cholecystoenteric fistula is felt to be caused by a combination of pressure, necrosis, and inflammation with chronic longstanding gallstone disease. Up to 25% of patients who develop gallstone ileus will harbor multiple stones in the alimentary tract; therefore, a close inspection of the entire intestine is important at the time of operation.
CLINICAL PRESENTATION
Over the past few years, however, the incidence of gallstone ileus has been shown to be greater than previously thought. Several recent large population-based series have found that gallstone ileus accounts for approximately 0.1% of all small bowel obstructions. The disease usually affects women (70%) and those in the seventh or eighth decade of life. Most patients present with bloating, crampy abdominal pain, and vomiting, symptoms typical of mechanical small bowel obstruction. A careful history may reveal earlier episodic colicky right upper quadrant abdominal pain consistent with gallstone disease. The classic finding on plain abdominal radiograph is that of Rigler’s triad (pneumobilia, dilated small bowel loops with air-fluid levels, and a large, calcified gallstone in the lumen of the small bowel). Currently, computed tomography (CT) is used ubiquitously. CT has 99% accuracy for diagnosing gallstone ileus. Typical CT findings include pneumobilia, dilated loops of small bowel with air-fluid levels consistent with small bowel obstruction, and transition point with the ectopic stone always visible radiologically.
MANAGEMENT
Operation is required for all patients with gallstone ileus, as spontaneous passage of these large stones is rare once the patient has become symptomatic. It is crucial to optimize the patient physiologically as much as possible in this semi urgent situation, with the understanding that most gallstone ileus patients are elderly and commonly have numerous medical comorbidities. Two contemporary series of registry data have expanded our understanding of gallstone ileus. This condition was once thought to be relatively rare; however, the National Inpatient Sample study identified 3268 gallstone ileus patients, which accounts for approximately 0.1% of all patients admitted to the hospital with mechanical small bowel obstruction during this time period. In this series, overall hospital mortality was substantial at 6.7%. Mortality was significantly higher in patients who underwent cholecystectomy and closure of the biliary fistula compared to those who simply had small bowel obstruction addressed by cholecystolithotomy. Overall, 77% of the 3268 patients had small bowel obstruction pathology treated and the remaining 23% had biliary fistula closed and cholecystectomy at the same operation. An interesting finding was the substantial incidence of postoperative renal insufficiency, or approximately 30% in the entire group of patients. The latter finding highlights the need for preoperative resuscitation and close attention to postoperative fluid management. The surgeon must consider carefully feasibility of same operation intervention to repair biliary pathology: It is safe to defer biliary operation to a later date with a second staged operation. When this strategy of two stage operation is selected, surgeons should consider and counsel their patient regarding the substantial incidence of recurrent biliary symptoms.
Tratamento Cirúrgico da ACALASIA ESOFÁGICA
A acalasia, definida como a falha ou relaxamento incompleto do esfíncter esofágico inferior (EEI), acompanhada de aperistalse do corpo esofágico na ausência de obstrução mecânica, é o tipo mais comum de distúrbio da motilidade esofágica. Tem uma incidência de 1 em 100.000 pessoas, com uma prevalência de 10 em 100.000. Não há diferença na prevalência de gênero entre as idades de 30 e 60 anos. A causa primária da acalasia ainda é indeterminada, mas acredita-se que surja da degeneração das células ganglionares inibitórias no plexo miontérico do EEI e corpo esofágico. Fatores associados a um risco aumentado de acalasia incluem distúrbios virais/neurodegenerativos, síndrome de Down, diabetes mellitus tipo 1, hipotireoidismo e condições autoimunes, como a síndrome de Sjögren, o lúpus eritematoso sistêmico e a uveíte. Os casos familiares são raros.
O diagnóstico de acalasia deve ser suspeitado em pacientes com disfagia para sólidos e líquidos que não melhora com o uso de inibidores da bomba de prótons. Se não for tratada, a acalasia é uma doença progressiva que pode evoluir para megaesôfago e está associada a um aumento do risco de carcinoma de células escamosas do esôfago. Embora não existam critérios padronizados para determinar a gravidade da doença, o diâmetro e a confirmação do esôfago dentro da cavidade torácica são geralmente considerados os dois principais fatores. A escala de Eckardt é uma escala frequentemente usada para avaliar a gravidade da doença e a eficácia da terapia. Qualitativamente, a acalasia grave é definida como um diâmetro esofágico maior que 6 cm; a acalasia em estágio avançado inclui a angulação distal, um esôfago sigmóide/tortuoso com diâmetro maior que 6 cm ou um megaesôfago com diâmetro maior que 10 cm. Aproximadamente 5% a 15% das pessoas com acalasia evoluem para acalasia em estágio avançado, são geralmente resistentes aos tratamentos endoscópicos e cirúrgicos iniciais e, em última instância, requerem uma esofagectomia.
MIOTOMIA Á HELLER
A miotomia laparoscópica de Heller (LHM) com fundoplicatura parcial, desenvolvida como uma alternativa minimamente invasiva à miotomia anterior aberta tradicional e posteriormente à miotomia toracoscópica, tem sido o padrão ouro para o tratamento da acalasia nas últimas três décadas. O objetivo da miotomia é abrir completamente o EEI e aliviar a disfagia. A LHM proporciona alívio sintomático inicial da disfagia em cerca de 90% dos pacientes com tipos I e II de acalasia e em 50% dos pacientes com tipo III de acalasia, enquanto diminui as taxas de refluxo pós-operatório. Em comparação com os procedimentos de miotomia aberta, a LHM está associada a menor dor pós-operatória, menor tempo de internação hospitalar e retorno mais precoce à função. A LHM é indicada como tratamento de primeira linha para todos os candidatos à cirurgia com acalasia que estejam dispostos a se submeter à cirurgia ou para aqueles que falharam na dilatação endoscópica. A operação consiste em dividir os músculos do EEI, seguida por uma fundoplicatura para diminuir o refluxo pós-operatório. A fundoplicatura parcial é favorecida em relação à fundoplicatura total porque reduz a falha do tratamento. As duas principais complicações da cirurgia são perfuração da mucosa e DRGE.
Surgical Management of Chronic Pancreatitis
Chronic pancreatitis is a progressive, destructive inflammatory process that ends in destruction of the pancreatic parenchyma resulting in malabsorption, diabetes mellitus, and severe pain. The etiology of chronic pancreatitis is multifactorial. About 65–70% of patients have a history of alcohol abuse, the remaining patients are classified as idiopathic chronic pancreatitis (20–25%), including tropical pancreatitis, a major cause of childhood chronic pancreatitis in tropical regions, or unusual causes including hereditary pancreatitis, cystic fibrosis, and chronic pancreatitis-associated metabolic and congenital factors. Current evidence suggests that a combination of predisposing factors, including environmental, toxic, and genetic, are involved in most patients rather than one single factor. The best-known hypotheses about the pathogenesis of chronic pancreatitis include necrosis-fibrosis, toxic-metabolic, oxidative stress, plug and stone formation with duct obstruction, and primary duct obstruction. Repeated episodes of inflammation initiated by autodigestion, one or more episodes of severe pancreatitis, oxidative stress, and/or toxic-metabolic factors lead to activation and continued stimulation of parenchymal pancreatic stellate cells. These stellate cells cause the fibrosis characteristic of chronic pancreatitis. Nevertheless, multiple hypotheses exist to explain the pathophysiology in the various subgroups of patients with chronic pancreatitis, but to date there is no single unifying theory.
OPERATIVE TREATMENT
There are several different concepts for the operative treatment of chronic pancreatitis. The concept of preservation of functioning pancreatic parenchyma (drainage operations) would be the goal for protection against further loss of pancreatic function. The second main concept is based on resective procedures either in the situation where there is no dilation of the pancreatic duct, if the pancreatic head is enlarged, or if a pancreatic carcinoma is suspected in the setting of chronic pancreatitis. These two concepts involve different operative procedures.
Drainage procedures
Sphincterotomy of the pancreatic duct was one of the first operative procedures proposed for patients with a dilated pancreatic duct in chronic pancreatitis with presumed obstruction or stenosis at the papilla Vater. Unfortunately, this procedure was associated with minimal lasting success for the amelioration of pain, indicating that a stenosis at the papilla of Vater is not the cause of pain in chronic pancreatitis nor the cause of ductal dilation. In contrast, direct ductal-enteric drainage by the original Puestow procedure or its modification by Partington and Rochelle is more successful in patients with chronic pancreatitis and a dilated pancreatic duct. The original Puestow procedure included resection of the tail of the pancreas with filleting open the pancreatic duct proximally along the body of the pancreas with anastomosis to a Roux-en-Y loop of jejunum. Partington and Rochelle modified the Puestow procedure by eliminating the resection of the pancreatic tail. A recent procedure involves a wedge-shaped opening of the pancreatic duct (even when the duct is <5 mm) with a subsequent pancreatico-jejunostomy. The preservation of functional tissue and reduction of operative mortality to less than 1% and morbidity to less than 10% are the goals and benefits of this operation. Unfortunately, large series have reported persistence or recurrence of pain at long-term follow up (>5 year) in 30–50% of patients; in addition, patients with a dominant mass in the head of the pancreas and a non-dilated pancreatic duct do not appear to profit from a drainage procedure at all. A recent randomized controlled trial demonstrated that operative drainage in selected patients with a large duct was more effective than endoscopic treatment in patients with obstruction of the pancreatic duct.
Pancreatic Resections
Pancreatoduodenectomy (Kausch-Whipple procedure). For many surgeons, a pancreatoduodenectomy is the gold standard for patients with the pain of chronic pancreatitis, although the newer, duodenum-preserving procedures are good (and possibly better) alternatives as well (see below). The approach of resection of the proximal gland is based on Longmire’s tenet that the ‘‘pacemaker’’ of pain is in the head of the pancreas. The indications for pancreatoduodenectomy in patients with chronic pancreatitis and pain are:
(1) a non-dilated pancreatic duct (diameter < 6 mm measured in the body of the gland),
(2) an enlarged head of the pancreas, often containing cysts and calcifications,
(3) a previous, ineffective ductal drainage procedure, and/or
(4) when there is the possibility of malignancy in the head of the gland.
This latter subgroup comprises up to 6–10% of patients undergoing operative intervention for chronic pancreatitis. After pancreatoduodenectomy, > 80% of patients have permanent pain relief, which is greater than after a drainage operation. In experienced centers, a pancreatoduodenectomy can be performed with a low operative mortality rate (< 2%), and a morbidity of 40%. Although the classic pancreatoduodenectomy has these advantages, there is some long-term morbidity in chronic pancreatitis patients, especially regarding quality of life. In addition to development of diabetes, patients experience postoperative digestive dysfunction, including dumping, diarrhea, peptic ulcer, and dyspeptic complaints. To address these effects of the classic pancreatoduodenectomy which involved a distal gastrectomy, ‘‘organ-preserving’’ operations like the pylorus-preserving pancreatoduodenectomy. Symptoms of dumping and bile-reflux gastritis can be decreased by preserving the stomach, the pylorus, and the first part of the duodenum. In addition, regarding quality of life, a pylorus-preserving technique provides better results than the classic pancreatoduodenectomy procedure; weight gain occurs in 90% of the patients postoperatively while still leading to long-lasting pain relief in 85–90% of the patients. Pylorus-preserving resections, however, appear to have a greater incidence of transient delayed gastric emptying postoperatively (20–30% of the patients) as well as the risk of cholangitis and the long-term occurrence of exocrine and endocrine pancreatic insufficiency (seen in >45% of patients), representing the possible drawbacks of this operation in chronic pancreatitis patients. The relevant studies (level I and II) comparing classic with pylorus preserving pancreatoduodenectomy could not demonstrate a clear advantage for either resection. One should remember, however, that pancreatoduodenectomy was originally introduced to treat malignant pancreatic or periampullary disease by an oncologic resection. Therefore, for a benign disorder such as chronic pancreatitis, there is no reason – other than the occasional inability to exclude pancreatic cancer definitely – to remove peripancreatic organs (the distal stomach, the duodenum, and the extrahepatic bile ducts), which are involved only secondarily in chronic pancreatitis. This concept stimulated the development of organ-preserving pancreatic resections.
Duodenum-preserving pancreatic head resection (DPPHR).
This procedure addresses patients with a dominant mass in the head of the pancreas with or without a dilated main pancreatic duct. The duodenum-preserving resection (Beger procedure) includes a ventral dissection and dorsal mobilization of the head of the pancreas. After division of the pancreas anterior to the porto-mesenteric vein (as with a pancreatoduodenectomy), the resection is carried out toward the papilla of Vater. A subtotal resection of the pancreatic head is carried out leaving a small margin of pancreatic tissue associated with the duodenum containing the common bile duct; a small rim of pancreatic tissue toward the vena cava should be preserved as well during removal of most all of the uncinate process. In most patients, it is possible to free the bile duct from the surrounding scarring without disrupting continuity with the ampulla of Vater, thereby avoiding the need for a bilio-digestive anastomosis.
In some patients (20%), the common bile duct is obstructed and should be opened, so that the bile will drain into the cavity of the resected pancreatic head which is drained into a Roux-en-Y limb of jejunum. The standard reconstruction consists of a Roux-en- pancreaticojejunostomy to the distal pancreatic remnant (body and tail of pancreas) and a pancreatojejunostomy to the rim of pancreas at the duodenum (including the opened bile duct if needed). In up to 10% of patients, this DPPHR procedure is combined with a lateral pancreaticojejunostomy to drain multiple stenoses of the main pancreatic duct. The mortality rate is low (1%), and the morbidity rate is around 15%, less than after pancreatoduodenectomy.
When compared with pancreatoduodenectomy in patients with chronic pancreatitis, the DPPHR offers the advantage of preserving the duodenum and extrahepatic biliary tree, and its superiority over even the pylorus-preserving resection has been shown in prospective studies. Patients who underwent the DPPHR had greater weight gain, a better glucose tolerance, and a higher capacity for insulin secretion. In long-term follow-up, about 20% of the patients developed new onset of diabetes mellitus, like the incidence of diabetes after pancreatoduodenectomy. There is some evidence that endocrine function may be better preserved after DPPHR when compared with patients with chronic pancreatitis not undergoing operation, secondary to the relief of pancreatic ductal obstruction/hypertension. Regarding pain status, 90% of patients after DPPHR have long-term relief of pain. Regarding quality of life, 69% of the patients in one study were rehabilitated professionally, 26% retired, and only 5% of the patients were unimproved. Considering the better pain status, a lesser frequency of acute episodes of chronic pancreatitis, especially in those patients with an enlarged pancreatic head, marked decrease in the need for further hospitalization, low early and late mortality rate, and the restoration of a better quality of life, evidence suggests that DPPHR may delay the natural course of the disease of chronic pancreatitis.
The DPPHR was modified by Frey and colleagues to include a longitudinal pancreatico-jejunostomy combined with a local ‘‘coring out’’ of the pancreatic head without the need for an extensive dissection near the superior mesenteric vessels as with the DPPHR. The Frey and DPPHR have undergone evaluation in multiple comparative trials, confirming their effectiveness as operative procedures for chronic pancreatitis. A modified technique (Bern procedure) of the Beger and Frey procedures has been described recently in patients with chronic pancreatitis. This extended Frey procedure combines the advantages of the Beger and Frey procedure by maintaining a non-anatomic, subtotal central pancreatic head resection but without the need for transsection of the gland over the superior mesenteric vein (SMV), the most tedious part of the DPPHR procedure which was the major advantage offered by the Frey procedure. This modified technique reduces the risk of intraoperative bleeding which is especially increased in the presence of portal hypertension.
Left-sided pancreatic resection (distal pancreatectomy)
Most surgeons believe that the pancreatic head is the pacemaker in chronic pancreatitis, and therefore, pancreatic head resections should be the target for most patients with chronic pancreatitis affecting the entire gland. There is, however, a small and carefully selected group of patients in whom a left-sided pancreatic resection is the appropriate treatment. This subgroup is selected by imaging techniques, including CT, ERCP, or MRI outlining inflammatory complications, such as pseudocysts, fistula, and pancreatic duct stenosis, involving only (or primarily) the body and/or tail region of the pancreas. A good example is the patient who develops a mid-ductal stricture after an episode of necrotizing pancreatitis secondary to gallstone pancreatitis. Similarly, suspicion of a neoplasm or recurrent acute pancreatitis believed secondary to an isolated, mid-ductal stricture may be justification for a left-sided pancreatic resection. Overall, about 10% of all patients who undergo operative intervention for chronic pancreatitis may be candidates for a distal pancreatectomy. These distal pancreatectomies for benign disease can be performed without splenectomy, but conservation of the splenic artery and vein can be difficult and is time-consuming. Nevertheless, the advantage of avoiding the possibility of overwhelming postsplenectomy sepsis should be taken into consideration as well as the importance of the spleen for maintenance of the host defense system. Thus, preservation of the spleen is desirable if there is no clear indication for splenectomy, such as perisplenic pseudocyst or inflammatory/fibrotic encasement of the splenic vessels.
Central pancreatectomy (middle segmentectomy).
Benign lesions of the neck and proximal body of the pancreas, such as the exceedingly rare focal chronic pancreatitis or post-traumatic pancreatitis, pose an interesting operative challenge. If the lesions are not amenable to simple enucleation, surgeons may be faced with the choice of performing a right-sided resection (pancreatoduodenectomy) or a left-sided resection (distal pancreatectomy) to include the lesion, resulting in resection of a substantial amount of otherwise functioning pancreatic parenchyma. Central pancreatic resections have been reported primarily for benign or low-grade neoplasms with Roux-en-Y pancreatojejunostomy reconstruction. Central pancreatectomy affords the possibility of saving functional pancreatic tissue in attempt to avoid the complications of pancreatic insufficiency. Further studies, however, must prove the effectiveness of such an operation for patients with chronic pancreatitis. Central resections in patients with chronic pancreatitis must be viewed with caution and considered only in highly selected cases.
Pancreatic resection with islet cell autotransplantation.
Because of the concerns of pancreatic endocrine insufficiency after any pancreatic resection for chronic pancreatitis, renewed interest has focused on the possibility of performing a total pancreatectomy, isolating the islets, and reinfusing (autotransplanting) the islets into the liver. Improvements in islet cell harvesting and preservation for islet cell allotransplantation for diabetics have allowed new enthusiasm in this technique for patients with chronic pancreatitis. Results to date are encouraging, but the inability to harvest reliably an adequate number of islets and to prove successful engraftment within the liver remain current limitations. This approach may be more effective early in the disease when islets have not been depleted.
OUTCOMES
In summary, definite evidence for the best operative method for treating painful chronic pancreatitis is still not fully accepted. The study designs in the few randomized controlled trials (evidencebased Level I data) available today have some limitations in design and reporting of morbidity and include only small numbers of patients. Nevertheless, the different variations of the DPPHR, Beger, Frey, and Bern procedures appear to be as equally successful in achieving long-term pain control as pancreatoduodenectomy, but they have fewer postoperative complications and appear to be superior with regard to preservation of pancreatic function and quality of life.
Aula de Anatomia do Dr Nicolaes Tulp (1632)
O valor da obra “A Aula de Anatomia do Dr. Nicolaes Tulp” é incalculável, pois ela pertence ao acervo do Mauritshuis, em Haia, na Holanda, e é considerada uma das mais importantes e valiosas obras do museu. Além disso, a pintura é uma das mais famosas obras de Rembrandt e uma das mais importantes do período Barroco holandês. Por isso, é considerada uma obra-prima da arte ocidental e tem um valor histórico, artístico e cultural inestimável. Embora não haja um valor monetário exato para a pintura, pode-se dizer que é uma das obras mais valiosas e procuradas do mundo da arte, tanto pelo seu significado histórico quanto pela sua qualidade artística.
“A Aula de Anatomia do Dr. Nicolaes Tulp” é uma pintura a óleo sobre tela, criada por Rembrandt van Rijn em 1632. A obra mede 169,5 cm x 216,5 cm e está atualmente exposta no Mauritshuis, em Haia, na Holanda. A composição da pintura apresenta um grupo de homens em torno de uma mesa de dissecação, liderados pelo médico Nicolaes Tulp, que está realizando uma demonstração de anatomia. O corpo sendo dissecado é o de um criminoso enforcado chamado Aris Kindt. A composição apresenta uma disposição simétrica e organizada das figuras em torno da mesa, com Tulp no centro da imagem.
A luz na pintura é focada no corpo sendo dissecado, destacando-o em relação ao fundo escuro da sala. A técnica de chiaroscuro usada por Rembrandt acentua o realismo e o drama da cena. As figuras são pintadas em tons de marrom, cinza e preto, com destaques de branco. A obra apresenta detalhes precisos e realistas da anatomia do corpo, bem como das ferramentas médicas utilizadas na dissecação. O corpo do criminoso apresenta uma ferida na cabeça e uma perna amputada, o que sugere que ele pode ter sido executado por um crime violento.
No canto inferior direito da pintura, há um livro aberto com o título “Spiegel der Konst” (“Espelho da Arte”), um tratado de anatomia escrito por Adriaan van de Spiegel e utilizada pelos médicos da época. Em geral, a “Aula de Anatomia do Dr. Nicolaes Tulp” é uma obra-prima devido à sua técnica precisa e detalhada, bem como à sua habilidade em transmitir um senso de realismo e drama. A pintura é considerada uma das obras mais importantes do período Barroco holandês e é frequentemente citada como um exemplo do estilo de pintura de Rembrandt.
A seguir estão algumas das características artísticas e estéticas da obra:
- Composição: A pintura apresenta uma composição equilibrada e organizada, com as figuras dos membros da guilda cirúrgica em torno da mesa de dissecação centralizada.
- Luz e Sombra: Rembrandt usa uma técnica conhecida como chiaroscuro, ou contraste entre luz e sombra, para dar profundidade e dimensão à cena. A luz focaliza no cadáver e no médico principal, destacando-os do fundo escuro.
- Realismo: A pintura é altamente realista, mostrando detalhes precisos das ferramentas cirúrgicas, do cadáver e das expressões dos personagens.
- Cores: O uso limitado de cores em tons de marrom e cinza dá à pintura uma atmosfera austera e solene.
- Simbolismo: A pintura inclui vários elementos simbólicos, como a presença de uma coruja, que representa sabedoria, e a posição da mão do cadáver, que simboliza a morte.
- Técnica: A pintura foi executada com uma técnica de pincelada solta e fluida, que enfatiza a textura e a superfície da pintura.
Em geral, a “Aula de Anatomia do Dr. Nicolaes Tulp” é considerada uma obra-prima devido à sua habilidade técnica e sua capacidade de transmitir um senso de realismo e drama. A pintura é considerada uma das obras mais importantes do período Barroco holandês e é frequentemente citada como um exemplo do estilo de pintura de Rembrandt.
“Ao CADÁVER DESCONHECIDO, todo nosso respeito e agradecimento!”
O estudo da anatomia humana é essencial para o ensino e avanço da medicina, e o cadáver desempenha um papel fundamental nesse processo. O cadáver é uma ferramenta valiosa que permite aos estudantes de medicina, anatomia e outras áreas da saúde aprenderem sobre a estrutura e a organização do corpo humano de uma maneira muito mais eficaz do que seria possível apenas com modelos em duas dimensões ou imagens de realidade virtual.
A dissecação de um cadáver permite que os estudantes examinem as diferentes camadas de tecido e órgãos que compõem o corpo humano, geralmente ensinadas na disciplina de ANATOMIA TOPOGRÁFICA. Isso pode ajudá-los a entender como as estruturas funcionam juntas para manter a homeostase. Ao dissecar um cadáver, os estudantes podem observar as relações anatômicas entre as estruturas, entender as diferenças entre os diferentes sistemas do corpo humano e identificar as variações individuais que ocorrem em cada pessoa. Essas informações são fundamentais para o diagnóstico e o tratamento de doenças e distúrbios.
Além disso, o estudo do cadáver permite que os estudantes observem as variações normais que ocorrem no corpo humano, bem como as anomalias e patologias que podem afetar diferentes sistemas e órgãos. Isso pode ajudar os estudantes a entender as causas e os efeitos de doenças e distúrbios e a desenvolver habilidades práticas para o diagnóstico e o tratamento. Por exemplo, ao estudar um cadáver com uma patologia específica, os estudantes podem aprender a identificar essa condição em pacientes vivos e determinar o melhor tratamento para cada caso.
Além de fornecer informações valiosas sobre a estrutura do corpo humano, o estudo do cadáver também é importante para a investigação científica e o desenvolvimento de novas técnicas e tecnologias médicas. Por exemplo, ao estudar a anatomia do sistema nervoso central de um cadáver, os pesquisadores podem identificar áreas do cérebro que controlam funções específicas, como a fala ou a visão. Isso pode levar ao desenvolvimento de novas técnicas cirúrgicas que preservam essas funções durante procedimentos no cérebro.
O estudo do cadáver também é fundamental para o treinamento de cirurgiões. Através da dissecação de cadáveres, os cirurgiões podem praticar técnicas cirúrgicas complexas e aprimorar suas habilidades antes de realizar procedimentos em pacientes vivos. Isso pode ajudar a reduzir o risco de complicações durante as cirurgias e melhorar os resultados para os pacientes. A anatomia é a disciplina fundamental da formação cirúrgica desde tempos remotos na história da medicina.
No entanto, o uso de cadáveres para o estudo da anatomia humana é uma questão ética e legal complexa. Para garantir que o uso de cadáveres seja feito de forma ética e responsável, muitos países têm leis e regulamentações rigorosas que governam a dissecação de corpos humanos. Essas leis e regulamentações visam garantir que os cadáveres sejam tratados com respeito e dignidade, que o consentimento adequado seja obtido antes da dissecação e que a privacidade e a confidencialidade dos doadores sejam protegidas.
Visão Crítica de Segurança (Colecistectomia)
A colecistectomia laparoscópica (CL) é o padrão-ouro para tratamento de cálculos biliares. No entanto, o risco de lesão do ducto biliar (BDI) continua a ser preocupação significativa, uma vez que CL ainda tem taxa de BDI maior do que a via laparotômica, apesar de muitos esforços propostos para aumentar sua segurança. A Visão Crítica da Segurança (CVS) proposta por Strasberg é a técnica para a identificação dos elementos críticos do triângulo de Calot durante a CL. Esta técnica foi adotada em vários programas de ensino e com a proposta de reduzir o risco de lesão acidental da via biliar (LAVB) e o uso da adequado da CVS está associado a menores taxas de LAVB. O objetivo deste #Webinar é abordar a Anatomia Cirúrgica Fundamental para a realização de uma Colecistectomia Laparoscópica.
Intraoperative cholangiography: Selective or Routine?
Intraoperative cholangiography (IOC), described by Mirizzi in 1932, represented a significant advance in the diagnosis of choledocolithiasis during cholecystectomy. The natural history of asymptomatic choledocolithiasis has been investigated in different populations and its therapeutic management continues to be controversial. IOC is traditionally advocated as a procedure to be adopted in all laparoscopic cholecystectomies since it permits to define the anatomy of the biliary tract and to detect common bile duct stones. However, in the laparoscopic era, technological advances in radiologic-endoscopic workup have markedly increased the costs of investigation of patients with suspected choledocolithiasis. The routine use of IOC has raised the question of which cases require the exploration of bile tract anatomy during surgery and whether there are methods to predict preoperatively unsuspected choledocolithiasis. Therefore, the most appropriate management of preoperatively unsuspected choledocolithiasis, i.e., the routine or selective use of IOC, still remains undefined. In this respect, well-defined criteria for the inclusion of patients with possible choledocolithiasis who should be submitted to cholangiography exist in the literature, but there is no safe approach to exclude asymptomatic patients without an indication for contrast examination.
A systematic literature search was performed by KOVACS N, et al (2022) using the following search keys: cholangiogra* and cholecystectomy. The primary outcomes were BDI and retained stone rate. To investigate the differences between the groups (routine IOC vs selective IOC and IOC vs no IOC), they calculated weighted mean differences (WMD) for continuous outcomes and relative risks (RR) for dichotomous outcomes, with 95% confidence intervals (CI). Of the 19,863 articles, 38 were selected and 32 were included in the quantitative synthesis. Routine IOC showed no superiority compared to selective IOC in decreasing BDI (RR = 0.91, 95% CI 0.66; 1.24). Comparing IOC and no IOC, no statistically significant differences were found in the case of BDI, retained stone rate, readmission rate, and length of hospital stay. They found an increased risk of conversion rate to open surgery in the no IOC group (RR = 0.64, CI 0.51; 0.78). The operation time was significantly longer in the IOC group compared to the no IOC group (WMD = 11.25 min, 95% CI 6.57; 15.93). So this data findings suggest that IOC may not be indicated in every case, however, the evidence is very uncertain.
Postpancreatectomy Hemorrhage
Postoperative complications represent one of the most debated topics in pancreatic surgery. Indeed, the rate of complications following pancreatectomy is among the highest in abdominal surgery, with morbidity ranging between 30 and 60%. They are often characterized by elevated clinical burden, with a consequent challenging postoperative management. Mortality rates can exceed 5%. The impact on patient recovery and hospital stay eventually leads to massive utilization of resources and increases costs for the health system. The International Study Group of Pancreatic Surgery (ISGPS) provides standardized definitions and clinically based classifications for the most common complications after pancreatectomy, including postoperative pancreatic fistula (POPF), post pancreatectomy hemorrhage (PPH), delayed gastric emptying (DGE), bile leakage, and chyle leak.
Post Pancreatectomy Hemorrhage
Despite its lower incidence compared with POPF with reported rates of 3–10% after pancreatectomy, PPH remains one of the major of postoperative complications, with mortality rates ranging from 30 to 50%. According to current ISGPS recommendations, PPH is classified in three grades (A, B, and C) based on two main criteria: timing of the hemorrhage and severity of the bleeding. The timing is dichotomized as early, occurring within 24 hours from the index surgery, and late, when it happens afterwards.
Management is tailored according to the clinical picture, the timing of onset, and the presumptive location. Early hemorrhage is generally due to either unsuccessful intraoperative hemostasis or to an underlying coagulopathy. The vast majority of patients are basically asymptomatic, with PPH having no influence on the postoperative course. However, when the bleeding is severe, re-laparotomy is recommended, with the aim of finding and controlling the source of bleeding. This approach usually guarantees an uneventful subsequent course. Late PPH is often challenging and the pathogenesis is diverse. Vascular erosion secondary to POPF or intraabdominal abscess, late failure of intraoperative hemostatic devices, arterial pseudoaneurysm, and intraluminal ulceration are some of the most common causes. In this setting, surgical access to the source of bleeding may be challenging. Angiography (if extraluminal) and endoscopy (if intraluminal) represent the primary approaches to treatment. Surgery is reserved for hemodynamically unstable patients and for those who present with deteriorating condition, multiorgan failure or sepsis. Given these assumptions, except for early mild events, contrast-enhanced abdominal CT is crucial in all cases of PPH, possibly allowing identification of the source and planning the management accordingly. Also, it should be noted that late massive hemorrhages may be preceded by mild self-limiting sentinel bleeds. A prompt abdominal CT scan aimed at excluding vascular lesions is therefore strongly recommended in these cases.
The incidence of complications following pancreatic resection remains high. The ISGPS established standardized definitions and clinical grading systems for POPF, PPH, DGE, and biliary and chyle leak. These classification systems have enabled unbiased comparisons of intraoperative techniques and management decisions. However, the management policies of these complications are most often driven by a patient’s condition and local surgical expertise and is not always based on the available high-level evidence. The development of high-volume specialized units with appropriate resources and multidisciplinary experience in complication management might further improve the evidence and the outcomes.
Subtotal cholecystectomy for difficult acute cholecystitis
Laparoscopic cholecystectomy is considered the gold standard for treatment of benign gallbladder diseases. Cholecystectomy using this method can be completed in 90% of elective cholecystectomies and 70% of emergency cholecystectomies. Acute cholecystitis, especially if difficult, can change the above paradigm, resulting in open conversion or change of technique. The conditions that define a difficult cholecystectomy are as follows: necessity of conversion from laparoscopic to open surgery; duration of procedure greater than 180 min; blood loss greater than 300 ml; and urgent need for involvement of a more experienced surgeon. One of the “rescue” procedures to complete the surgery safely (both for the surgeons and patients) is subtotal cholecystectomy (STC). Open and laparoscopic subtotal cholecystectomy have been reported. For many surgeons, this is considered a bail out technique, and the timing of decision making is crucial to avoid catastrophic complications. Te capability to perform STC in laparoscopy is increasingly requested during difficult laparoscopic cholecystectomy. Difficult LC has a risk of BDI from 3 to 5 times higher in laparoscopy than open surgery. In case of operative difficulties of young surgeons mostly trained in laparoscopy the help of senior surgeons is strongly recommended. The purpose of the present study is to clarify how laparoscopic subtotal cholecystectomy may be used to complete a difficult cholecystectomy for acute cholecystitis without serious complications.

Biliary leakage represents the most frequent complication of incomplete resection of the gallbladder wall in cases of difficult acute cholecystectomy treated with subtotal cholecystectomy. This complication is rarely fatal but requires correct treatment. If bile leakage does not stop spontaneously seven days postoperatively, the possible treatments are endoscopic biliary sphincterotomy, endoscopic plastic stent, and a fully covered self-expanding metal stent. When performing closure of the gallbladder stump, suturing the anterior residual of both anterior and posterior wall represents the best method to have fewer complications. Complications, if not lethal, decrease the patient’s quality of life. Intraoperatively, it is of utmost importance to carefully expose the gallbladder stump to avoid left-in-place stones, wash the entire cavity and drain the abdomen. Bile duct injuries can be a significant complication in this type of surgery. Prevention of the lesions with conversion from laparoscopic to open, or the opinion of older surgeon in case of difficulties is strongly recommended. Mortality is a very rare complication. The limitations of our study are given by the heterogeneity of the techniques used for LSC and the lack of a long-term follow-up analyzing the related complications.
Timing of Elective Surgery after #COVID19 Infection
The options to clear a recovered COVID-19 patient for surgery could be a symptom or test-based strategy. The studies recommend a hybrid strategy until more information is available. Surgical procedures should be delayed if possible for 4–6 weeks. The scientific data recommend against routine testing in this group as patients are likely not infectious any-more and risk of airway reactivity or ARDS will be reduced. Between 2 and 4 weeks after symptom onset, scientific data recommend a test-based strategy due to insufficient data that recovered COVID patients are completely uninfectious. Data from China suggest that patients with severe symptoms have higher viral burden and prolonged viral shedding. For surgery completion<2 weeks after COVID-19 symptoms or diagnosis, scientific data recommend no testing and operating under COVID isolation precautions in the operating room. Immunocompromised patients are likely to shed longer and may be at higher risk of other infectious or pulmonary complications. The scientific data suggest individualized case-based decisions by involved providers until more information is available.
Aderências Pós-Operatórias
Do Manejo Conservador à Decisão Cirúrgica
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 10 minutos)
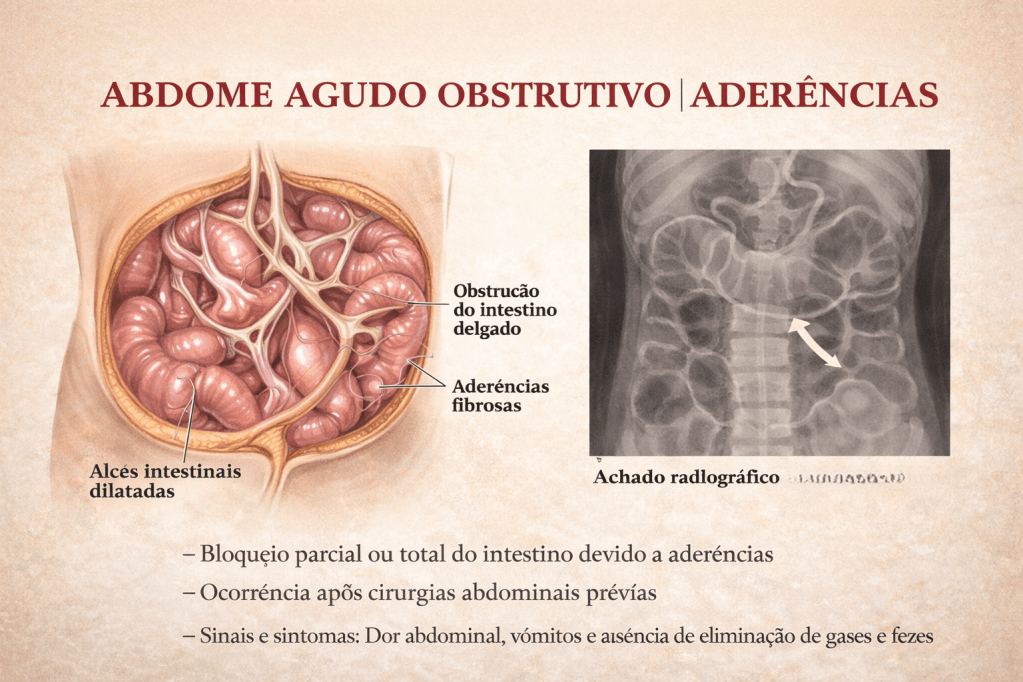
Introdução
A obstrução intestinal é um dos cenários mais desafiadores e frequentes no pronto-socorro cirúrgico. Historicamente, o abdome agudo obstrutivo impunha o dilema imediato da exploração cirúrgica. Hoje, compreendemos que a etiologia dita o compasso da intervenção. Entre todas as causas de obstrução do intestino delgado, as aderências pós-operatórias (bridas) assumem o protagonismo absoluto. Para o estudante de medicina, o residente e o cirurgião do aparelho digestivo, dominar o manejo da obstrução por bridas é essencial. O desafio não reside apenas na técnica operatória da enterólise, mas sim no raciocínio clínico aguçado para responder à pergunta fundamental: quando persistir no tratamento conservador e quando a indicação cirúrgica se torna imperativa?
Cenário Brasileiro
A formação de aderências peritoniais é uma resposta fisiológica quase universal ao trauma cirúrgico (isquemia, manipulação, coágulos), ocorrendo em até 90% dos pacientes submetidos a laparotomias. Contudo, em uma parcela significativa, essa cicatrização aberrante leva à obstrução mecânica. Na prática da cirurgia geral no Brasil, a obstrução por bridas responde por cerca de 60% a 75% das admissões por obstrução do intestino delgado. Dados do Sistema Único de Saúde (DataSUS) indicam que milhares de laparotomias exploradoras são realizadas anualmente no país devido a quadros obstrutivos agudos, gerando elevado custo financeiro e considerável morbimortalidade, além de reinternações frequentes.
Fisiopatologia e Diagnóstico
O peritônio, quando lesado, inicia uma cascata inflamatória rica em fibrina. Em condições normais, a fibrinólise degrada essa matriz em poucos dias. O desequilíbrio nesse sistema — com falha na fibrinólise peritonia — resulta na organização da fibrina em tecido conjuntivo vascularizado: a aderência.
A Tríade Diagnóstica
O diagnóstico é eminentemente clínico e radiológico:
- Clínica: Dor abdominal em cólica, distensão, vômitos (fecaloides em fases tardias) e parada de eliminação de flatus e fezes.
- Laboratório: Hemograma, eletrólitos, lactato e gasometria são cruciais para rastrear sinais de isquemia ou necrose (leucocitose importante, acidose metabólica).
- Imagem: A Tomografia Computadorizada (TC) de abdome com contraste venoso substituiu o raio-X simples como padrão-ouro. A TC identifica o ponto de transição (zona de calibres distintos), o grau de obstrução, a presença de sofrimento de alça (espessamento parietal, pneumatose, gás no sistema porta) e descarta outras etiologias (hérnias internas, neoplasias).
Estratégias de Tratamento
O manejo da obstrução por bridas sofreu uma evolução paradigmática, distanciando-se de intervenções intempestivas em favor de protocolos baseados em evidências.
1. Tratamento Conservador (Não Operatório)
Na ausência de sinais de isquemia intestinal, perfuração ou peritonite localizada, a conduta inicial deve ser conservadora:
- Jejum (NPO) e descompressão com Sonda Nasogástrica (SNG).
- Ressuscitação volêmica vigorosa e correção de distúrbios hidroeletrolíticos.
- Protocolo com Contraste Hidrossolúvel: A administração de contraste hiperosmolar (ex: Gastrografin) via SNG não possui apenas valor diagnóstico (avaliar se o contraste atinge o cólon em 24h), mas também efeito terapêutico. A hiperosmolaridade atrai líquido para a luz intestinal, reduzindo o edema da parede e estimulando o peristaltismo, resolvendo o quadro obstrutivo em uma parcela significativa dos casos.
2. Tratamento Cirúrgico: O Timing Ideal
A falha do tratamento conservador (geralmente avaliada após 48 a 72 horas) ou o surgimento de sinais de alarme (taquicardia, febre, irritação peritoneal, acidose) indicam cirurgia de urgência.
- Laparotomia vs. Laparoscopia: A via aberta continua sendo o padrão em pacientes com múltiplas cirurgias prévias ou grande distensão abdominal. A abordagem laparoscópica é excelente em mãos experientes, mas reserva-se a casos selecionados (ex: suspeita de brida única, distensão moderada), exigindo extrema cautela na introdução do primeiro trocarte (técnica aberta de Hasson) para evitar enterotomias iatrogênicas.
- O Ato Cirúrgico: A lise de aderências deve ser restrita ao necessário para resolver a obstrução. Múltiplas enterólises desnecessárias aumentam o risco de lesões e a formação de novas bridas. Em caso de necrose, a ressecção do segmento isquêmico com anastomose primária ou estomia (a depender da estabilidade hemodinâmica) é imperativa.
Pontos-Chave para a Prática Diária
- Suspeição de Isquemia: Dor refratária a analgésicos, taquicardia inexplicada e acidose metabólica são gritos de socorro de uma alça intestinal isquêmica. O tratamento conservador é contraindicado.
- O Valor da TC: A tomografia não apenas confirma o diagnóstico, mas é o farol que guia a decisão entre a enfermaria e o centro cirúrgico.
- Prevenção Primária: O uso de barreiras antiaderentes ainda carece de evidências universais robustas, mas a técnica cirúrgica apurada, manipulação tecidual delicada e controle rigoroso da hemostasia continuam sendo a melhor profilaxia.
Conclusões Aplicadas
O tratamento da obstrução intestinal por bridas é a quintessência do bom senso cirúrgico. O cirurgião moderno deve possuir a paciência de um clínico para conduzir o tratamento conservador respaldado pelo contraste hidrossolúvel e, simultaneamente, a assertividade de um intervencionista para indicar a sala de cirurgia ao primeiro sinal de estrangulamento.Compreender que “operar a radiografia” é um erro crasso e que a reavaliação clínica seriada (de preferência pelo mesmo examinador) é a bússola do tratamento, molda o profissional de excelência que minimiza ressecções intestinais desnecessárias e reduz a morbidade pós-operatória.
“Na obstrução intestinal aguda, não deixe o sol se pôr ou nascer sobre o paciente sem uma decisão. Mas lembre-se: a cirurgia sem indicação precipita o desastre que buscava evitar.” — Aforismo clássico derivado dos ensinamentos de Sir Zachary Cope, pioneiro no estudo e diagnóstico do abdome agudo.
Gostou ❔Nos deixe um comentário ✍️ , compartilhe em suas redes sociais e|ou mande sua dúvida pelo 💬 Chat On-line em nossa DM do Instagram.
The “BAD” Gallbladder
Once the decision for surgery has been made, an operative plan needs to be discussed and implemented. Should one initially start with laparoscopic surgery for the “bad gallbladder”? If a laparoscopic approach is taken, when should bail-out maneuvers be attempted? Is converting to open operation still the standard next step? A 2016 study published by Ashfaq and colleagues sheds some light on our first question. They studied 2212 patients who underwent laparoscopic cholecystectomy, of which 351 were considered “difficult gallbladders.” A difficult gallbladder was considered one that was necrotic or gangrenous, involved Mirizzi syndrome, had extensive adhesions, was converted to open, lasted more than 120 minutes, had a prior tube cholecystostomy, or had known gallbladder perforation. Seventy of these 351 operations were converted to open. The indications for conversion included severe inflammation and adhesions around the gallbladder rendering dissection of triangle of Calot difficult (n 5 37 [11.1%]), altered anatomy (n 5 14 [4.2%]), and intraoperative bleeding that was difficult to control laparoscopically (n 5 6 [1.8%]). The remaining 13 patients (18.5%) included a combination of cholecystoenteric fistula, concern for malignancy, common bile duct exploration for stones, and inadvertent enterotomy requiring small bowel repair. Comparing the total laparoscopic cholecystectomy group and the conversion groups, operative time and length of hospital stay were significantly different; 147 +- 47 minutes versus 185 +- 71 minutes (P<.005) and 3+-2 days versus 5+-3 days (P 5 .011), respectively. There was no significant difference in postoperative hemorrhage, subhepatic collection, cystic duct leak, wound infection, reoperation, and 30-day mortality.2 From these findings, we can glean that most cholecystectomies should be started laparoscopically, because it is safe to do so. It is the authors’ practice to start laparoscopically in all cases.
BAILOUT PROCEDURES
Despite the best efforts of experienced surgeons, it is sometimes impossible to safely obtain the critical view of safety in a bad gallbladder with dense inflammation and even scarring in the hepatocystic triangle. Continued attempts to dissect in this hazardous region can lead to devastating injury, including transection of 1 or both hepatic ducts, the common bile duct, and/or a major vascular injury (usually the right hepatic artery). Therefore, it is imperative that any surgeon faced with a bad gallbladder have a toolkit of procedures to safely terminate the operation while obtaining maximum symptom and source control, rather than continue to plunge blindly into treacherous terrain. If the critical view of safety cannot be achieved owing to inflammation, and when further dissection in the hepatocystic triangle is dangerous, these authors default to laparoscopic subtotal cholecystectomy as our bail-out procedure of choice. The rationale for this approach is that it resolves symptoms by removing the majority of the gallbladder, leading to low (although not zero) rates of recurrent symptoms. It is safe, and can be easily completed laparoscopically, thus avoiding the longer hospital stay and morbidity of an open operation. There is now significant data supporting this approach. In a series of 168 patients (of whom 153 were laparoscopic) who underwent subtotal cholecystectomy for bad gallbladders, the mean operative time was 150 minutes (range, 70–315 minutes) and the average blood loss was 170 mL (range, 50–1500 mL). The median length of stay for these patients was 4 days (range, 1–68 days), and there were no common bile duct injuries.23 There were 12 postoperative collections (7.1%), 4 wound infections (2.4%), 1 bile leak (0.6%), and 7 retained stones (4.2%), but the 30-day mortality was similar to those who underwent a total laparoscopic cholecystectomy. A systematic review and meta-analysis by Elshaer and colleagues showed that subtotal cholecystectomy achieves comparable morbidity rates compared with total cholecystectomy. These data support the idea that we should move away from the idea that the only acceptable outcome for a cholecystectomy is the complete removal of a gallbladder, especially when it is not safe to do so. This shift toward subtotal cholecystectomy has been appropriately referred to as the safety first, total cholecystectomy second approach.
Management of HEPATIC ADENOMA
MANAGEMENT
At a minimum, cessation of exogenous hormones is recommended, followed by serial imaging, as these lesions will shrink when the inciting hormone has been removed. In general, to obtain either CT or MRI every 6 months for the first 2 years and then once annually for lesions that we are following. Intervention is dictated by the risk of hemorrhage and malignant transformation, which is directly correlated to size. Hepatocellular carcinoma is seen in 5% of patients with lesions exceeding 8 cm. The rate of rupture is strongly associated with size > 5 cm.
Surgical resection is recommended for lesions ≥ 5 cm, those that are enlarging, those for which malignancy cannot be excluded, and those that are symptomatic. Additionally, adenomas in men, regardless of size, and especially those that are associated with anabolic steroid use, those with glycogen storage disease, and those that harbor ß-catenin mutations should also be given serious consideration for surgical extirpation.
Pregnant women with HA pose a management challenge. Since adenomas respond to hormones, they can increase in size with pregnancy and thus put the patient at risk for rupturing and bleeding. For women who would like to become pregnant, close monitoring without discouraging the patient’s wish is
recommended for lesions < 5 cm. However, for those patients with lesions > 5 cm or those who had complications from the adenomas from previous pregnancies, we recommend surgical resection prior to proceeding with the pregnancy. In situations where HA is discovered during pregnancy, we recommend close monitoring for lesions < 5 cm. However, for lesions > 5 cm, surgical resection should be considered during the second trimester but delayed in the third trimester since surgical intervention during this trimester is fraught with complications.
Managing the “difficult” gallbladder
The gold standard for the surgical treatment of symptomatic cholelithiasis is conventional laparoscopic cholecystectomy (LC). The “difficult gallbladder” is a scenario in which a cholecystectomy turns into an increased surgical risk compared with standard cholecystectomy. The procedure may be difficult due to processes that either obscure normal biliary anatomy (such as acute or chronic inflammation) or operative exposure (obesity or adhesions caused by prior upper abdominal surgery). So, when confronted with a difficult cholecystectomy, the surgeon has a must: to turn the operation into a safe cholecystectomy, which can mean conversion (to an open procedure), cholecystostomy, or partial/ subtotal cholecystectomy. The surgeon should understand that needs to rely on damage control, to prevent more serious complications if choosing to advance and progress to a complete cholecystectomy.
When to Predict a Difficult Laparoscopic Cholecystectomy
A difficult cholecystectomy may be predicted preoperatively based on patient characteristics and ultrasound and laboratory findings. This is probably a very important step in mitigating the high risk associated with a difficult procedure and may serve either to reschedule the procedure or design intraoperative strategies of management to guarantee a safe performance of the surgical procedure.
The following situations are associated with a higher chance of a difficult cholecystectomy:
• Acute cholecystitis (more than 5 days of onset)
• Previous cholecystitis episode
• Male sex
• Obesity
• Cirrhosis
• Sclero-atrophic gallbladder
• Thick walls (>5 mm)
• Previous signs of canalicular dwelling (clinical and laboratory)
Through multivariate analysis, Bourgoin identified these elements of predictive help to identify difficult LC: male sex, previous cholecystitis attack, fibrinogen, neutrophil, and alkaline phosphatase levels. Another important point is the fact of conversion from a laparoscopic procedure to an open and traditional cholecystectomy, usually through a right subcostal incision. Conversion should not be considered as a personal failure, and the surgeon needs to understand the concept of “safety first,” considering that conversion is performed in order to complete the procedure without additional risks and preventing complications and not solving intraoperative complications. It is also useful to define a time threshold to aid in the decision to convert. It is not worth taking an hour and a half and still dissecting adhesions, preventing the correct visualization of the cystic pedicle. This time limit represents a method to prevent inefficiencies in the operating room (OR) schedule as well as additional expenditures.
A smart surgeon should rely to conversion in the following situations:
• Lack of progress in the procedure
• Unclear anatomy/any grade of uncertainty
• CVS not achieved
• Bleeding/vascular injury
• BD injury
• Lack of infrastructure, expertise, and support
Final Remarks
The primary goal of a laparoscopic cholecystectomy in the treatment of symptomatic cholelithiasis is the safe remotion of the gallbladder and the absence of common bile duct injury. Some tips to take into account:
– Never perform a laparoscopic cholecystectomy without a skilled surgeon close by.
– Beware of the easy gallbladder.
– Slow down, take your time.
– Knowledge is power, conversión can be the salvation!
– Do not repair a bile duct injury (unless you have performed at least 25 hepaticojejunostomies).
– Do not ignore postoperative complaints (pain, jaundice, major abdominal discomfort, fever)
Other options when confronted with a difficult laparoscopic cholecystectomy are:
– A percutaneous cholecystostomy, if the risk was identified preoperatively or the patient is a poor surgical candidate;
– An intraoperative cholangiography, which may aid in identifying an injury to the bile duct and solve it, if you are an experienced surgeon;
– A subtotal or partial cholecystectomy;
– Ask for help;
– Conversion to an open procedure;
Liver Procurement
Donor Evaluation and Management
There are very few absolute contraindications for abdominal organ donation, which can be summarized in the short form CHUMP: (1) Creutzfeldt-Jakob disease, (2) active HIV infection, (3) uncontrolled donor sepsis, (4) history of melanoma or other malignancy that poses a risk for transmission regardless of the apparent disease – free period, and (5) past history of non-curable malignancy (curable malignancy such as localized small kidney tumors, localized prostate cancer, localized colon malignancy >5 years previously may be considered after careful risk/benefit assessment). In addition to these general criteria, there are organ-specific criteria for guiding the acceptance of a liver for transplantation. A history of hepatitis or alcoholism is certainly a warning sign, but both livers from HBsAg-positive and/or HCV-positive donors are currently used worldwide, and suitability for transplant must be judged on a case-by-case basis. In general, in the case of a marginal liver donor, the intraoperative assessment by the donor surgeon, in addition to liver biopsy pathological evaluation, is the best single piece of information.
Technical Aspects of Liver Procurement
A midline laparotomy from the xyphoid to the pubis is performed and the round ligament divided. The intra-abdominal organs are explored to check for eventual malignancies, and the quality of the liver is assessed: in the absence of contraindications for a transplant, a sternotomy can be performed. Of note, in the presence of prior heart surgery, the complete warm dissection should be made prior to the sternotomy. It is also prudential to isolate and encircle the aorta prior to sternotomy in order to be ready to cannulate in the event of cardiac arrest/injury at thoracotomy. A blunt dissection behind the sternum just below the jugular notch should be performed until the fingertip can be placed retrosternal around the jugular notch. The sternotomy is then performed in a cranial to caudal direction with the sternum saw to avoid left innominate vein injury. The division of the left triangular ligament allows the mobilization of the left lateral segments of the liver and the exposure of the supraceliac aorta just below the diaphragm to be encircled. The division of the falciform ligament up to the suprahepatic inferior vena cava (IVC) provides more mobility of the liver, necessary when the IVC must be divided from a cardiac graft. Before starting the dissection of the hepatoduodenal ligament, the hepatogastric ligament must be inspected by dividing the lesser omentum. This ligament is usually very thin and transparent so that any replaced or accessory left hepatic artery should be easily visible. In addition, palpation of the ventral border of the foramen of Winslow makes it possible to identify a possible accessory or replaced right hepatic artery. Variations in the hepatic arterial supply can complicate the hilar dissection in up to one third of donors.
THE HILAR STRUCTURES
The hilar structures of the liver are then dissected free; the common bile duct (CBD) is dissected on the level of the edge of the second duodenal portion after opening of the peritoneum and visualization of the duct. In difficult cases, due to a high BMI, following the cystic duct out of the gall bladder can help to identify the CBD. The CBD should be encircled from the lateral border of the hepatoduodenal ligament in order to avoid injury of the portal vein. The CBD and the gallbladder are opened and flushed with normosaline solution. The origins of the gastroduodenal, gastric, and splenic arteries are then identified and encircled and, in the case of liver only procurement, will be taped just before cross-clamping in order to increase flushing through the hepatic artery to the liver.
VASCULAR CANULATION / SOLUTION PRESERVATION
The aorta can be isolated by two approaches. One approach requires mobilization of the right colon on top of Gerota’s fascia and should be extended into a Kocher maneuver to uncover both the inferior vena cava and the abdominal aorta; the other approach is performed by opening the root of the mesentery from the Treitz fascia, along the margin of the duodenum until visualization of the right iliac vessels and ureter is achieved. The inferior mesenteric artery can be tied and divided, and the abdominal aorta, just 2–3 cm above the bifurcation, isolated and encircled. The lumbar arteries could be either tied or clipped and then cut in order to provide mobility of the aorta and facilitate the cannulation. Two umbilical tapes are placed around the dissected segment of the aorta and secured by clamps and will be used to secure aortic cannulae to the vessel. The inferior mesenteric vein (IMV) is most commonly used for access into the portal system by ligating the distal part of it but leaving it uncut to retract the vein with a mosquito clamp. Another tie is then placed around the cranial portion of the vein, using it for occlusion of the vein by retracting it while a partial incision of the vein is performed. The portal cannula can be inserted into the IMV while the tension of the occluding tie is decreased before tying it around the vein and inserted cannula. At this point, 30,000-IU heparin should be given to prevent the blood from clotting after the cross-clamping. Once these preliminary procedures have been completed, the aortic cannulae (20-F armed cannulae) can be inserted into the distal abdominal aorta and secured with the umbilical tapes.
CROSS-CLAMP
The subdiaphragmatic aorta is now clamped (cross-clamp), and cold preservation solution is then rapidly infused through the aortic and portal cannulae; the liver flow is decompressed by dividing the inferior vena cava in the chest. The abdomen is filled with water and ice. The choice of solution for infusion and its amount varies from center to center. The quality of the flush can be assessed by evaluating the outflow of the supradiaphragmatic IVC which should become more transparent with time as the blood in the abdominal organs is replaced by the preservation solution. After the flush is completed, some of the ice is removed from the abdomen to allow the cold dissection of the structures. The gastroduodenal, gastric, and splenic arteries can now be divided. Just below the gastroduodenal artery, the portal vein can be found and can be followed back, if pancreas procurement is not performed, by dividing the head of the pancreas. The cannulae in the IMV can now be removed, the splenic vein ligated and divided, and the venous cannulae replaced in the superior mesenteric vein once it is divided from its distal branches. The superior mesenteric artery (SMA) can now be found in the retro-pancreatic laminae and should be ligated, secured to a clamp and divided in order to find the aortic plane by following back the SMA. This dissection must be made on the left side of the SMA in order to avoid damage to a possible replaced or accessory right hepatic artery. The renal arteries are usually just below the SMA. They should be visualized before the suprarenal aorta is divided. This section must be made in 45°, first looking for ostia of accessory renal arteries before performing complete separation of the aorta. By following back the splenic and gastric arteries, the celiac trunk can be visualized. The aorta must now be divided just below the diaphragm, obtaining a patch containing the celiac trunk and the origin of the mesenteric artery. At this time point, a finger is placed in the supradiaphragmatic IVC helping to identify it while the diaphragm is cut. A portion of the diaphragm should be kept with the liver to ensure that this gross and fast dissection does not damage the organ. The diaphragm is cut to the right, and the incision is then continued between the right kidney and the liver, usually dividing the adrenal gland which is a good sign that none of the adjacent organs are damaged. The location for division of the infrahepatic IVC depends on the renal veins. These are identified on both sides, and the IVC can be safely divided on the virtual line about 1 cm above the renal veins. The only structures now holding the liver in the abdomen are the diaphragmatic pillars. By keeping the liver to the right thoracic cavity and holding the aortic patch, the resected IVC, and the portal vein with its cannulae, the liver removal can be completed by cutting the diaphragmatic muscles. The liver is freed and taken out of the abdomen. A further perfusion with cool preservation solution should be performed on the back table before packing the liver in the transportation box usually with 1 l of preservation solution. The liver can now be packed in the transportation box.
Liver Disease and Perioperative Risk
The liver is the biggest intestinal organ and plays a central role in the homeostasis of different physiological systems including nutrition and drug metabolism, the synthesis of plasma proteins and haemostatic factors, as well as the elimination of different endogenous and exogenous substances. Although the liver contributes with only 3% to total body weight, given its major role in homeostasis and high energy consumption, it receives 25% of total cardiac output (CO). Two vessels contribute to the perfusion of the liver. The majority (70%) of the hepatic perfusion is provided by the portal vein, which contributes 50% of the organ’s oxygen demand. The other 50% is provided by the hepatic artery, which makes up around 30% of total liver perfusion. Hepatic arterial blood flow is mainly dependent on the organ’s metabolic demands and controlled via autoregulatory mechanisms, whereas blood supply through the portal vein depends on the perfusion throughout the whole gastrointestinal tract and the spleen. This unique, dual perfusion system provides constant perfusion rates and oxygen supply, which is crucial for adequate liver function. These high oxygen demands are reflected in a hepatic vein saturation of almost 30%.
The liver is also unique in its ability of regeneration, which allows the performance of major surgery including, amongst others, extended resections of liver tumours, living donations and so on. Many patients have normal liver function parameters when they present for liver surgery, especially when the reason for resection is metastasis or a benign liver tumour. The most common causes of liver resections are the hepatocellular carcinoma (HCC) and the cholangiocellular carcinoma (CCC). Hepatocellular carcinoma (HCC) often develops in patients with underlying liver cirrhosis; many of these patients show signs of chronic liver dysfunction (CLD).
As explained previously, the liver plays a central role in a great deal of physiological systems. Therefore, in case of chronic liver dysfunction (CLD) or liver failure, several effects on other organ systems have to be expected. Consequently liver resections and bile duct surgery as having a high risk for perioperative cardiac events, with an estimated 30-day cardiac event rate (cardiac death and myocardial infarction) of more than 5%. Patients undergoing liver surgery pose a significant challenge to treating physicians in the perioperative period. Due to the improvement of surgical techniques, the “liver patient” is becoming more and more complex, confronting surgeons, anaesthetists and intensive care personnel with difficult intra- and postoperative courses, and considerable multiorgan disorders. The cornerstones of an optimal management are careful selection of the patients, appropriate monitoring and protection of the liver and other vital organs.
Role of The SURGEON
Attributes of a Good Surgeon
Realising the benefits that good leadership and teamwork can deliver requires commitment from all those involved in patient care. From the surgeon’s viewpoint there are numerous desirable attributes which are developed through medical school education, foundation training, core training and into professional practice. These are outlined below:
1. Clinical Care
An obvious consideration of what makes a “good surgeon” is the care provided to patients throughout the patient journey. This includes technical ability in the operating theatre and non-technical skills.
2. Maintenance and Improvement
Remaining up-to-date with innovations in surgical practice and patient are is an important attribute of a good surgeon. In doing so, one is able to inform patients and explain the reasons for and against procedures, allowing them to make an informed decision. Willingness to learn from others and improve from others by reviewing personal practice forms part of Continuing Professional Development; this is a requirement in a portfolio to meet revalidation and recertification criteria.
3. Teaching, Training and Supervision
Educating others forms part of professional development and surgeons frequently oversee projects for medical students or trainees. This requires knowledge of the objectives of the tasks undertaken, knowledge of what technical and non-technical skills should be improved and knowledge of how to encourage the development of these skills. The mentormentee relationship should work both ways, such that the mentee is able to approach their supervisor for assistance and is accepting of any constructive criticism delivered.
4. Relationships with Patients
Relationships with patients are fundamentally based on trust; the patient trusts that the surgeon will do all in their power to help them and their surgical journey. Obtaining informed consent prior to clinical care is based on trust and allows patient autonomy to be upheld. Developing relationships with patients begins from the first consultation and is continued after the day of an operation being undertaken. Acknowledging the needs of the individual and employing effective communication helps in developing an open relationship. In this way patients disclose their medical history and admit underlying fears, allowing better patient care to be delivered.
5. Relationships with Colleagues
Partnership with all members of the multidisciplinary clinical team, management, technicians and support staff fosters healthy working relationships. Consequently, patient care is enhanced through communication, enhanced productivity and an improved team dynamic. Understanding how a colleague works and taking action to facilitate a positive working environment is beneficial to all. Emotional intelligence forms an important component of working relationships, through the ability “to understand and recognize emotional states and to use that understanding to manage one’s self and other individuals or teams”.
6. Health
Maintenance of good personal health and knowing when you must stop working is important in the protection of patient safety. The relevant senior staff must be informed of communicable disease or blood-borne disease transmission. In addition, being vigilant of the health of colleagues forms part of protecting patient safety, for example, failure to report suspicion that the consultant consistently operates after several glasses of wine or that the CT2 has been seen smoking drugs can facilitate the propagation of errors in the workplace. Finally, surgeons are renowned for working at all hours, however acknowledgement that we all need rest is crucial in good patient care.
“Not Only SURGEONS…”
SURGERY, A NOBLE PROFESSION
Surgery is, indeed, one of the noblest of professions. Here is how Dictionary defines the word noble: 1) possessing outstanding qualities such as eminence, dignity; 2) having power of transmitting by inheritance; 3) indicating superiority or commanding excellence of mind, character, or high ideals or morals. These three attributes befit the profession of surgery. Over centuries, the surgical profession has set the standards of ethical and humane practice. Surgeons have made magnificent contributions in education, clinical care, and science. Their landmark accomplishments in surgical science and innovations in operative technique have revolutionized surgical care, saved countless lives, and significantly improved longevity and the quality of human life. Generations of surgeons have developed their craft and passed it on to succeeding generations, as they have to me and to each one of you, to take into the future.
Beyond its scientific and technical contributions, surgery is uniquely fulfilling as a profession. It has disciplined itself over the centuries and dedicated its practice to the best welfare of all human beings. In return, it has been accorded the respect of society, of other professions, and of policy makers. Its conservative stance has served it well and has been the reason for its constancy and consistency. At the beginning of the 21st century, however, profound changes are taking place at all levels and at a dizzying pace, providing both challenges and opportunities to the surgical profession. These changes are occurring on a global level, on the national level, in science and technology, in healthcare, and in surgical education and practice.
To retain its leadership position in innovation and its attractiveness as a career choice for students, surgery must evolve with the times. It is my belief that surgery needs to introduce changes to create new priorities in clinical practice, education, and research; to increase the morale and prestige of surgeons; and to preserve general surgery as a profession. I am reminded of a Chinese aphorism that says, “You cannot prevent the birds of unhappiness from flying over your head, but you can prevent them from building a nest in your hair.”
ADVANCES IN SCIENCE
The coalescence of major advances in science and technology made the end of the 20th century unique in human history. Notable among the achievements are the development of microchips and miniaturization, which fueled the explosion in information technology. The structure of the human genome is nearly completely elucidated, ushering in the genomic era in which genetic information will be used to predict, on an individual basis, susceptibility to disease and responsiveness to drug therapy. The field of nanotechnology allows scientists to work at a resolution of less than one nanometer, the size of the atom. By comparison, the DNA molecule is 2.5 nanometers.
In the last 50 years, biomedical research became increasingly reductionist, turning physiologists and anatomists into molecular biologists. As a result, two basic science fields—integrative physiology and gross anatomy—now have a lower standing in medical education and surgical science than they once did. Surgery and surgical departments can and possibly should claim these fields, but the window of opportunity is narrow. Research is now moving back from discipline-based reductionist science to multidisciplinary science of complexity, in which biomedical scientists work side by side with engineers, mathematicians, and bioinformatists. The ability of high-speed computers to quickly process tens of millions of pieces of data now allows for data-driven rather than hypothesis-based research. This collaboration among different disciplines has already been successful.
TRANSFORMATION OF HEALTHCARE SYSTEM
During the past 75 years, we have seen the entire healthcare system undergo a profound transformation. In the 1930s and for a considerable period thereafter, medical practice was fee-for-service, the doctor–patient relationship was strong, and the physician perceived himself or herself as being responsible nearly exclusively to his or her individual patients. The texture of medical practice started to change when the federal government became involved in the provision of healthcare in 1965. The committee on “Crossing the Quality Chasm” identified six key attributes of the 21st-century healthcare system. It must be:
- Safe, avoiding injuries to patients;
- Effective, providing services based on scientific knowledge;
- Patient-oriented, respectful of and responsive to individual patients’ needs, values, and preferences;
- Timely, reducing waits, eliminating harmful delays for both care receiver and caregiver;
- Efficient, avoiding wasted equipment, supplies, ideas, and energy;
- Equitable, providing equal care across genders, ethnicities, geographic locations, and socioeconomic strata;
No one knows at present what this 21st-century healthcare system will look like. While care in the old system was reactive, in the new system it will be proactive. The “find it, fix it” approach of the old system will be replaced by a “predict it, prevent it, and if you cannot prevent it, fix it” approach. Sporadic intervention, provided only when patients present with illness, will give way to a system in which physicians and other healthcare providers plan 1-, 5-, and 10-year care programs for each patient. Care will be more interactive, with patients taking a more important role in their own care. The technology-oriented system will become a system that provides graded intervention. Delivery systems will not be fractionated but integrated. Even more importantly, care will not be based simply on experience and clinical impression but on evidence of proven outcome measures. If the old system was cost-insensitive, the new system will be cost-sensitive.
SURGICAL PRACTICE
There are many reasons for the declining interest in general surgery, some of which parallel reasons for the drop in medical school applicants in general. One problem specific to surgery is that medical students are given less and less exposure to surgery, due to the shortening of required surgical rotations. Most important, however, is their perception that the life of the surgical resident is stressful, the work hours too long, and the time for personal and family needs inadequate. The workload of the surgical resident over the years has increased significantly both in amount and intensity, without concomitant increase in the number of residents and at a time when hospitals have significantly reduced the support personnel on the surgical ward and in the operating rooms. Students graduating with debts close to $100,000 simply find the years of training in surgery too long, followed by uncertain practice income after graduation.
From several recent studies, lifestyle is the critical and most pressing issue in surgical residency. Some studies have also shown that the best students tend to select specialties that provide controllable lifestyles, such as radiology, dermatology, and ophthalmology. We have a problem not only in the declining number of students applying for surgical training but also in the declining quality of those who do apply. In a preliminary survey of 153 responding general surgery programs, we found that attrition (i.e., categorical residents leaving the training programs) occurred at a rate of 13% to 19% in the last 5 years. In 2001, 46% of those leaving general surgery training programs cited lifestyle as the major reason.
Unless these trends are reversed, general surgery as a specialty is threatened, and a future shortage of general surgeons is inevitable. I know that the Council of the American Surgical Association is most concerned about the crisis in general surgery. We must do a better job of communicating to students and residents that the practice of surgery is as rewarding as ever and full of opportunities in this new era. Innovations in minimal access and computer-assisted surgery and simulation technology provide exciting new possibilities in surgical training. We must also look very carefully at the demands of surgical residency and improve the life of residents without compromising their surgical experience. Unless we deal with work hours and quality of life issues, we are likely to see continuing decline in the interest of medical students in surgical training.
CONCLUSIONS
In conclusion, the noble profession of surgery must rise to meet numerous challenges as the world in which it operates continues to undergo profound change. These challenges represent opportunities for the profession to develop an international perspective and a global outreach and to address the growing needs of an aging population undergoing major demographic and workforce shifts. The leadership of American surgery has a unique role to play in the formulation of a new healthcare system for the 21st century. This task will require commitment to quality of care and patient safety, and it will depend on harnessing the trust and support of the American public. Advances in science and technology—particularly in minimal access surgery, robotics, and simulation technology—provide unprecedented opportunity for surgeons to continue to make landmark contributions that will improve surgical care and the human condition. I believe it is also crucially important that we train surgeon-scientists who will keep surgery at the cutting edge in the genomic and bioinformatics era. Ours is a noble profession imbued with eminence, dignity, high ideals, and ethical values. It has a rich and proud heritage… and I quote, “The highest intellects, like the tops of mountains, are the first to catch and reflect the dawn.”
Source: Lecture from Haile T. Debas, MD (UCSF School of Medicine, San Francisco, California) Presented at the 122nd Annual Meeting of the American Surgical Association, April 25, 2002, The Homestead, Hot Springs, Virginia.
Vascularização da Árvore Biliar
O Fio da Navalha na Prevenção da Isquemia e Estenose
Autor: Prof. Dr. Ozimo Gama (Tempo de Leitura: 11 minutos)
Introdução
No campo minado que é a cirurgia hepatobiliar, existe um princípio fisiológico que não admite margem para erro: enquanto o parênquima hepático é um órgão privilegiado, nutrido por um duplo suprimento sanguíneo (arterial e venoso portal), a árvore biliar é alimentada exclusivamente pelo sistema arterial. Para o estudante de medicina e o residente de cirurgia, a compreensão desta premissa muda drasticamente a forma como o bisturi e o cautério são manuseados em torno do hilo hepático. A esqueletização excessiva de um ducto biliar não é um sinal de dissecção meticulosa, mas sim a antessala anatómica de uma complicação catastrófica: a isquemia, seguida de necrose e estenose biliar. Neste artigo, dissecaremos a anatomia vascular da árvore biliar, as armadilhas do ducto cístico e os fundamentos anatómicos da cirurgia oncológica da vesícula.
1. A Vesícula Biliar: Topografia e Peculiaridades Histológicas
A vesícula biliar repousa no equador que divide o fígado direito do esquerdo — uma linha imaginária conhecida como Linha de Cantlie (ou linha de Rex-Cantlie), que cursa entre os segmentos IVb e V em direção à veia cava inferior posteriormente.
A vesícula é maioritariamente peritonizada, exceto na sua face posterior, que assenta diretamente na placa cística. No entanto, o seu verdadeiro “Calcanhar de Aquiles” reside na sua histologia: a vesícula biliar não possui muscularis mucosae, não possui submucosa e apresenta uma camada muscular descontínua.
- Implicação Cirúrgica Oncológica: Estas especificidades anatómicas removem as barreiras naturais contra a progressão tumoral, facilitando a invasão direta do cancro da vesícula biliar para o parênquima hepático. É por este motivo que o tratamento cirúrgico curativo exige uma Colecistectomia Radical (que inclui a ressecção em cunha dos segmentos hepáticos IVb e V) sempre que o estadiamento tumoral (T) for superior ou igual a T1b.
2. O Labirinto Anatómico do Ducto Cístico e da Via Biliar Principal
A partir do infundíbulo cónico da vesícula, o ducto cístico estende-se como o limite inferior do triângulo hepatocístico em direção ao hilo, unindo-se ao ducto hepático comum (DHC) para formar o ducto colédoco (VBP). Contudo, na via biliar, a variação anatómica é a regra, não a exceção. O ducto cístico pode correr paralelamente ao DHC, espiralar posteriormente a este e inserir-se na sua face medial. Mais perigoso ainda é quando o cístico se insere no Ducto Setorial Posterior Direito (RPD), uma variação que ocorre em cerca de 4% dos fígados. Esta configuração é notoriamente perigosa, expondo o RPD a um risco altíssimo de secção inadvertida durante uma colecistectomia de rotina. Distalmente, o colédoco entra na cabeça do pâncreas e une-se ao ducto pancreático (Wirsung) para formar a ampola hepatopancreática, controlada pelo Esfíncter de Oddi.
- O Canal Comum Longo: Quando a junção entre o colédoco e o ducto pancreático ocorre de forma anómala (antes do complexo esfincteriano), ocorre o refluxo de enzimas pancreáticas ativadas para a árvore biliar. Este trauma químico crónico é o principal fator de risco para a formação de cistoss do colédoco e para o desenvolvimento de Colangiocarcinoma.
3. O “Tendão de Aquiles”: A Vascularização Arterial da Via Biliar
Como mencionado, a árvore biliar depende de um fluxo arterial ininterrupto. O suprimento segue um padrão de perfusão que ascende e descende ao longo dos ductos:
- O Suprimento Distal (Ascendente): O ducto colédoco recebe a sua irrigação inferior a partir de arteríolas emparelhadas provenientes da Artéria Gastroduodenal (GDA) e da Artéria Pancreatoduodenal Superior Posterior (PSPDA). Esta última representa o suprimento arterial mais importante e constante para o colédoco distal.
- O Suprimento Proximal (Descendente): Proximamente, o colédoco é alimentado por arteríolas originadas da artéria hepática direita (AHD).
- As Artérias Marginais: Estes vasos superiores e inferiores anastomosam-se para formar as artérias marginais, que correm paralelamente ao longo do ducto colédoco, situando-se tipicamente nas posições das 3 e 9 horas (lateral e medialmente). Desnudar (esqueletizar) o colédoco deste suprimento arterial para criar uma anastomose ou durante uma linfadenectomia não regrada acarreta um risco altíssimo de isquemia e estenose cicatricial a longo prazo.
4. O Plexo Epicoledociano Hilar e as Variações Arteriais
No interior do hilo hepático, uma rica rede de capilares liga as artérias hepáticas direita e esquerda. Este leito vascular, denominado Plexo Epicoledociano Hilar, fornece uma circulação colateral crítica que pode manter o suprimento arterial para um lado do fígado caso o vaso ipsilateral seja lesado. A preservação deste plexo e do suprimento sanguíneo arterial para o fígado remanescente é absolutamente crucial na criação de uma anastomose biliodigestiva (hepaticojejunostomia). A isquemia hilar resulta na temível colangiopatia isquémica e no desenvolvimento de abcessos hepáticos intratáveis.
O Papel das Variações Arteriais no Colangiocarcinoma Hilar
Na cirurgia do colangiocarcinoma hilar (Tumor de Klatskin), a complexidade aumenta devido às variações arteriais. A artéria hepática direita cruza posteriormente ao ducto hepático na maioria dos casos, mas em 25% cruza anteriormente. Mais relevante ainda é a presença de uma Artéria Hepática Direita Substituída (ou Acessória), com origem na Artéria Mesentérica Superior (AMS). Ao contrário da anatomia clássica, este vaso cursa lateralmente ao ducto colédoco (e não medialmente), estando extremamente vulnerável à iatrogenia se o cirurgião não o identificar durante a dissecção do ligamento hepatoduodenal. Combinações destas variações com a localização exata do tumor ditarão se uma lesão hilar é ressecável ou irresecável.
Pontos-Chave para a Prática Diária
- A Regra das 3 e 9 Horas: Lembre-se sempre de que o suprimento axial do colédoco viaja nos seus flancos laterais. Evite dissecções circunferenciais extensas da via biliar principal.
- Histologia e Neoplasias: A ausência de submucosa na vesícula biliar dita a necessidade de cirurgia hepática radical (ressecção em cunha do leito) em tumores T1b ou superiores.
- Inserções Anómalas do Cístico: A inserção num ducto setorial direito (RPD) é uma armadilha clássica. A Visão Crítica de Segurança (CVS) e a colangiografia são as únicas formas de evitar a transecção de um ducto principal aberrante.
- O Perigo do Canal Comum: A junção biliopancreática anómala exige vigilância ou tratamento cirúrgico devido ao elevado risco de colangiocarcinoma derivado da inflamação enzimática crónica.
Conclusões Aplicadas à Prática do Cirurgião Digestivo
O respeito pela árvore biliar transcende a mera identificação do tubo que transporta a bílis; exige uma veneração profunda pela sua microvascularização. O cirurgião que descasca a via biliar como se tratasse de um cabo elétrico condena o seu doente a um futuro de estenoses isquémicas, colangites de repetição e reintervenções de extrema complexidade. O domínio da variabilidade anatómica vascular (como as artérias substituídas da AMS) e o conhecimento tático do plexo epicoledociano não são meros detalhes de rodapé dos livros de anatomia; são a linha que separa o triunfo cirúrgico oncológico da catástrofe iatrogénica irreparável.
“A anatomia biliar não segue regras absolutas, apenas tendências. Operar no hilo hepático assumindo a normalidade é o primeiro passo para o desastre. O cirurgião deve dissecar com a expectativa constante da variação.” — Aforismo clássico da cirurgia hepatobiliar.
Gostou ❔Deixe-nos um comentário ✍️, partilhe nas suas redes sociais e|ou envie a sua dúvida pelo 💬 Chat On-line na nossa DM do Instagram.
Surgical Management of PANCREATIC CANCER
INTRODUCTION
Pancreatic ductal adenocarcinoma (PDAC) carries one of the poorest overall prognosis of all human malignancies. The 5-year survival in patients with PDAC, for all stages, remains as low as 6–7%. The low survival rate is attributed to several factors, of which the two most important are aggressive tumor biology and late stage at which most patients are diagnosed. Only 10–20% of patients are eligible for resection at presentation, 30–40% are unresectable/locally advanced, and 50–60% are metastatic. Pancreatic cancer without distant metastasis can be divided into three categories: resectable, borderline resectable, and locally advanced. In absence of metastatic disease, the most important factor for improving survival and possibly offer cure is to achieve a margin-negative resection. Even after potential curative resection, most patients develop recurrences eventually, and 5-year survival of completely resected patients is only up to 25%. The aggressive tumor biology and its inherent resistance to chemotherapy and radiotherapy contributes to early recurrence and metastasis.
SURGICAL ADVANCES
Pancreatic cancer surgery has evolved over the past few decades and remains the cornerstone of treatment of resectable and borderline resectable tumors. Advances in modern imaging give precise information on disease extension and vascular involvement that aids in surgical planning in order to achieve a margin-negative resection.
SURGICAL TECHNIQUES
Surgical techniques for pancreatic cancer include pancreaticoduodenectomy, distal pancreatectomy with splenectomy, and total pancreatectomy. Standard lymphadenectomy for pancreatoduodenectomy should include removal of lymph node stations 5, 6, 8a, 12b1, 12b2, 12c, 13a, 13b, 14a, 14b, 17a, and 17b. Involvement of superior mesenteric vein (SMV)/portal vein(PV) was previously considered as a contraindication for resection. However, curative resection along with SMV/PV with vascular reconstruction has now become a standard practice in specialized high-volume centers. To improve margin-negative resections, specially in borderline resectable tumors with proximity to vascular structures, SMA first approach was proposed as a new modification of standard pancreatico-duodenectomy. In a systematic review, SMA first approach was shown to be associated with better perioperative outcomes, such as blood loss, transfusion requirements, pancreatic fistula, delayed gastric emptying, and reduced local and metastatic recurrence rates. In case of arterial involvement, there is no good evidence at present to justify arterial resections for right-sided pancreatic tumors. However, the modified Appleby procedure, which includes en bloc removal of celiac axis with or without arterial reconstruction, when used in appropriately selected patients, offers margin negative resection with survival benefit for locally advanced pancreatic body and tail tumors and should be performed in high-volume centers. Most evidence does not support advantage of more extended resections such as removal of the para-aortic lymph nodes and nerve plexus and multivisceral resections routinely. Such extended resections are associated with compromised quality of life because of associated higher perioperative morbidity and intractable diarrhea. However, in highly selected patients, with preserved performance status and stable or nonprogressive disease on neoadjuvant treatment, such extended resections can provide survival advantage over palliative treatments. Radical surgery in the presence of oligometastatic disease has also been reported to prolong survival in highly selected patients.
Adverse events in SURGERY
The surgical domain can be seen as more complex and high risk in its delivery of care than other non-interventional specialities. It is therefore not surprising that in the majority of studies of adverse events in healthcare, at least 50% occurred within the surgical domain and the majority of these in the operating theatre. Furthermore, at least half of these adverse events were also deemed preventable. Just as the multiple studies in the developed world have similar figures for adverse events in hospitalised patients across all specialities, there appears to be a similar rate of harm in surgery. A review of 14 studies, incorporating more than 16000 surgical patients, quoted an adverse event occurring in 14.4% of surgical patients. This was not simply minor harm; a full 3.6% of these adverse events were fatal, 10% severe and 34% moderately harmful. Gawande, a surgeon from Boston, made one of the first attempts to clarify the source of these adverse events.

This paper pioneered the concept that the majority of these adverse events were not due to lack of technical expertise or surgical skill on the part of the surgeon, finding instead that ‘systems factors’ were the main contributing factor in 86% of adverse events. The most common system factors quoted were related to the people involved and how they were functioning in their environment. Communication breakdown was a factor in 43% of incidents, individual cognitive factors (such as decision-making) were cited in 86%, with excessive workload, fatigue and the design or ergonomics of the environment also contributing.
These findings were confirmed in the systematic review of surgical adverse events, where it was found that errors in what were described as ‘nonoperative management’ were implicated in 8.32% of the study population versus only 2.5% contributed to by technical surgical error. In accordance with other high-risk industries, such as commercial aviation, the majority of these adverse events are therefore not caused by failures of technical skill on the part of the individual surgeon, but rather lie within the wider healthcare team, environment and system. Lapses and errors in communication, teamworking, leadership, situational awareness or decision-making all feature highly in post-hoc analysis of surgical adverse events. This knowledge of error causation has been prominent and acknowledged in most other high-risk industries for many years, but it is only recently that healthcare has appreciated this.
Hepatocellular ADENOMA
Benign liver tumours are common and are frequently found coincidentally. Most benign liver lesions are asymptomatic, although larger lesions can cause non-specific complaints such as vague abdominal pain. Although rare, some of the benign lesions, e.g. large hepatic adenomas, can cause complications such as rupture or bleeding. Asymptomatic lesions are often managed conservatively by observation. Surgical resection can be performed for symptomatic lesions or when there is a risk of malignant transformation. The type of resection is variable, from small, simple, peripheral resections or enucleations, to large resections or even liver transplantation for severe polycystic liver disease.
General Considerations
Hepatocellular adenomas (HCA) are rare benign hepatic neoplasms in otherwise normal livers with a prevalence of around 0.04% on abdominal imaging. HCAs are predominantly found in women of child-bearing age (2nd to 4th decade) with a history of oral contraceptive use; they occur less frequently in men. The association between oral contraceptive usage and HCA is strong and the risk for a HCA increases if an oral contraceptive with high hormonal potency is used, and if it is used for over 20 months. Long-term users of oral contraceptives have an estimated annual incidence of HCA of 3–4 per 100000. More recently, an increase in incidence in men has been reported, probably related to the increase in obesity, which is reported as another risk factor for developing HCA. In addition, anabolic steroid usage by body builders and metabolic disorders such as diabetes mellitus or glycogen storage disease type I are associated with HCAs. HCAs in men are generally smaller but have a higher risk of developing into a malignancy. In the majority of patients, only one HCA is found, but in a minority of patients more than 10 lesions have been described (also referred to as liver adenomatosis).
Clinical presentation
Small HCAs are often asymptomatic and found on abdominal imaging being undertaken for other purposes, during abdominal surgery or at autopsy. Some patients present with abdominal discomfort, fullness or (right upper quadrant) pain due to an abdominal mass. It is not uncommon that the initial symptoms of a HCA are acute onset of abdominal pain and hypovolaemic shock due to intraperitoneal rupture. In a series of patients who underwent resection, bleeding was reported in up to 25%. The risk of rupture is related to the size of the adenoma. Exophytic lesions (protruding from the liver) have a higher chance of bleeding compared to intrahepatic or subcapsular lesions (67% vs 11% and 19%, respectively, P<0.001). Lesions in segments II and III are also at higher risk of bleeding compared to lesions in the right liver (35% vs 19%, P = 0.049).
Management
There is no guideline for the treatment of HCAs, although there are general agreements. In men, all lesions should be considered for surgical resection independent of size, given the high risk of malignant transformation, while taking into account comorbidity and location of the lesion. Resection should also be considered in patients with HCAs due to a metabolic disorder. In women, lesions <5 cm can be observed with sequential imaging after cessation of oral contraceptive treatment. In larger tumours, treatment strategies vary. Some clinicians have proposed non-surgical management if hormone therapy is stopped and patients are followed up with serial radiological examinations. The time period of waiting is still under debate, however recent studies indicate that a waiting period of longer than 6 months could be justified.
More recently, the subtypes of the Bordeaux classification of HCA have been studied related to their risk of complications. Some groups report that percutaneous core needle biopsy is of limited value because the therapeutic strategy is based primarily on patient sex and tumour size. Others report a different therapeutic approach based on subtype. Thomeer et al. concluded that there was no evidence to support the use of subtype classification in the stratification and management of individual patients related to risk of bleeding. Size still remains the most important feature to predict those at risk of bleeding during follow-up. However, malignant transformation does seem to be related to differences in subtypes. β-catenin-mutated HCAs trigger a potent mitogenic signalling pathway that is prominent in HCC. Cases of inflammatory HCAs can also show activation of the β-catenin pathway with a risk of developing malignancy. Therefore, β-catenin-mutated and inflammatory HCAs are prone to malignant degeneration, and particularly if >5cm. In these circumstances, invasive treatment should be considered.
Liver Uptake on Cadaveric Donors for Transplant
Since the initial descriptions of orthotopic liver transplantation (OLT) in the 1960s, both the number of patients receiving transplants and the indications for the procedure have increased significantly. OLT represents the only treatment modality for many patients with a diverse spectrum of disease, with the predominant common factor end-stage liver failure. Advances in perioperative care of the donor and recipient, organ preservation methods, and surgical techniques have resulted in a 5 year overall survival of 78% for all recipients (Kim et al, 2015).
OPERATIVE TECHNIQUES
The first published description of human liver transplantation was by Starzl and colleagues in 1963 at the University of Colorado. In this seminal paper, the dismal outcomes of three OLT recipients were described, including one intraoperative death from uncorrectable coagulopathy and two survivors of 7 and 22 days. In addition to the pioneering conceptual framework and implementation of LT, the advanced techniques included grafts from non–heart-beating donors, venovenous bypass in the recipients, choledochocholedochostomy, and coagulation monitoring by using thromboelastography (TEG). Many of these concepts remain or have reentered the realm of LT more than 40 years after their initial description. Based largely on the initial body of work by Starzl and colleagues, this section describes the surgical procedures commonly used worlwide.
The typical deceased donor has had a catastrophic head injury or an intracerebral bleed, with brain death but without multisystem organ failure. Electrolyte imbalance and hepatic steatosis in the donor are predictors of graft nonfunction. A “donor risk index” has been derived to assess the likelihood of good graft function. Key adverse factors include older donor age (especially >60 years of age), use of a split or partial graft, and a non–heart-beating donor, from which the organs are harvested after the donor’s cardiac output ceases, in contrast to the more typical deceased donation in which the organs are harvested prior to cardiovascular collapse. Use of non–heart-beating donors is associated with reduced rates of long-term graft survival and increased risk of biliary complications, which correlate with the duration of “warm ischemia” after cardiovascular collapse and before retrieval of the organ. With the critical shortage of deceased organ donors, expansion of the donor pool has included acceptance of donors 70 years of age and older for selected recipients. Prior to hepatectomy, the harvesting team makes a visual and, if necessary, histologic assessment of the donor organ. Particular attention is paid to anatomic variants in the hepatic artery that may complicate the graft arterial anastomosis in the recipient. Once donor circulation is interrupted, the organ is rapidly infused with a cold preservation solution (e.g., University of Wisconsin, histidine-tryptophan-ketoglutarate, or Institut Georges Lopez solution). Donor iliac arteries and veins are also retrieved in case vascular grafting is required. After its arrival at the recipient institution, further vascular dissection, with arterial reconstruction if necessary, is performed before implantation.
Future Perspectives
Major challenges remain in LT, including the shortage of donor organs, threat of recurrent disease, and morbidity associated with lifelong therapeutic immunosuppression. Nevertheless, the availability of LT has transformed the lives of patients with advancing liver disease and their health care providers from an ultimately futile effort to manage the complications of cirrhosis into a life-prolonging and life-enhancing intervention.