Minimally Invasive Approach to Choledocholithiasis
Introduction
The incidence of choledocholithiasis in patients undergoing cholecystectomy is estimated to be 10 %. The presence of common bile duct stones is associated with several known complications including cholangitis, gallstone pancreatitis, obstructive jaundice, and hepatic abscess. Making the diagnosis early and prompt management is crucial. Traditionally, when choledocholithiasis is identified with intraoperative cholangiography during the cholecystectomy, it has been managed surgically by open choledochotomy and place- ment of a T-tube. This open surgical approach has a morbidity rate of 10–15 %, mortality rate of <1 %, with a <6 % incidence of retained stones. Patients who fail endoscopic retrieval of CBD stones, as well as cases in which an endoscopic approach is not appropriate, should be explored surgically.
Clinical Manifestation
Acute obstruction of the bile duct by a stone causes a rapid distension of the biliary tree and activation of local pain fibers. Pain is the most common presenting symptom for choledocholithiasis and is localized to either the right upper quadrant or to the epigastrium. The obstruction will also cause bile stasis which is a risk factor for bacterial over- growth. The bacteria may originate from the duodenum or the stone itself. The combination of biliary obstruction and colo- nization of the biliary tree will lead to the development of fevers, the second most common presenting symptom of cho- ledocholithiasis. Biliary obstruction, if unrelieved, will lead to jaundice. When these three symptoms (pain, fever, and jaundice) are found simultaneously, it is known as Charcot’s triad. This triad suggests the diagnosis of acute ascending cholangitis, a potentially life-threatening condition. If not treated promptly, this can lead to hypotension and decreased metal status, both signs of severe sepsis. When combined with Charcot’s triad, this constellation of symptoms is commonly referred to as Reynolds pentad.
Laparoscopic common bile duct exploration
Laparoscopic common bile duct exploration (LCBDE) allows for single stage treatment of gallstone disease, reducing overall hospital stay, improving safety and cost-effectiveness when compared to the two-stage approach of ERCP and laparoscopic cholecystectomy. Bile duct clearance can be confirmed by direct visualization with a choledochoscope. But, before the advent of choledochoscope, bile duct clearance was uncertain, and blind instrumentation of the duct resulted in accentuated edema and inflammation. Due to advancement in instruments, optical magnification, and direct visualization, laparoscopic exploration of the CBD results in fewer traumas to the bile duct. This has led to an increasing tendency to close the duct primarily, reducing the need for placement of T-tubes. Still, laparoscopic bile duct exploration is being done in only a few centers. Apart from the need for special instruments, there is also a significant learning curve to acquire expertise to be able to perform a laparoscopic bile duct surgery.
Morbidity and mortality rates of laparoscopic exploration are comparable to ERCP (2–17 and 1–5 %), and there is no clear difference in primary success rates between the two approaches. However, the endoscopic approach may be preferable for elderly and frail patients, who are at higher risk with surgery. Patients older than 70–80 years of age have a 4–10 % mortality rate with open duct exploration. It may be as high as 20 % in elderly patients undergoing urgent procedures. In comparison, advanced age and comor- bidities do not have a significant impact on overall complication rates for ERCP. A success rate of over 90 % has been reported with laparoscopic CBD exploration. Availability of surgical expertise and appropriate equipment affect the success rate of laparoscopic exploration, as does the size, number of the CBD stones, as well as biliary anatomy. Over the years, laparoscopic exploration has become efficient, safe, and cost effective. Complications include CBD laceration, stricture formation, bile leak, abscess, pancreatitis, and retained stones.
In cases of failure of laparoscopic CBD exploration, a guidewire or stent can be passed through the cystic duct, common bile duct, and through the ampulla into the duodenum followed by cholecystectomy. This makes the identification and cannulation of the ampulla easier during the post- operative ERCP. Laparoscopic common bile duct exploration is traditionally performed through a transcystic or transductal approach. The transcystic approach is appropriate under certain circumstances. These include a small stone (<10 mm) located in the CBD, presence of small common bile duct (<6 mm), or if there is poor access to the common duct. The transductal approach is preferable in cases of large stones, stones in proximal ducts (hepatic ducts), large occluding stones in a large duct, presence of multiple stones, or if the cystic duct is small (<4 mm) or tortuous. Contraindications for laparoscopic approach include lack of training, and severe inflammation in the porta hepatis making the exploration difficult and risky.
Key Points
With advancement in imaging technology, laparoscopic and endoscopic techniques, management of common bile duct stone has changed drasti- cally in recent years. This has made the treatment of this condition safe and more efficient. Many options are now available to manage this condition, and any particular modality for treatment should be chosen carefully based on the patient related factors, institutional protocol, available expertise, resources, and cost-effectiveness.
Classroom: M.I.A. of Choledocholithiasis
Management of Complicated Appendicitis: Open or Laparoscopic Surgery?
Patients with acute appendicitis can present at different stages of the disease process, ranging from mild mucosal inflammation to frank perforation with abscess formation. The reported overall incidence of acute appendicitis varies with age, gender, and geographical differences. Interestingly, while the incidence of non-perforated appendicitis in the United States decreased between 1970 and 2004, no significant decline in the rate of perforated appendicitis was observed despite the increasng use of computed tomography (CT) and fewer negative appendectomies.
Of 32,683 appendectomies sampled from the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) hospitals between 2005 and 2008, 5,405 patients (16.5%) had a preoperative diagnosis of acute appendicitis with peritonitis/abscess.
The definition of complicated appendicitis varies slightly in the literature. Clinicopathological diagnoses (gangrenous, perforated, appendiceal abscess/phlegmon) of acute appendicitis are commonly used for its definition. Classically, patients at the extremes of age are more likely to present with complicated appendicitis. Similarly, pre-morbid conditions including diabetes and type of medical insurance are significantly associated with the risk of perforation.
The importance of early appendectomy has also been emphasized to prevent perforation of the appendix and the sub- sequent negative impact on patient outcomes. However, more recent meta-analysis data supports the safety of a relatively short (12–24 h) delay before appendectomy, which was not significantly associated with increased rate of complicated appendicitis. Teixeira et al. also showed that the time to appendectomy was not a significant risk factor for perforated appendicitis but did result in a significantly increased risk of surgical site infection.
The outcome of patients with complicated appendicitis is significantly worse than patients with uncomplicated appendicitis. A population-based study from Sweden showed that, in a risk-adjusted model, patients with perforated appendicitis were 2.34 times more likely to die after appendectomy than non- perforated appendicitis patients. Because of its higher mortality and morbidity in patients with complicated appendicitis, the management of complicated appendicitis has evolved significantly over the last few decades.
Open or Laparoscopic Surgery
Since the first laparoscopic appendectomy was described by Semm in 1983, multiple studies have compared operative time, complication rates, length of hospital stay, hospital cost, and other outcomes between open and laparoscopic appendectomy for acute appendicitis. The most recent Cochrane review included 67 studies showing that laparoscopic appendectomy was associated with a lower incidence of wound infection, reduced postoperative pain, shorter postoperative length of hospital stay, and faster recovery to daily activity. In contrast, reduced risk of intra-abdominal abscesses and shorter operative time were found as the advantages of open appendectomy.
Due to increased surgeon experience in uncomplicated appendicitis, laparoscopic appendectomy is more frequently attempted even in complicated appendicitis cases as an alternative approach to open appendectomy. Although the general surgical steps for complicated appendicitis are similar to those for uncomplicated appendicitis, the laparoscopic procedure can be more technically demanding. Therefore, conversion from laparoscopic appendectomy to open appendectomy can be expected.
Despite these concerns, the laparoscopic approach in patients with com- plicated appendicitis has been proven to be safe and comparable to open appendectomy. Retrospective studies using a large database in the United States uniformly showed more favorable clinical outcomes (mortality, morbidity, length of hospital stay, readmission rate) and hospital costs in patients who underwent laparoscopic appendectomy when compared to open appendectomy. The real risk of developing an intra- abdominal abscess after laparoscopic appendectomy remains unclear. A meta-analysis by Markides et al. found no significant difference in the intra-abdominal abscess rate between laparoscopic and open appendectomy for complicated appendicitis, whereas Ingraham et al. showed a higher likelihood of developing an organ-space surgical site infection in patients undergoing laparoscopic appendectomy.
Non Operative Management of SPLENIC TRAUMA

The spleen, an important component of the reticuloendothelial system in normal adults, is a highly vascular solid organ that arises as a mass of differentiated mesenchymal tissue during early embryonic development. The normal adult spleen weighs between 75 and 100 g and receives an average blood flow of 300 mL/min. It functions as the primary filter of the reticuloendothelial system by sequestering and removing antigens, bacteria, and senescent or damaged cellular elements from the circulation. In addition, the spleen has an important role in humoral immunity because it produces immunoglobulin M and opsonins for the complement activation system.
The increased availability of high-resolution CT scan and advances in arterial angiography and embolization techniques have contributed to the success of nonoperative management of splenic injuries.
The hemodynamically stable patient with blunt splenic trauma can be adequately managed with bed rest, serial abdominal exams, and hemoglobin and hematocrit monitoring. This approach, in combination with occasional angiography, especially for grade III and IV injuries, confers a splenic salvage rate of up to 95%.
In the setting of expectant management, indications for angiography have been delineated by several studies and include the following CT scan features: contrast extravasation, the presence of a pseudoaneurysm, significant hemoperitoneum, high-grade injury, and evidence of a vascular injury. The goal of angiography is to localize bleeding and embolize the source with coils or a gelatin foam product. Embolization can occur either at the main splenic artery just distal to the dorsal pancreatic portion of the vessel—known as proximal embolization—or selectively at the distal branch of the injured vessel. The goal behind the former technique is to decrease the perfusion pressure to the spleen to encourage hemostasis. The disadvantage to this technique is global splenic ischemia, and many have questioned the spleen’s immunocompetence following proximal embolization.
Malhotra et al. examined the effects of angioembolization on splenic function by examining serum levels of a particular T-cell line. T-cell proportions between patients who had undergone splenic embolization with asplenic patients and healthy controls were similar suggesting some degree of splenic immunocompetency was maintained. A Norwegian study comparing blood samples from patients who had undergone angioembolization with healthy controls demonstrated that the study samples had similar levels of pneumococcal immunoglobulins and no Howell-Jolly bodies, suggesting normal splenic function. Although these preliminary studies remain encouraging, there is no definitive evidence that splenic immunocompetency is fully maintained following angio-embolization.
There is no question that advancements in interventional techniques have contributed to the successful nonoperative management of splenic injuries. This has certainly changed the strategy, but it has not completely replaced operative intervention. The challenge now remains predicting those patients who will ultimately require splenectomy.
Many groups have studied potential predictors of nonoperative failure. Earlier studies found that a higher injury grade, increased transfusion requirement, and hypotension on initial presentation consistently predicted failure of nonoperative management. More recent literature reflects the use of advanced imaging techniques for predicting which patients will ultimately require splenectomy. Haan looked at the overall outcomes of patients admitted with blunt splenic trauma and reported several radiographic findings that were prevalent among patients requiring splenectomy after angioembolization:
- contrast extravasation,
- pseudoaneurysm,
- significant hemoperitoneum,
- and arteriovenous fistula.
Among these characteristics, an arteriovenous fistula had the highest rate of nonoperative failure at 40%. Nonradiographic features associated with significant risk of nonoperative failure include age greater than 40, injury severity score of 25 or greater, or presence of large-volume hemoperitoneum.
Aside from radiographic findings, some groups have also examined the mechanism of injury and its association with nonoperative failure. Plurad et al. conducted a retrospective review over a 15-year period and found that patients who were victims of blunt assault were more likely to fail nonoperative management: 36% of these patients required splenectomy versus 11.5% of patients from all other mechanisms combined. These findings suggest that regardless of overall injury severity, individuals who sustain a direct transfer of injury to the left torso are more likely to require splenectomy.
Currently, the accepted standard of care for most splenic trauma is expectant management with close observation. Operative intervention is reserved for the hemodynamically labile patient who shows signs of active hemorrhage and who does not respond appropriately to fluid resuscitation. Although these clinical scenarios seem straightforward, it is often the condition of the patient who falls in between the two ends of the spectrum that can be the most challenging to manage. In the setting of advanced imaging techniques and interventional radiology, the trauma surgeon has more diagnostic information and more treatment options for the patient with splenic trauma.
IPMN Surgical Management

INTRODUCTION
IPMNs were first recognized in 1982 by Ohashi, but the term IPMN was not officially used until 1993. IPMNs are defined in the WHO Classification of Tumors of the Digestive System as an intraductal, grossly visible epithelial neoplasm of mucin-producing cells. Using imaging and histology, IPMNs can be classified into three types based on duct involvement:
1. Main-duct IPMN (approximately 25% of IPMNs): Segmental or diffuse dilation of the main pancreatic duct (>5 mm) in the absence of other causes of ductal obstruction.
2. Branch-duct IPMN (approximately 57% of IPMNs): Pancreatic cysts (>5 mm) that communicate with the main pancreatic duct.
3. Mixed type IPMN (approximately 18% of IPMNs): Meets criteria for both main and branch duct.
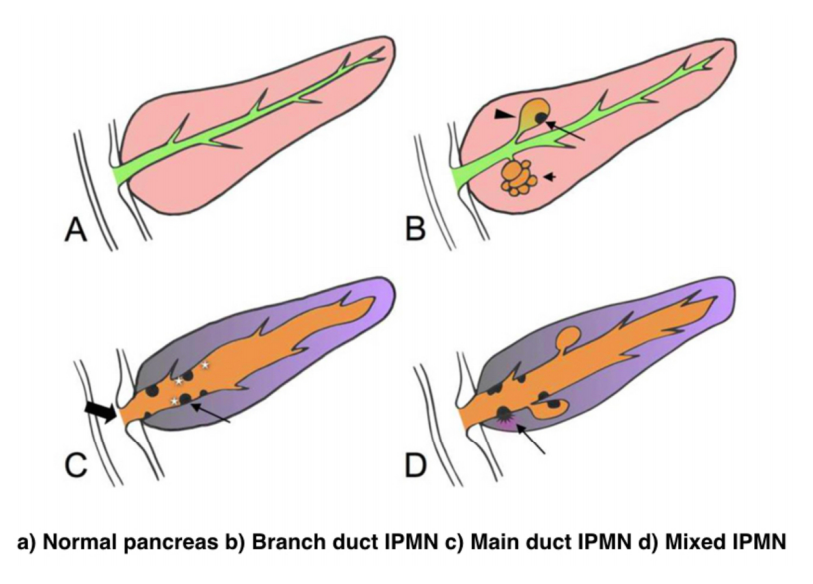
Due to the asymptomatic nature of the disease, the overall incidence of IPMNs is difficult to define but is thought to account for approximately 3% to 5% of all pancreatic tumors. Most IPMNs are discovered as incidental lesions from the workup of an unrelated process by imaging or endoscopy. IPMNs are slightly more prevalent in males than in females, with a peak incidence of 60 to 70 years of age. Branch-duct IPMNs tend to occur in a slightly younger population and are less associated with malignancy compared with main-duct or mixed variants.
Because a majority of IPMNs are discovered incidentally, most are asymptomatic. When symptoms do occur, they tend to be nonspecific and include unexplained weight loss, anorexia, abdominal pain, and back pain. Jaundice can occur with mucin obstructing the ampulla or with an underlying invasive carcinoma. The obstruction of the pancreatic duct can also lead to pancreatitis. IPMNs may represent genomic instability of the entire pancreas. This concept, known as a “field defect,” has been described as a theoretical risk of developing a recurrent IPMN or pancreatic adenocarcinoma at a site remote from the original IPMN. The three different types of IPMNs, main duct, branch duct, and mixed duct, dictate different treatment algorithms.
MAIN DUCT IPMNs
Main-duct IPMNs should be resected in all patients unless the risks of existing comorbidities outweigh the benefits of resection. The goal of operative management of IPMNs is to remove all adenomatous or potentially malignant epithelium to minimize recurrence in the pancreas remnant. There are two theories on the pathophysiologic basis of IPMNs. The first groups IPMNs into a similar category as an adenocarcinoma, a localized process involving only a particular segment of the pancreas. The thought is that removal of the IPMN is the only treatment necessary. In contrast, some believe IPMNs to represent a field defect of the pancreas. All of the ductal epithelium remains at risk of malignant degeneration despite removal of the cyst. Ideally, a total pancreatectomy would eliminate all risk, but this is a radical procedure that is associated with metabolic derangements and exocrine insufficiency. Total pancreatectomy should be limited to the most fit patients, with a thorough preoperative assessment and proper risk stratification prior to undertaking this surgery.
There is less uncertainty with treatment of main-duct IPMNs. The high incidence of underlying malignancy associated with the IPMNs warrants surgical resection. IPMNs localized to the body and tail (approximately 33%) can undergo a distal pancreatectomy with splenectomy. At the time of surgery, a frozen section of the proximal margin should be interpreted by a pathologist to rule out high-grade dysplasia. A prospective study identified a concordance rate of 94% between frozen section and final pathologic examination. If the margin is positive (high-grade dysplasia, invasion) additional margins may be resected from the pancreas until no evidence of disease is present. However, most surgeons will proceed to a total pancreatectomy after two subsequent margins demonstrate malignant changes. This more extensive procedure should be discussed with the patient prior to surgery, and the patient should be properly consented regarding the risks of a total pancreatectomy.
IPMNs localized to the head or uncinate process of the pancreas should undergo a pancreaticoduodenectomy. A frozen section of the distal margin should be analyzed by pathology for evidence of disease. As mentioned before, after two additional margins reveal malignant changes, a total pancreatectomy is usually indicated (approximately 5%). The absence of abnormal changes in frozen sections does not equate to negative disease throughout the pancreas remnant. Rather, skip lesions involving the remainder of the pancreas can exist and thus patients ultimately still require imaging surveillance after successful resection. A prophylactic total pancreatectomy is rarely performed because the subsequent pancreatic endocrine (diabetes mellitus) and exocrine deficits (malnutrition) carry an increased morbidity.
BRANCH DUCT IPMNs
Localized branch-duct IPMN can be treated with a formal anatomic pancreatectomy, pancreaticoduodenectomy, or distal pancreatectomy, depending on the location of the lesion. However, guidelines were established that allow for nonoperative management with certain branch- type IPMN characteristics.
These include asymptomatic patients with a cyst size less than 3 cm and lack of mural nodules. The data to support this demonstrate a very low incidence of malignancy (approximately 2%) in this patient group. Which nearly matches the anticipated mortality of undergoing a formal anatomic resection. In approximately 20% to 30% of patients with branch- duct IPMNs, there is evidence of multifocality. The additional IPMNs can be visualized on high-resolution CT or MRI imaging. Ideally, patients with multifocal branch-duct IPMNs should undergo a total pancreatectomy. However, as previously mentioned, the increased morbidity and lifestyle alterations associated with a total pancreatectomy allows for a more conservative approach. This would include removing the most suspicious or dominant of the lesions in an anatomic resection and follow-up imaging surveillance of the remaining pancreas remnant. If subsequent imaging demonstrates malignant charac- teristics, a completion pancreatectomy is usually indicated.
RECURRENCE RATES
Recurrence rates with IPMNs are variable. An anatomic resection of a branch-duct IPMN with negative margins has been shown to be curative. The recurrence of a main- duct IPMN in the remnant gland is anywhere from 0% to 10% if the margins are negative and there is no evidence of invasion. Most case series cite a 5-year survival rate of at least 70% after resection of noninvasive IPMNs. In contrast, evidence of invasive disease, despite negative margins, decreases 5-year survival to 30% to 50%. The recurrence rate in either the pancreatic remnant or distant sites approaches 50% to 90% in these patients. Histopathologic subtype of the IPMN is correlated with survival. The aggressive tubular subtype has a 5-year survival ranging from 37% to 55% following surgical resection, whereas the colloid subtype has 5-year survival ranging from 61% to 87% post resection. Factors associated with decreased survival include tubular subtype, lymph node metastases, vascular invasion, and positive margins. IPMNs with evidence of invasion should be treated similar to pancreatic adenocarcinomas. Studies show that IPMNs tend to have better survival than pancreatic adenocarcinoma. This survival benefit may be secondary to the less aggressive tumor biology or the earlier diagnosis of IPMNs.
SURVEILLANCE
All patients who have a resected IPMN should undergo imaging surveillance. There is continual survival benefit with further resection if an IPMN does recur. International Consensus Guidelines published in 2017 offer recom- mendations for the frequency and modality of imaging surveillance after resection. Routine serum measurement of CEA and CA 19-9 has a limited role for detection of an IPMN recurrence. Of note, a new pancreatic lesion discovered on imaging after resection could represent a postoperative pseudocyst, a recurrence of the IPMN from inadequate resection, a new IPMN, or an unrelated new neoplastic process. IPMNs may also be associated with extrapancreatic neoplasms (stomach, colon, rectum, lung, breast) and pancreatic ductal adenocarcinoma. It is unclear if this represents a true genetic syndrome. However, patients with IPMNs should have a discussion about the implications of their disease with their physician and are encouraged to undergo colonoscopy to exclude a synchronous neoplastic process.
The incidence of PANCREATIC CYSTIC LESIONS will continue to increase as imaging technology improves. EUS, cytology, and molecular panels have made differentiating the type of PCN less problematic. The importance of an accurate preoperative diagnosis ensures that operative management is selectively offered to those with high-risk lesions. Management beyond surgery, including adjuvant therapy and surveillance, continue to be active areas of research.
Perioperative Medicine

Surgery and anesthesia profoundly alter the normal physiologic and metabolic states. Estimating the patient’s ability to respond to these stresses in the postoperative period is the task of the preoperative evaluation. Perioperative complications are often the result of failure, in the preoperative period, to identify underlying medical conditions, maximize the patient’s preoperative health, or accurately assess perioperative risk. Sophisticated laboratory studies and specialized testing are no substitute for a thoughtful and careful history and physical examination. Sophisticated technology has merit primarily in confirming clinical suspicion.
Classroom: Perioperative Medicine
Hepatocellular Carcinoma: Resection Versus Transplantation

Hepatocellular carcinoma is the second most common cause of cancer mortality worldwide and its incidence is rising in North America, with an estimated 35,000 cases in the U.S. in 2014. The best chance for cure is surgical resection in the form of either segmental removal or whole organ transplantation although recent survival data on radiofrequency ablation approximates surgical resection and could be placed under the new moniker of “thermal resection”. The debate between surgical resection and transplantation focuses on patients with “within Milan criteria” tumors, single tumors, and well compensated cirrhosis who can safely undergo either procedure. Although transplantation historically has had better survival outcomes, early diagnosis, reversal of liver disease, and innovations in patient selection and neo-adjuvant therapies have led to similar 5-year survival. Transplantation clearly has less risk of tumor recurrence but exposes recipients to long term immunosuppression and its side effects. Liver transplantation is also limited by the severe global limit on the supply of organ donors whereas resection is readily available. The current data does not favor one treatment over the other for patients with minimal or no portal hypertension and normal synthetic function. Instead, the decision to resect or transplant for HCC relies on multiple factors including tumor characteristics, biology, geography, co-morbidities, location, organ availability, social support and practice preference.
Resection Versus Transplantation
The debate between resection and transplantation revolves around patients who have well compensated cirrhosis with Milan criteria resectable tumors. Patients within these criteria represent a very small proportion of those who initially present with HCC. This is especially true in western countries where hepatitis C is the most common cause of liver failure and HCC is a result of the progressive and in most cases advanced cirrhosis.
Given the need for a large number of patients to show statistical significance, it would be difficult to perform a high-quality prospective randomized controlled trial comparing resection and transplantation. In fact the literature revealed that no randomized controlled trials addressing this issue exist. Instead, outcomes of surgical treatment for HCC stem from retrospective analyses that have inherent detection, selection and attrition biases.
Given the numerous articles available on this subject, several meta-analyses have been published to delineate the role of transplantation and resection for treatment of HCC. However, there is reason to be wary of these meta-analyses because they pool data from heterogeneous populations with variable selection criteria and treatment protocols. One such meta-analysis by Dhir et al. focused their choice of articles to strict criteria which excluded studies with non-cirrhotic patients, fibrolamellar HCC and hepato-cholangiocarcinomas but included those with HCC within Milan criteria and computation of 5-year survival; between 1990 and 2011 they identified ten articles that fit within these criteria, of which six were ITT analyses, six included only well-compensated cirrhotics (Child-Pugh Class A without liver dysfunction) and three were ITT analyses of well-compensated cirrhotics.
Analysis of the six ITT studies that included all cirrhotics (n = 1118) (Child-Pugh Class A through C) showed no significant difference in survival at 5 years (OR = 0.600, 95 % CI 0.291– 1.237 l; p=0.166) but ITT analysis of only well-compensated cirrhotics (Child- Pugh Class A) revealed that patients undergoing transplant had a significantly higher 5-year survival as compared to those with resection (OR=0.521, 95 % CI 0.298–0.911; p=0.022).
A more recent ITT retrospective analysis from Spain assessed long-term survival and tumor recurrence following resection or transplant for tumors <5 cm in 217 cirrhotics (Child-Pugh Class A, B and C) over the span of 16 years. Recurrence at 5 years was significantly higher in the resection group (71.6 % vs. 16 % p<0.001) but survival at 4 years was similar (60 % vs. 62 %) which is likely explained by the evolving role of adjuvant therapies to treat post-resection recurrence.
Conclusions
- Patients with anatomically resectable single tumors and no cirrhosis or Child-Pugh Class A cirrhosis with normal bilirubin, HVPG (<10 mmHg), albumin and INR can be offered resection (evidence quality moderate; strong recommendation).
- Patients with Milan criteria tumors in the setting of Child- Pugh Class A with low platelets and either low albumin or high bilirubin or Child-Pugh Class B and C cirrhosis, especially those with more than one tumor, should be offered liver transplantation over resection (evidence quality moderate; strong recommendation).
- Those with Milan criteria tumors and Child-Pugh Class A cirrhosis without liver dysfunction should be considered for transplantation over resection (evidence quality low; weak recommendation).
- No recommendation can be made in regard to transplanting tumors beyond Milan criteria (evidence quality low) except to follow regional review board criteria.
- Pre-transplant therapies such as embolic or thermal ablation are safe and by expert opinion considered to be effective in decreasing transplant waitlist dropout and bridging patients to transplant (evidence quality low, weak recommendation). These interventions should be considered for those waiting longer than 6 months (evi- dence quality low, moderate recommendation).
- Living donor liver transplantation is a safe and effective option for treatment of HCC that are within and exceed Milan criteria (evidence quality moderate, weak recommendation).
The century of THE SURGEONS
Surgery is a profession defined by its authority to cure by means of bodily invasion. The brutality and risks of opening a living person’s body have long been apparent, the benefits only slowly and haltingly worked out. Nonetheless, over the past two centuries, surgery has become radically more effective, and its violence substantially reduced — changes that have proved central to the development of mankind’s abilities to heal the sick.
Consider, for instance, amputation of the leg.
The procedure had long been recognized as lifesaving, in particular for compound fractures and other wounds prone to sepsis, and at the same time horrific. Before the discovery of anesthesia, orderlies pinned the patient down while an assistant exerted pressure on the femoral artery or applied a tourniquet on the upper thigh.
Surgeons using the circular method proceeded through the limb in layers, taking a long curved knife in a circle through the skin first, then, a few inches higher up, through the muscle, and finally, with the assistant retracting the muscle to expose the bone a few inches higher still, taking an amputation saw smoothly through the bone so as not to leave splintered protrusions. Surgeons using the flap method, popularized by the British surgeon Robert Liston, stabbed through the skin and muscle close to the bone and cut swiftly through at an oblique angle on one side so as to leave a flap covering the stump.
The limits of patients’ tolerance for pain forced surgeons to choose slashing speed over precision. With either the flap method or the circular method, amputation could be accomplished in less than a minute, though the subsequent ligation of the severed blood vessels and suturing of the muscle and skin over the stump sometimes required 20 or 30 minutes when performed by less experienced surgeons.
No matter how swiftly the amputation was performed, however, the suffering that patients experienced was terrible. Few were able to put it into words. Among those who did was Professor George Wilson. In 1843, he underwent a Syme amputation — ankle disarticulation — performed by the great surgeon James Syme himself. Four years later, when opponents of anesthetic agents attempted to dismiss them as “needless luxuries,” Wilson felt obliged to pen a description of his experience:
“The horror of great darkness, and the sense of desertion by God and man, bordering close on despair, which swept through my mind and overwhelmed my heart, I can never forget, however gladly I would do so. During the operation, in spite of the pain it occasioned, my senses were preternaturally acute, as I have been told they generally are in patients in such circumstances. I still recall with unwelcome vividness the spreading out of the instruments: the twisting of the tourniquet: the first incision: the fingering of the sawed bone: the sponge pressed on the flap: the tying of the blood-vessels: the stitching of the skin: the bloody dismembered limb lying on the floor.”
It would take a little while for surgeons to discover that the use of anesthesia allowed them time to be meticulous. Despite the advantages of anesthesia, Liston, like many other surgeons, proceeded in his usual lightning-quick and bloody way. Spectators in the operating-theater gallery would still get out their pocket watches to time him. The butler’s operation, for instance, took an astonishing 25 seconds from incision to wound closure. (Liston operated so fast that he once accidentally amputated an assistant’s fingers along with a patient’s leg, according to Hollingham. The patient and the assistant both died of sepsis, and a spectator reportedly died of shock, resulting in the only known procedure with a 300% mortality.)
How to Choose a Mesh in Hernia Repair

Since the introduction of polypropylene (PP) mesh for hernia repair, surgeons continue to discuss the use of mesh in a variety of settings for one of the most common operations performed by general surgeons—hernia repair. This discussion has involved raw materials, cost, and outcomes and for many years referred to only a few products, as manufacturing was limited. Nowadays, with multiple permanent, absorbable, biologic, and hybrid products on the market, the choice of mesh for a hernia repair can be daunting. Increasing clinical complexity further emphasizes the need for individualizing care, but more frequently, hospital supply chain personnel institute product procurement procedures for cost control, limiting mesh choice for surgeons. This can force surgeons into a “one-size-fits-all” practice regarding mesh choice, which may not be ideal for some patients. Conversely, current literature lacks definitive evidence supporting the use of one mesh over another, a fact that has not escaped the radar screen of the hospital supply chain and mesh industry, both of which attempt to limit vendor and mesh choice for financial gain. It is unlikely that this type of “proof” will ever come to fruition. This leaves us with choosing a mesh based on an algorithm that is centered on the patient and the patient’s unique clinical scenario.
Article: Mesh in Hernia Repair
The Surgical Personality

Surgical stereotypes are remnants of the days of pre-anaesthesia surgery and include impulsivity, narcissism, authoritativeness, decisiveness, and thinking hierarchically. Medical students hold these stereotypes of surgeons early in their medical training. As Pearl Katz says in the The Scalpel’s Edge: ‘Each generation perpetuates the culture and passes it on by recruiting surgical residents who appear to resemble them and training these residents to emulate their thinking and behaviour.’ The culture of surgery has evolved, and certain behaviours are rightly no longer seen as acceptable, Non-technical skills such as leadership and communication have become incorporated into surgical training. Wen Shen, Associate Professor of Clinical Surgery at University of California San Francisco, argues that this has gone too far: ‘Putting likeability before surgical outcomes is like judging a restaurant by the waiters and ignoring the food,’ I would argue that operative and communication skills are indivisible, An aggressive surgeon is a threat to patient safety if colleagues are frightened to speak up for fear of a colleague shouting or, worse, throwing instruments. Conversely, a flattened hierarchy promotes patient safety.
Read More










